Advances in S. cerevisiae Engineering for Xylose Fermentation and Biofuel Production: Balancing Growth, Metabolism, and Defense
Abstract
:1. Microbes Serve as Outstanding Chassis for Biochemical Production
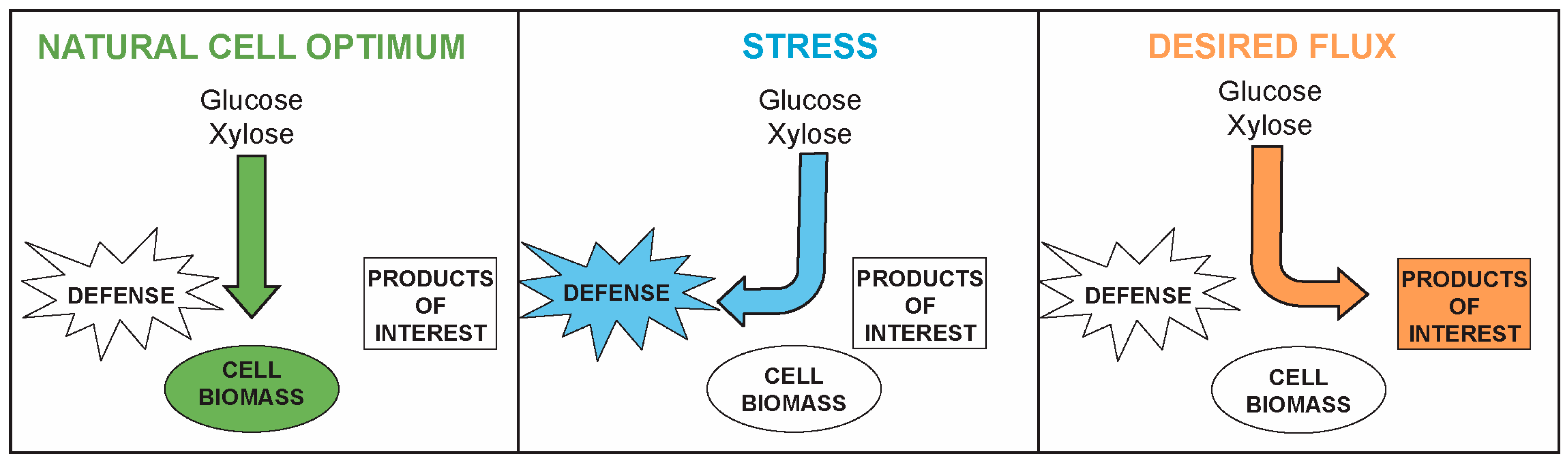
2. Rapid Growth and Maximal Stress Tolerance Are Competing Interests in Cells
3. Engineering Yeast for Ethanol Production from Lignocellulosic Biomass
4. Key Regulators That Govern Physiological Pathways Rewired for Xylose Fermentation
4.1. Protein Kinase A Pathway
4.2. Snf1 Pathway
4.3. High Osmolarity Glycerol Pathway
4.4. Cell Cycle Regulation by PKA, Snf1, and HOG Pathways
5. Future Prospects
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Cravens, A.; Payne, J.; Smolke, C.D. Synthetic biology strategies for microbial biosynthesis of plant natural products. Nat. Commun. 2019, 10, 2142. Available online: https://www.nature.com/articles/s41467-019-09848-w (accessed on 14 February 2023). [CrossRef] [Green Version]
- Money, N.P. The Rise of Yeast: How the Sugar Fungus Shaped Civilization. 2018, p. 210. Available online: https://global.oup.com/academic/product/the-rise-of-yeast-9780198749707 (accessed on 17 February 2023).
- Samuel, D. Investigation of Ancient Egyptian Baking and Brewing Methods by Correlative Microscopy. Science 1996, 273, 488–490. Available online: https://pubmed.ncbi.nlm.nih.gov/8662535/ (accessed on 17 February 2023). [CrossRef] [PubMed] [Green Version]
- Nielsen, J. Yeast Systems Biology: Model Organism and Cell Factory. Biotechnol. J. 2019, 14, 1800421. Available online: https://onlinelibrary.wiley.com/doi/full/10.1002/biot.201800421 (accessed on 14 February 2023). [CrossRef] [PubMed] [Green Version]
- Schindler, D. Genetic Engineering and Synthetic Genomics in Yeast to Understand Life and Boost Biotechnology. Bioengineering 2020, 7, 137. Available online: https://www.mdpi.com/2306-5354/7/4/137/htm (accessed on 14 February 2023). [CrossRef] [PubMed]
- Ding, J.; Huang, X.; Zhang, L.; Zhao, N.; Yang, D.; Zhang, K. Tolerance and stress response to ethanol in the yeast Saccharomyces cerevisiae. Appl. Microbiol. Biotechnol. 2009, 85, 253–263. Available online: https://link.springer.com/article/10.1007/s00253-009-2223-1 (accessed on 20 February 2023). [CrossRef]
- Liu, J.; Wang, X.; Dai, G.; Zhang, Y.; Bian, X. Microbial chassis engineering drives heterologous production of complex secondary metabolites. Biotechnol. Adv. 2022, 59, 107966. [Google Scholar] [CrossRef]
- Zhang, C.; Ottenheim, C.; Weingarten, M.; Ji, L.H. Microbial Utilization of Next-Generation Feedstocks for the Biomanufacturing of Value-Added Chemicals and Food Ingredients. Front. Bioeng. Biotechnol. 2022, 10, 486. [Google Scholar] [CrossRef]
- Shah, A.M.; Yang, W.; Mohamed, H.; Zhang, Y.; Song, Y. Microbes: A Hidden Treasure of Polyunsaturated Fatty Acids. Front. Nutr. 2022, 9, 415. [Google Scholar] [CrossRef]
- Joshi, S.; Mishra, S.D. Recent advances in biofuel production through metabolic engineering. Bioresour. Technol. 2022, 352, 127037. [Google Scholar] [CrossRef]
- Lau, M.W.; Gunawan, C.; Dale, B.E. The impacts of pretreatment on the fermentability of pretreated lignocellulosic biomass: A comparative evaluation between ammonia fiber expansion and dilute acid pretreatment. Biotechnol. Biofuels 2009, 2, 30. Available online: https://link.springer.com/articles/10.1186/1754-6834-2-30 (accessed on 18 February 2023). [CrossRef] [Green Version]
- Singh, K.K.; Rasmussen, A.K.; Lene, A.; Rasmussen, J. Genome-Wide Analysis of Signal Transducers and Regulators of Mitochondrial Dysfunction in Saccharomyces cerevisiae. Ann. N. Y. Acad. Sci. 2004, 1011, 284–298. Available online: http://mips.gsf.de (accessed on 11 June 2020). [CrossRef] [PubMed]
- Liu, Z.L. Molecular mechanisms of yeast tolerance and in situ detoxification of lignocellulose hydrolysates. Appl. Microbiol. Biotechnol. 2011, 90, 809–825. Available online: https://link.springer.com/article/10.1007/s00253-011-3167-9 (accessed on 18 February 2023). [CrossRef]
- Cunha, J.T.; Romaní, A.; Costa, C.E.; Sá-Correia, I.; Domingues, L. Molecular and physiological basis of Saccharomyces cerevisiae tolerance to adverse lignocellulose-based process conditions. Appl. Microbiol. Biotechnol. 2018, 103, 159–175. Available online: https://link.springer.com/article/10.1007/s00253-018-9478-3 (accessed on 18 February 2023). [CrossRef] [PubMed] [Green Version]
- Fletcher, E.; Baetz, K. Multi-Faceted Systems Biology Approaches Present a Cellular Landscape of Phenolic Compound Inhibition in Saccharomyces cerevisiae. Front. Bioeng. Biotechnol. 2020, 8, 1126. [Google Scholar] [CrossRef] [PubMed]
- Baruah, J.; Nath, B.K.; Sharma, R.; Kumar, S.; Deka, R.C.; Baruah, D.C.; Kalita, E. Recent trends in the pretreatment of lignocellulosic biomass for value-added products. Front. Energy Res. 2018, 6, 141. [Google Scholar] [CrossRef]
- Luterbacher, J.S.; Rand, J.M.; Alonso, D.M.; Han, J.; Youngquist, J.T.; Maravelias, C.T.; Pfleger, B.F.; Dumesic, J.A. Nonenzymatic sugar production from biomass using biomass-derived γ-valerolactone. Science 2014, 343, 277–280. Available online: https://www.science.org/doi/10.1126/science.1246748 (accessed on 18 February 2023). [CrossRef]
- Hou, Q.; Ju, M.; Li, W.; Liu, L.; Chen, Y.; Yang, Q. Pretreatment of Lignocellulosic Biomass with Ionic Liquids and Ionic Liquid-Based Solvent Systems. Molecules 2017, 22, 490. Available online: https://www.mdpi.com/1420-3049/22/3/490/htm (accessed on 18 February 2023). [CrossRef]
- Ouellet, M.; Datta, S.; Dibble, D.C.; Tamrakar, P.R.; Benke, P.I.; Li, C.; Singh, S.; Sale, K.L.; Adams, P.D.; Keasling, J.D.; et al. Impact of ionic liquid pretreated plant biomass on Saccharomyces cerevisiae growth and biofuel production. Green. Chem. 2011, 13, 2743–2749. Available online: https://pubs.rsc.org/en/content/articlehtml/2011/gc/c1gc15327g (accessed on 18 February 2023). [CrossRef]
- Vanacloig-Pedros, E.; Fisher, K.J.; Liu, L.; Debrauske, D.J.; Young, M.K.M.; Place, M.; Hittinger, C.T.; Sato, T.K.; Gasch, A.P. Comparative chemical genomic profiling across plant-based hydrolysate toxins reveals widespread antagonism in fitness contributions. FEMS Yeast Res. 2022, 22, foac036. Available online: https://academic.oup.com/femsyr/article/22/1/foac036/6650360 (accessed on 18 February 2023). [CrossRef]
- Piotrowski, J.S.; Zhang, Y.; Bates, D.M.; Keating, D.H.; Sato, T.K.; Ong, I.M.; Landick, R. Death by a thousand cuts: The challenges and diverse landscape of lignocellulosic hydrolysate inhibitors. Front. Microbiol. 2014, 5, 90. Available online: http://journal.frontiersin.org/article/10.3389/fmicb.2014.00090/abstract (accessed on 24 January 2019). [CrossRef]
- Palmqvist, E.; Hahn-Hägerdal, B. Fermentation of lignocellulosic hydrolysates. II: Inhibitors and mechanisms of inhibition. Bioresour. Technol. 2000, 74, 25–33. [Google Scholar] [CrossRef]
- Almeida, J.R.M.; Modig, T.; Petersson, A.; Hähn-Hägerdal, B.; Lidén, G.; Gorwa-Grauslund, M.F. Increased tolerance and conversion of inhibitors in lignocellulosic hydrolysates by Saccharomyces cerevisiae. J. Chem. Technol. Biotechnol. 2007, 82, 340–349. Available online: https://onlinelibrary.wiley.com/doi/full/10.1002/jctb.1676 (accessed on 18 February 2023). [CrossRef]
- Chundawat, S.P.S.; Vismeh, R.; Sharma, L.N.; Humpula, J.F.; da Costa Sousa, L.; Chambliss, C.K.; Jones, A.D.; Balan, V.; Dale, B.E. Multifaceted characterization of cell wall decomposition products formed during ammonia fiber expansion (AFEX) and dilute acid based pretreatments. Bioresour. Technol. 2010, 101, 8429–8438. Available online: https://www.sciencedirect.com/science/article/pii/S0960852410010084 (accessed on 24 January 2019). [CrossRef]
- Klinke, H.B.; Thomsen, A.B.; Ahring, B.K. Inhibition of ethanol-producing yeast and bacteria by degradation products produced during pre-treatment of biomass. Appl. Microbiol. Biotechnol. 2004, 66, 10–26. Available online: http://link.springer.com/10.1007/s00253-004-1642-2 (accessed on 24 January 2019). [CrossRef]
- Bunnell, K.; Rich, A.; Luckett, C.; Wang, Y.J.; Martin, E.; Carrier, D.J. Plant maturity effects on the physicochemical properties and dilute acid hydrolysis of switchgrass (Panicum virgatum, L.) hemicelluloses. ACS Sustain. Chem. Eng. 2013, 1, 649–654. Available online: https://pubs.acs.org/doi/full/10.1021/sc4000175 (accessed on 18 February 2023). [CrossRef]
- Jönsson, L.J.; Martín, C. Pretreatment of lignocellulose: Formation of inhibitory by-products and strategies for minimizing their effects. Bioresour. Technol. 2016, 199, 103–112. [Google Scholar] [CrossRef] [Green Version]
- Ong, R.G.; Higbee, A.; Bottoms, S.; Dickinson, Q.; Xie, D.; Smith, S.A.; Serate, J.; Pohlmann, E.; Jones, A.D.; Coon, J.J.; et al. Inhibition of microbial biofuel production in drought-stressed switchgrass hydrolysate. Biotechnol. Biofuels 2016, 9, 237. Available online: https://link.springer.com/articles/10.1186/s13068-016-0657-0 (accessed on 18 February 2023). [CrossRef] [Green Version]
- Wehrs, M.; Thompson, M.G.; Banerjee, D.; Prahl, J.P.; Morella, N.M.; Barcelos, C.A.; Moon, J.; Costello, Z.; Keasling, J.D.; Shih, P.M.; et al. Investigation of Bar-seq as a method to study population dynamics of Saccharomyces cerevisiae deletion library during bioreactor cultivation. Microb. Cell Fact. 2020, 19, 167. Available online: https://link.springer.com/articles/10.1186/s12934-020-01423-z (accessed on 18 February 2023). [CrossRef] [PubMed]
