Ras2 Is Responsible for the Environmental Responses, Melanin Metabolism, and Virulence of Botrytis cinerea
Abstract
:1. Introduction
2. Materials and Methods
2.1. Fungal Strains and Culture Conditions
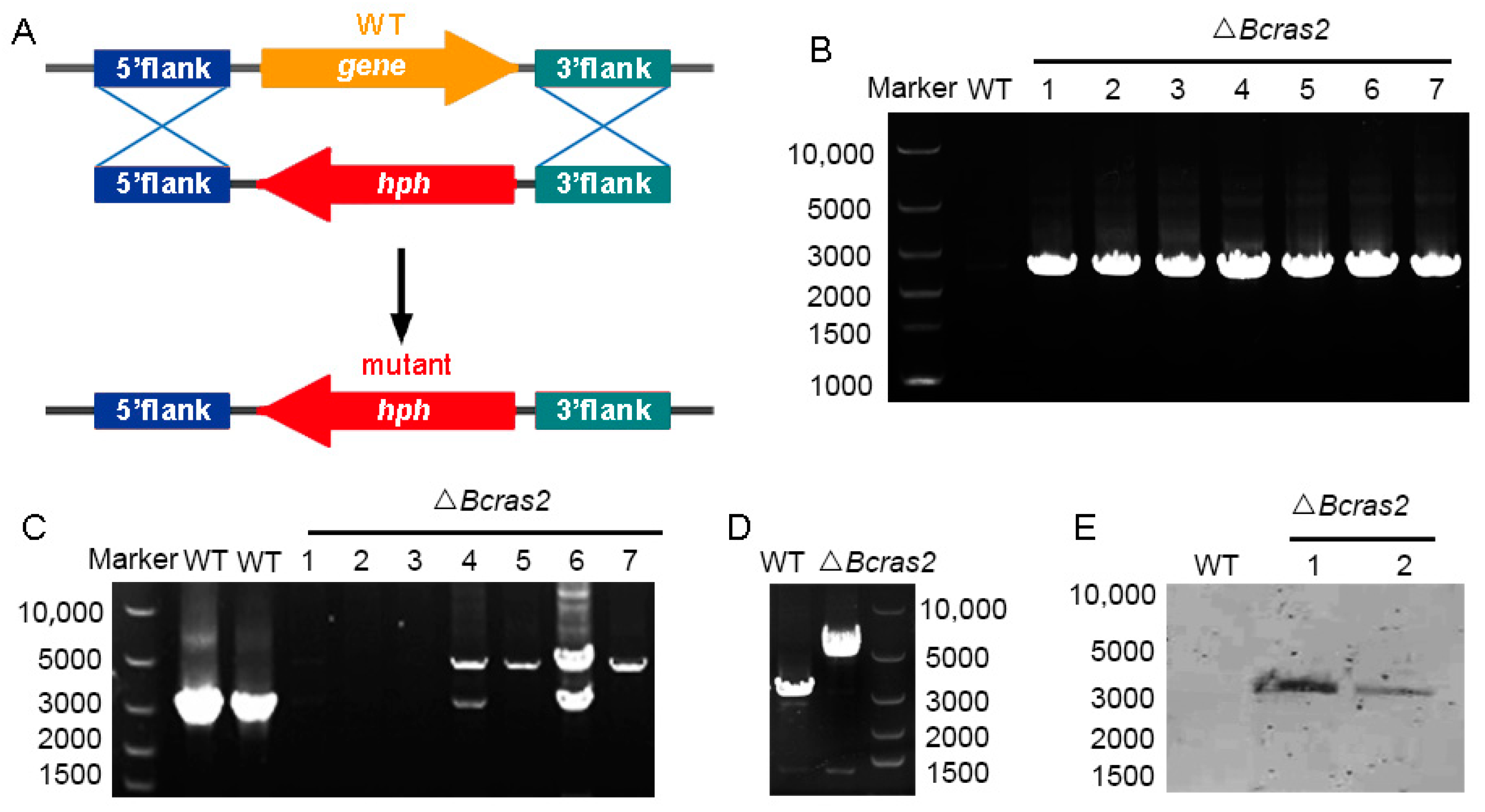
2.2. Generation of Gene Deletion Mutants
2.3. Phenotype Analysis
2.4. Virulence Assays
2.5. RNA Sequencing
2.6. Gene Expression Analysis
2.7. Statistical Analysis
3. Results
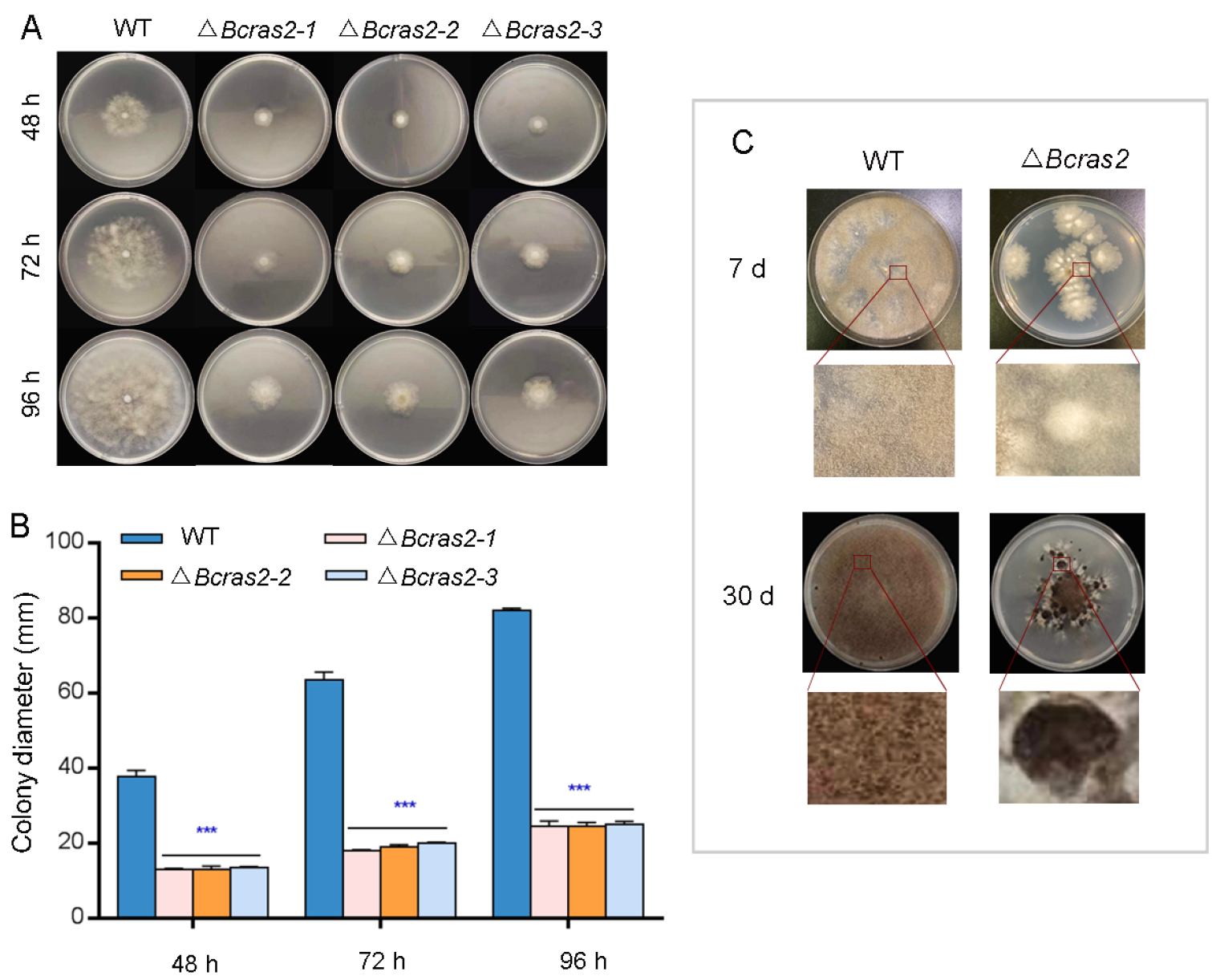
3.1. Bcras2 Is Required for Mycelial Growth and Development
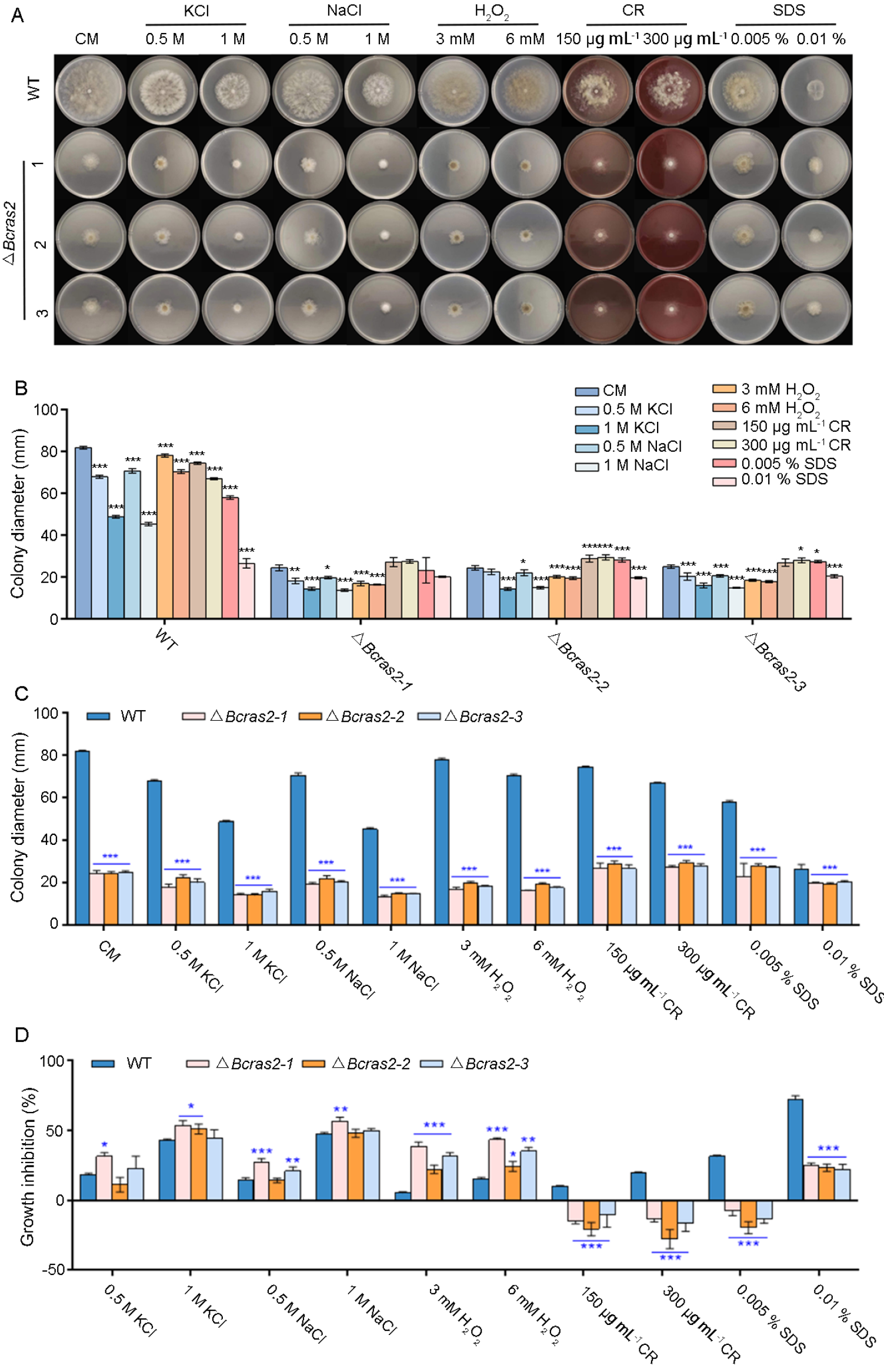
3.2. Bcras2 Is Responsible for Oxidative Stress Responses and Cell Wall Integrity
3.3. Bcras2 Is Essential for Virulence
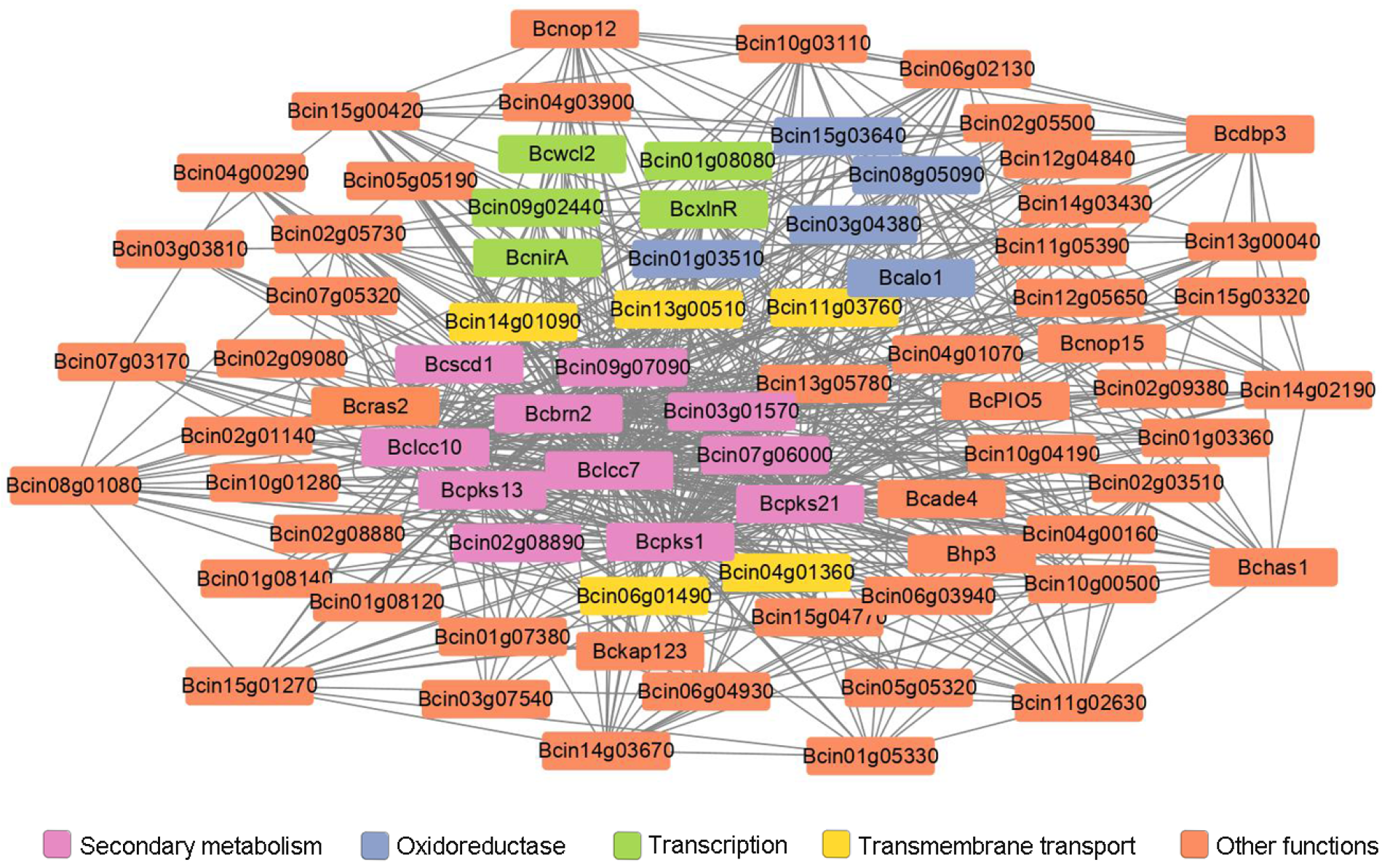
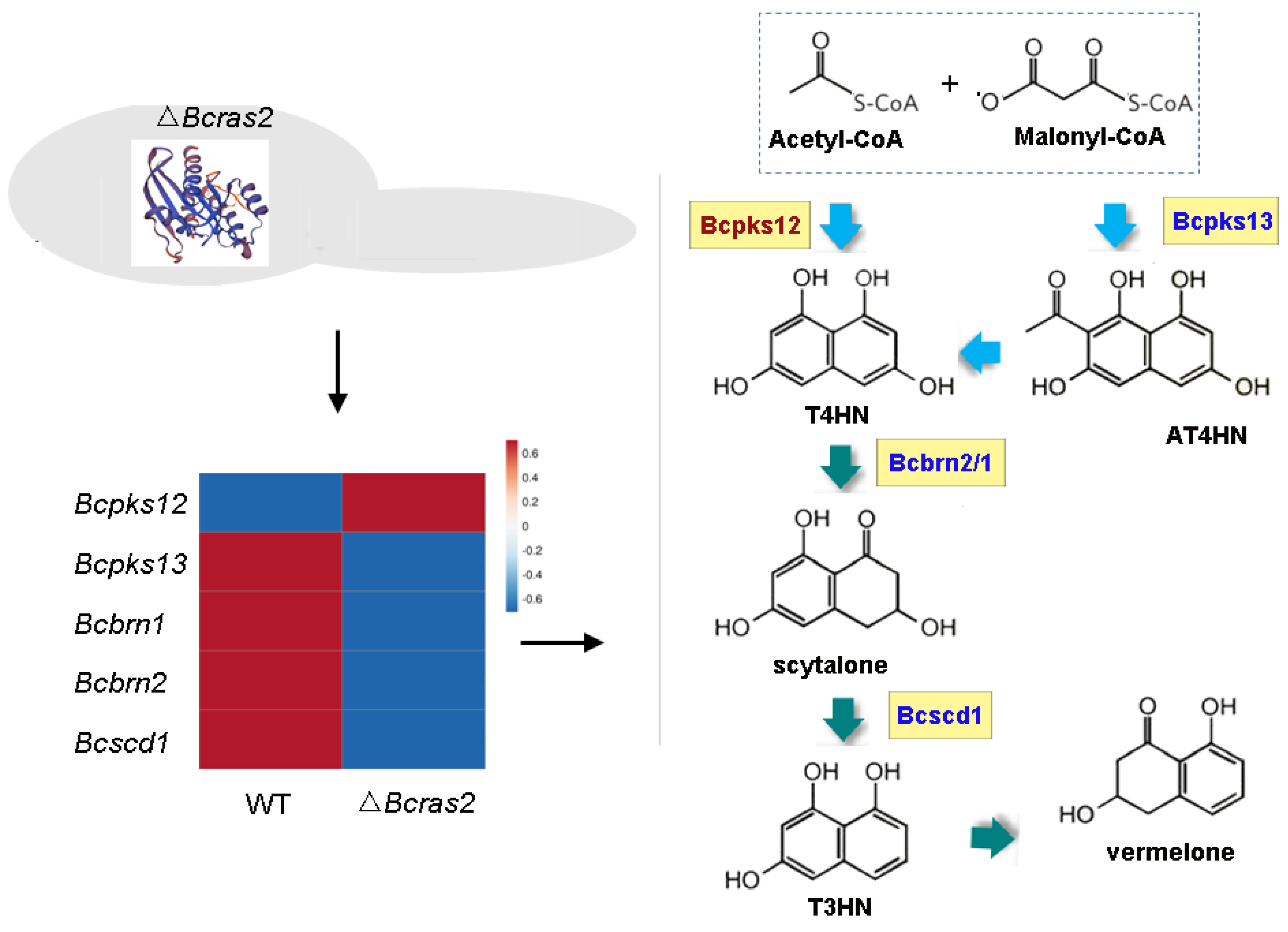
3.4. Identification of the Potential Downstream Targets of Bcras2 by RNA Sequencing
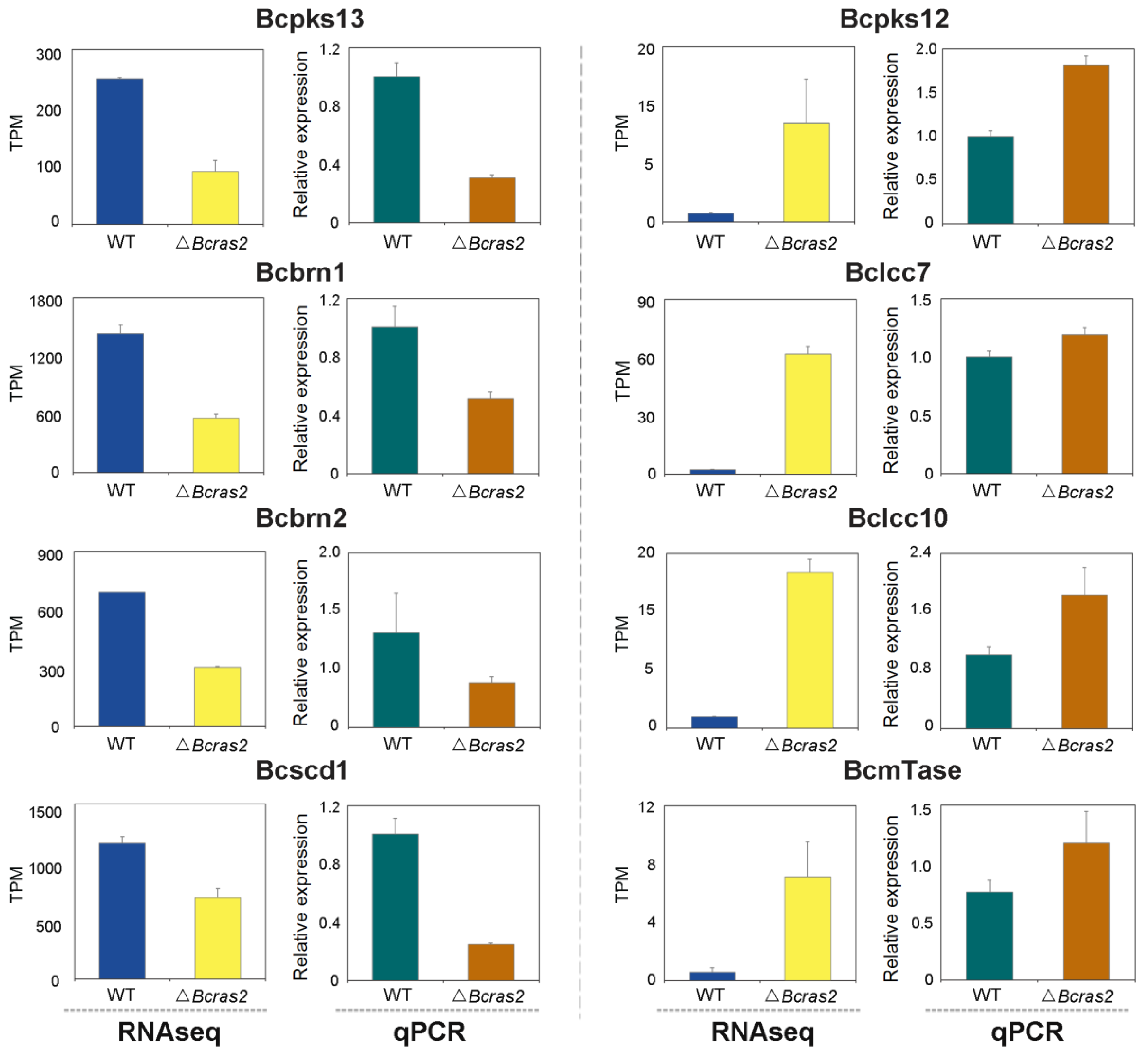
3.5. Validation of the Differentially Expressed Genes by RT-qPCR
3.6. The Melanin Metabolic Pathway Is Regulated by Bcras2
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Weiberg, A.; Wang, M.; Lin, F.M.; Zhao, H.; Zhang, Z.; Kaloshian, I.; Huang, H.D.; Jin, H.L. Fungal small RNAs suppress plant immunity by hijacking host RNA interference pathways. Science 2013, 342, 118–123. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, H.; Chen, Y.; Zhang, Z.Q.; Li, B.Q.; Qin, G.Z.; Tian, S.P. Pathogenic mechanisms and control strategies of Botrytis cinerea causing post-harvest decay in fruits and vegetables. Food Qual. Saf. 2018, 2, 111–119. [Google Scholar] [CrossRef] [Green Version]
- He, C.; Zhang, Z.; Li, B.; Xu, Y.; Tian, S. Effect of natamycin on Botrytis cinerea and Penicillium expansum—Postharvest pathogens of grape berries and jujube fruit. Postharvest Biol. Technol. 2019, 151, 134–141. [Google Scholar] [CrossRef]
