Tuning the Interfacial Deformation of Gliadin-Flaxseed Gum Complex Particles for Improving the Foam Stability
Abstract
1. Introduction
2. Results and Discussion
2.1. Structural Properties of Gliadin-Flaxseed Gum Particles
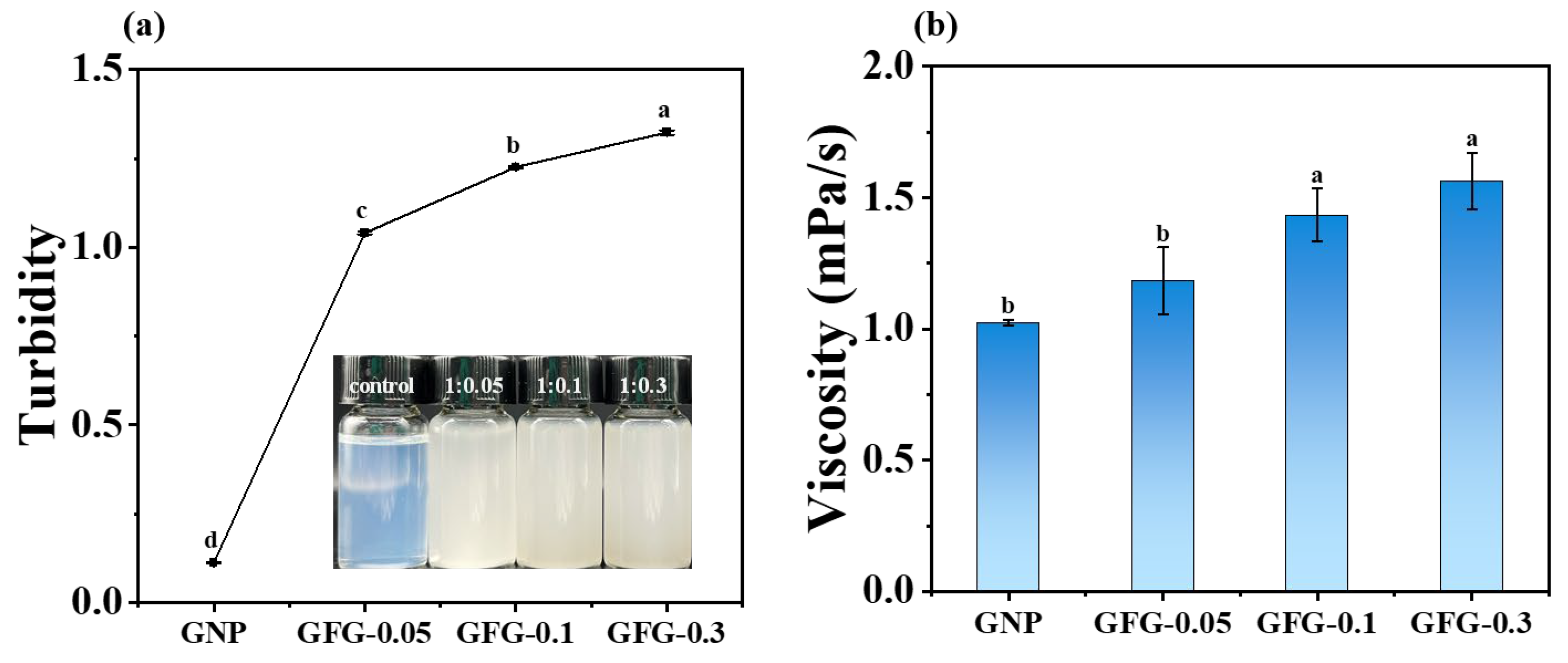
2.1.1. Appearance, Turbidity, and Viscosity
2.1.2. Particle Size, Zeta Potential, and Morphology
2.1.3. Flexibility and Surface Wettability
2.2. Interfacial Properties of Gliadin-Flaxseed Gum Particles
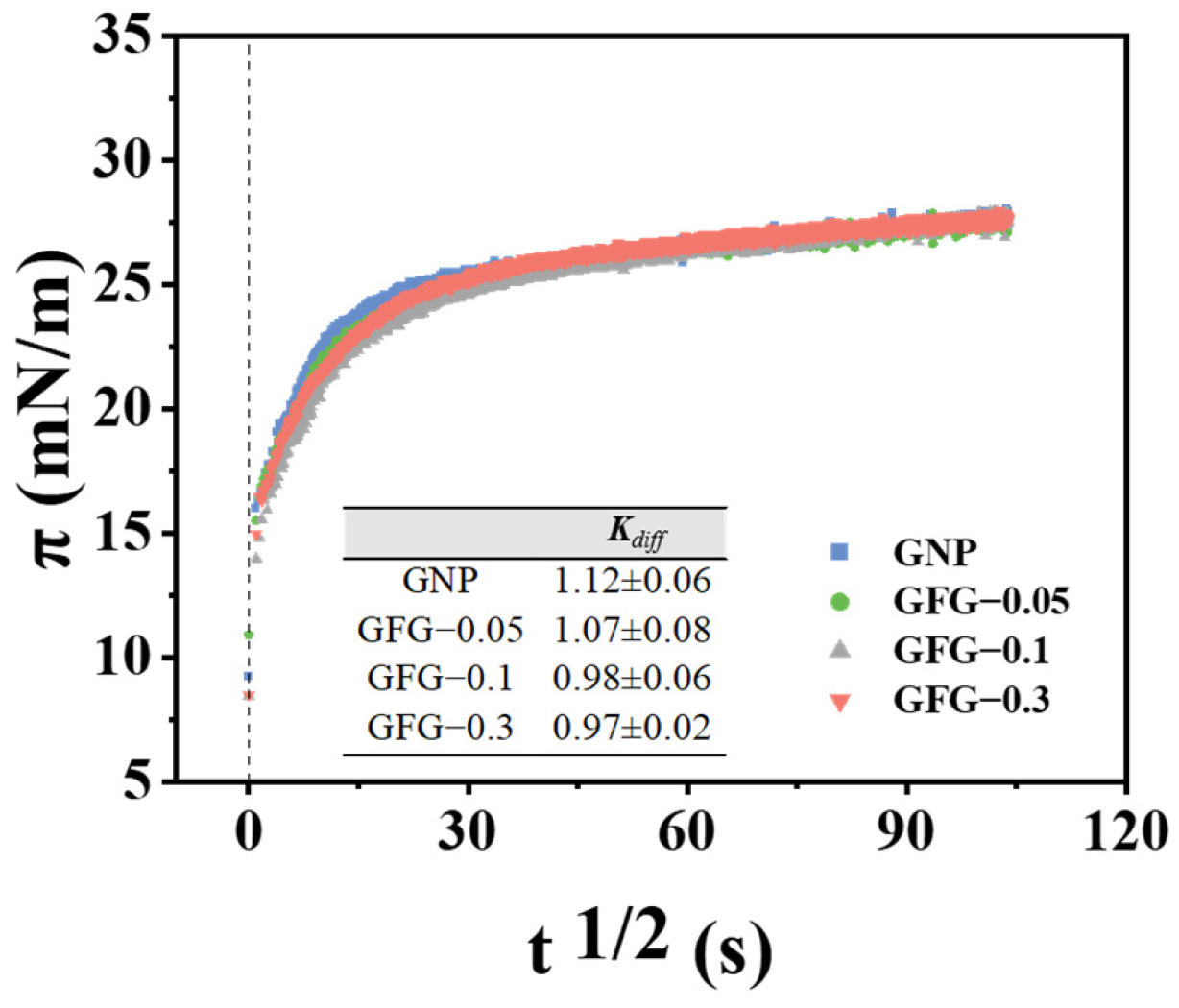
2.2.1. Interfacial Adsorption Kinetics
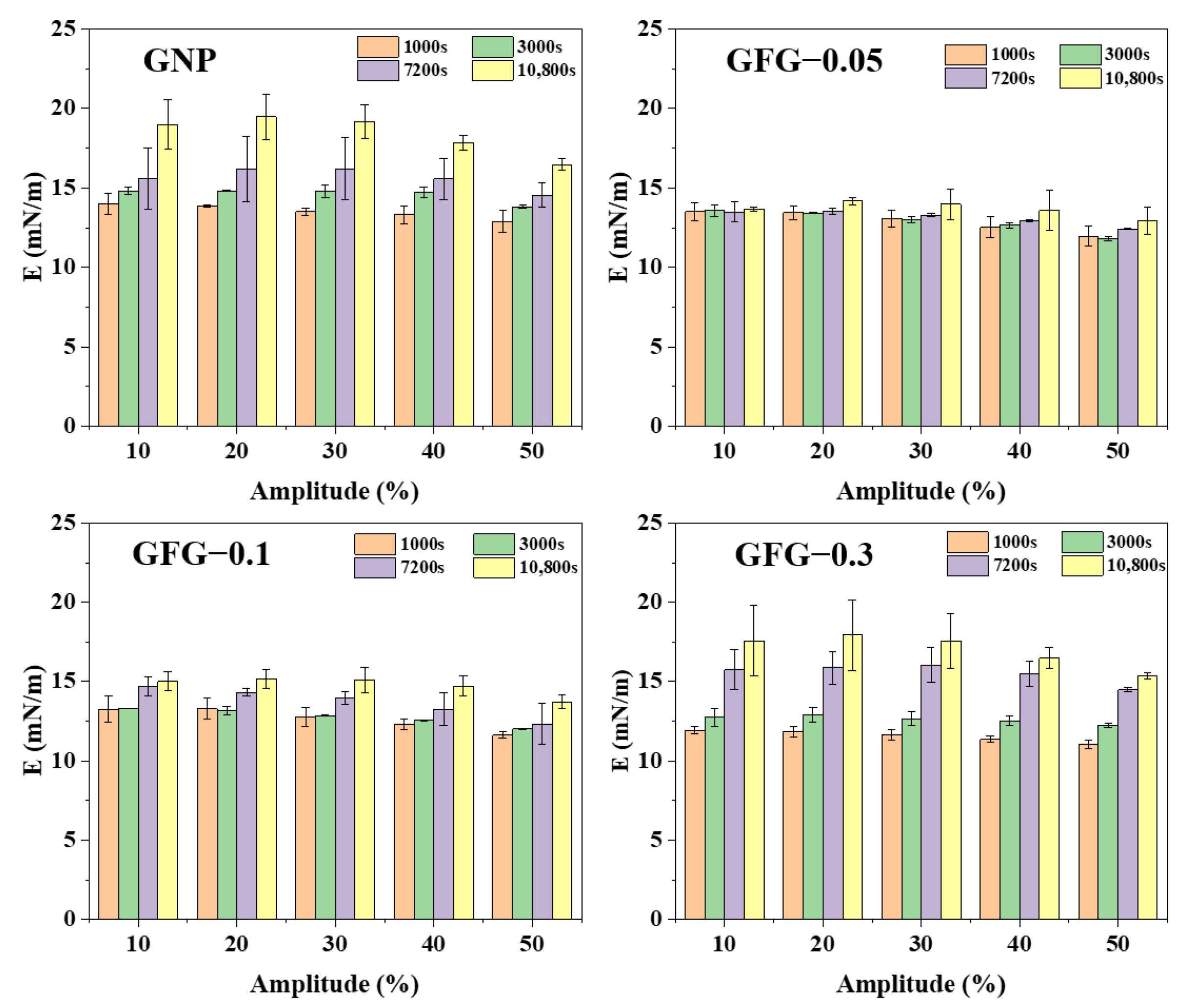
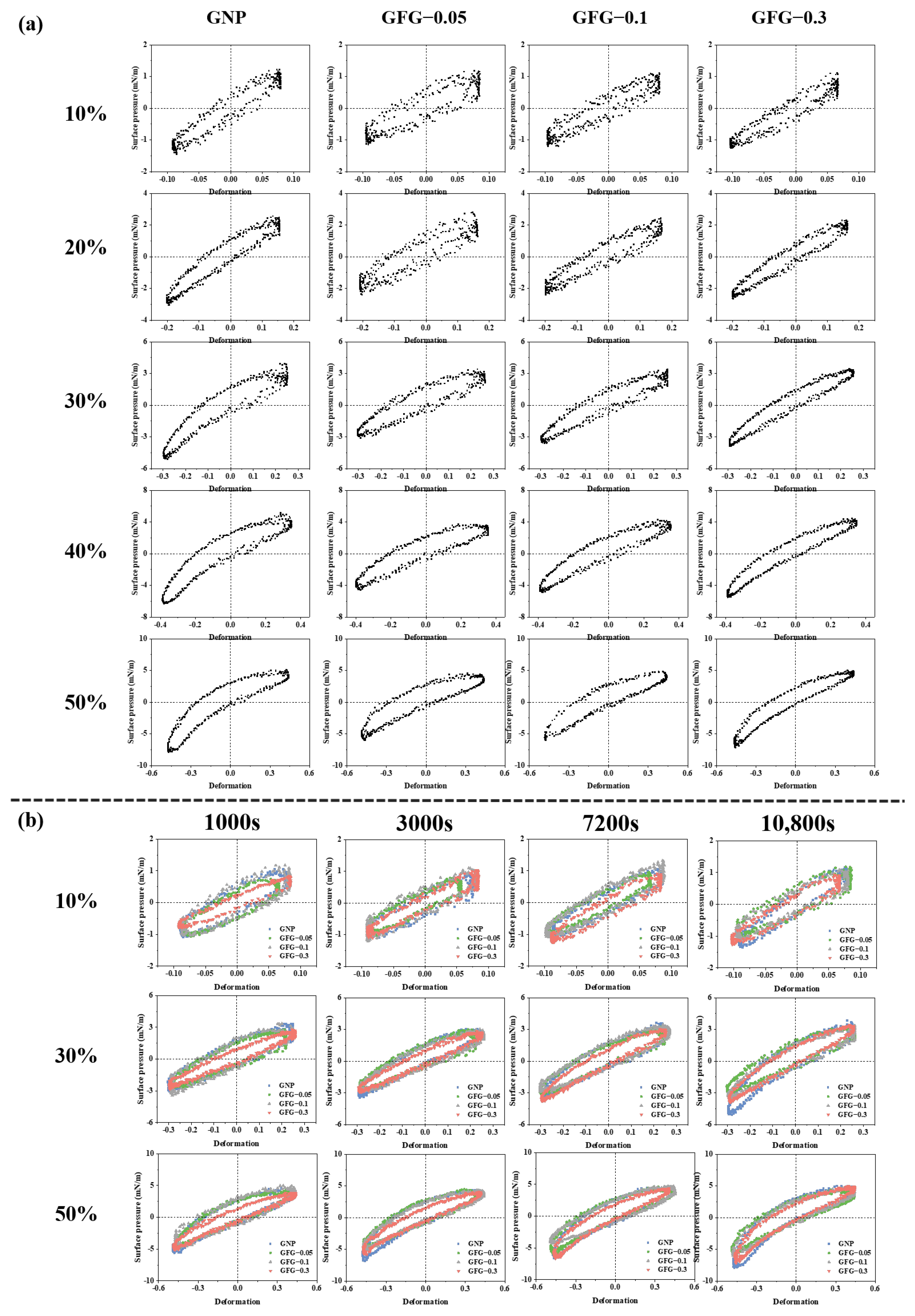
2.2.2. Interfacial Dilatational Rheology
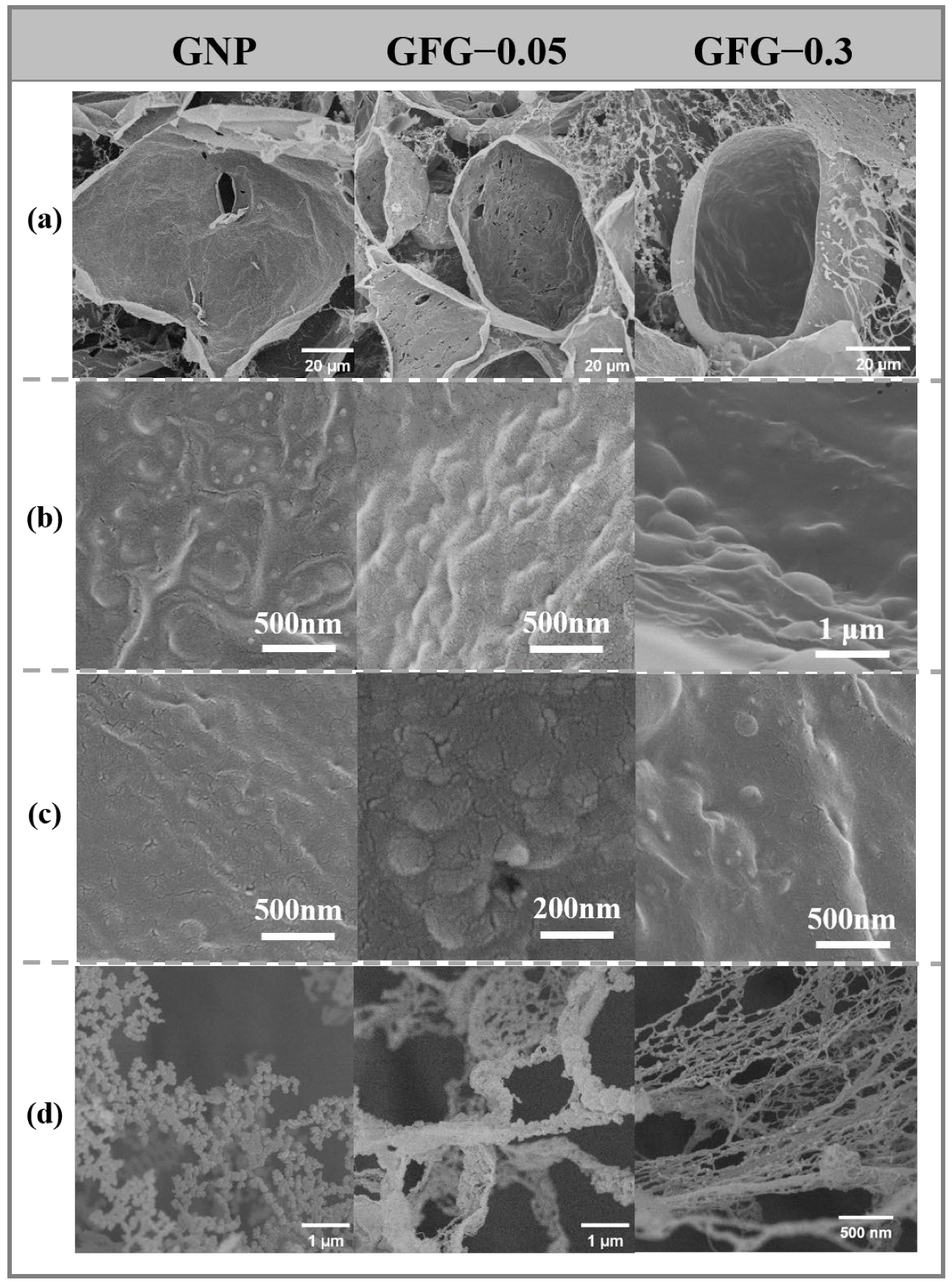
2.2.3. Interfacial Morphology of Foams
2.3. Foaming Properties of Complex Particles
3. Conclusions
4. Materials and Methods
4.1. Materials
4.2. Flaxseed Gum Extraction
4.3. Preparation of Gliadin-Flaxseed Gum Composite Particles
4.4. Turbidity and Viscosity Measurements
4.5. Particle Size and Zeta-Potential Measurements
4.6. Microstructure Observations
4.7. Surface Wettability Measurement
4.8. Flexibility Measurement
4.9. Visualization of Interfacial Morphology by Cryo-SEM
4.10. Interfacial Adsorption Behavior Measurement
4.11. Interfacial Dilatational Rheology Measurement
4.12. Foaming Properties
4.13. Statistical Analysis
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Murray, B.S. Recent developments in food foams. Curr. Opin. Colloid Interface Sci. 2020, 50, 101394. [Google Scholar] [CrossRef]
- Peng, D.; Jin, W.; Tang, C.; Lu, Y.; Wang, W.; Li, J.; Li, B. Foaming and surface properties of gliadin nanoparticles: Influence of pH and heating temperature. Food Hydrocoll. 2018, 77, 107–116. [Google Scholar] [CrossRef]