- Brauer, M.J.; Huttenhower, C.; Airoldi, E.M.; Rosenstein, R.; Matese, J.C.; Gresham, D.; Boer, V.M.; Troyanskaya, O.G.; Botstein, D. Coordination of growth rate, cell cycle, stress response, and metabolic activity in yeast. Mol. Biol. Cell 2008, 19, 352–367. Available online: https://www.molbiolcell.org/doi/10.1091/mbc.e07-08-0779 (accessed on 8 September 2022). [CrossRef] [PubMed] [Green Version]
- Gibney, P.A.; Lu, C.; Caudy, A.A.; Hess, D.C.; Botstein, D. Yeast metabolic and signaling genes are required for heat-shock survival and have little overlap with the heat-induced genes. Proc. Natl. Acad. Sci. USA 2013, 110, E4393–E4402. Available online: https://pubmed.ncbi.nlm.nih.gov/24167267/ (accessed on 1 June 2023). [CrossRef] [PubMed]
- Slavov, N.; Botstein, D. Coupling among growth rate response, metabolic cycle, and cell division cycle in yeast. Mol. Biol. Cell 2011, 22, 1997–2009. Available online: https://pubmed.ncbi.nlm.nih.gov/21525243/ (accessed on 1 June 2023). [CrossRef]
- Chasman, D.; Ho, Y.; Berry, D.B.; Nemec, C.M.; MacGilvray, M.E.; Hose, J.; Merrill, A.E.; Lee, M.V.; Will, J.L.; Coon, J.J.; et al. Pathway connectivity and signaling coordination in the yeast stress-activated signaling network. Mol. Syst. Biol. 2014, 10, 759. Available online: https://onlinelibrary.wiley.com/doi/abs/10.15252/msb.20145120 (accessed on 14 April 2020). [CrossRef] [PubMed]
- Ho, Y.H.; Shishkova, E.; Hose, J.; Coon, J.J.; Gasch, A.P. Decoupling Yeast Cell Division and Stress Defense Implicates mRNA Repression in Translational Reallocation during Stress. Curr. Biol. 2018, 28, 2673–2680.e4. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ho, Y.-H.; Gasch, A.P. Exploiting the yeast stress-activated signaling network to inform on stress biology and disease signaling. Curr. Genet. 2015, 61, 503–511. Available online: http://link.springer.com/10.1007/s00294-015-0491-0 (accessed on 24 January 2019). [CrossRef] [PubMed] [Green Version]
- Lu, C.; Brauer, M.J.; Botstein, D. Slow growth induces heat-shock resistance in normal and respiratory-deficient yeast. Mol. Biol. Cell 2009, 20, 891–903. Available online: https://pubmed.ncbi.nlm.nih.gov/19056679/ (accessed on 1 June 2023). [CrossRef] [Green Version]
- Gasch, A.P.; Spellman, P.T.; Kao, C.M.; Carmel-Harel, O.; Eisen, M.B.; Storz, G.; Botstein, D.; Brown, P.O. Genomic Expression Programs in the Response of Yeast Cells to Environmental Changes. Mol. Biol. Cell 2000, 11, 4241–4257. Available online: http://www.molbiolcell.org/doi/10.1091/mbc.11.12.4241 (accessed on 19 March 2019). [CrossRef]
- Gasch, A.P. Yeast genomic expression studies using DNA microarrays. Methods Enzymol. 2002, 350, 393–414. [Google Scholar]
- Pascual-Ahuir, A.; Manzanares-Estreder, S.; Timón-Gómez, A.; Proft, M. Ask yeast how to burn your fats: Lessons learned from the metabolic adaptation to salt stress. Curr. Genet. 2018, 64, 63–69. Available online: https://pubmed.ncbi.nlm.nih.gov/28631015/ (accessed on 1 June 2023). [CrossRef] [Green Version]
- Martínez-Montañés, F.; Pascual-Ahuir, A.; Proft, M. Toward a genomic view of the gene expression program regulated by osmostress in yeast. OMICS 2010, 14, 619–627. Available online: https://pubmed.ncbi.nlm.nih.gov/20726780/ (accessed on 1 June 2023). [CrossRef]
- Estruch, F.; Carlson, M. Two homologous zinc finger genes identified by multicopy suppression in a SNF1 protein kinase mutant of Saccharomyces cerevisiae. Mol. Cell Biol. 1993, 13, 3872–3881. Available online: https://pubmed.ncbi.nlm.nih.gov/8321194/ (accessed on 1 June 2023).
- Schmitt, A.P.; Mcentee, K. Msn2p, a zinc finger DNA-binding protein, is the transcriptional activator of the multistress response in Saccharomyces cerevisiae. Proc. Natl. Acad. Sci. USA 1996, 93, 5777–5782. Available online: https://pubmed.ncbi.nlm.nih.gov/8650168/ (accessed on 1 June 2023). [CrossRef] [PubMed]
- Martínez-Pastor, M.T.; Marchler, G.; Schüller, C.; Marchler-Bauer, A.; Ruis, H.; Estruch, F. The Saccharomyces cerevisiae zinc finger proteins Msn2p and Msn4p are required for transcriptional induction through the stress response element (STRE). EMBO J. 1996, 15, 2227–2235. Available online: http://www.ncbi.nlm.nih.gov/pubmed/8641288 (accessed on 14 March 2019). [CrossRef] [PubMed]
- Castrillo, J.I.; Zeef, L.A.; Hoyle, D.C.; Zhang, N.; Hayes, A.; Gardner, D.C.J.; Cornell, M.J.; Petty, J.; Hakes, L.; Wardleworth, L.; et al. Growth control of the eukaryote cell: A systems biology study in yeast. J. Biol. 2007, 6, 1–25. Available online: https://jbiol.biomedcentral.com/articles/10.1186/jbiol54 (accessed on 16 June 2022). [CrossRef] [PubMed]
- Regenberg, B.; Grotkjær, T.; Winther, O.; Fausbøll, A.; Åkesson, M.; Bro, C.; Hansen, L.K.; Brunak, S.; Nielsen, J. Growth-rate regulated genes have profound impact on interpretation of transcriptome profiling in Saccharomyces cerevisiae. Genome Biol. 2006, 7, R107. Available online: https://genomebiology.biomedcentral.com/articles/10.1186/gb-2006-7-11-r107 (accessed on 8 September 2022). [CrossRef] [Green Version]
- Airoldi, E.M.; Huttenhower, C.; Gresham, D.; Lu, C.; Caudy, A.A.; Dunham, M.J.; Broach, J.R.; Botstein, D.; Troyanskaya, O.G. Predicting Cellular Growth from Gene Expression Signatures. PLoS Comput. Biol. 2009, 5, e1000257. Available online: https://journals.plos.org/ploscompbiol/article?id=10.1371/journal.pcbi.1000257 (accessed on 8 September 2022). [CrossRef]
- Lee, M.V.; Topper, S.E.; Hubler, S.L.; Hose, J.; Wenger, C.D.; Coon, J.J.; Gasch, A.P. A dynamic model of proteome changes reveals new roles for transcript alteration in yeast. Mol. Syst. Biol. 2011, 7, 514. Available online: https://pubmed.ncbi.nlm.nih.gov/21772262/ (accessed on 1 June 2023). [CrossRef]
- Bergen, A.C.; Kocik, R.A.; Hose, J.; McClean, M.N.; Gasch, A.P. Modeling single-cell phenotypes links yeast stress acclimation to transcriptional repression and pre-stress cellular states. Elife 2022, 11, e82017. Available online: https://pubmed.ncbi.nlm.nih.gov/36350693/ (accessed on 1 June 2023). [CrossRef]
- Görner, W.; Durchschlag, E.; Martinez-Pastor, M.T.; Estruch, F.; Ammerer, G.; Hamilton, B.; Ruis, H.; Schüller, C. Nuclear localization of the C2H2 zinc finger protein Msn2p is regulated by stress and protein kinase A activity. Genes. Dev. 1998, 12, 586–597. Available online: http://www.ncbi.nlm.nih.gov/pubmed/9472026 (accessed on 14 March 2019). [CrossRef] [Green Version]
- Lippman, S.I.; Broach, J.R. Protein kinase A and TORC1 activate genes for ribosomal biogenesis by inactivating repressors encoded by Dot6 and its homolog Tod6. Proc. Natl. Acad. Sci. USA 2009, 106, 19928–19933. Available online: http://www.ncbi.nlm.nih.gov/pubmed/19901341 (accessed on 25 March 2019). [CrossRef]
- Pokholok, D.K.; Zeitlinger, J.; Hannett, N.M.; Reynolds, D.B.; Young, R.A. Activated signal transduction kinases frequently occupy target genes. Science 2006, 313, 533–536. Available online: http://www.ncbi.nlm.nih.gov/pubmed/16873666 (accessed on 13 February 2019). [CrossRef]
- Pelechano, V.; Jimeno-González, S.; Rodríguez-Gil, A.; García-Martínez, J.; Pérez-Ortín, J.E.; Chávez, S. Regulon-Specific Control of Transcription Elongation across the Yeast Genome. PLoS Genet. 2009, 5, e1000614. [Google Scholar] [CrossRef] [PubMed]
- Baccarini, L.; Martínez-Montañés, F.; Rossi, S.; Proft, M.; Portela, P. PKA-chromatin association at stress responsive target genes from Saccharomyces cerevisiae. Biochim. Biophys. Acta—Gene Regul. Mech. 2015, 1849, 1329–1339. Available online: https://www.sciencedirect.com/science/article/pii/S1874939915001935?via%3Dihub (accessed on 29 January 2019). [CrossRef] [PubMed]
- Chang, Y.-W.; Howard, S.C.; Herman, P.K. The Ras/PKA Signaling Pathway Directly Targets the Srb9 Protein, a Component of the General RNA Polymerase II Transcription Apparatus. Mol. Cell 2004, 15, 107–116. Available online: https://www.sciencedirect.com/science/article/pii/S1097276504003077?via%3Dihub (accessed on 28 March 2019). [CrossRef]
- De Wever, V.; Reiter, W.; Ballarini, A.; Ammerer, G.; Brocard, C. A dual role for PP1 in shaping the Msn2-dependent transcriptional response to glucose starvation. EMBO J. 2005, 24, 4115–4123. Available online: https://pubmed.ncbi.nlm.nih.gov/16281053/ (accessed on 25 January 2023). [CrossRef] [PubMed] [Green Version]
- Li, L.; Kaplan, J.; Ward, D.M. The glucose sensor Snf1 and the transcription factors Msn2 and Msn4 regulate transcription of the vacuolar iron importer gene CCC1 and iron resistance in yeast. J. Biol. Chem. 2017, 292, 15577–15586. Available online: https://pubmed.ncbi.nlm.nih.gov/28760824/ (accessed on 25 January 2023). [CrossRef] [PubMed] [Green Version]
- Bodenmiller, B.; Wanka, S.; Kraft, C.; Urban, J.; Campbell, D.; Pedrioli, P.G.; Gerrits, B.; Picotti, P.; Lam, H.; Vitek, O.; et al. Phosphoproteomic analysis reveals interconnected system-wide responses to perturbations of kinases and phosphatases in yeast. Sci. Signal 2010, 3, rs4. Available online: https://pubmed.ncbi.nlm.nih.gov/21177495/ (accessed on 1 June 2023). [CrossRef] [Green Version]
- Ho, N.W.; Chen, Z.; Brainard, A.P.; Sedlak, M. Successful design and development of genetically engineered Saccharomyces yeasts for effective cofermentation of glucose and xylose from cellulosic biomass to fuel ethanol. Adv. Biochem. Eng. Biotechnol. 1999, 65, 163–192. Available online: https://pubmed.ncbi.nlm.nih.gov/10533435/ (accessed on 1 June 2023).