- Petrasch, S.; Knapp, S.J.; Van Kan, J.A.L.; Blanco-Ulate, B. Grey mould of strawberry, a devastating disease caused by the ubiquitous necrotrophic fungal pathogen Botrytis cinerea. Mol. Plant Pathol. 2019, 20, 877–892. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mehari, Z.; Malacarne, G.; Pilat, S. The molecular dialogue between grapevine inflorescence/berry and Botrytis cinerea during initial, quiescent and egression infection stages. Acta Hortic. 2019, 1248, 587–594. [Google Scholar] [CrossRef]
- Fournier, E.; Gladieux, P.; Giraud, T. The ‘Dr Jekyll and Mr Hyde fungus’: Noble rot versus gray mold symptoms of Botrytis cinerea on grapes. Evol. Appl. 2013, 6, 960–969. [Google Scholar] [CrossRef] [PubMed]
- Lovato, A.; Zenoni, S.; Tornielli, G.B.; Colombo, T.; Vandelle, E.; Polverari, A. Specific molecular interactions between Vitis vinifera and Botrytis cinerea are required for noble rot development in grape berries. Postharvest Biol. Technol. 2019, 156, 110924. [Google Scholar] [CrossRef]
- Wang, X.; Tao, Y.; Wu, Y.; An, R.Y.; Yue, Z. Aroma compounds and characteristics of noble-rot wines of Chardonnay grapes artificially botrytized in the vineyard. Food Chem. 2017, 226, 41–50. [Google Scholar] [CrossRef]
- Dautt-Castro, M.; Rosendo-Vargas, M.; Casas-Flores, S. The small GTPases in fungal signaling conservation and function. Cells 2021, 10, 1039. [Google Scholar] [CrossRef]
- Arkowitz, R.A.; Bassilana, M. Regulation of hyphal morphogenesis by Ras and Rho small GTPases. Fungal Biol. Rev. 2015, 29, 7–19. [Google Scholar] [CrossRef]
