Phytosterol-Loaded Surface-Tailored Bioactive-Polymer Nanoparticles for Cancer Treatment: Optimization, In Vitro Cell Viability, Antioxidant Activity, and Stability Studies
Abstract
:1. Introduction
2. Results and Discussion
2.1. Optimization
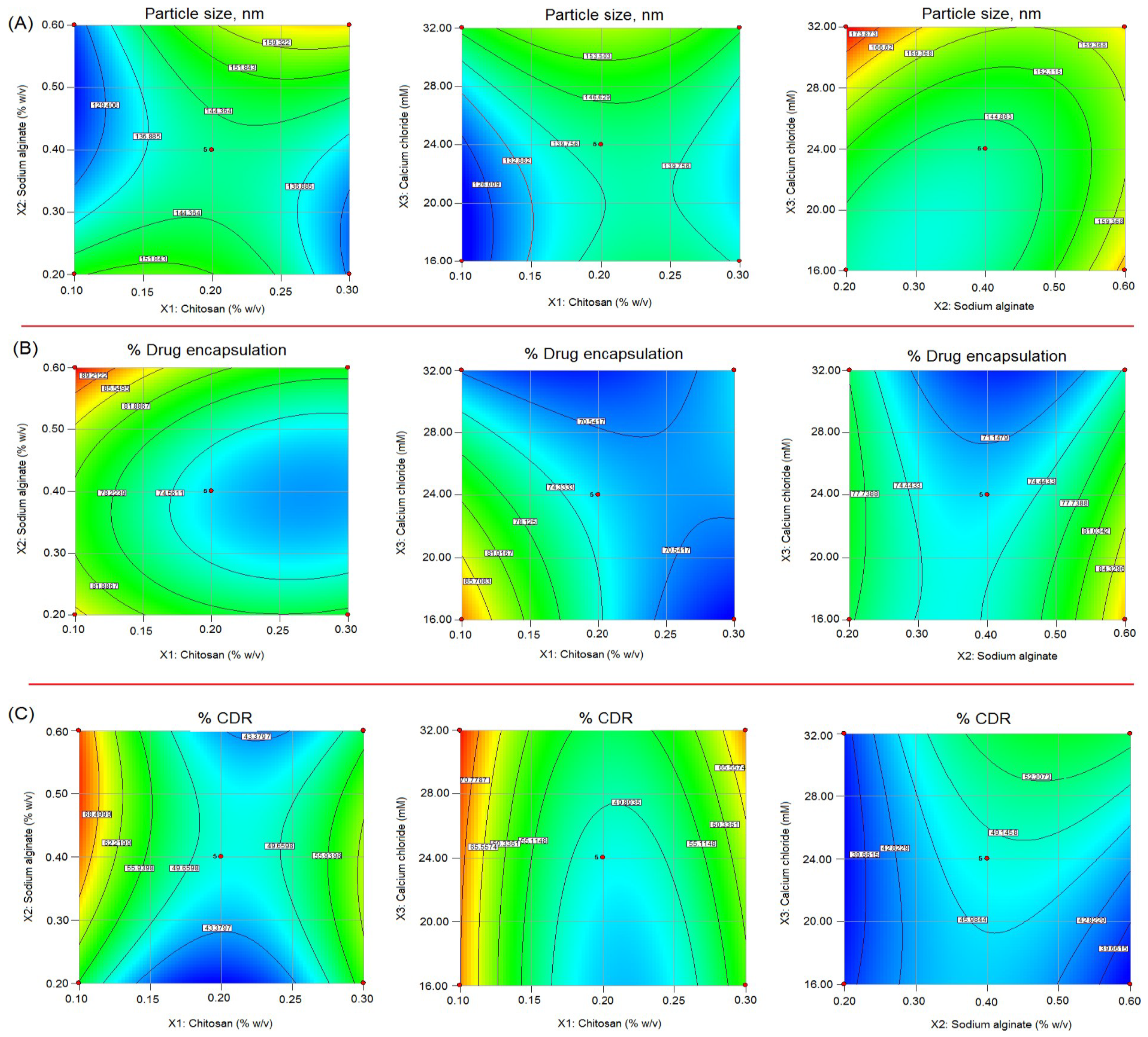
2.2. Impact of Independent Variables X1, X2, and X3 on Particle Size (Y1)
2.3. Impact of Independent Variables X1, X2, and X3 on %Drug Encapsulation (Y2)
2.4. Impact of Independent Variables X1, X2, and X3 on %Cumulative Drug Release (Y3)
2.5. Checkpoint Analysis of Optimum Formulation
2.6. Characterization of Nanosystem
2.6.1. Nanoparticlecharacterization
2.6.2. Thermal Behavior
2.6.3. FT-IR Spectral Analysis
2.6.4. Drug Release and Kinetics
2.6.5. Everted Gut Sac Model
2.6.6. Cell-Viability Assay
2.6.7. Antioxidant Activity
2.6.8. Stability Study
3. Conclusions
4. Materials and Methods
4.1. Materials
4.2. Analytical Methodology
4.3. Formulation Optimization
4.4. Preparation of β-Sitosterol-Loaded Alg/Ch-NPs
4.5. Folate Modification of β-Sitosterol-Loaded Alg/Ch-NPs
4.6. In Vitro Characterization of Nanosize System
4.6.1. Particle Sizes and Their Distribution
4.6.2. Drug Encapsulation and Loading
4.6.3. Fourier Transform Infrared Spectroscopy (FT-IR)
4.6.4. Differential Scanning Calorimetry
4.6.5. Drug-Release Study
4.6.6. In Vitro Permeation Study
4.6.7. Cell-Viability Study
4.6.8. Radical Scavenging Power
4.7. Stability Study
4.8. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Siegel, R.L.; Miller, K.D.; Fuchs, H.E.; Jemal, A. Erratum to Cancer statistics, 2021. CA A Cancer J. Clin. 2021, 71, 7–33. [Google Scholar] [CrossRef] [PubMed]
- Jahan, S.; Karim, E.; Chowdhury, E. Nanoparticles Targeting Receptors on Breast Cancer for Efficient Delivery of Chemotherapeutics. Biomedicines 2021, 9, 114. [Google Scholar] [CrossRef] [PubMed]
- Ji, X.; Lu, Y.; Tian, H.; Meng, X.; Wei, M.; Cho, W.C. Chemoresistance mechanisms of breast cancer and their countermeasures. Biomed. Pharmacother. 2019, 114, 108800. [Google Scholar] [CrossRef] [PubMed]
- Huda, S.; Alam, A.; Sharma, P.K. Smart nanocarriers-based drug delivery for cancer therapy: An innovative and developing strategy. J. Drug Deliv. Sci. Technol. 2020, 60, 102018. [Google Scholar] [CrossRef]
- Bertrand, N.; Wu, J.; Xu, X.; Kamaly, N.; Farokhzad, O.C. Cancer nanotechnology: The impact of passive and active targeting in the era of modern cancer biology. Adv. Drug Deliv. Rev. 2014, 66, 2–25. [Google Scholar] [CrossRef] [Green Version]
- Blanco, E.; Shen, H.; Ferrari, M. Principles of nanoparticle design for overcoming biological barriers to drug delivery. Nat. Biotechnol. 2015, 33, 941–951. [Google Scholar] [CrossRef]