- Jagger, A. Biofuels for transport in 2050. Biofuels Bioprod Biorefining 2011, 5, 481–485. Available online: https://onlinelibrary.wiley.com/doi/full/10.1002/bbb.330 (accessed on 17 February 2023). [CrossRef]
- Eisentraut, A. Sustainable Production of Second-Generation Biofuels: Potential and Perspectives in Major Economies and Developing Countries | IEA Energy Papers | OECD iLibrary IEA Energy Papers. 2010. Available online: https://www.oecd-ilibrary.org/energy/sustainable-production-of-second-generation-biofuels_5kmh3njpt6r0-en (accessed on 17 February 2023).
- Sims, R.; Taylor, M.; Saddler, J.; Mabee, W. FROM 1st-TO 2nd-GENERATION BIOFUEL TECHNOLOGIES An Overview of Current Industry and RD&D Activities. Available online: www.ieabioenergy.com (accessed on 17 February 2023).
- Buijs, N.A.; Siewers, V.; Nielsen, J. Advanced biofuel production by the yeast Saccharomyces cerevisiae. Curr. Opin. Chem. Biol. 2013, 17, 480–488. [Google Scholar] [CrossRef]
- Jin, C.; Yao, M.; Liu, H.; Lee, C.F.F.; Ji, J. Progress in the production and application of n-butanol as a biofuel. Renew. Sustain. Energy Rev. 2011, 15, 4080–4106. [Google Scholar] [CrossRef]
- Azambuja, S.P.H.; Goldbeck, R. Butanol production by Saccharomyces cerevisiae: Perspectives, strategies and challenges. World J. Microbiol. Biotechnol. 2020, 36, 48. Available online: https://link.springer.com/article/10.1007/s11274-020-02828-z (accessed on 17 February 2023). [CrossRef] [PubMed]
- Sakuragi, H.; Morisaka, H.; Kuroda, K.; Ueda, M. Enhanced butanol production by eukaryotic Saccharomyces cerevisiae engineered to contain an improved pathway. Biosci. Biotechnol. Biochem. 2015, 79, 314–320. Available online: https://academic.oup.com/bbb/article/79/2/314/5939446 (accessed on 17 February 2023). [CrossRef] [PubMed]
- Choi, Y.J.; Lee, J.; Jang, Y.S.; Lee, S.Y. Metabolic engineering of microorganisms for the production of higher alcohols. MBio 2014, 5. Available online: https://journals.asm.org/doi/10.1128/mBio.01524-14 (accessed on 17 February 2023). [CrossRef] [PubMed] [Green Version]
- Si, T.; Luo, Y.; Xiao, H.; Zhao, H. Utilizing an endogenous pathway for 1-butanol production in Saccharomyces cerevisiae. Metab. Eng. 2014, 22, 60–68. [Google Scholar] [CrossRef] [PubMed]
- Swidah, R.; Wang, H.; Reid, P.J.; Ahmed, H.Z.; Pisanelli, A.M.; Persaud, K.C.; Grant, C.M.; Ashe, M.P. Butanol production in S. cerevisiae via a synthetic ABE pathway is enhanced by specific metabolic engineering and butanol resistance. Biotechnol. Biofuels 2015, 8, 97. [Google Scholar] [CrossRef] [Green Version]
- Schadeweg, V.; Boles, E. N-Butanol production in Saccharomyces cerevisiae is limited by the availability of coenzyme A and cytosolic acetyl-CoA. Biotechnol. Biofuels 2016, 9, 44. Available online: https://biotechnologyforbiofuels.biomedcentral.com/articles/10.1186/s13068-016-0456-7 (accessed on 17 February 2023). [CrossRef] [Green Version]
- Krivoruchko, A.; Serrano-Amatriain, C.; Chen, Y.; Siewers, V.; Nielsen, J. Improving biobutanol production in engineered Saccharomyces cerevisiae by manipulation of acetyl-CoA metabolism. J. Ind. Microbiol. Biotechnol. 2013, 40, 1051–1056. Available online: https://pubmed.ncbi.nlm.nih.gov/23760499/ (accessed on 17 February 2023). [CrossRef]
- Schadeweg, V.; Boles, E. Increasing n-butanol production with Saccharomyces cerevisiae by optimizing acetyl-CoA synthesis, NADH levels and trans-2-enoyl-CoA reductase expression. Biotechnol. Biofuels 2016, 9, 257. Available online: https://biotechnologyforbiofuels.biomedcentral.com/articles/10.1186/s13068-016-0673-0 (accessed on 17 February 2023). [CrossRef] [Green Version]
- Gambacorta, F.V.; Wagner, E.R.; Jacobson, T.B.; Tremaine, M.; Muehlbauer, L.K.; McGee, M.A.; Baerwald, J.J.; Wrobel, R.L.; Wolters, J.F.; Place, M.; et al. Comparative functional genomics identifies an iron-limited bottleneck in a Saccharomyces cerevisiae strain with a cytosolic-localized isobutanol pathway. Synth. Syst. Biotechnol. 2022, 7, 738–749. Available online: https://pubmed.ncbi.nlm.nih.gov/35387233/ (accessed on 17 February 2023). [CrossRef]
- Kuroda, K.; Ueda, M. Cellular and molecular engineering of yeast Saccharomyces cerevisiae for advanced biobutanol production. FEMS Microbiol. Lett. 2016, 363, 247. Available online: https://academic.oup.com/femsle/article/363/3/fnv247/2594551 (accessed on 17 February 2023). [CrossRef] [Green Version]
- Hong, K.K.; Nielsen, J. Metabolic engineering of Saccharomyces cerevisiae: A key cell factory platform for future biorefineries. Cell Mol. Life Sci. 2012, 69, 2671–2690. Available online: https://link.springer.com/article/10.1007/s00018-012-0945-1 (accessed on 17 February 2023). [CrossRef] [PubMed]
- Generoso, W.C.; Schadeweg, V.; Oreb, M.; Boles, E. Metabolic engineering of Saccharomyces cerevisiae for production of butanol isomers. Curr. Opin. Biotechnol. 2015, 33, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Steen, E.J.; Chan, R.; Prasad, N.; Myers, S.; Petzold, C.J.; Redding, A.; Ouellet, M.; Keasling, J.D. Metabolic engineering of Saccharomyces cerevisiae for the production of n-butanol. Microb. Cell Fact. 2008, 7, 36. Available online: https://microbialcellfactories.biomedcentral.com/articles/10.1186/1475-2859-7-36 (accessed on 17 February 2023). [CrossRef] [PubMed] [Green Version]
- Lian, J.; Si, T.; Nair, N.U.; Zhao, H. Design and construction of acetyl-CoA overproducing Saccharomyces cerevisiae strains. Metab. Eng. 2014, 24, 750–760. Available online: https://pubmed.ncbi.nlm.nih.gov/24853351/ (accessed on 17 February 2023). [CrossRef]
- Kim, S.R.; Ha, S.-J.; Wei, N.; Oh, E.J.; Jin, Y.-S. Simultaneous co-fermentation of mixed sugars: A promising strategy for producing cellulosic ethanol. Trends Biotechnol. 2012, 30, 274–282. Available online: https://www.sciencedirect.com/science/article/pii/S0167779912000157 (accessed on 24 January 2019). [CrossRef]
- Lee, J.W.; Yook, S.; Koh, H.; Rao, C.V.; Jin, Y.S. Engineering xylose metabolism in yeasts to produce biofuels and chemicals. Curr. Opin. Biotechnol. 2021, 67, 15–25. [Google Scholar] [CrossRef]
- Wohlbach, D.J.; Kuo, A.; Sato, T.K.; Potts, K.M.; Salamov, A.A.; LaButti, K.M.; Sun, H.; Clum, A.; Pangilian, J.L.; Lindquist, E.A.; et al. Comparative genomics of xylose-fermenting fungi for enhanced biofuel production. Proc. Natl. Acad. Sci. USA 2011, 108, 13212–13217. Available online: https://pubmed.ncbi.nlm.nih.gov/21788494/ (accessed on 18 February 2023). [CrossRef]
- Harner, N.K.; Wen, X.; Bajwa, P.K.; Austin, G.D.; Ho, C.Y.; Habash, M.B.; Trevors, J.T.; Lee, H. Genetic improvement of native xylose-fermenting yeasts for ethanol production. J. Ind. Microbiol. Biotechnol. 2015, 42, 1–20. Available online: https://pubmed.ncbi.nlm.nih.gov/25404205/ (accessed on 16 May 2023). [CrossRef]
- Trichez, D.; Steindorff, A.S.; de Morais Júnior, W.G.; Vilela, N.; Bergmann, J.C.; Formighieri, E.F.; Gonçalves, S.B.; de Almeida, J.R.M. Identification of traits to improve co-assimilation of glucose and xylose by adaptive evolution of Spathaspora passalidarum and Scheffersomyces stipitis yeasts. Appl. Microbiol. Biotechnol. 2023, 107, 1143–1157. Available online: https://pubmed.ncbi.nlm.nih.gov/36625916/ (accessed on 16 May 2023). [CrossRef]
- Kim, S.R.; Skerker, J.M.; Kang, W.; Lesmana, A.; Wei, N.; Arkin, A.P.; Jin, Y. Rational and Evolutionary Engineering Approaches Uncover a Small Set of Genetic Changes Efficient for Rapid Xylose Fermentation in Saccharomyces cerevisiae. PLoS ONE 2013, 8, e57048. [Google Scholar] [CrossRef] [Green Version]
- Kwak, S.; Jin, Y.S. Production of fuels and chemicals from xylose by engineered Saccharomyces cerevisiae: A review and perspective. Microb. Cell Fact. 2017, 16, 82. Available online: https://pubmed.ncbi.nlm.nih.gov/28494761/ (accessed on 1 June 2023). [CrossRef] [Green Version]
- Kuyper, M.; Harhangi, H.; Stave, A.; Winkler, A.; Jetten, M.; Delaat, W.; den Ridder, J.J.J.; Op den Camp, H.J.M.; van Dijken, J.P.; Pronk, J.Y. High-level functional expression of a fungal xylose isomerase: The key to efficient ethanolic fermentation of xylose by Saccharomyces cerevisiae ? FEMS Yeast Res. 2003, 4, 69–78. Available online: https://academic.oup.com/femsyr/article-lookup/doi/10.1016/S1567-1356(03)00141-7 (accessed on 24 January 2019). [CrossRef] [PubMed] [Green Version]
- Kuyper, M.; Hartog, M.; Toirkens, M.; Almering, M.; Winkler, A.; Vandijken, J.; Pronk, J.T. Metabolic engineering of a xylose-isomerase-expressing strain for rapid anaerobic xylose fermentation. FEMS Yeast Res. 2005, 5, 399–409. Available online: https://academic.oup.com/femsyr/article-lookup/doi/10.1016/j.femsyr.2004.09.010 (accessed on 24 January 2019). [CrossRef] [PubMed] [Green Version]
- Parreiras, L.S.; Breuer, R.J.; Avanasi Narasimhan, R.; Higbee, A.J.; La Reau, A.; Tremaine, M.; Qin, L.; Willis, L.B.; Bice, B.D.; Bonfert, B.L.; et al. Engineering and Two-Stage Evolution of a Lignocellulosic Hydrolysate-Tolerant Saccharomyces cerevisiae Strain for Anaerobic Fermentation of Xylose from AFEX Pretreated Corn Stover. PLoS ONE 2014, 9, e107499. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kim, S.R.; Park, Y.C.; Jin, Y.S.; Seo, J.H. Strain engineering of Saccharomyces cerevisiae for enhanced xylose metabolism. Biotechnol. Adv. 2013, 31, 851–861. [Google Scholar] [CrossRef]