- Peng, D.; Jin, W.; Zhou, P.; Ren, C.; Li, B. Foaming and surface rheological behaviors of gliadin particles: Effect of solvent and concentration of gliadin stock solution. Food Hydrocoll. 2020, 106, 105868. [Google Scholar] [CrossRef]
- Tyowua, A.T.; Binks, B.P. Growing a particle-stabilized aqueous foam. J. Colloid Interface Sci. 2020, 561, 127–135. [Google Scholar] [CrossRef]
- Yang, L.L.; Wang, T.D.; Yang, X.; Jiang, G.C.; Luckham, P.F.; Xu, J.P.; Li, X.L.; Ni, X.X. Highly Stabilized Foam by Adding Amphiphilic Janus Particles for Drilling a High-Temperature and High-Calcium Geothermal Well. Ind. Eng. Chem. Res. 2019, 58, 9795–9805. [Google Scholar] [CrossRef]
- Han, Y.; Zhu, L.; Karrar, E.; Qi, X.; Zhang, H.; Wu, G. Pickering foams stabilized by protein-based particles: A review of characterization, stabilization, and application. Trends Food Sci. Technol. 2023, 133, 148–159. [Google Scholar] [CrossRef]
- Huang, Y.; Yang, J.; Chen, L.; Zhang, L. Chitin Nanofibrils to Stabilize Long-Life Pickering Foams and Their Application for Lightweight Porous Materials. ACS Sustain. Chem. Eng. 2018, 6, 10552–10561. [Google Scholar] [CrossRef]
- Li, X.; Murray, B.S.; Yang, Y.; Sarkar, A. Egg white protein microgels as aqueous Pickering foam stabilizers: Bubble stability and interfacial properties. Food Hydrocoll. 2020, 98, 105292. [Google Scholar] [CrossRef]
- Li, M.; He, S. Utilization of zein-based particles in Pickering emulsions: A review. Food Rev. Int. 2023, 39, 4040–4060. [Google Scholar] [CrossRef]
- Wouters, A.G.B.; Schaefer, S.; Joye, I.J.; Delcour, J.A. Relating the structural, air-water interfacial and foaming properties of wheat (Triticum aestivum L.) gliadin and maize (Zea mays L.) zein based nanoparticle suspensions. Colloids Surf. A Physicochem. Eng. Asp. 2019, 567, 249–259. [Google Scholar] [CrossRef]