- Ahmad, J.; Ameeduzzafar; Ahmad, M.Z.; Akhter, H. Surface-Engineered Cancer Nanomedicine: Rational Design and Recent Progress. Curr. Pharm. Des. 2020, 26, 1181–1190. [Google Scholar] [CrossRef]
- Akhter, H.; Ahsan, M.J.; Rahman, M.; Anwar, S.; Rizwanullah, M. Advancement in Nanotheranostics for Effective Skin Cancer Therapy: State of the Art. Curr. Nanomed. 2020, 10, 90–104. [Google Scholar] [CrossRef]
- Assaraf, Y.G.; Leamon, C.P.; Reddy, J.A. The folate receptor as a rational therapeutic target for personalized cancer treatment. Drug Resist. Updat. 2014, 17, 89–95. [Google Scholar] [CrossRef]
- Salehi, B.; Quispe, C.; Sharifi-Rad, J.; Cruz-Martins, N.; Nigam, M.; Mishra, A.P.; Konovalov, D.A.; Orobinskaya, V.; Abu-Reidah, I.M.; Zam, W.; et al. Phytosterols: From Preclinical Evidence to Potential Clinical Applications. Front. Pharmacol. 2021, 11, 599959. [Google Scholar] [CrossRef]
- Ogbe, R.J.; Ochalefu, D.O.; Mafulul, S.G.; Olaniru, O.B. A review on dietary phytosterols: Their occurrence, metabolism and health benefits. Asian J. Plant Sci. Res. 2015, 5, 10–21. [Google Scholar]
- Zhong, S.; Zhang, H.; Liu, Y.; Wang, G.; Shi, C.; Li, Z.; Feng, Y.; Cui, X. Folic acid functionalized reduction-responsive magnetic chitosan nanocapsules for targeted delivery and triggered release of drugs. Carbohydr. Polym. 2017, 168, 282–289. [Google Scholar] [CrossRef] [PubMed]
- Di Martino, A.; Trusova, M.E.; Postnikov, P.S.; Sedlarik, V. Folic acid-chitosan-alginate nanocomplexes for multiple delivery of chemotherapeutic agents. J. Drug Deliv. Sci. Technol. 2018, 47, 67–76. [Google Scholar] [CrossRef]
- Loquercio, A.; Castell-Perez, E.; Gomes, C.; Moreira, R.G. Preparation of Chitosan-Alginate Nanoparticles for Trans-cinnamaldehyde Entrapment. J. Food Sci. 2015, 80, N2305–N2315. [Google Scholar] [CrossRef] [PubMed]
- Murata, Y.; Jinno, D.; Liu, D.; Isobe, T.; Kofuji, K.; Kawashima, S. The Drug Release Profile from Calcium-induced Alginate Gel Beads Coated with an Alginate Hydrolysate. Molecules 2007, 12, 2559–2566. [Google Scholar] [CrossRef]
- Sarmento, B.; Ribeiro, A.J.; Veiga, F.; Ferreira, D.C.; Neufeld, R.J. Insulin-loaded nanoparticles are prepared by alginate iono-tropic pre-gelation followed by chitosan polyelectrolyte complexation. J. Nanosci. Nanotechnol. 2007, 7, 2833–2841. [Google Scholar] [CrossRef]
- Caetano, L.A.; Almeida, A.J.; Gonçalves, L.M. Effect of Experimental Parameters on Alginate/Chitosan Microparticles for BCG Encapsulation. Mar. Drugs 2016, 14, 90. [Google Scholar] [CrossRef] [Green Version]