- Osiro, K.O.; Brink, D.P.; Borgström, C.; Wasserstrom, L.; Carlquist, M.; Gorwa-Grauslund, M.F. Assessing the effect of d-xylose on the sugar signaling pathways of Saccharomyces cerevisiae in strains engineered for xylose transport and assimilation. FEMS Yeast Res. 2018, 18, 1. Available online: https://academic.oup.com/femsyr/article/doi/10.1093/femsyr/fox096/4791530 (accessed on 3 April 2019). [CrossRef] [Green Version]
- Osiro, K.O.; Borgström, C.; Brink, D.P.; Fjölnisdóttir, B.L.; Gorwa-Grauslund, M.F. Exploring the xylose paradox in Saccharomyces cerevisiae through in vivo sugar signalomics of targeted deletants. Microb. Cell Fact. 2019, 18, 88. Available online: https://microbialcellfactories.biomedcentral.com/articles/10.1186/s12934-019-1141-x (accessed on 3 September 2020). [CrossRef] [Green Version]
- Endalur Gopinarayanan, V.; Nair, N.U. Pentose Metabolism in Saccharomyces cerevisiae: The Need to Engineer Global Regulatory Systems. Biotechnol. J. 2019, 14, 1800364. Available online: https://onlinelibrary.wiley.com/doi/full/10.1002/biot.201800364 (accessed on 17 February 2023). [CrossRef] [Green Version]
- Wei, S.; Liu, Y.; Wu, M.; Ma, T.; Bai, X.; Hou, J.; Shen, Y.; Bao, X. Disruption of the transcription factors Thi2p and Nrm1p alleviates the post-glucose effect on xylose utilization in Saccharomyces cerevisiae. Biotechnol. Biofuels 2018, 11, 112. Available online: https://biotechnologyforbiofuels.biomedcentral.com/articles/10.1186/s13068-018-1112-1 (accessed on 17 February 2023). [CrossRef] [Green Version]
- Wei, S.; Bai, P.; Liu, Y.; Yang, M.; Ma, J.; Hou, J.; Liu, W.; Bao, X.; Shen, Y. A Thi2p Regulatory Network Controls the Post-glucose Effect of Xylose Utilization in Saccharomyces cerevisiae. Front. Microbiol. 2019, 10, 1649. [Google Scholar] [CrossRef] [Green Version]
- Wu, M.; Li, H.; Wei, S.; Wu, H.; Wu, X.; Bao, X.; Hou, J.; Liu, W.; Shen, Y. Simulating Extracellular Glucose Signals Enhances Xylose Metabolism in Recombinant Saccharomyces cerevisiae. Microorganisms 2020, 8, 100. Available online: https://www.mdpi.com/2076-2607/8/1/100 (accessed on 4 June 2020). [CrossRef] [PubMed] [Green Version]
- Endalur Gopinarayanan, V.; Nair, N.U. A semi-synthetic regulon enables rapid growth of yeast on xylose. Nat. Commun. 2018, 9, 1233. Available online: https://www.nature.com/articles/s41467-018-03645-7 (accessed on 17 February 2023). [CrossRef] [Green Version]
- Sato, T.K.; Tremaine, M.; Parreiras, L.S.; Hebert, A.S.; Myers, K.S.; Higbee, A.J.; Sardi, M.; McIlwain, S.J.; Ong, I.M.; Breuer, R.J.; et al. Directed Evolution Reveals Unexpected Epistatic Interactions That Alter Metabolic Regulation and Enable Anaerobic Xylose Use by Saccharomyces cerevisiae. PLOS Genet. 2016, 12, e1006372. [Google Scholar] [CrossRef] [Green Version]
- dos Santos, L.V.; Carazzolle, M.F.; Nagamatsu, S.T.; Sampaio, N.M.V.; Almeida, L.D.; Pirolla, R.A.S.; Borelli, G.; Corrêa, T.L.R.; Argueso, J.L.; Pereira, G.A.G. Unraveling the genetic basis of xylose consumption in engineered Saccharomyces cerevisiae strains. Sci. Rep. 2016, 6, 38676. Available online: http://www.nature.com/articles/srep38676 (accessed on 24 January 2019). [CrossRef] [PubMed]
- Myers, K.S.; Riley, N.M.; MacGilvray, M.E.; Sato, T.K.; McGee, M.; Heilberger, J.; Coon, J.J.; Gasch, A.P. Rewired cellular signaling coordinates sugar and hypoxic responses for anaerobic xylose fermentation in yeast. PLoS Genet. 2019, 15, e1008037. [Google Scholar] [CrossRef] [Green Version]
- Conrad, M.; Schothorst, J.; Kankipati, H.N.; Van Zeebroeck, G.; Rubio-Texeira, M.; Thevelein, J.M. Nutrient sensing and signaling in the yeast Saccharomyces cerevisiae. FEMS Microbiol. Rev. 2014, 38, 254–299. Available online: https://academic.oup.com/femsre/article-lookup/doi/10.1111/1574-6976.12065 (accessed on 24 January 2019). [CrossRef] [Green Version]
- Coccetti, P.; Nicastro, R.; Tripodi, F. Conventional and emerging roles of the energy sensor Snf1/AMPK in Saccharomyces cerevisiae. Microb. Cell 2018, 5, 482. [Google Scholar] [CrossRef] [Green Version]
- Vallejo, M.C.; Mayinger, P. Delayed Turnover of Unphosphorylated Ssk1 during Carbon Stress Activates the Yeast Hog1 Map Kinase Pathway. PLoS ONE 2015, 10, e0137199. Available online: https://pubmed.ncbi.nlm.nih.gov/26340004/ (accessed on 31 May 2023). [CrossRef]
- Piao, H.; Maclean Freed, J.; Mayinger, P. Metabolic Activation of the HOG MAP Kinase Pathway by Snf1/AMPK Regulates Lipid Signaling at the Golgi. Traffic 2012, 13, 1522–1531. Available online: https://onlinelibrary.wiley.com/doi/full/10.1111/j.1600-0854.2012.01406.x (accessed on 17 February 2023). [CrossRef] [Green Version]
- Saito, H.; Posas, F. Response to hyperosmotic stress. Genetics 2012, 192, 289–318. Available online: http://www.ncbi.nlm.nih.gov/pubmed/9383053 (accessed on 24 January 2019). [CrossRef] [Green Version]
- Thevelein, J.M.; de Winde, J.H. Novel sensing mechanisms and targets for the cAMP-protein kinase A pathway in the yeast Saccharomyces cerevisiae. Mol. Microbiol. 1999, 33, 904–918. Available online: http://doi.wiley.com/10.1046/j.1365-2958.1999.01538.x (accessed on 24 January 2019). [CrossRef] [PubMed]
- Santangelo, G.M. Glucose signaling in Saccharomyces cerevisiae. Microbiol. Mol. Biol. Rev. 2006, 70, 253–282. Available online: http://www.ncbi.nlm.nih.gov/pubmed/16524925 (accessed on 9 April 2019). [CrossRef] [PubMed] [Green Version]
- Smets, B.; Ghillebert, R.; De Snijder, P.; Binda, M.; Swinnen, E.; De Virgilio, C.; Winderickx, J. Life in the midst of scarcity: Adaptations to nutrient availability in Saccharomyces cerevisiae. Curr. Genet. 2010, 56, 1–32. Available online: http://link.springer.com/10.1007/s00294-009-0287-1 (accessed on 1 April 2019). [CrossRef] [Green Version]
- Schepers, W.; Van Zeebroeck, G.; Pinkse, M.; Verhaert, P.; Thevelein, J.M. In vivo phosphorylation of Ser21 and Ser83 during nutrient-induced activation of the yeast protein kinase A (PKA) target trehalase. J. Biol. Chem. 2012, 287, 44130–44142. Available online: http://www.ncbi.nlm.nih.gov/pubmed/23155055 (accessed on 24 January 2019). [CrossRef] [PubMed] [Green Version]
- Dihazi, H.; Kessler, R.; Eschrich, K. Glucose-Induced Stimulation of the Ras-cAMP Pathway in Yeast Leads to Multiple Phosphorylations and Activation of 6-Phosphofructo-2-Kinase. 2003. Available online: https://pubs.acs.org/doi/abs/10.1021/bi034167r (accessed on 24 January 2019).
- Portela, P.; Moreno, S.; Rossi, S. Characterization of yeast pyruvate kinase 1 as a protein kinase A substrate, and specificity of the phosphorylation site sequence in the whole protein. Biochem. J. 2006, 396, 117–126. Available online: http://www.ncbi.nlm.nih.gov/pubmed/16426231 (accessed on 1 April 2019). [CrossRef]
- Cameroni, E.; Hulo, N.; Roosen, J.; Winderickx, J.; De Virgilio, C. The Novel Yeast PAS Kinase Rim15 Orchestrates G0-Associated Antioxidant Defense Mechanisms. Cell Cycle 2004, 3, 462–468. [Google Scholar] [CrossRef] [Green Version]
- Toda, T.; Cameron, S.; Sass, P.; Zoller, M.; Scott, J.D.; McMullen, B.; Hurwitz, M.; Krebs, E.G.; Wigler, M. Cloning and characterization of BCY1, a locus encoding a regulatory subunit of the cyclic AMP-dependent protein kinase in Saccharomyces cerevisiae. Mol. Cell Biol. 1987, 7, 1371–1377. Available online: http://www.ncbi.nlm.nih.gov/pubmed/3037314 (accessed on 7 March 2019).
- Toda, T.; Cameron, S.; Sass, P.; Zoller, M.; Wigler, M. Three different genes in S. cerevisiae encode the catalytic subunits of the cAMP-dependent protein kinase. Cell 1987, 50, 277–287. Available online: https://www.sciencedirect.com/science/article/pii/0092867487902236?via%3Dihub (accessed on 7 March 2019). [CrossRef]
- Cannon, J.F.; Tatchell, K. Characterization of Saccharomyces cerevisiae genes encoding subunits of cyclic AMP-dependent protein kinase. Mol. Cell Biol. 1987, 7, 2653–2663. Available online: http://www.ncbi.nlm.nih.gov/pubmed/2823100 (accessed on 8 March 2019).
- Matsumoto, K.; Uno, I.; Oshima, Y.; Ishikawa, T. Isolation and characterization of yeast mutants deficient in adenylate cyclase and cAMP-dependent protein kinase. Proc. Natl. Acad. Sci. USA 1982, 79, 2355–2359. Available online: http://www.ncbi.nlm.nih.gov/pubmed/6285379 (accessed on 8 March 2019). [CrossRef]
- Kataoka, T.; Broek, D.; Wigler, M. DNA sequence and characterization of the S. cerevisiae gene encoding adenylate cyclase. Cell 1985, 43, 493–505. [Google Scholar] [CrossRef] [PubMed]
- Casperson, G.F.; Walker, N.; Bourne, H.R. Isolation of the gene encoding adenylate cyclase in Saccharomyces cerevisiae. Proc. Natl. Acad. Sci. USA 1985, 82, 5060–5063. Available online: https://pubmed.ncbi.nlm.nih.gov/2991907/ (accessed on 18 February 2023). [CrossRef] [PubMed]
- Broach, J.R.; Deschenes, R.J. The Function of Ras Genes in Saccharomyces cerevisiae. Adv. Cancer Res. 1990, 54, 79–139. Available online: https://www.sciencedirect.com/science/article/pii/S0065230X0860809X?via%3Dihub (accessed on 7 March 2019).
