Synthesis of pH-Sensitive Cross-Linked Basil Seed Gum/Acrylic Acid Hydrogels by Free Radical Copolymerization Technique for Sustained Delivery of Captopril
Abstract
:1. Introduction
2. Experimental
2.1. Materials
2.2. Basil Seed Gum Extraction and Purification
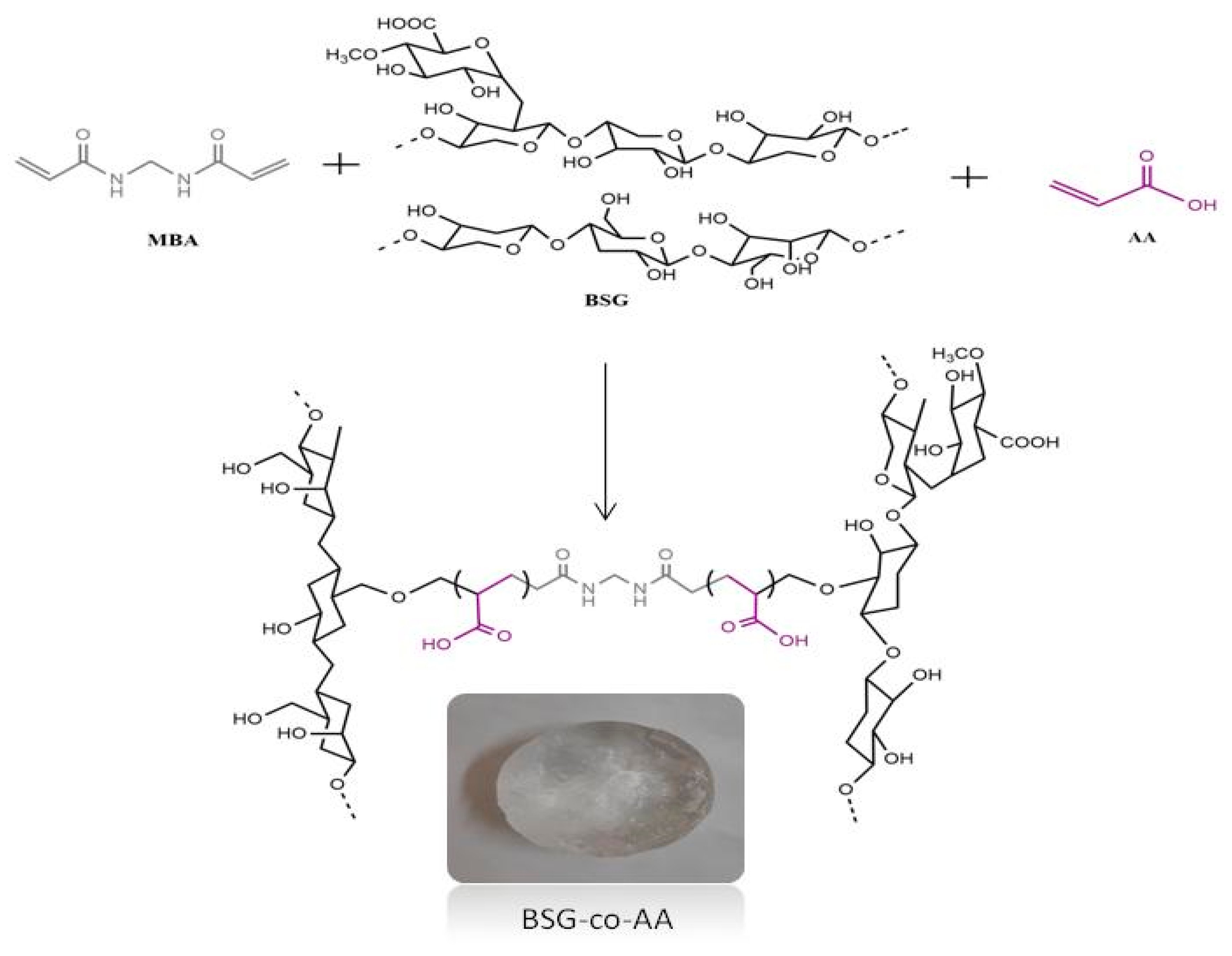