- Hillig, R.C.; Sautier, B.; Schroeder, J.; Moosmayer, D.; Hilpmann, A.; Stegmann, C.M.; Werbeck, N.D.; Briem, H.; Boemer, U.; Weiske, J. PNAS Plus: Discovery of potent SOS1 inhibitors that block RAS activation via disruption of the RAS–SOS1 interaction. Proc. Natl. Acad. Sci. USA 2019, 116, 2551–2560. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Norton, T.S.; Fortwendel, J.R. Control of Ras-mediated signaling in Aspergillus fumigatus. Mycopathologia 2014, 178, 325–330. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sun, S.; Deng, Y.; Cai, E.; Yang, M.; Li, L.; Chen, B.; Chang, C.; Jiang, Z. The farnesyltransferase beta-subunit Ram1 regulates sporisorium scitamineum mating, pathogenicity and cell wall integrity. Front. Microbiol. 2019, 10, 976. [Google Scholar] [CrossRef] [Green Version]
- Xie, X.Q.; Guan, Y.; Ying, S.H.; Feng, M.G. Differentiated functions of Ras1 and Ras2 proteins in regulating the germination, growth, conidiation, multi-stress tolerance and virulence of Beauveria bassiana. Environ. Microbiol. 2013, 15, 447–462. [Google Scholar] [CrossRef] [PubMed]
- Guan, Y.; Wang, D.-Y.; Ying, S.-H.; Feng, M.-G. A novel Ras GTPase (Ras3) regulates conidiation, multi-stress tolerance and virulence by acting upstream of Hog1 signaling pathway in Beauveria bassiana. Fungal Genet. Biol. 2015, 82, 85–94. [Google Scholar] [CrossRef]
- Minz-Dub, A.M.; Kokkelink, L.; Tudzynski, B.; Tudzynski, P.; Sharon, A. Involvement of Botrytis cinerea small GTPases BcRAS1 and BcRAC in differentiation, virulence, and the cell cycle. Eukaryot. Cell 2013, 12, 1609–1618. [Google Scholar] [CrossRef] [Green Version]
