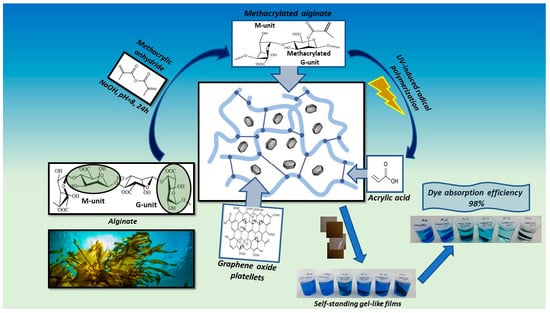
Graphene-Enhanced Methacrylated Alginate Gel Films for Sustainable Dye Removal in Water Purification
Abstract
:1. Introduction
2. Results and Discussion
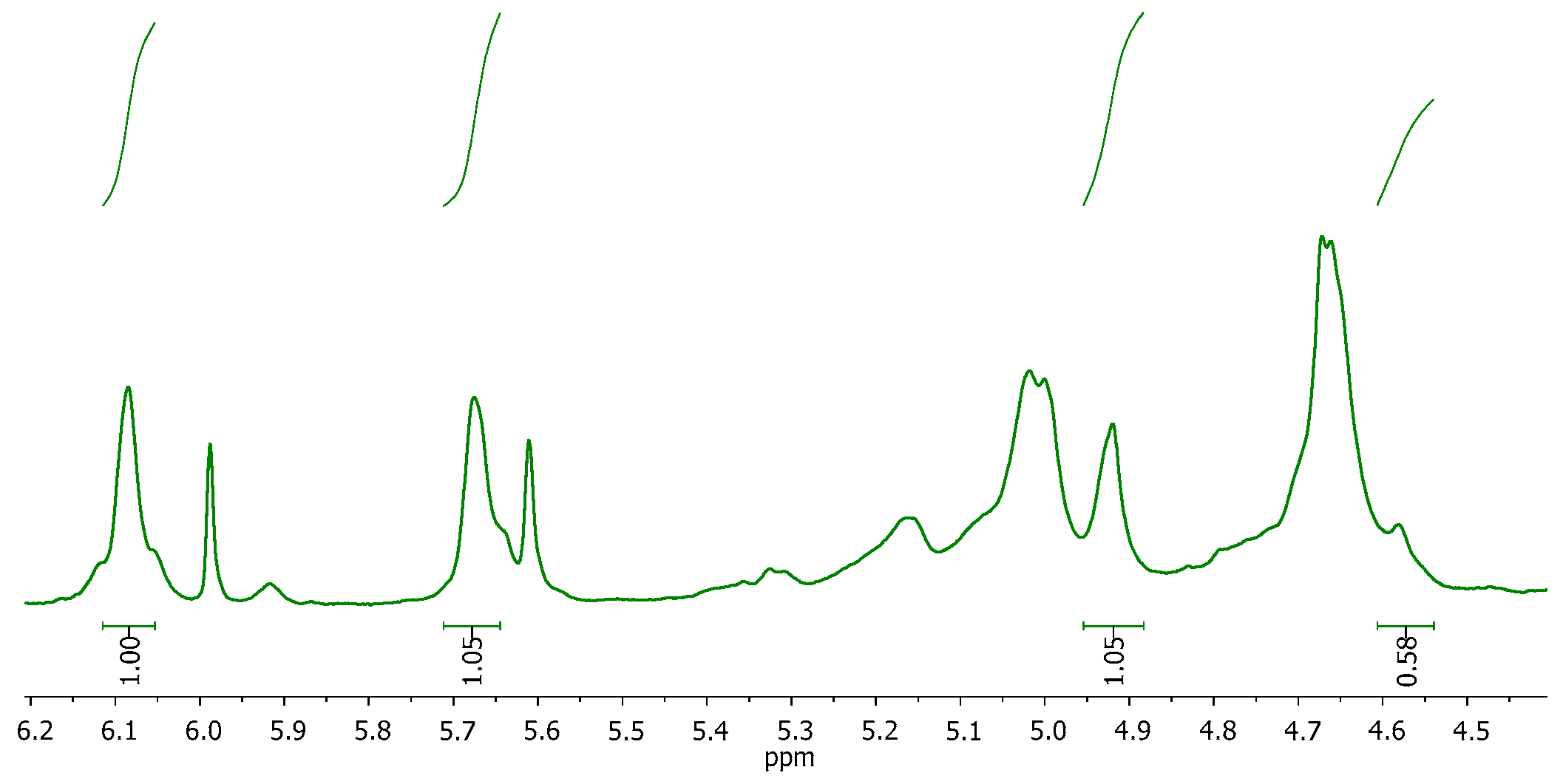
2.1. Synthesis and Characterization of Alginate and Its Methacrylated Derivative
2.2. Contact Angle Measurements
2.3. Mechanical Properties of MALG/AA and MALG/AA/GO Films
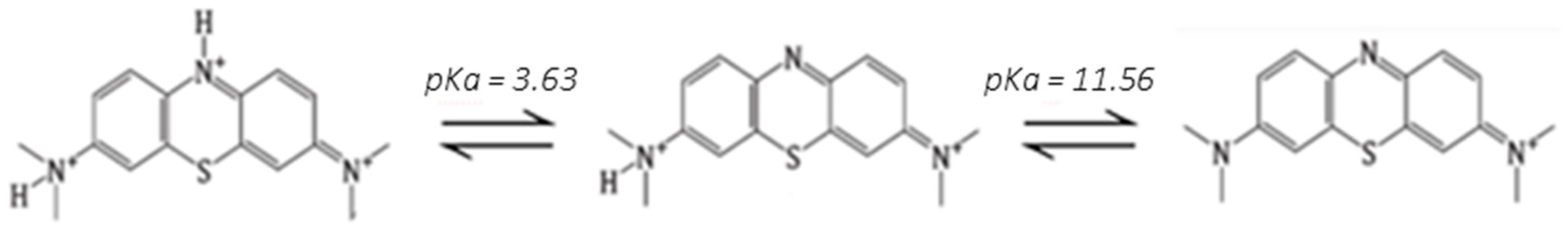
2.4. Dye Adsorption Onto MALG/AA and MALG/AA/GO Films
2.5. MALG/AA/GO Films Reusability
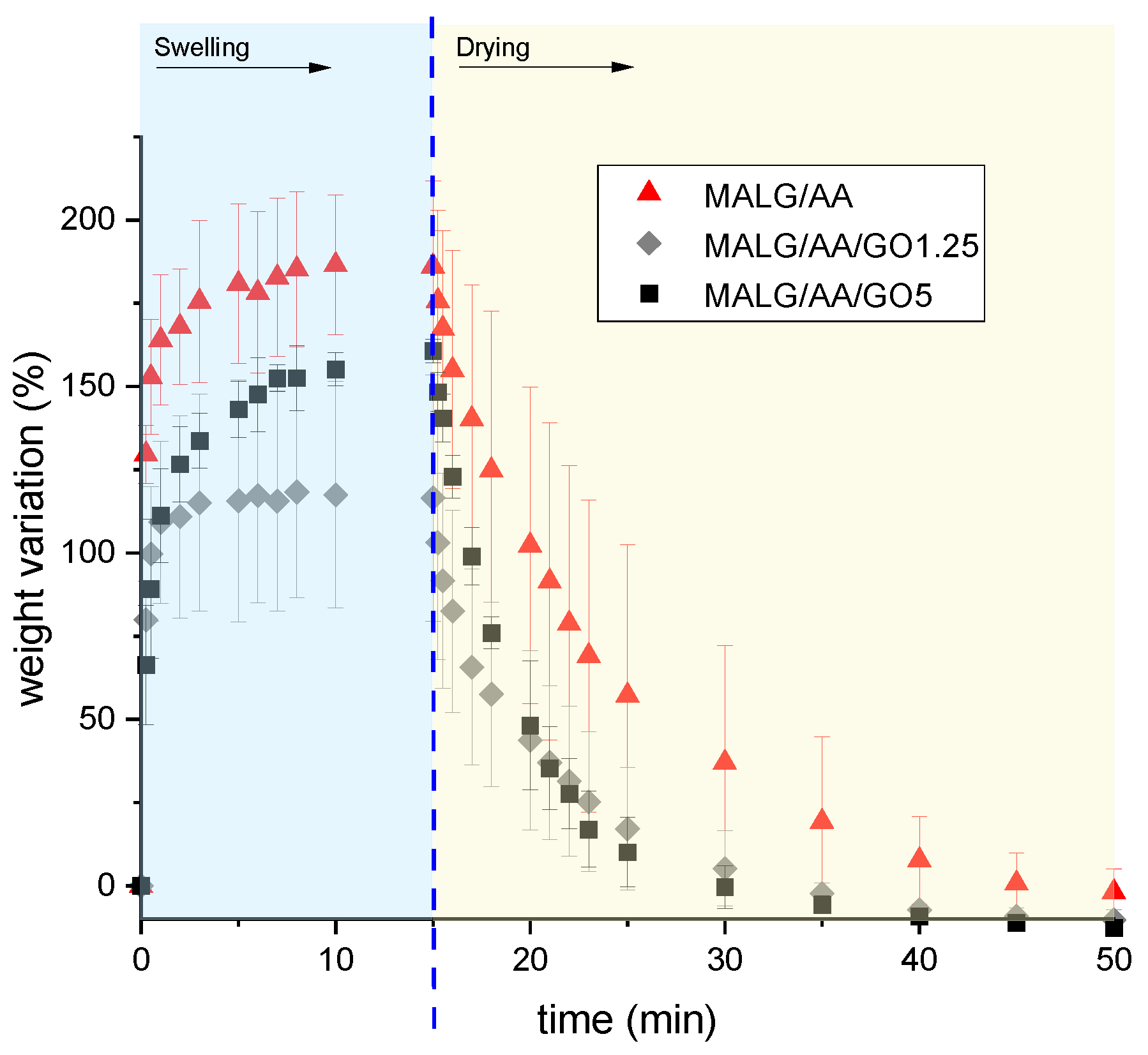
2.5.1. Swelling–Drying Tests of the Prepared MALG Films
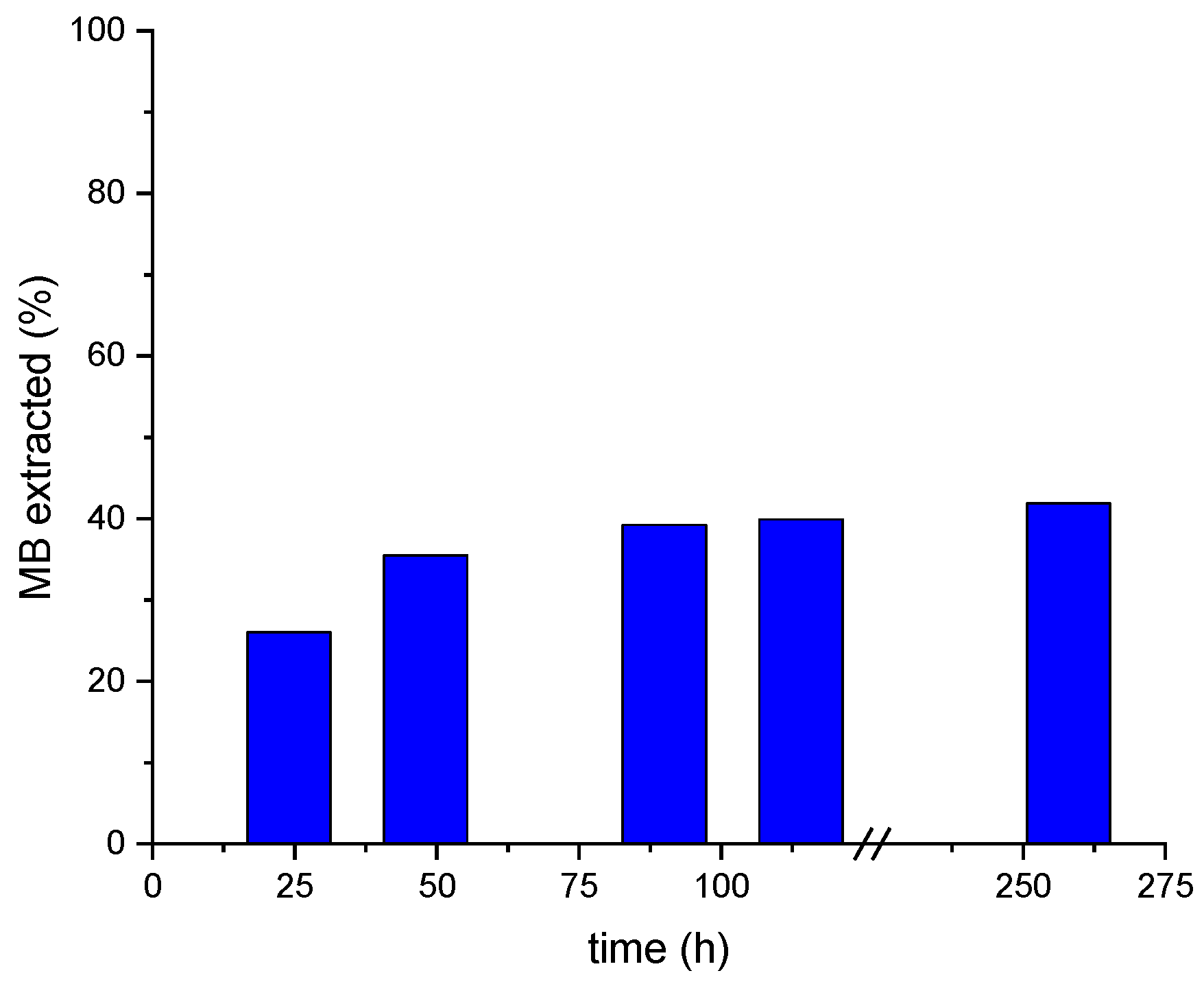
2.5.2. Absorbed MB Extraction from MALG Films in Acidic Conditions
2.6. Additional Nanocomposite MALG/AA/GO Films Characterizations
3. Conclusions
4. Materials and Methods
4.1. Materials and Products
4.2. Methacrylation of Polysaccharides
4.3. Films Preparation
4.4. Alginate and Methacrylated Alginate Characterization
4.5. Water Contact Angle Measurements
4.6. Dynamic Mechanical Analysis
4.7. Dye Adsorption Experiment
4.8. MALG/AA Films Reutilization
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Costa, J.A.V.; Lucas, B.F.; Alvarenga, A.G.P.; Moreira, J.B.; de Morais, M.G. Microalgae Polysaccharides: An Overview of Production, Characterization, and Potential Applications. Polysaccharides 2021, 2, 759–772. [Google Scholar] [CrossRef]