- Thai, H.; Nguyen, C.T.; Thach, L.T.; Tran, M.T.; Mai, H.D.; Nguyen, T.T.T.; Le, G.D.; Van Can, M.; Tran, L.D.; Bach, G.L.; et al. Characterization of chitosan/alginate/lovastatin nanoparticles and investigation of their toxic effects in vitro and in vivo. Sci. Rep. 2020, 10, 909–915. [Google Scholar] [CrossRef]
- Borges, O.; Cordeiro-Da-Silva, A.; Romeijn, S.G.; Amidi, M.; de Sousa, A.; Borchard, G.; Junginger, H.E. Uptake studies in rat Peyer’s patches, cytotoxicity and release studies of alginate coated chitosan nanoparticles for mucosal vaccination. J. Control Release 2006, 114, 348–358. [Google Scholar] [CrossRef] [Green Version]
- Mokhtari, S.; Jafari, S.M.; Assadpour, E. Development of a nutraceutical Nano-delivery system through emulsification/internal gelation of alginate. Food Chem. 2017, 229, 286–295. [Google Scholar] [CrossRef]
- Ahdyani, R.; Novitasari, L.; Martien, R. Formulation and characterization of timolol maleate-loaded nanoparticles gel by ionic gelation method using chitosan and sodium alginate. Int. J. Appl. Pharm. 2019, 11, 48–54. [Google Scholar] [CrossRef]
- Sabbagh, H.A.K.; Hussein-Al-Ali, S.H.; Hussein, M.Z.; Abudayeh, Z.; Ayoub, R.; Abudoleh, S.M. A Statistical Study on the Development of Metronidazole-Chitosan-Alginate Nanocomposite Formulation Using the Full Factorial Design. Polymers 2020, 12, 772. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bi, Y.G.; Lin, Z.T.; Deng, S.T. Fabrication and characterization of hydroxyapatite/sodium alginate/chitosan composite microspheres for drug delivery and bone tissue engineering. Mater. Sci. Eng. C Mater. Biol. Appl. 2019, 100, 576–583. [Google Scholar] [CrossRef] [PubMed]
- Hasnain, M.S.; Nayak, A.K. Alginates: Versatile Polymers in Biomedical Applications and Therapeutics; Apple Academic Press: Cambridge, MA, USA, 2020. [Google Scholar]
- Seyam, S.; Nordin, N.A.; Alfatama, M. Recent Progress of Chitosan and Chitosan Derivatives-Based Nanoparticles: Pharmaceutical Perspectives of Oral Insulin Delivery. Pharmaceuticals 2020, 13, 307. [Google Scholar] [CrossRef] [PubMed]
- Rahaiee, S.; Shojaosadati, S.A.; Hashemi, M.; Moini, S.; Razavi, S.H. Improvement of crocin stability by biodegradeble nanoparticles of chitosan-alginate. Int. J. Biol. Macromol. 2015, 79, 423–432. [Google Scholar] [CrossRef]
- Choukaife, H.; Doolaanea, A.A.; Alfatama, M. Alginate Nanoformulation: Influence of Process and Selected Variables. Pharmaceuticals 2020, 13, 335. [Google Scholar] [CrossRef]
- Kausar, H.; Mujeeb, M.; Ahad, A.; Moolakkadath, T.; Aqil, M.; Ahmad, A.; Akhter, M.H. Optimization of ethosomes for topical thymoquinone delivery for the treatment of skin acne. J. Drug Deliv. Sci. Technol. 2019, 49, 177–187. [Google Scholar] [CrossRef]