- Schumacher, J.; Kokkelink, L.; Huesmann, C.; Jimenez-Teja, D.; Collado, I.G.; Barakat, R.; Tudzynski, P.; Tudzynski, B. The cAMP-dependent signaling pathway and its role in conidial germination, growth, and virulence of the gray mold Botrytis cinerea. Mol. Plant-Microbe Interact. 2008, 21, 1443–1459. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, H.; Zhang, Z.; Qin, G.; He, C.; Li, B.; Tian, S. Actin is required for cellular development and virulence of Botrytis cinerea via the mediation of secretory proteins. Msystems 2020, 5, e00732-19. [Google Scholar] [CrossRef] [Green Version]
- An, B.; Li, B.Q.; Li, H.; Zhang, Z.Q.; Qin, G.Z.; Tian, S.P. Aquaporin8 regulates cellular development and reactive oxygen species production, a critical component of virulence in Botrytis cinerea. New Phytol. 2016, 209, 1668–1680. [Google Scholar] [CrossRef]
- Cao, S.N.; Yuan, Y.; Qin, Y.H.; Zhang, M.Z.; de Figueiredo, P.; Li, G.H.; Qin, Q.M. The pre-rRNA processing factor Nop53 regulates fungal development and pathogenesis via mediating production of reactive oxygen species. Environ. Microbiol. 2018, 20, 1531–1549. [Google Scholar] [CrossRef]
- Kim, D.; Langmead, B.; Salzberg, S.L. HISAT: A fast spliced aligner with low memory requirements. Nat. Methods 2015, 12, 357–360. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Love, M.I.; Huber, W.; Anders, S. Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol. 2014, 15, 550. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Klopfenstein, D.V.; Zhang, L.; Pedersen, B.S.; Ramirez, F.; Vesztrocy, A.W.; Naldi, A.; Mungall, C.J.; Yunes, J.M.; Botvinnik, O.; Weigel, M.; et al. GOATOOLS: A python library for gene ontology analyses. Sci. Rep. 2018, 8, 10872. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ren, H.; Wu, X.; Lyu, Y.; Zhou, H.; Xie, X.; Zhang, X.; Yang, H. Selection of reliable reference genes for gene expression studies in Botrytis cinerea. J. Microbiol. Methods 2017, 142, 71–75. [Google Scholar] [CrossRef]