- Broek, D.; Toda, T.; Michaeli, T.; Levin, L.; Birchmeier, C.; Zoller, M.; Powers, S.; Wigler, M. The S. cerevisiae CDC25 gene product regulates the RAS/adenylate cyclase pathway. Cell 1987, 48, 789–799. Available online: https://pubmed.ncbi.nlm.nih.gov/3545497/ (accessed on 18 February 2023). [CrossRef]
- Boy-Marcotte, E.; Ikonomi, P.; Jacquet, M. SDC25, a dispensable Ras guanine nucleotide exchange factor of Saccharomyces cerevisiae differs from CDC25 by its regulation. Mol. Biol. Cell 1996, 7, 529. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Colombo, S.; Ronchetti, D.; Thevelein, J.M.; Winderickx, J.; Martegani, E. Activation state of the Ras2 protein and glucose-induced signaling in Saccharomyces cerevisiae. J. Biol. Chem. 2004, 279, 46715–46722. Available online: http://www.ncbi.nlm.nih.gov/pubmed/15339905 (accessed on 1 April 2019). [CrossRef] [Green Version]
- Colombo, S.; Ma, P.; Cauwenberg, L.; Winderickx, J.; Crauwels, M.; Teunissen, A.; Nauwelaers, D.; de Winde, J.H.; Gorwa, M.F.; Colavizza, D.; et al. Involvement of distinct G-proteins, Gpa2 and Ras, in glucose- and intracellular acidification-induced cAMP signalling in the yeast Saccharomyces cerevisiae. EMBO J. 1998, 17, 3326–3341. Available online: https://pubmed.ncbi.nlm.nih.gov/9628870/ (accessed on 18 February 2023). [CrossRef] [Green Version]
- Dechant, R.; Binda, M.; Lee, S.S.; Pelet, S.; Winderickx, J.; Peter, M. Cytosolic pH is a second messenger for glucose and regulates the PKA pathway through V-ATPase. EMBO J. 2010, 29, 2515–2526. Available online: http://emboj.embopress.org/cgi/doi/10.1038/emboj.2010.138 (accessed on 2 June 2020). [CrossRef] [Green Version]
- Peeters, K.; Van Leemputte, F.; Fischer, B.; Bonini, B.M.; Quezada, H.; Tsytlonok, M.; Haesen, D.; Vanthienen, W.; Bernardes, N.; Gonzalez-Blas, C.B.; et al. Fructose-1,6-bisphosphate couples glycolytic flux to activation of Ras. Nat. Commun. 2017, 8, 922. Available online: https://pubmed.ncbi.nlm.nih.gov/29030545/ (accessed on 18 February 2023). [CrossRef] [Green Version]
- Tanaka, K.; Lin, B.K.; Wood, D.R.; Tamanoi, F. IRA2, an upstream negative regulator of RAS in yeast, is a RAS GTPase-activating protein. Proc. Natl. Acad. Sci. USA 1991, 88, 468–472. Available online: http://www.ncbi.nlm.nih.gov/pubmed/1988946 (accessed on 1 April 2019). [CrossRef]
- Tanaka, K.; Nakafuku, M.; Satoh, T.; Marshall, M.S.; Gibbs, J.B.; Matsumoto, K.; Kaziro, Y.; Toh-e, A.S. cerevisiae genes IRA1 and IRA2 encode proteins that may be functionally equivalent to mammalian ras GTPase activating protein. Cell 1990, 60, 803–807. Available online: https://www.sciencedirect.com/science/article/pii/009286749090094U?via%3Dihub (accessed on 13 March 2019). [CrossRef] [PubMed]
- Tanaka, K.; Nakafuku, M.; Tamanoi, F.; Kaziro, Y.; Matsumoto, K.; Toh-e, A. IRA2, a second gene of Saccharomyces cerevisiae that encodes a protein with a domain homologous to mammalian ras GTPase-activating protein. Mol. Cell Biol. 1990, 10, 4303–4313. Available online: http://www.ncbi.nlm.nih.gov/pubmed/2164637 (accessed on 7 March 2019). [PubMed] [Green Version]
- Tanaka, K.; Matsumoto, K.; Toh-E, A. IRA1, an inhibitory regulator of the RAS-cyclic AMP pathway in Saccharomyces cerevisiae. Mol. Cell Biol. 1989, 9, 757–768. Available online: http://www.ncbi.nlm.nih.gov/pubmed/2540426 (accessed on 1 April 2019). [PubMed] [Green Version]
- Sass, P.; Field, J.; Nikawa, J.; Toda, T.; Wigler, M. Cloning and characterization of the high-affinity cAMP phosphodiesterase of Saccharomyces cerevisiae. Proc. Natl. Acad. Sci. USA 1986, 83, 9303. [Google Scholar] [CrossRef]
- Nikawa, J.; Sass, P.; Wigler, M. Cloning and characterization of the low-affinity cyclic AMP phosphodiesterase gene of Saccharomyces cerevisiae. Mol. Cell Biol. 1987, 7, 3629. [Google Scholar] [CrossRef] [PubMed]
- Ma, P.; Wera, S.; Van Dijck, P.; Thevelein, J.M. The PDE1-encoded low-affinity phosphodiesterase in the yeast Saccharomyces cerevisiae has a specific function in controlling agonist-induced cAMP signaling. Mol. Biol. Cell 1999, 10, 91–104. Available online: https://pubmed.ncbi.nlm.nih.gov/9880329/ (accessed on 18 February 2023). [CrossRef] [Green Version]
- Hu, Y.; Liu, E.; Bai, X.; Zhang, A. The localization and concentration of the PDE2 -encoded high-affinity cAMP phosphodiesterase is regulated by cAMP-dependent protein kinase A in the yeast Saccharomyces cerevisiae. FEMS Yeast Res. 2010, 10, 177–187. Available online: https://academic.oup.com/femsyr/article-lookup/doi/10.1111/j.1567-1364.2009.00598.x (accessed on 26 May 2020). [CrossRef] [Green Version]
- Griffioen, G.; Thevelein, J.M. Molecular mechanisms controlling the localisation of protein kinase A. In Current Genetics; Springer: Berlin/Heidelberg, Germany, 2002; Volume 41, pp. 199–207. [Google Scholar]
- Griffioen, G.; Anghileri, P.; Imre, E.; Baroni, M.D.; Ruis, H. Nutritional control of nucleocytoplasmic localization of cAMP-dependent protein kinase catalytic and regulatory subunits in Saccharomyces cerevisiae. J. Biol. Chem. 2000, 275, 1449–1456. Available online: http://www.ncbi.nlm.nih.gov/pubmed/10625697 (accessed on 24 January 2019). [CrossRef] [Green Version]
- Tudisca, V.; Recouvreux, V.; Moreno, S.; Boy-Marcotte, E.; Jacquet, M.; Portela, P. Differential localization to cytoplasm, nucleus or P-bodies of yeast PKA subunits under different growth conditions. Eur. J. Cell Biol. 2010, 89, 339–348. Available online: https://www.sciencedirect.com/science/article/pii/S017193350900301X?via%3Dihub (accessed on 11 February 2019). [CrossRef]
- Colledge, M.; Scott, J.D. AKAPs: From structure to function. Trends Cell Biol. 1999, 9, 216–221. Available online: https://www.sciencedirect.com/science/article/pii/S0962892499015585?via%3Dihub (accessed on 7 March 2019). [CrossRef]
- Dolinsky, W.; Paolillo, R.; D’apice, S.; Schiattarella, G.G.; Ameri, P.; Borzacchiello, D.; Catalucci, D.; Chimenti, C.; Crott, L.; Sciarretta, S.; et al. Mitochondrial a Kinase Anchor Proteins in Cardiovascular Health and Disease: A Review Article on Behalf of the Working Group on Cellular and Molecular Biology of the Heart of the Italian Society of Cardiology. Int. J. Mol. Sci. 2022, 23, 7691. Available online: https://www.mdpi.com/1422-0067/23/14/7691/htm (accessed on 5 September 2022).
- Søberg, K.; Skålhegg, B.S. The Molecular Basis for Specificity at the Level of the Protein Kinase a Catalytic Subunit. Front. Endocrinol 2018, 9, 538. Available online: https://www.frontiersin.org/article/10.3389/fendo.2018.00538/full (accessed on 26 March 2019). [CrossRef] [Green Version]
- Taskén, K.; Aandahl, E.M. Localized Effects of cAMP Mediated by Distinct Routes of Protein Kinase, A. Physiol. Rev. 2004, 84, 137–167. Available online: http://www.physiology.org/doi/10.1152/physrev.00021.2003 (accessed on 19 July 2019). [CrossRef] [Green Version]
- Kocik, R.A.; Gasch, A.P. Breadth and Specificity in Pleiotropic Protein Kinase A Activity and Environmental Responses. Front. Cell Dev. Biol. 2022, 10, 334. [Google Scholar] [CrossRef]
- Langeberg, L.K.; Scott, J.D. A-kinase-anchoring proteins. J. Cell Sci. 2005, 118, 3217–3220. Available online: https://journals.biologists.com/jcs/article/118/15/3217/28454/A-kinase-anchoring-proteins (accessed on 11 November 2022). [CrossRef] [Green Version]
- Houslay, M.D. Underpinning compartmentalised cAMP signalling through targeted cAMP breakdown. Trends Biochem. Sci. 2010, 35, 91–100. Available online: https://www.sciencedirect.com/science/article/pii/S0968000409001923?via%3Dihub (accessed on 13 March 2019). [CrossRef]
- Torres-Quesada, O.; Mayrhofer, J.E.; Stefan, E. The many faces of compartmentalized PKA signalosomes. Cell Signal 2017, 37, 1–11. Available online: https://www.sciencedirect.com/science/article/pii/S0898656817301420?via%3Dihub (accessed on 26 March 2019). [CrossRef] [PubMed]
- Baillie, G.S.; Scott, J.D.; Houslay, M.D. Compartmentalisation of phosphodiesterases and protein kinase A: Opposites attract. FEBS Lett. 2005, 579, 3264–3270. Available online: http://doi.wiley.com/10.1016/j.febslet.2005.03.089 (accessed on 8 March 2019). [CrossRef] [PubMed] [Green Version]
- Sample, V.; DiPilato, L.M.; Yang, J.H.; Ni, Q.; Saucerman, J.J.; Zhang, J. Regulation of nuclear PKA revealed by spatiotemporal manipulation of cyclic AMP. Nat. Chem. Biol. 2012, 8, 375–382. Available online: http://www.nature.com/articles/nchembio.799 (accessed on 13 March 2019). [CrossRef] [PubMed] [Green Version]
- Hess, K.C.; Liu, J.; Manfredi, G.; Mühlschlegel, F.A.; Buck, J.; Levin, L.R.; Barrientos, A. A mitochondrial CO 2 -adenylyl cyclase-cAMP signalosome controls yeast normoxic cytochrome c oxidase activity. FASEB J. 2014, 28, 4369–4380. Available online: http://www.fasebj.org/doi/10.1096/fj.14-252890 (accessed on 9 April 2019). [CrossRef] [PubMed] [Green Version]
- Galello, F.; Moreno, S.; Rossi, S. Interacting proteins of protein kinase A regulatory subunit in Saccharomyces cerevisiae. J. Proteomics. 2014, 109, 261–275. [Google Scholar] [CrossRef]
- Filteau, M.; Diss, G.; Torres-Quiroz, F.; Dubé, A.K.; Schraffl, A.; Bachmann, V.A. Gagnon-Arsenault, I.; Chrétien, A.; Steunou, A.; Dionne, U.; et al. Systematic identification of signal integration by protein kinase, A. Proc. Natl. Acad. Sci. USA 2015, 112, 4501–4506. Available online: http://www.ncbi.nlm.nih.gov/pubmed/25831502 (accessed on 31 March 2019). [CrossRef] [PubMed]