- Bilal, M.; Gul, I.; Basharat, A.; Qamar, S.A. Polysaccharides-Based Bio-Nanostructures and Their Potential Food Applications. Int. J. Biol. Macromol. 2021, 176, 540–557. [Google Scholar] [CrossRef] [PubMed]
- Yao, Y.; Xu, B. Skin Health Promoting Effects of Natural Polysaccharides and Their Potential Application in the Cosmetic Industry. Polysaccharides 2022, 3, 818–830. [Google Scholar] [CrossRef]
- Venkatesan, J.; Nithya, R.; Sudha, P.N.; Kim, S.K. Role of Alginate in Bone Tissue Engineering, 1st ed.; Elsevier Inc.: Amsterdam, The Netherlands, 2014; Volume 73, ISBN 9780128002681. [Google Scholar]
- Enobakhare, B.; Bader, D.I.; Lee, D. Concentration and M/G Ratio Influence the Physiochemical and Mechanical Properties of Alginate Constructs for Tissue Engineering. J. Appl. Biomater. Biomech. 2006, 4, 87–96. [Google Scholar] [CrossRef] [PubMed]
- Ramos, P.E.; Silva, P.; Alario, M.M.; Pastrana, L.M.; Teixeira, J.A.; Cerqueira, M.A.; Vicente, A.A. Effect of Alginate Molecular Weight and M/G Ratio in Beads Properties Foreseeing the Protection of Probiotics. Food Hydrocoll. 2018, 77, 8–16. [Google Scholar] [CrossRef]
- Bausch, B.; Frankl, S.; Becher, D.; Menz, F.; Baier, T.; Bauer, M.; Böse, O.; Hölzle, M. Naturally-Derived Thermal Barrier Based on Fiber-Reinforced Hydrogel for the Prevention of Thermal Runaway Propagation in High-Energetic Lithium-Ion Battery Packs. J. Energy Storage 2023, 61, 106841. [Google Scholar] [CrossRef]