- Terhem, R.B.; van Kan, J.A.L. Functional analysis of hydrophobin genes in sexual development of Botrytis cinerea. Fungal Genet. Biol. 2014, 71, 42–51. [Google Scholar] [CrossRef]
- Cheung, N.; Tian, L.; Liu, X.; Li, X. The destructive fungal pathogen Botrytis cinerea—Insights from genes studied with mutant analysis. Pathogens 2020, 9, 923. [Google Scholar] [CrossRef]
- Zhang, Z.; He, C.; Chen, Y.; Li, B.; Tian, S. DNA methyltransferases regulate pathogenicity of Botrytis cinerea to horticultural crops. J. Fungi 2021, 7, 659. [Google Scholar] [CrossRef]
- Aboelfotoh Hendy, A.; Xing, J.; Chen, X.; Chen, X.L. The farnesyltransferase β-subunit RAM1 regulates localization of RAS proteins and appressorium-mediated infection in Magnaporthe oryzae. Mol. Plant Pathol. 2019, 20, 1264–1278. [Google Scholar] [CrossRef] [Green Version]
- Phasha, M.M.; Wingfield, M.J.; Wingfield, B.D.; Coetzee, M.; Steenkamp, E.T. Ras2 is important for growth and pathogenicity in Fusarium circinatum. Fungal Genet. Biol. 2021, 150, 103541. [Google Scholar] [CrossRef]
- Paulo, C.; Julia, S.; Hevia, M.A.; Paul, T.; Larrondo, L.F.; Michael, F. Assessing the effects of light on differentiation and virulence of the plant pathogen Botrytis cinerea: Characterization of the white collar complex. PLoS ONE 2013, 8, e84223. [Google Scholar] [CrossRef] [Green Version]
- Rasiukeviiūt, N.; Rasiukeviiūt, N.; Brazaityt, A.; Brazaityt, A.; Vatakait-Kairien, V.; Vatakait-Kairien, V.; Kupinskien, A.; Kupinskien, A.; Duchovskis, P.; Duchovskis, P. The effect of monochromatic LED light wavelengths and photoperiods on Botrytis cinerea. J. Fungi 2021, 7, 970. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Song, J.; Wang, Y.; Yang, L.; Wu, M.; Li, G.; Zhang, J. Biological characterization of the melanin biosynthesis gene Bcscd1 in the plant pathogenic fungus Botrytis cinerea. Fungal Genet. Biol. 2022, 160, 103693. [Google Scholar] [CrossRef] [PubMed]
- Ha, S.T.T.; Kim, Y.-T.; Jeon, Y.H.; Choi, H.W.; In, B.-C. Regulation of Botrytis cinerea infection and gene expression in cut roses by using nano silver and salicylic acid. Plants 2021, 10, 1241. [Google Scholar] [CrossRef]
- Li, H.; James, A.; Shen, X.; Wang, Y. Roles of microbiota in the formation of botrytized grapes and wines. CyTA-J. Food 2021, 19, 656–667. [Google Scholar] [CrossRef]
- Kim, J. Development of carbazole derivatives compounds against Candida albicans: Candidates to prevent hyphal formation via the Ras1-MAPK pathway. J. Fungi 2021, 7, 688. [Google Scholar] [CrossRef]
- Pentland, D.R.; Piper-Brown, E.; Mühlschlegel, F.A.; Gourlay, C.W. Ras signalling in pathogenic yeasts. Microb. Cell 2018, 5, 63. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Feng, Q.H.; Summers, E.; Bing, G.; Fink, G. Ras Signaling Is Required for Serum-Induced Hyphal Differentiation in Candida albicans. J. Bacteriol. 1999, 181, 6339–6346. [Google Scholar] [CrossRef] [Green Version]
- Conceição, P.M.; Chaves, A.F.A.; Navarro, M.V.; Castilho, D.G.; Calado, J.C.P.; Haniu, A.E.C.J.; Xander, P.; Batista, W.L. Cross-talk between the Ras GTPase and the Hog1 survival pathways in response to nitrosative stress in Paracoccidioides brasiliensis. Nitric Oxide 2019, 86, 1–11. [Google Scholar] [CrossRef]
- De Mendoza, D.; Pilon, M. Control of membrane lipid homeostasis by lipid-bilayer associated sensors: A mechanism conserved from bacteria to humans. Prog. Lipid Res. 2019, 76, 100996. [Google Scholar] [CrossRef]