- Griffioen, G.; Branduardi, P.; Ballarini, A.; Anghileri, P.; Norbeck, J.; Baroni, M.D.; Ruis, H. Nucleocytoplasmic distribution of budding yeast protein kinase A regulatory subunit Bcy1 requires Zds1 and is regulated by Yak1-dependent phosphorylation of its targeting domain. Mol. Cell Biol. 2001, 21, 511–523. Available online: http://www.ncbi.nlm.nih.gov/pubmed/11134339 (accessed on 25 January 2019). [CrossRef] [Green Version]
- Wagner, E.R.; Nightingale, N.M.; Jen, A.; Overmyer, K.A.; McGee, M.; Coon, J.J.; Gasch, A.P. PKA regulatory subunit Bcy1 couples growth, lipid metabolism, and fermentation during anaerobic xylose growth in Saccharomyces cerevisiae. PLoS Genet 2023, 19, e1010593. [Google Scholar] [CrossRef]
- Ye, T.; Elbing, K.; Hohmann, S. The pathway by which the yeast protein kinase Snf1p controls acquisition of sodium tolerance is different from that mediating glucose regulation. Microbiology 2008, 154, 2814–2826. Available online: https://www.microbiologyresearch.org/content/journal/micro/10.1099/mic.0.2008/020149-0 (accessed on 13 June 2023). [CrossRef] [Green Version]
- Shashkova, S.; Welkenhuysen, N.; Hohmann, S. Molecular communication: Crosstalk between the Snf1 and other signaling pathways. FEMS Yeast Res. 2015, 15, 26. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ashrafi, K.; Lin, S.S.; Manchester, J.K.; Gordon, J.I. Sip2p and its partner Snf1p kinase affect aging in, S. cerevisiae. Genes. Dev. 2000, 14, 1872–1885. Available online: http://genesdev.cshlp.org/content/14/15/1872.full (accessed on 25 January 2023). [CrossRef] [PubMed]
- Lin, S.S.; Manchester, J.K.; Gordon, J.I. Sip2, an N-myristoylated β subunit of Snf1 kinase, regulates aging in Saccharomyces cerevisiae by affecting cellular histone kinase activity, recombination at rDNA loci, and silencing. J. Biol. Chem. 2003, 278, 13390–13397. [Google Scholar] [CrossRef] [Green Version]
- Hedbacker, K.; Carlson, M. SNF1/AMPK pathways in yeast. Front. Biosci. 2008, 13, 2408–2420. Available online: https://pubmed.ncbi.nlm.nih.gov/17981722/ (accessed on 25 January 2023). [CrossRef] [Green Version]
- Usaite, R.; Jewett, M.C.; Oliveira, A.P.; Yates, J.R.; Olsson, L.; Nielsen, J. Reconstruction of the yeast Snf1 kinase regulatory network reveals its role as a global energy regulator. Mol. Syst. Biol. 2009, 5, 319. Available online: https://pubmed.ncbi.nlm.nih.gov/19888214/ (accessed on 25 January 2023). [CrossRef] [Green Version]
- Simpson-Lavy, K.J.; Kupiec, M. The polyHIS Tract of Yeast AMPK Coordinates Carbon Metabolism with Iron Availability. Int. J. Mol. Sci. 2023, 24, 1368. Available online: https://www.mdpi.com/1422-0067/24/2/1368/htm (accessed on 30 January 2023). [CrossRef] [PubMed]
- Kuchin, S.; Treich, I.; Carlson, M. A regulatory shortcut between the Snf1 protein 7 kinase and RNA polymerase II holoenzyme. Proc. Natl. Acad. Sci. USA 2000, 97, 7916–7920. Available online: https://www.pnas.org/doi/abs/10.1073/pnas.140109897 (accessed on 19 January 2023). [CrossRef] [PubMed]
- Young, E.T.; Zhang, C.; Shokat, K.M.; Parua, P.K.; Braun, K.A. The AMP-activated protein kinase Snf1 regulates transcription factor binding, RNA polymerase II activity, and mRNA stability of glucose-repressed genes in Saccharomyces cerevisiae. J. Biol. Chem. 2012, 287, 29021–29034. Available online: http://www.jbc.org/article/S0021925820684332/fulltext (accessed on 19 January 2023). [CrossRef] [Green Version]
- Jiang, R.; Carlson, M. The Snf1 protein kinase and its activating subunit, Snf4, interact with distinct domains of the Sip1/Sip2/Gal83 component in the kinase complex. Mol. Cell Biol. 1997, 17, 2099–2106. Available online: https://journals.asm.org/doi/10.1128/MC.B.17.4.2099 (accessed on 25 January 2023). [CrossRef] [PubMed] [Green Version]
- Celenza, J.L.; Eng, F.J.; Carlson, M. Molecular analysis of the SNF4 gene of Saccharomyces cerevisiae: Evidence for physical association of the SNF4 protein with the SNF1 protein kinase. Mol. Cell Biol. 1989, 9, 5045–5054. Available online: https://pubmed.ncbi.nlm.nih.gov/2481228/ (accessed on 25 January 2023).
- Leech, A.; Nath, N.; McCartney, R.R.; Schmidt, M.C. Isolation of mutations in the catalytic domain of the snf1 kinase that render its activity independent of the snf4 subunit. Eukaryot. Cell 2003, 2, 265–273. Available online: https://pubmed.ncbi.nlm.nih.gov/12684376/ (accessed on 25 January 2023). [CrossRef] [PubMed] [Green Version]
- Momcilovic, M.; Iram, S.H.; Liu, Y.; Carlson, M. Roles of the glycogen-binding domain and Snf4 in glucose inhibition of SNF1 protein kinase. J. Biol. Chem. 2008, 283, 19521–19529. Available online: https://pubmed.ncbi.nlm.nih.gov/18474591/ (accessed on 25 January 2023). [CrossRef] [PubMed] [Green Version]
- Mayer, F.V.; Heath, R.; Underwood, E.; Sanders, M.J.; Carmena, D.; McCartney, R.R.; Leiper, F.C.; Xiao, B.; Jing, C.; Walker, P.A.; et al. ADP regulates SNF1, the Saccharomyces cerevisiae homolog of AMP-activated protein kinase. Cell Metab. 2011, 14, 707–714. Available online: https://pubmed.ncbi.nlm.nih.gov/22019086/ (accessed on 25 January 2023). [CrossRef] [Green Version]
- MC, S.; RR, M. beta-subunits of Snf1 kinase are required for kinase function and substrate definition. EMBO J. 2000, 19, 4936–4943. Available online: https://pubmed.ncbi.nlm.nih.gov/10990457/ (accessed on 25 January 2023).
- Simpson-Lavy, K.J.; Kupiec, M. Regulation of yeast Snf1 (AMPK) by a polyhistidine containing pH sensing module. iScience. 2022, 25, 105083. [Google Scholar] [CrossRef]
- Wilson, W.A.; Hawley, S.A.; Hardie, D.G. Glucose repression/derepression in budding yeast: SNF1 protein kinase is activated by phosphorylation under derepressing conditions, and this correlates with a high AMP:ATP ratio. Curr. Biol. 1996, 6, 1426–1434. Available online: https://pubmed.ncbi.nlm.nih.gov/8939604/ (accessed on 25 January 2023). [CrossRef] [Green Version]
- Simpson-Lavy, K.; Xu, T.; Johnston, M.; Kupiec, M. The Std1 Activator of the Snf1/AMPK Kinase Controls Glucose Response in Yeast by a Regulated Protein Aggregation. Mol. Cell. 2017, 68, 1120–1133.e3. [Google Scholar] [CrossRef] [Green Version]
- Zhang, M.; Galdieri, L.; Vancura, A. The yeast AMPK homolog SNF1 regulates acetyl coenzyme A homeostasis and histone acetylation. Mol. Cell Biol. 2013, 33, 4701–4717. Available online: https://pubmed.ncbi.nlm.nih.gov/24081331/ (accessed on 25 January 2023). [CrossRef] [PubMed] [Green Version]
- Sanz, P. Snf1 protein kinase: A key player in the response to cellular stress in yeast. Biochem. Soc. Trans. 2003, 31 Pt 1, 178–181. Available online: https://pubmed.ncbi.nlm.nih.gov/12546680/ (accessed on 25 January 2023). [CrossRef] [PubMed]
- Hahn, J.S.; Thiele, D.J. Activation of the Saccharomyces cerevisiae heat shock transcription factor under glucose starvation conditions by Snf1 protein kinase. J. Biol. Chem. 2004, 279, 5169–5176. Available online: https://pubmed.ncbi.nlm.nih.gov/14612437/ (accessed on 25 January 2023). [CrossRef] [Green Version]
- Mizuno, T.; Masuda, Y.; Irie, K. The Saccharomyces cerevisiae AMP.K.; Snf1, Negatively Regulates the Hog1 MAPK Pathway in ER Stress Response. PLoS Genet. 2015, 11, e1005491. Available online: https://pubmed.ncbi.nlm.nih.gov/26394309/ (accessed on 25 January 2023). [CrossRef] [Green Version]
- Back, S.H.; Schröder, M.; Lee, K.; Zhang, K.; Kaufman, R.J. ER stress signaling by regulated splicing: IRE1/HAC1/XBP1. Methods 2005, 35, 395–416. Available online: https://pubmed.ncbi.nlm.nih.gov/15804613/ (accessed on 25 January 2023). [CrossRef] [PubMed]
- Portillo, F.; Mulet, J.M.; Serrano, R. A role for the non-phosphorylated form of yeast Snf1: Tolerance to toxic cations and activation of potassium transport. FEBS Lett. 2005, 579, 512–516. Available online: https://pubmed.ncbi.nlm.nih.gov/15642368/ (accessed on 25 January 2023). [CrossRef] [Green Version]
- Dubacq, C.; Chevalier, A.; Mann, C. The protein kinase Snf1 is required for tolerance to the ribonucleotide reductase inhibitor hydroxyurea. Mol. Cell Biol. 2004, 24, 2560–2572. Available online: https://pubmed.ncbi.nlm.nih.gov/14993292/ (accessed on 25 January 2023). [CrossRef] [Green Version]
- Pérez-Sampietro, M.; Casas, C.; Herrero, E. The AMPK Family Member Snf1 Protects Saccharomyces cerevisiae Cells upon Glutathione Oxidation. PLoS ONE 2013, 8, e58283. [Google Scholar] [CrossRef] [Green Version]
- Hong, S.P.; Carlson, M. Regulation of snf1 protein kinase in response to environmental stress. J. Biol. Chem. 2007, 282, 16838–16845. Available online: https://pubmed.ncbi.nlm.nih.gov/17438333/ (accessed on 25 January 2023). [CrossRef] [PubMed] [Green Version]
- Platara, M.; Ruiz, A.; Serrano, R.; Palomino, A.; Moreno, F.; Ariño, J. The transcriptional response of the yeast Na(+)-ATPase ENA1 gene to alkaline stress involves three main signaling pathways. J. Biol. Chem. 2006, 281, 36632–36642. Available online: https://pubmed.ncbi.nlm.nih.gov/17023428/ (accessed on 25 January 2023). [CrossRef] [Green Version]
- Mori, K. Signalling pathways in the unfolded protein response: Development from yeast to mammals. J. Biochem. 2009, 146, 743–750. Available online: https://pubmed.ncbi.nlm.nih.gov/19861400/ (accessed on 25 January 2023). [CrossRef]
- Walter, P.; Ron, D. The unfolded protein response: From stress pathway to homeostatic regulation. Science 2011, 334, 1081–1086. Available online: https://pubmed.ncbi.nlm.nih.gov/22116877/ (accessed on 25 January 2023). [CrossRef] [PubMed] [Green Version]
- Ferrer-Dalmau, J.; Randez-Gil, F.; Marquina, M.; Prieto, J.A.; Casamayor, A. Protein kinase Snf1 is involved in the proper regulation of the unfolded protein response in Saccharomyces cerevisiae. Biochem. J. 2015, 468, 33–47. Available online: https://pubmed.ncbi.nlm.nih.gov/25730376/ (accessed on 25 January 2023). [CrossRef]