- Yan, C.; McClements, D.J.; Zou, L.; Liu, W. A stable high internal phase emulsion fabricated with OSA-modified starch: An improvement in β-carotene stability and bioaccessibility. Food Funct. 2019, 10, 5446–5460. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.; Huan, S.; Bai, L.; Ketola, A.; Shi, X.; Zhang, X.; Ketoja, J.A.; Rojas, O.J. High Internal Phase Oil-in-Water Pickering Emulsions Stabilized by Chitin Nanofibrils: 3D Structuring and Solid Foam. ACS Appl. Mater. Interfaces 2020, 12, 11240–11251. [Google Scholar] [CrossRef] [PubMed]
- Li, S.-Q.; Huang, X.-N.; Wei, S.-H.; Yu, X.-H.; Yang, X.-Q.; Zhu, J.-H.; Yin, S.-W. Gliadin-based nanoparticles used for pickering stabilization: Current status and future perspective. Food Hydrocoll. 2023, 145, 109020. [Google Scholar] [CrossRef]
- Peng, D.; Shang, W.; Yang, J.; Li, K.; Shen, W.; Wan, C.; Geng, F.; Deng, Q.; Jin, W. Interfacial arrangement of tunable gliadin nanoparticles via electrostatic assembly with pectin: Enhancement of foaming property. Food Hydrocoll. 2023, 143, 108852. [Google Scholar] [CrossRef]
- El Hariri El Nokab, M.; Lasorsa, A.; Sebakhy, K.O.; Picchioni, F.; van der Wel, P.C.A. Solid-state NMR spectroscopy insights for resolving different water pools in alginate hydrogels. Food Hydrocoll. 2022, 127, 107500. [Google Scholar] [CrossRef]
- Huang, X.; Huang, X.; Gong, Y.; Xiao, H.; McClements, D.J.; Hu, K. Enhancement of curcumin water dispersibility and antioxidant activity using core–shell protein–polysaccharide nanoparticles. Food Res. Int. 2016, 87, 1–9. [Google Scholar] [CrossRef]
- Zhang, W.; Gu, X.; Liu, X.; Wang, Z. Fabrication of Pickering emulsion based on particles combining pectin and zein: Effects of pectin methylation. Carbohydr. Polym. 2021, 256, 117515. [Google Scholar] [CrossRef]