- Md, S.; Alhakamy, N.A.; Aldawsari, H.M.; Husain, M.; Khan, N.; Alfaleh, M.A.; Asfour, H.Z.; Riadi, Y.; Bilgrami, A.L.; Akhter, M.H. Plumbagin-Loaded Glycerosome Gel as Topical Delivery System for Skin Cancer Therapy. Polymers 2021, 13, 923. [Google Scholar] [CrossRef]
- Gazzali, A.M.; Lobry, M.; Colombeau, L.; Acherar, S.; Azaïs, H.; Mordon, S.; Arnoux, P.; Baros, F.; Vanderesse, R.; Frochot, C. Stability of folic acid under several parameters. Eur. J. Pharm. Sci. 2016, 93, 419–430. [Google Scholar] [CrossRef]
- Trivedi, D. Evaluation of the Physicochemical and Thermal Properties of Folic Acid: Influence of the Energy of Consciousness Healing Treatment. Sch. J. Food Nutr. 2019, 1, 733. [Google Scholar] [CrossRef]
- Kavithaa, K.; Paulpandi, M.; Ramya, S.; Ramesh, M.; Balachandar, V.; Ramasamy, K.; Narayanasamy, A. Sitosterol-fabricated chitosan nanocomplex induces apoptotic cell death through mitochondrial dysfunction in lung cancer animal model: An enhanced synergetic drug delivery system for lung cancer therapy. New J. Chem. 2021, 45, 9251–9263. [Google Scholar] [CrossRef]
- Lee, S.; Lee, K. pH-Sensitive Folic Acid Conjugated Alginate Nanoparticle for Induction of Cancer-Specific Fluorescence Imaging. Pharmaceutics 2020, 12, 537. [Google Scholar] [CrossRef] [PubMed]
- Wibowo, F.R.; Saputra, O.A.; Lestari, W.W.; Koketsu, M.; Mukti, R.R.; Martien, R. pH-Triggered Drug Release Controlled by Poly(Styrene Sulfonate) Growth Hollow Mesoporous Silica Nanoparticles. ACS Omega 2020, 5, 4261–4269. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nasab, N.A.; Kumleh, H.H.; Beygzadeh, M.; Teimourian, S.; Kazemzad, M. Delivery of curcumin by a pH-responsive chitosan mesoporous silica nanoparticles for cancer treatment. Artif. Cells Nanomed. Biotechnol. 2018, 46, 75–81. [Google Scholar] [CrossRef] [PubMed]
- Ochoa, L.; Igartua, M.; Hernandez, R.M.; Rodríguez-Gascón, A.; Solinis, M.A.; Pedraz, J. Novel extended-release formulation of lovastatin by one-step melt granulation: In vitro and in vivo evaluation. Eur. J. Pharm. Biopharm. 2011, 77, 306–312. [Google Scholar] [CrossRef]
- Tarafder, S.; Nansen, K.; Bose, S. Lovastatin release from polycaprolactone coated β-tricalcium phosphate: Effects of pH, concentration and drug–polymer interactions. Mater. Sci. Eng. C 2013, 33, 3121–3128. [Google Scholar] [CrossRef] [Green Version]
- Li, P.; Dai, Y.-N.; Zhang, J.-P.; Wang, A.-Q.; Wei, Q. Chitosan-Alginate Nanoparticles as a Novel Drug Delivery System for Nifedipine. Int. J. Biomed. Sci. 2008, 4, 221–228. [Google Scholar]