- Lu, J.Y.; Lin, Y.Y.; Sheu, J.C.; Wu, J.T.; Lee, F.J.; Chen, Y.; Lin, M.; Chiang, F.T.; Tai, T.; Berger, S.L.; et al. Acetylation of yeast AMPK controls intrinsic aging independently of caloric restriction. Cell 2011, 146, 969–979. Available online: https://pubmed.ncbi.nlm.nih.gov/21906795/ (accessed on 25 January 2023). [CrossRef] [PubMed] [Green Version]
- Wierman, M.B.; Maqani, N.; Strickler, E.; Li, M.; Smith, J.S. Caloric Restriction Extends Yeast Chronological Life Span by Optimizing the Snf1 (AMPK) Signaling Pathway. Mol. Cell Biol. 2017, 37, e00562-16. Available online: https://pubmed.ncbi.nlm.nih.gov/28373292/ (accessed on 25 January 2023). [CrossRef] [Green Version]
- Maqani, N.; Fine, R.D.; Shahid, M.; Li, M.; Enriquez-Hesles, E.; Smith, J.S. Spontaneous mutations in CYC8 and MIG1 suppress the short chronological lifespan of budding yeast lacking SNF1/AMPK. Microb. Cell 2018, 5, 233–248. Available online: https://pubmed.ncbi.nlm.nih.gov/29796388/ (accessed on 25 January 2023). [CrossRef] [Green Version]
- Hedbacker, K.; Townley, R.; Carlson, M. Cyclic AMP-Dependent Protein Kinase Regulates the Subcellular Localization of Snf1-Sip1 Protein Kinase. Mol. Cell Biol. 2004, 24, 1836–1843. [Google Scholar] [CrossRef] [Green Version]
- Barrett, L.; Orlova, M.; Maziarz, M.; Kuchin, S. Protein kinase A contributes to the negative control of Snf1 protein kinase in Saccharomyces cerevisiae. Eukaryot. Cell 2012, 11, 119–128. Available online: https://pubmed.ncbi.nlm.nih.gov/22140226/ (accessed on 25 January 2023). [CrossRef] [Green Version]
- Nicastro, R.; Tripodi, F.; Gaggini, M.; Castoldi, A.; Reghellin, V.; Nonnis, S.; Tedeschi, G.; Coccetti, P. Snf1 phosphorylates adenylate cyclase and negatively regulates protein kinase A-dependent transcription in Saccharomyces cerevisiae. J. Biol. Chem. 2015, 290, 24715–24726. [Google Scholar] [CrossRef] [Green Version]
- Maeda, T.; Wurgler-Murphy, S.M.; Saito, H. A two-component system that regulates an osmosensing MAP kinase cascade in yeast. Nature 1994, 369, 242–245. Available online: https://pubmed.ncbi.nlm.nih.gov/8183345/ (accessed on 19 February 2023). [CrossRef] [PubMed]
- Maeda, T.; Takekawa, M.; Saito, H. Activation of Yeast PBS2 MAPKK by MAPKKKs or by Binding of an SH3-Containing Omosensor. Science 1995, 269, 554–558. Available online: https://www.science.org/doi/10.1126/science.7624781 (accessed on 10 February 2023). [CrossRef] [PubMed]
- Ota, I.M.; Varshavsky, A. A yeast protein similar to bacterial two-component regulators. Science 1993, 262, 566–569. Available online: https://pubmed.ncbi.nlm.nih.gov/8211183/ (accessed on 1 June 2023). [CrossRef]
- Brewster, J.L.; De Valoir, T.; Dwyer, N.D.; Winter, E.; Gustin, M.C. An osmosensing signal transduction pathway in yeast. Science 1993, 259, 1760–1763. Available online: https://pubmed.ncbi.nlm.nih.gov/7681220/ (accessed on 19 February 2023). [CrossRef] [PubMed]
- Tatebayashi, K.; Takekawa, M.; Saito, H. A docking site determining specificity of Pbs2 MAPKK for Ssk2/Ssk22 MAPKKKs in the yeast HOG pathway. EMBO J. 2003, 22, 3624. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Boguslawski, G. PBS2, a yeast gene encoding a putative protein kinase, interacts with the RAS2 pathway and affects osmotic sensitivity of Saccharomyces cerevisiae. J. Gen. Microbiol. 1992, 138, 2425–2432. Available online: https://pubmed.ncbi.nlm.nih.gov/1479360/ (accessed on 1 June 2023). [CrossRef] [Green Version]
- Posas, F.; Saito, H. Osmotic activation of the HOG MAPK pathway via Ste11p MAPKKK: Scaffold role of Pbs2p MAPKK. Science 1997, 276, 1702–1708. Available online: https://pubmed.ncbi.nlm.nih.gov/9180081/ (accessed on 19 February 2023). [CrossRef]
- De Nadal, E.; Posas, F. The HOG pathway and the regulation of osmoadaptive responses in yeast. FEMS Yeast Res. 2022, 22, 1. Available online: https://academic.oup.com/femsyr/article/22/1/foac013/6543702 (accessed on 20 January 2023). [CrossRef]
- Albertyn, J.; Hohmann, S.; Thevelein, J.M.; Prior, B.A. GPD1, which encodes glycerol-3-phosphate dehydrogenase, is essential for growth under osmotic stress in Saccharomyces cerevisiae, and its expression is regulated by the high-osmolarity glycerol response pathway. Mol. Cell Biol. 1994, 14, 4135–4144. Available online: http://www.ncbi.nlm.nih.gov/pubmed/8196651 (accessed on 24 January 2019).
- Klipp, E.; Nordlander, B.; Krüger, R.; Gennemark, P.; Hohmann, S. Integrative model of the response of yeast to osmotic shock. Nat. Biotechnol. 2005, 23, 975–982. Available online: https://pubmed.ncbi.nlm.nih.gov/16025103/ (accessed on 10 February 2023). [CrossRef]
- Muzzey, D.; Gómez-Uribe, C.A.; Mettetal, J.T.; van Oudenaarden, A. A systems-level analysis of perfect adaptation in yeast osmoregulation. Cell 2009, 138, 160–171. Available online: https://pubmed.ncbi.nlm.nih.gov/19596242/ (accessed on 1 June 2023). [CrossRef] [PubMed] [Green Version]
- Hao, N.; Behar, M.; Parnell, S.C.; Torres, M.P.; Borchers, C.H.; Elston, T.C.C.; Dohlman, H.G. A systems-biology analysis of feedback inhibition in the Sho1 osmotic-stress-response pathway. Curr. Biol. 2007, 17, 659–667. Available online: https://pubmed.ncbi.nlm.nih.gov/17363249/ (accessed on 10 February 2023). [CrossRef] [PubMed] [Green Version]
- Hao, N.; Zeng, Y.; Elston, T.C.; Dohlman, H.G. Control of MAPK specificity by feedback phosphorylation of shared adaptor protein Ste50. J. Biol. Chem. 2008, 283, 33798–33802. Available online: https://pubmed.ncbi.nlm.nih.gov/18854322/ (accessed on 1 June 2023). [CrossRef] [PubMed] [Green Version]
- Yamamoto, K.; Tatebayashi, K.; Tanaka, K.; Saito, H. Dynamic control of yeast MAP kinase network by induced association and dissociation between the Ste50 scaffold and the Opy2 membrane anchor. Mol. Cell 2010, 40, 87–98. Available online: https://pubmed.ncbi.nlm.nih.gov/20932477/ (accessed on 1 June 2023). [CrossRef] [PubMed] [Green Version]
- Molin, C.; Jauhiainen, A.; Warringer, J.; Nerman, O.; Sunnerhagen, P.E.R. mRNA stability changes precede changes in steady-state mRNA amounts during hyperosmotic stress. RNA 2009, 15, 600. Available online: http://www.rnajournal.org/cgi/doi/10.1261/rna.1403509 (accessed on 10 February 2023). [CrossRef] [Green Version]
- Romero-Santacreu, L.; Moreno, J.; Pérez-Ortín, J.E.; Alepuz, P. Specific and global regulation of mRNA stability during osmotic stress in Saccharomyces cerevisiae. RNA 2009, 15, 1110–1120. Available online: https://pubmed.ncbi.nlm.nih.gov/19369426/ (accessed on 10 February 2023). [CrossRef] [Green Version]
- Miller, C.; Schwalb, B.; Maier, K.; Schulz, D.; Dümcke, S.; Zacher, B.; Mayer, A.; Sydow, J.; Marcinowski, L. Dölken, L.; et al. Dynamic transcriptome analysis measures rates of mRNA synthesis and decay in yeast. Mol. Syst. Biol. 2011, 7, 458. Available online: https://pubmed.ncbi.nlm.nih.gov/21206491/ (accessed on 10 February 2023). [CrossRef]
- Alepuz, P.M.; De Nadal, E.; Zapater, M.; Ammerer, G.; Posas, F. Osmostress-induced transcription by Hot1 depends on a Hog1-mediated recruitment of the RNA Pol II. EMBO J. 2003, 22, 2433. [Google Scholar] [CrossRef] [Green Version]
- Proft, M.; Struhl, K. Hog1 kinase converts the Sko1-Cyc8-Tup1 repressor complex into an activator that recruits SAGA and SWI/SNF in response to osmotic stress. Mol. Cell 2002, 9, 1307–1317. Available online: https://pubmed.ncbi.nlm.nih.gov/12086627/ (accessed on 1 June 2023). [CrossRef]
- Proft, M.; Struhl, K. MAP kinase-mediated stress relief that precedes and regulates the timing of transcriptional induction. Cell 2004, 118, 351–361. Available online: https://pubmed.ncbi.nlm.nih.gov/15294160/ (accessed on 1 June 2023). [CrossRef] [PubMed] [Green Version]
- Proft, M.; Mas, G.; de Nadal, E.; Vendrell, A.; Noriega, N.; Struhl, K.; Posas, F. The stress-activated Hog1 kinase is a selective transcriptional elongation factor for genes responding to osmotic stress. Mol. Cell 2006, 23, 241–250. Available online: https://pubmed.ncbi.nlm.nih.gov/16857590/ (accessed on 1 June 2023). [CrossRef] [PubMed]
- Dihazi, H.; Kessler, R.; Eschrich, K. High osmolarity glycerol (HOG) pathway-induced phosphorylation and activation of 6-phosphofructo-2-kinase are essential for glycerol accumulation and yeast cell proliferation under hyperosmotic stress. J. Biol. Chem. 2004, 279, 23961–23968. Available online: http://www.ncbi.nlm.nih.gov/pubmed/15037628 (accessed on 24 January 2019). [CrossRef] [PubMed] [Green Version]
- Mollapour, M.; Piper, P.W. Hog1 mitogen-activated protein kinase phosphorylation targets the yeast Fps1 aquaglyceroporin for endocytosis, thereby rendering cells resistant to acetic acid. Mol. Cell Biol. 2007, 27, 6446–6456. Available online: https://pubmed.ncbi.nlm.nih.gov/17620418/ (accessed on 25 January 2023). [CrossRef] [Green Version]
- Westfall, P.J.; Patterson, J.C.; Chen, R.E.; Thorner, J. Stress resistance and signal fidelity independent of nuclear MAPK function. Proc. Natl. Acad. Sci. USA 2008, 105, 12212–12217. Available online: https://pubmed.ncbi.nlm.nih.gov/18719124/ (accessed on 10 February 2023). [CrossRef]
- Beese, S.E.; Negishi, T.; Levin, D.E. Identification of positive regulators of the yeast fps1 glycerol channel. PLoS Genet. 2009, 5, 11. Available online: https://pubmed.ncbi.nlm.nih.gov/19956799/ (accessed on 10 February 2023). [CrossRef] [Green Version]
- Bouwman, J.; Kiewiet, J.; Lindenbergh, A.; Van Eunen, K.; Siderius, M.; Bakker, B.M. Metabolic regulation rather than de novo enzyme synthesis dominates the osmo-adaptation of yeast. Yeast 2011, 28, 43–53. Available online: https://pubmed.ncbi.nlm.nih.gov/20803479/ (accessed on 10 February 2023). [CrossRef]