- Shen, C.; Chen, W.; Li, C.; Chen, X.; Cui, H.; Lin, L. Pickering emulsion stabilized by gliadin/soybean polysaccharide composite colloidal nanoparticle: Physicochemical properties and its application on washing of fresh-cut cabbage. Food Res. Int. 2022, 161, 111886. [Google Scholar] [CrossRef]
- Peng, D.; Yang, J.; de Groot, A.; Jin, W.; Deng, Q.; Li, B.; Sagis, L.M.C. Soft gliadin nanoparticles at air/water interfaces: The transition from a particle-laden layer to a thick protein film. J. Colloid Interface Sci. 2024, 669, 236–247. [Google Scholar] [CrossRef]
- Tian, Y.; Pi, J.; Lv, J.; Chen, Y.; Ma, M.; Fu, X. The impact of ultrasound treatment combined with flaxseed gum on the foaming properties of egg white. Food Hydrocoll. 2024, 148, 109507. [Google Scholar] [CrossRef]
- Liu, J.; Shim, Y.Y.; Tse, T.J.; Wang, Y.; Reaney, M.J.T. Flaxseed gum a versatile natural hydrocolloid for food and non-food applications. Trends Food Sci. Technol. 2018, 75, 146–157. [Google Scholar] [CrossRef]
- Chen, M.; Bleeker, R.; Sala, G.; Meinders, M.B.J.; van Valenberg, H.J.F.; van Hooijdonk, A.C.M.; van der Linden, E. Particle size determines foam stability of casein micelle dispersions. Int. Dairy J. 2016, 56, 151–158. [Google Scholar] [CrossRef]
- Wasilewska, A.; Bielicka, M.; Klekotka, U.; Kalska-Szostko, B. Nanoparticle applications in food—A review. Food Funct. 2023, 14, 2544–2567. [Google Scholar] [CrossRef]
- Zhu, X.; Chen, Y.; Hu, Y.; Han, Y.; Xu, J.; Zhao, Y.; Chen, X.; Li, B. Tuning the molecular interactions between gliadin and tannic acid to prepare Pickering stabilizers with improved emulsifying properties. Food Hydrocoll. 2021, 111, 106179. [Google Scholar] [CrossRef]
- Sun, H.; Ma, Y.; Huang, X.; Song, L.; Guo, H.; Sun, X.; Li, N.; Qiao, M. Stabilization of flaxseed oil nanoemulsions based on flaxseed gum: Effects of temperature, pH and NaCl on stability. LWT 2023, 176, 114512. [Google Scholar] [CrossRef]