- Thomas, D.; Mathew, N.; Nath, M.S. Starch Modified Alginate Nanoparticles for Drug Delivery Application. Int. J. Biol. Macromol. 2021, 173, 277–284. [Google Scholar] [CrossRef]
- Guo, H.; Qin, Q.; Chang, J.S.; Lee, D.J. Modified Alginate Materials for Wastewater Treatment: Application Prospects. Bioresour. Technol. 2023, 387, 129639. [Google Scholar] [CrossRef]
- Zhang, M.K.; Zhang, X.H.; Han, G.Z. Magnetic Alginate/PVA Hydrogel Microspheres with Selective Adsorption Performance for Aromatic Compounds. Sep. Purif. Technol. 2022, 278, 119547. [Google Scholar] [CrossRef]
- Pandey, S.; Ramontja, J. Natural Bentonite Clay and Its Composites for Dye Removal: Current State and Future Potential. Am. J. Chem. Appl. 2016, 3, 8–19. [Google Scholar]
- Makhado, E.; Pandey, S.; Modibane, K.D.; Kang, M.; Hato, M.J. Sequestration of Methylene Blue Dye Using Sodium Alginate Poly(Acrylic Acid)@ZnO Hydrogel Nanocomposite: Kinetic, Isotherm, and Thermodynamic Investigations. Int. J. Biol. Macromol. 2020, 162, 60–73. [Google Scholar] [CrossRef] [PubMed]
- Zhang, G.; Yi, L.; Deng, H.; Sun, P. Dyes Adsorption Using a Synthetic Carboxymethyl Cellulose-Acrylic Acid Adsorbent. J. Environ. Sci. 2014, 26, 1203–1211. [Google Scholar] [CrossRef] [PubMed]
- Gong, X.L.; Lu, H.Q.; Li, K.; Li, W. Effective Adsorption of Crystal Violet Dye on Sugarcane Bagasse–Bentonite/Sodium Alginate Composite Aerogel: Characterisation, Experiments, and Advanced Modelling. Sep. Purif. Technol. 2022, 286, 120478. [Google Scholar] [CrossRef]