- Zhang, F.; Liu, Z.; He, X.; Li, Z.; Shi, B.; Cai, F. β-Sitosterol-loaded solid lipid nanoparticles ameliorate complete Freund’s adjuvant-induced arthritis in rats: Involvement of NF-κB and HO-1/Nrf-2 pathway. Drug Deliv. 2020, 27, 1329–1341. [Google Scholar] [CrossRef]
- Akhter, H.; Beg, S.; Tarique, M.; Malik, A.; Afaq, S.; Choudhry, H.; Hosawi, S. Receptor-based targeting of engineered nanocarrier against solid tumors: Recent progress and challenges ahead. Biochim. Biophys. Acta Gen. Subj. 2021, 1865, 129777. [Google Scholar] [CrossRef]
- Akhter, H.; Rizwanullah, M.; Ahmad, J.; Ahsan, M.J.; Mujtaba, A.; Amin, S. Nanocarriers in advanced drug targeting: Setting novel paradigm in cancer therapeutics. Artif. Cells Nanomed. Biotechnol. 2018, 46, 873–884. [Google Scholar] [CrossRef] [Green Version]
- Akhter, H.; Khalilullah, H.; Gupta, M.; Alfaleh, M.A.; Alhakamy, N.A.; Riadi, Y.; Md, S. Impact of Protein Corona on the Biological Identity of Nanomedicine: Understanding the Fate of Nanomaterials in the Biological Milieu. Biomedicines 2021, 9, 1496. [Google Scholar] [CrossRef] [PubMed]
- Akhter, H.; Rizwanullah, M.; Ahmad, J.; Amin, S.; Ahmad, M.Z.; Minhaj, A.; Mujtaba, A.; Ali, J. Molecular Targets and Nanoparticulate Systems Designed for the Improved Therapeutic Intervention in Glioblastoma Multiforme. Drug Res. 2021, 71, 122–137. [Google Scholar] [CrossRef] [PubMed]
- Akhter, H.; Madhav, N.S.; Ahmad, J. Epidermal growth factor receptor based active targeting: A paradigm shift towards advance tumor therapy. Artif. Cells Nanomed. Biotechnol. 2018, 46, 1188–1198. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Awad, A.; Chinnam, M.; Fink, C.; Bradford, P. β-Sitosterol activates Fas signaling in human breast cancer cells. Phytomedicine 2007, 14, 747–754. [Google Scholar] [CrossRef]
- Mohammad, N.A.; Zaidel, D.N.A.; Muhamad, I.I.; Hamid, M.A.; Yaakob, H.; Jusoh, Y.M.M. Optimization of the antioxidant-rich xanthone extract from mangosteen (Garcinia mangostana L.) pericarp via microwave-assisted extraction. Heliyon 2019, 5, e02571. [Google Scholar] [CrossRef] [Green Version]
- Soni, K.; Mujtaba, A.; Akhter, H.; Zafar, A.; Kohli, K. Optimisation of ethosomal nanogel for topical nano-CUR and sulphoraphane delivery in effective skin cancer therapy. J. Microencapsul. 2019, 37, 91–108. [Google Scholar] [CrossRef]
- Akhter, H.; Kumar, S.; Nomani, S. Sonication tailored enhance cytotoxicity of naringenin nanoparticle in pancreatic cancer: Design, optimization, and in vitro studies. Drug Dev. Ind. Pharm. 2020, 46, 659–672. [Google Scholar] [CrossRef]