- Jin, W.; Zhu, J.; Jiang, Y.; Shao, P.; Li, B.; Huang, Q. Gelatin-Based Nanocomplex-Stabilized Pickering Emulsions: Regulating Droplet Size and Wettability through Assembly with Glucomannan. J. Agric. Food Chem. 2017, 65, 1401–1409. [Google Scholar] [CrossRef]
- Chen, S.; Du, Y.; Zhang, H.; Wang, Q.; Gong, Y.; Chang, R.; Zhang, J.; Zhang, J.; Yuan, Y.; Liu, B.; et al. The lipid digestion behavior of oil-in-water Pickering emulsions stabilized by whey protein microgels of various rigidities. Food Hydrocoll. 2022, 130, 107735. [Google Scholar] [CrossRef]
- Diao, Y.; Zhang, Y.; Zhang, W.; Xu, W.; Hu, Z.; Yi, Y.; Wang, Y. Acid-thermal-induced formation of rice bran protein nano-particles: Foaming properties and physicochemical characteristics. Int. J. Food Sci. Technol. 2022, 57, 3624–3633. [Google Scholar] [CrossRef]
- Langevin, D. Influence of interfacial rheology on foam and emulsion properties. Adv. Colloid Interface Sci. 2000, 88, 209–222. [Google Scholar] [CrossRef]
- Yang, J.; Duan, Y.; Geng, F.; Cheng, C.; Wang, L.; Ye, J.; Zhang, H.; Peng, D.; Deng, Q. Ultrasonic-assisted pH shift-induced interfacial remodeling for enhancing the emulsifying and foaming properties of perilla protein isolate. Ultrason. Sonochemistry 2022, 89, 106108. [Google Scholar] [CrossRef]
- Yang, J.; Duan, Y.; Zhang, H.; Huang, F.; Wan, C.; Cheng, C.; Wang, L.; Peng, D.; Deng, Q. Ultrasound coupled with weak alkali cycling-induced exchange of free sulfhydryl-disulfide bond for remodeling interfacial flexibility of flaxseed protein isolates. Food Hydrocoll. 2023, 140, 108597. [Google Scholar] [CrossRef]
- Perez, A.A.; Carrara, C.R.; Sánchez, C.C.; Santiago, L.G.; Patino, J.M.R. Interfacial dynamic properties of whey protein concentrate/polysaccharide mixtures at neutral pH. Food Hydrocoll. 2009, 23, 1253–1262. [Google Scholar] [CrossRef]