- Cohen, B.E. Membrane thickness as a key factor contributing to the activation of osmosensors and essential Ras signaling pathways. Front. Cell Dev. Biol. 2018, 6, 76. [Google Scholar] [CrossRef] [Green Version]
- Schumacher, J. DHN melanin biosynthesis in the plant pathogenic fungus Botrytis cinerea is based on two developmentally regulated key enzyme (PKS)-encoding genes. Mol. Microbiol. 2016, 99, 729–748. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, X.; Zhu, C.; Na, Y.; Ren, D.; Zhang, C.; He, Y.; Wang, Y.; Xiang, S.; Ren, W.; Jiang, Y. Compartmentalization of melanin biosynthetic enzymes contributes to self-defense against intermediate compound scytalone in Botrytis cinerea. Mbio 2021, 12, e00007–e00021. [Google Scholar] [CrossRef] [PubMed]
- Slj, A.; Zhe, C.; Lu, C.; Glla, B.; Zhong, H.C.; Zmca, B. Molecular evolution and regulation of DHN melanin-related gene clusters are closely related to adaptation of different melanin-producing fungi. Genomics 2021, 113, 1962–1975. [Google Scholar]
- Zhu, P.; Li, Q.; Zhang, C.; Na, Y.; Xu, L. Bcpks12 gene inactivation substantiates biological functions of sclerotium melanization in Botrytis cinerea. Physiol. Mol. Plant Pathol. 2017, 98, 80–84. [Google Scholar] [CrossRef]
- Zhang, C.; He, Y.; Zhu, P.; Chen, L.; Wang, Y.; Ni, B.; Xu, L. Loss of bcbrn1 and bcpks13 in Botrytis cinerea not only blocks melanization but also increases vegetative growth and virulence. Mol. Plant-Microbe Interact. 2015, 28, 1091–1101. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sun, W.; Yu, Y.; Chen, J.; Yu, B.; Chen, T.; Ying, H.; Zhou, S.; Ouyang, P.; Liu, D.; Chen, Y. Light signaling regulates Aspergillus niger biofilm formation by affecting melanin and extracellular polysaccharide biosynthesis. mBio 2021, 12, e03434-20. [Google Scholar] [CrossRef]