- ALSamman, M.T.; Sánchez, J. Recent Advances on Hydrogels Based on Chitosan and Alginate for the Adsorption of Dyes and Metal Ions from Water. Arab. J. Chem. 2021, 14, 103455. [Google Scholar] [CrossRef]
- Santander, P.; Butter, B.; Oyarce, E.; Yáñez, M.; Xiao, L.P.; Sánchez, J. Lignin-Based Adsorbent Materials for Metal Ion Removal from Wastewater: A Review. Ind. Crops Prod. 2021, 167, 113510. [Google Scholar] [CrossRef]
- Singh, V.P.; Sharma, M.; Vaish, R. Enhanced Dye Adsorption and Rapid Photo Catalysis in Candle Soot Coated Bi2WO6 ceramics. Eng. Res. Express 2019, 1, 025056. [Google Scholar] [CrossRef]
- Singh, V.P.; Sharma, M.; Vaish, R. Enhanced Dye Adsorption and Rapid Photocatalysis of Candle Soot Coated BaTiO3 Ceramics. Mater. Chem. Phys. 2020, 252, 123311. [Google Scholar] [CrossRef]
- Yadav, J.; Sahu, O. Dye Removal of Cationic Dye from Aqueous Solution through Acid Functionalized Ceramic. Total Environ. Res. Themes 2023, 6, 100038. [Google Scholar] [CrossRef]
- Al-Shemy, M.T.; Al-Sayed, A.; Dacrory, S. Fabrication of Sodium Alginate/Graphene Oxide/Nanocrystalline Cellulose Scaffold for Methylene Blue Adsorption: Kinetics and Thermodynamics Study. Sep. Purif. Technol. 2022, 290, 120825. [Google Scholar] [CrossRef]
- ALSamman, M.M.; Sánchez, J. Chitosan- and Alginate-Based Hydrogels for the Adsorption of Anionic and Cationic Dyes from Water. Polymers 2022, 14, 1498. [Google Scholar] [CrossRef]