- Qiao, X.; Miller, R.; Schneck, E.; Sun, K. Foaming properties and the dynamics of adsorption and surface rheology of silk fibroin at the air/water interface. Colloids Surf. A Physicochem. Eng. Asp. 2020, 591, 124553. [Google Scholar] [CrossRef]
- Shen, P.; Peng, J.; Sagis, L.M.C.; Landman, J. Air-water interface properties and foam stabilization by mildly extracted lentil protein. Food Hydrocoll. 2024, 147, 109342. [Google Scholar] [CrossRef]
- Shen, Q.; Luo, Y.; Zheng, W.; Xiong, T.; Han, F.; Zuo, J.; Dai, J.; Li, B.; Chen, Y. Nonlinear rheological behavior and quantitative proteomic analysis of pea protein isolates at the air-water interface. Food Hydrocoll. 2023, 135, 108115. [Google Scholar] [CrossRef]
- Sagis, L.M.C.; Humblet-Hua, K.N.P.; van Kempen, S.E.H.J. Nonlinear stress deformation behavior of interfaces stabilized by food-based ingredients. J. Phys. Condens. Matter 2014, 26, 464105. [Google Scholar] [CrossRef]
- Yang, J.; Yang, Q.; Waterink, B.; Venema, P.; de Vries, R.; Sagis, L.M.C. Physical, interfacial and foaming properties of different mung bean protein fractions. Food Hydrocoll. 2023, 143, 108885. [Google Scholar] [CrossRef]
- Van Kempen, S.E.H.J.; Schols, H.A.; van der Linden, E.; Sagis, L.M.C. Non-linear surface dilatational rheology as a tool for understanding microstructures of air/water interfaces stabilized by oligofructose fatty acid esters. Soft Matter 2013, 9, 9579–9592. [Google Scholar] [CrossRef]
- Yang, J.; Waardenburg, L.C.; Berton-Carabin, C.C.; Nikiforidis, C.V.; van der Linden, E.; Sagis, L.M.C. Air-water interfacial behaviour of whey protein and rapeseed oleosome mixtures. J. Colloid Interface Sci. 2021, 602, 207–221. [Google Scholar] [CrossRef]