- Gao, X.; Wang, Q.; Feng, Q.; Zhang, B.; He, C.; Luo, H.; An, B. Heat shock transcription factor CgHSF1 is required for melanin biosynthesis, appressorium formation, and pathogenicity in Colletotrichum gloeosporioides. J. Fungi 2022, 8, 175. [Google Scholar] [CrossRef]
- Schumacher, J.; Simon, A.; Cohrs, K.C.; Viaud, M.; Tudzynski, P. The transcription factor BcLTF1 regulates virulence and light responses in the necrotrophic plant pathogen Botrytis cinerea. PLoS Genet. 2014, 10, e1004040. [Google Scholar] [CrossRef] [Green Version]
- Zhang, Z.; Li, H.; Qin, G.; He, C.; Li, B.; Tian, S. The MADS-Box transcription factor Bcmads1 is required for growth, sclerotia production and pathogenicity of Botrytis cinerea. Sci. Rep. 2016, 6, 33901. [Google Scholar] [CrossRef] [Green Version]


Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, H.; Shen, X.; Wu, W.; Zhang, W.; Wang, Y. Ras2 Is Responsible for the Environmental Responses, Melanin Metabolism, and Virulence of Botrytis cinerea. J. Fungi 2023, 9, 432. https://doi.org/10.3390/jof9040432
Li H, Shen X, Wu W, Zhang W, Wang Y. Ras2 Is Responsible for the Environmental Responses, Melanin Metabolism, and Virulence of Botrytis cinerea. Journal of Fungi. 2023; 9(4):432. https://doi.org/10.3390/jof9040432
Chicago/Turabian StyleLi, Hua, Xuemei Shen, Wenjia Wu, Wanyu Zhang, and Yousheng Wang. 2023. "Ras2 Is Responsible for the Environmental Responses, Melanin Metabolism, and Virulence of Botrytis cinerea" Journal of Fungi 9, no. 4: 432. https://doi.org/10.3390/jof9040432