- Katuwavila, N.P.; Perera, A.D.L.C.; Samarakoon, S.R.; Soysa, P.; Karunaratne, V.; Amaratunga, G.A.J.; Karunaratne, D.N. Chi-tosan-alginate nanoparticle system efficiently delivers doxorubicin to MCF-7 Cells. J. Nanomater. 2016, 2016, 3178904. [Google Scholar] [CrossRef] [Green Version]
- Jha, S.K.; Karki, R.; Puttegowda, V.D.; Harinarayana, D. In Vitro Intestinal Permeability Studies and Pharmacokinetic Evaluation of Famotidine Microemulsion for Oral Delivery. Int. Sch. Res. Not. 2014, 2014, 452051. [Google Scholar] [CrossRef] [Green Version]
- Gilani, S.J.; Imam, S.S.; Ahmed, A.; Chauhan, S.; Mirza, M.A.; Taleuzzaman, M. Formulation and evaluation of thymoquinone niosomes: Application of developed and validated RP-HPLC method in delivery system. Drug Dev. Ind. Pharm. 2019, 45, 1799–1806. [Google Scholar] [CrossRef]
- Bhattamisra, S.K.; Shak, A.T.; Xi, L.W.; Safian, N.H.; Choudhury, H.; Lim, W.M.; Shahzad, N.; Alhakamy, N.; Anwer, K.; Radhakrishnan, A.K.; et al. Nose to brain delivery of rotigotine loaded chitosan nanoparticles in human SH-SY5Y neuroblastoma cells and animal model of Parkinson’s disease. Int. J. Pharm. 2020, 579, 119148. [Google Scholar] [CrossRef] [PubMed]
- Imam, S.S.; AlShehri, S.; Alzahrani, T.A.; Hussain, A.; Altamimi, M.A. Formulation and Evaluation of Supramolecular Food-Grade Piperine HP β CD and TPGS Complex: Dissolution, Physicochemical Characterization, Molecular Docking, In Vitro Antioxidant Activity, and Antimicrobial Assessment. Molecules 2020, 25, 4716. [Google Scholar] [CrossRef] [PubMed]
- ICH Q1A(R2): Stability Testing Guidelines: Stability Testing of New Drug Substances and Products; European Medicines Agency: Amsterdam, The Netherlands, 2003.
- Akhter, M.H.; Ahmad, A.; Ali, J.; Mohan, G. Formulation and Development of CoQ10-Loaded s-SNEDDS for Enhancement of Oral Bioavailability. J. Pharm. Innov. 2014, 9, 121–131. [Google Scholar] [CrossRef]










| Independent Variables | Level Employed | ||
|---|---|---|---|
| Low (−1) | Medium (0) | High (+1) | |
| X1: Chitosan (% w/v) | 0.1 | 0.2 | 0.3 |
| X2: Sodium alginate (% w/v) | 0.2 | 0.4 | 0.6 |
| X3: Calcium chloride (mM) | 16 | 24 | 32 |
| Dependent Variables | |||
| Y1: Particle size (nm) | Minimize | ||
| Y2: Drug encapsulation (%) | Maximize | ||
| Y3: Drug release (%) | Maximize | ||
| Formulation Code | Independent Variables | Responses | ||||
|---|---|---|---|---|---|---|
| X1 (% w/v) | X2 (% w/v) | X3 (mM) | Y1 (nm) | Y2 (%) | Y3 (%) | |
| FN1 | 0.10 | 0.40 | 16.00 | 121 | 90 | 70 |
| *FN2 | 0.20 | 0.40 | 24.00 | 142 | 72 | 49 |
| FN3 | 0.30 | 0.60 | 24.00 | 166 | 80 | 54 |
| FN4 | 0.10 | 0.20 | 24.00 | 151 | 86 | 55 |
| *FN5 | 0.20 | 0.40 | 24.00 | 137 | 73 | 47 |
| FN6 | 0.20 | 0.60 | 16.00 | 170 | 87 | 37 |