- Chen, J.H.; Li, G.P.; Liu, Q.L.; Ni, J.C.; Wu, W.B.; Lin, J.M. Cr(III) Ionic Imprinted Polyvinyl Alcohol/Sodium Alginate (PVA/SA) Porous Composite Membranes for Selective Adsorption of Cr(III) Ions. Chem. Eng. J. 2010, 165, 465–473. [Google Scholar] [CrossRef]
- Azizi-Lalabadi, M.; Jafari, S.M. Bio-Nanocomposites of Graphene with Biopolymers; Fabrication, Properties, and Applications. Adv. Colloid Interface Sci. 2021, 292, 102416. [Google Scholar] [CrossRef] [PubMed]
- Rocha, L.S.; Nogueira, J.; Daniel-Da-Silva, A.L.; Marques, P.; Fateixa, S.; Pereira, E.; Trindade, T. Water Softening Using Graphene Oxide/Biopolymer Hybrid Nanomaterials. J. Environ. Chem. Eng. 2021, 9, 105045. [Google Scholar] [CrossRef]
- Max Jensen, H.; Hofmann Larsen, F.; Balling Engelsen, S. Characterization of Alginates by Nuclear Magnetic Resonance (NMR) and Vibrational Spectroscopy (IR, NIR, Raman) in Combination with Chemometrics. In Natural Products From Marine Algae: Methods and Protocols; Stengel, D.B., Conan, S., Eds.; Springer Science+Business Media: New York, NY, USA, 2015; Volume 1308, pp. 1–439. ISBN 9781493926848. [Google Scholar]
- Nissanka, B.; Kottegoda, N.; Jayasundara, D.R. Probing Structural Variations of Graphene Oxide and Reduced Graphene Oxide Using Methylene Blue Adsorption Method. J. Mater. Sci. 2020, 55, 1996–2005. [Google Scholar] [CrossRef]
- Qian, L.W.; Yang, M.X.; Zhang, S.F.; Hou, C.; Song, W.Q.; Yang, J.F.; Tang, R.H. Preparation of a Sustainable Bioadsorbent by Modifying Filter Paper with Sodium Alginate, with Enhanced Mechanical Properties and Good Adsorption of Methylene Blue from Wastewaters. Cellulose 2018, 25, 2021–2036. [Google Scholar] [CrossRef]