- Hernández-Elvira, M.; Martínez-Gómez, R.; Domínguez-Martin, E.; Méndez, A.; Kawasaki, L.; Ongay-Larios, L.; Coria, R. Tunicamycin Sensitivity-Suppression by High Gene Dosage Reveals New Functions of the Yeast Hog1 MAP Kinase. Cells 2019, 8, 710. Available online: https://pubmed.ncbi.nlm.nih.gov/31336877/ (accessed on 31 May 2023). [CrossRef] [Green Version]
- Wagner, E.R.; Myers, K.S.; Riley, N.M.; Coon, J.J.; Gasch, A.P. PKA and HOG signaling contribute separable roles to anaerobic xylose fermentation in yeast engineered for biofuel production. PLoS ONE 2019, 14, e0212389. Available online: https://journals.plos.org/plosone/article?id=10.1371/journal.pone.0212389 (accessed on 15 December 2022). [CrossRef] [Green Version]
- Klein, C.; Struhl, K. Protein Kinase A Mediates Growth-Regulated Expression of Yeast Ribosomal Protein Genes by Modulating RAP1 Transcriptional Activity. Mol. Cell. Biol. 1994, 14, 1920–1928. [Google Scholar] [CrossRef] [PubMed]
- de la Cruz, J.; Gómez-Herreros, F.; Rodríguez-Galán, O.; Begley, V.; de la Cruz Muñoz-Centeno, M.; Chávez, S. Feedback regulation of ribosome assembly. Curr. Genet. 2018, 64, 393–404. Available online: https://pubmed.ncbi.nlm.nih.gov/29022131/ (accessed on 1 June 2023). [CrossRef]
- Jorgensen, P.; Rupeš, I.; Sharom, J.R.; Schneper, L.; Broach, J.R.; Tyers, M. A dynamic transcriptional network communicates growth potential to ribosome synthesis and critical cell size. Genes. Dev. 2004, 18, 2491–2505. Available online: http://genesdev.cshlp.org/content/18/20/2491.full (accessed on 25 June 2020). [CrossRef] [PubMed] [Green Version]
- Neuman-Silberberg, F.S.; Bhattacharya, S.; Broach, J.R. Nutrient availability and the RAS/cyclic AMP pathway both induce expression of ribosomal protein genes in Saccharomyces cerevisiae but by different mechanisms. Mol. Cell Biol. 1995, 15, 3187–3196. Available online: http://www.ncbi.nlm.nih.gov/pubmed/7760815 (accessed on 24 January 2019). [CrossRef] [PubMed]
- Zurita-Martinez, S.A.; Cardenas, M.E. Tor and cyclic AMP-protein kinase A: Two parallel pathways regulating expression of genes required for cell growth. Eukaryot. Cell 2005, 4, 63–71. Available online: https://pubmed.ncbi.nlm.nih.gov/15643061/ (accessed on 10 February 2023). [CrossRef] [PubMed] [Green Version]
- Jorgensen, P.; Nishikawa, J.L.; Breitkreutz, B.J.; Tyers, M. Systematic identification of pathways that couple cell growth and division in yeast. Science 2002, 297, 395–400. Available online: https://science.sciencemag.org/content/297/5580/395 (accessed on 2 July 2020). [CrossRef] [PubMed]
- Wang, Y.; Pierce, M.; Schneper, L.; Güldal, C.G.; Zhang, X.; Tavazoie, S.; Broach, J.R. Ras and Gpa2 mediate one branch of a redundant glucose signaling pathway in yeast. PLoS Biol. 2004, 2, 610–622. Available online: https://pubmed.ncbi.nlm.nih.gov/15138498/ (accessed on 10 February 2023). [CrossRef]
- Fingerman, I.; Nagaraj, V.; Norris, D.; Vershon, A.K. Sfp1 plays a key role in yeast ribosome biogenesis. Eukaryot. Cell 2003, 2, 1061–1068. Available online: https://pubmed.ncbi.nlm.nih.gov/14555489/ (accessed on 10 February 2023). [CrossRef] [Green Version]
- Marion, R.M.; Regev, A.; Segal, E.; Barash, Y.; Koller, D.; Friedman, N.; O’Shea, E.K. Sfp1 is a stress- and nutrient-sensitive regulator of ribosomal protein gene expression. Proc. Natl. Acad. Sci. USA 2004, 101, 14315–14322. Available online: https://pubmed.ncbi.nlm.nih.gov/15353587/ (accessed on 10 February 2023). [CrossRef]
- Pessina, S.; Tsiarentsyeva, V.; Busnelli, S.; Vanoni, M.; Alberghina, L.; Coccetti, P. Snf1/AMPK promotes S-phase entrance by controlling CLB5 transcription in budding yeast. Cell Cycle 2010, 9, 2189–2200. Available online: https://pubmed.ncbi.nlm.nih.gov/20505334/ (accessed on 10 February 2023). [CrossRef] [Green Version]
- Koch, C.; Moll, T.; Neuberg, M.; Ahorn, H.; Nasmyth, K. A role for the transcription factors Mbp1 and Swi4 in progression from G1 to S phase. Science 1993, 261, 1551–1557. Available online: https://pubmed.ncbi.nlm.nih.gov/8372350/ (accessed on 10 February 2023). [CrossRef]
- Busnelli, S.; Tripodi, F.; Nicastro, R.; Cirulli, C.; Tedeschi, G.; Pagliarin, R.; Alberghina, L.; Coccetti, P. Snf1/AMPK promotes SBF and MBF-dependent transcription in budding yeast. Biochim. Biophys. Acta 2013, 1833, 3254–3264. Available online: https://pubmed.ncbi.nlm.nih.gov/24084603/ (accessed on 10 February 2023). [CrossRef] [PubMed] [Green Version]
- Tripodi, F.; Fraschini, R.; Zocchi, M.; Reghellin, V.; Coccetti, P. Snf1/AMPK is involved in the mitotic spindle alignment in Saccharomyces cerevisiae. Sci. Rep. 2018, 8, 5853. Available online: http://www.nature.com/articles/s41598-018-24252-y (accessed on 3 June 2019). [CrossRef] [Green Version]
- Moore, J.K.; Cooper, J.A. Coordinating mitosis with cell polarity: Molecular motors at the cell cortex. Semin. Cell Dev. Biol. 2010, 21, 283–289. Available online: https://pubmed.ncbi.nlm.nih.gov/20109571/ (accessed on 10 February 2023). [CrossRef] [Green Version]
- Vendrell, A.; Martánez-Pastor, M.; González-Novo, A.; Pascual-Ahuir, A.; Sinclair, D.A.; Proft, M.; Posas, F. Sir2 histone deacetylase prevents programmed cell death caused by sustained activation of the Hog1 stress-activated protein kinase. EMBO Rep. 2011, 12, 1062. [Google Scholar] [CrossRef] [PubMed]
- Maeda, T.; Tsai, A.Y.; Saito, H. Mutations in a protein tyrosine phosphatase gene (PTP2) and a protein serine/threonine phosphatase gene (PTC1) cause a synthetic growth defect in Saccharomyces cerevisiae. Mol. Cell Biol. 1993, 13, 5408–5417. Available online: https://pubmed.ncbi.nlm.nih.gov/8395005/ (accessed on 19 February 2023).
- Escoté, X.; Zapater, M.; Clotet, J.; Posas, F. Hog1 mediates cell-cycle arrest in G1 phase by the dual targeting of Sic1. Nat. Cell Biol. 2004, 6, 997–1002. Available online: https://pubmed.ncbi.nlm.nih.gov/15448699/ (accessed on 19 February 2023). [CrossRef]
- Adrover, M.À.; Zi, Z.; Duch, A.; Schaber, J.; González-Novo, A.; Jimenez, J.; Nadal-Ribelles, M.; Clotet, J.; Klipp, E.; Posas, F. Time-dependent quantitative multicomponent control of the G1-S network by the stress-activated protein kinase Hog1 upon osmostress. Sci. Signal 2011, 4, 192. Available online: https://pubmed.ncbi.nlm.nih.gov/21954289/ (accessed on 19 February 2023). [CrossRef] [PubMed] [Green Version]
- González-Novo, A.; Jiménez, J.; Clotet, J.; Nadal-Ribelles, M.; Cavero, S.; de Nadal, E.; Posas, F. Hog1 Targets Whi5 and Msa1 Transcription Factors To Downregulate Cyclin Expression upon Stress. Mol. Cell Biol. 2015, 35, 1606. [Google Scholar] [CrossRef] [Green Version]
- Yaakov, G.; Duch, A.; García-Rubio, M.; Clotet, J.; Jimenez, J.; Aguilera, A.; Posas, F. The stress-activated protein kinase Hog1 mediates S phase delay in response to osmostress. Mol. Biol. Cell 2009, 20, 3572–3582. Available online: https://pubmed.ncbi.nlm.nih.gov/19477922/ (accessed on 19 February 2023). [CrossRef] [Green Version]
- Duch, A.; Canal, B.; Barroso, S.I.; García-Rubio, M.; Seisenbacher, G.; Aguilera, A.; de Nadal, E.; Posas, F. Multiple signaling kinases target Mrc1 to prevent genomic instability triggered by transcription-replication conflicts. Nat. Commun. 2018, 9, 379. Available online: https://www.nature.com/articles/s41467-017-02756-x (accessed on 19 February 2023). [CrossRef] [Green Version]
- Duch, A.; Felipe-Abrio, I.; Barroso, S.; Yaakov, G.; García-Rubio, M.; Aguilera, A.; de Nadal, E.; Posas, F. Coordinated control of replication and transcription by a SAPK protects genomic integrity. Nature 2013, 493, 116–121. Available online: https://pubmed.ncbi.nlm.nih.gov/23178807/ (accessed on 19 February 2023). [CrossRef]
- Alexander, M.R.; Tyers, M.; Perret, M.; Craig, B.M.; Fang, K.S.; Gustin, M.C. Regulation of Cell Cycle Progression by Swe1p and Hog1p Following Hypertonic Stress. Mol. Biol. Cell 2001, 12, 53. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Clotet, J.; Escoté, X.; Adrover, M.Á.; Yaakov, G.; Garí, E.; Aldea, M.; de Nadal, E.; Posas, F. Phosphorylation of Hsl1 by Hog1 leads to a G2 arrest essential for cell survival at high osmolarity. EMBO J. 2006, 25, 2338. [Google Scholar] [CrossRef]
- Jiménez, J.; Queralt, E.; Posas, F.; de Nadal, E. The regulation of Net1/Cdc14 by the Hog1 MAPK upon osmostress unravels a new mechanism regulating mitosis. Cell Cycle 2020, 19, 2105. [Google Scholar] [CrossRef]
- Tognetti, S.; Jiménez, J.; Viganò, M.; Duch, A.; Queralt, E.; de Nadal, E.; Posas, F. Hog1 activation delays mitotic exit via phosphorylation of Net1. Proc. Natl. Acad. Sci. USA 2020, 117, 8924–8933. Available online: https://pubmed.ncbi.nlm.nih.gov/32265285/ (accessed on 19 February 2023). [CrossRef] [Green Version]



Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wagner, E.R.; Gasch, A.P. Advances in S. cerevisiae Engineering for Xylose Fermentation and Biofuel Production: Balancing Growth, Metabolism, and Defense. J. Fungi 2023, 9, 786. https://doi.org/10.3390/jof9080786
Wagner ER, Gasch AP. Advances in S. cerevisiae Engineering for Xylose Fermentation and Biofuel Production: Balancing Growth, Metabolism, and Defense. Journal of Fungi. 2023; 9(8):786. https://doi.org/10.3390/jof9080786
Chicago/Turabian StyleWagner, Ellen R., and Audrey P. Gasch. 2023. "Advances in S. cerevisiae Engineering for Xylose Fermentation and Biofuel Production: Balancing Growth, Metabolism, and Defense" Journal of Fungi 9, no. 8: 786. https://doi.org/10.3390/jof9080786