| *FN7 | 0.20 | 0.40 | 24.00 | 145 | 74 | 49 |
| FN8 | 0.10 | 0.60 | 24.00 | 123 | 93 | 74 |
| FN9 | 0.30 | 0.40 | 32.00 | 146 | 72 | 72 |
| FN10 | 0.20 | 0.60 | 32.00 | 168 | 75 | 52 |
| FN11 | 0.20 | 0.20 | 16.00 | 139 | 78 | 40 |
| *FN12 | 0.20 | 0.40 | 24.00 | 140 | 74 | 48 |
| FN13 | 0.10 | 0.40 | 32.00 | 149 | 70 | 77 |
| FN14 | 0.20 | 0.20 | 32.00 | 180 | 78 | 38 |
| *FN15 | 0.20 | 0.40 | 24.00 | 146 | 71 | 47 |
| FN16 | 0.30 | 0.40 | 16.00 | 138 | 67 | 58 |
| FN17 | 0.30 | 0.20 | 24.00 | 131 | 79 | 58 |
| Model | R2 | Adjusted R2 | Predicted R2 | SD | CV % | Desirability |
|---|---|---|---|---|---|---|
| Response: Y1 | 0.958 | |||||
| Quadratic | 0.9805 | 0.9554 | 0.8706 | 3.45 | 2.35 | |
| 2FI | 0.5886 | 0.3417 | −0.9643 | 13.25 | ||
| Linear | 0.2245 | 0.0456 | −0.5873 | 15.96 | ||
| Response: Y2 | 0.958 | |||||
| Quadratic | 0.9904 | 0.9780 | 0.9565 | 1.11 | 1.42 | |
| 2FI | 0.5934 | 0.3494 | −0.5409 | 6.01 | ||
| Linear | 0.3668 | 0.2207 | −0.1936 | 6.58 | ||
| Response: Y3 | 0.958 | |||||
| Quadratic | 0.9943 | 0.9870 | 0.9326 | 1.41 | 3.46 | |
| 2FI | 0.23391 | −0.2174 | −2.5687 | 13.70 | ||
| Linear | 0.1440 | −0.0536 | −0.7143 | 12.69 |
| Independent Variables | Optimized Composition | Observed Responses | Predicted Response | ||||
|---|---|---|---|---|---|---|---|
| Y1 (nm) | Y2 (%) | Y3 (%) | Y1 (nm) | Y2 (%) | Y3 (%) | ||
| X1:X2:X3 | 0.1%:0.48%:16.32 mM | 126 ± 8.70 | 91.06 ± 2.6 | 71.50 ± 6.5 | 121 | 93 | 72.21 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Karim, S.; Akhter, M.H.; Burzangi, A.S.; Alkreathy, H.; Alharthy, B.; Kotta, S.; Md, S.; Rashid, M.A.; Afzal, O.; Altamimi, A.S.A.; et al. Phytosterol-Loaded Surface-Tailored Bioactive-Polymer Nanoparticles for Cancer Treatment: Optimization, In Vitro Cell Viability, Antioxidant Activity, and Stability Studies. Gels 2022, 8, 219. https://doi.org/10.3390/gels8040219
Karim S, Akhter MH, Burzangi AS, Alkreathy H, Alharthy B, Kotta S, Md S, Rashid MA, Afzal O, Altamimi ASA, et al. Phytosterol-Loaded Surface-Tailored Bioactive-Polymer Nanoparticles for Cancer Treatment: Optimization, In Vitro Cell Viability, Antioxidant Activity, and Stability Studies. Gels. 2022; 8(4):219. https://doi.org/10.3390/gels8040219
Chicago/Turabian StyleKarim, Shahid, Md Habban Akhter, Abdulhadi S. Burzangi, Huda Alkreathy, Basma Alharthy, Sabna Kotta, Shadab Md, Md Abdur Rashid, Obaid Afzal, Abdulmalik S. A. Altamimi, and et al. 2022. "Phytosterol-Loaded Surface-Tailored Bioactive-Polymer Nanoparticles for Cancer Treatment: Optimization, In Vitro Cell Viability, Antioxidant Activity, and Stability Studies" Gels 8, no. 4: 219. https://doi.org/10.3390/gels8040219