- Ajeel, S.J.; Beddai, A.A.; Almohaisen, A.M.N. Preparation of Alginate/Graphene Oxide Composite for Methylene Blue Removal. Mater. Today Proc. 2021, 51, 289–297. [Google Scholar] [CrossRef]
- Thakur, S.; Pandey, S.; Arotiba, O.A. Development of a Sodium Alginate-Based Organic/Inorganic Superabsorbent Composite Hydrogel for Adsorption of Methylene Blue. Carbohydr. Polym. 2016, 153, 34–46. [Google Scholar] [CrossRef]
- Nishikiori, H.; Nagaya, S.; Tanaka, N.; Katsuki, A.; Fujii, T. Acid-Base and Monomer-Dimer Equilibria of Methylene Blue in Dip-Coated Thin Films. Bull. Chem. Soc. Jpn. 1999, 72, 915–921. [Google Scholar] [CrossRef]
- Othman, I.; Abu Haija, M.; Kannan, P.; Banat, F. Adsorptive Removal of Methylene Blue from Water Using High-Performance Alginate-Based Beads. Water. Air. Soil Pollut. 2020, 231, 396. [Google Scholar] [CrossRef]
- Fang, Y.; Liu, Q.; Zhu, S. Selective Biosorption Mechanism of Methylene Blue by a Novel and Reusable Sugar Beet Pulp Cellulose/Sodium Alginate/Iron Hydroxide Composite Hydrogel. Int. J. Biol. Macromol. 2021, 188, 993–1002. [Google Scholar] [CrossRef]







| Film Formulation | E* (MPa) at 25 °C | E* (MPa) at 100 °C |
|---|---|---|
| MALG/AA | 676.6 | 886.9 |
| MALG/AA/GO1.25 | 1178.2 | 1305.3 |
| MALG/AA/GO5 | 1538.7 | 2066.6 |