- Kontogiorgos, V.; Prakash, S. Adsorption kinetics and dilatational rheology of plant protein concentrates at the air- and oil-water interfaces. Food Hydrocoll. 2023, 138, 108486. [Google Scholar] [CrossRef]
- Sorgentini, D.A.; Wagner, J.R. Comparative study of foaming properties of whey and isolate soybean proteins. Food Res. Int. 2002, 35, 721–729. [Google Scholar] [CrossRef]
- Lajnaf, R.; Picart-Palmade, L.; Cases, E.; Attia, H.; Marchesseau, S.; Ayadi, M.A. The foaming properties of camel and bovine whey: The impact of pH and heat treatment. Food Chem. 2018, 240, 295–303. [Google Scholar] [CrossRef] [PubMed]
- Li, R.; Zhang, Y.; Chang, Y.; Wu, Z.; Wang, Y.; Chen, X.E.; Wang, T. Role of foam drainage in producing protein aggregates in foam fractionation. Colloids Surf. B Biointerfaces 2017, 158, 562–568. [Google Scholar] [CrossRef] [PubMed]
- Hunter, T.N.; Pugh, R.J.; Franks, G.V.; Jameson, G.J. The role of particles in stabilising foams and emulsions. Adv. Colloid Interface Sci. 2008, 137, 57–81. [Google Scholar] [CrossRef] [PubMed]
- Alavi, F.; Emam-Djomeh, Z.; Mohammadian, M.; Salami, M.; Moosavi-Movahedi, A.A. Physico-chemical and foaming properties of nanofibrillated egg white protein and its functionality in meringue batter. Food Hydrocoll. 2020, 101, 105554. [Google Scholar] [CrossRef]
- Rüegg, R.; Schmid, T.; Hollenstein, L.; Müller, N. Effect of particle characteristics and foaming parameters on resulting foam quality and stability. LWT 2022, 167, 113859. [Google Scholar] [CrossRef]
- Sun, M.; Li, X.; McClements, D.J.; Xiao, M.; Chen, H.; Zhou, Q.; Xu, S.; Chen, Y.; Deng, Q. Reducing off-flavors in plant-based omega-3 oil emulsions using interfacial engineering: Coating algae oil droplets with pea protein/flaxseed gum. Food Hydrocoll. 2022, 122, 107069. [Google Scholar] [CrossRef]