| Composite | Mechanical Properties | Adsorption Yield (%) | Ref. |
|---|---|---|---|
| Alginate/Ca2+/filter paper | Elastic modulus 773 (1 wt.% ALG) MPa 1907 (4 wt.% ALGSA) MPa | 93.9 | [27] |
| Alginate/Ca2+/graphene oxide | Not provided | 91.3 | [28] |
| Alginate/poly(acrylic acid)/TiO2 NPs | Storage modulus (G′) 971 MPa | 80 (no TiO2NPs) 94.1 (0.05 g TiO2NPs) 99.4 (0.2 g TiO2NPs) | [29] |
| Methacrylated Alginate/poly(acrylic acid)/graphene oxide | Storage modulus (E′) 677 (no GO) MPa 1158 (1.25 wt.% GO) MPa 1539 (5 wt.% GO) MPa | 88 (no GO) 90 (1.25 wt.% GO) 97 (5 wt.% GO) | Current work |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Teijido, R.; Zhang, Q.; Blanco, M.; Pérez-Álvarez, L.; Lanceros-Méndez, S.; Vilas-Vilela, J.L.; Ruiz-Rubio, L. Graphene-Enhanced Methacrylated Alginate Gel Films for Sustainable Dye Removal in Water Purification. Gels 2024, 10, 25. https://doi.org/10.3390/gels10010025
Teijido R, Zhang Q, Blanco M, Pérez-Álvarez L, Lanceros-Méndez S, Vilas-Vilela JL, Ruiz-Rubio L. Graphene-Enhanced Methacrylated Alginate Gel Films for Sustainable Dye Removal in Water Purification. Gels. 2024; 10(1):25. https://doi.org/10.3390/gels10010025
Chicago/Turabian StyleTeijido, Rubén, Qi Zhang, Miren Blanco, Leyre Pérez-Álvarez, Senentxu Lanceros-Méndez, José Luis Vilas-Vilela, and Leire Ruiz-Rubio. 2024. "Graphene-Enhanced Methacrylated Alginate Gel Films for Sustainable Dye Removal in Water Purification" Gels 10, no. 1: 25. https://doi.org/10.3390/gels10010025