- Wang, Y.-H.; Wan, Z.-L.; Yang, X.-Q.; Wang, J.-M.; Guo, J.; Lin, Y. Colloidal complexation of zein hydrolysate with tannic acid: Constructing peptides-based nanoemulsions for alga oil delivery. Food Hydrocoll. 2016, 54, 40–48. [Google Scholar] [CrossRef]
- Shao, J.; Peng, D.; Yang, J.; Huang, F.; Jin, W.; Xiao, J.; Sun, S.; Li, F.; Li, T.; Deng, Q. Improvement of foaming and emulsifying properties of SPI via weak-base synchronized membrane separation: Insight from tuning the interfacial flexibility. Food Hydrocoll. 2024, 154, 110110. [Google Scholar] [CrossRef]
- Peng, D.; Jin, W.; Arts, M.; Yang, J.; Li, B.; Sagis, L.M.C. Effect of CMC degree of substitution and gliadin/CMC ratio on surface rheology and foaming behavior of gliadin/CMC nanoparticles. Food Hydrocoll. 2020, 107, 105955. [Google Scholar] [CrossRef]



Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wu, P.; Shang, W.; Shao, J.; Deng, Q.; Zhou, J.; Xiang, X.; Peng, D.; Jin, W. Tuning the Interfacial Deformation of Gliadin-Flaxseed Gum Complex Particles for Improving the Foam Stability. Gels 2024, 10, 677. https://doi.org/10.3390/gels10110677
Wu P, Shang W, Shao J, Deng Q, Zhou J, Xiang X, Peng D, Jin W. Tuning the Interfacial Deformation of Gliadin-Flaxseed Gum Complex Particles for Improving the Foam Stability. Gels. 2024; 10(11):677. https://doi.org/10.3390/gels10110677
Chicago/Turabian StyleWu, Ping, Wei Shang, Jiaqi Shao, Qianchun Deng, Jisong Zhou, Xia Xiang, Dengfeng Peng, and Weiping Jin. 2024. "Tuning the Interfacial Deformation of Gliadin-Flaxseed Gum Complex Particles for Improving the Foam Stability" Gels 10, no. 11: 677. https://doi.org/10.3390/gels10110677
APA StyleWu, P., Shang, W., Shao, J., Deng, Q., Zhou, J., Xiang, X., Peng, D., & Jin, W. (2024). Tuning the Interfacial Deformation of Gliadin-Flaxseed Gum Complex Particles for Improving the Foam Stability. Gels, 10(11), 677. https://doi.org/10.3390/gels10110677