2.3. Basil-Seed-Gum-Based Hydrogels Fabrication
2.4. Characterization
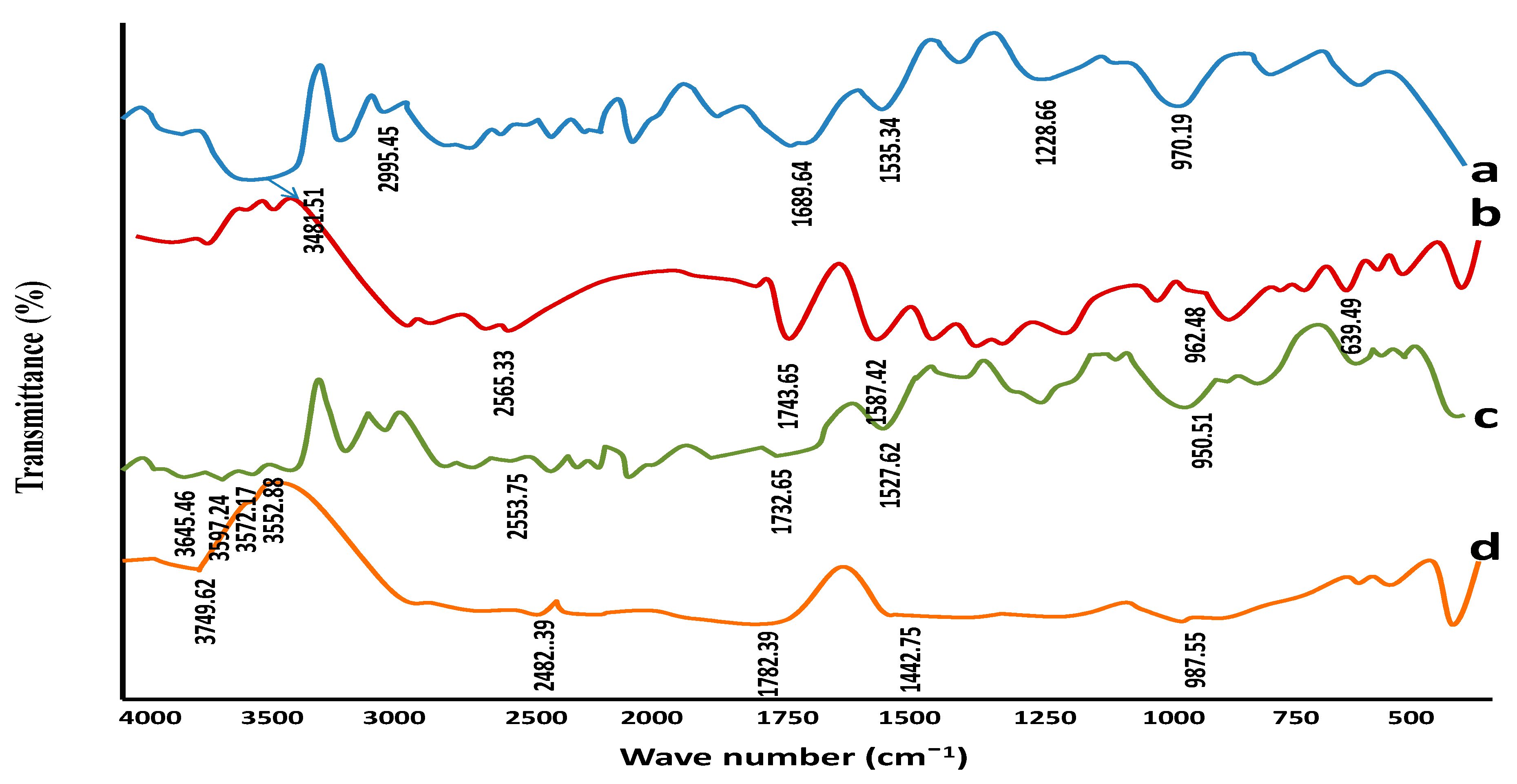
2.4.1. Fourier Transformed Infrared (FTIR) Spectroscopic Studies
2.4.2. Differential Scanning Calorimetric (DSC) and Thermo Gravimetric Analysis (TGA)
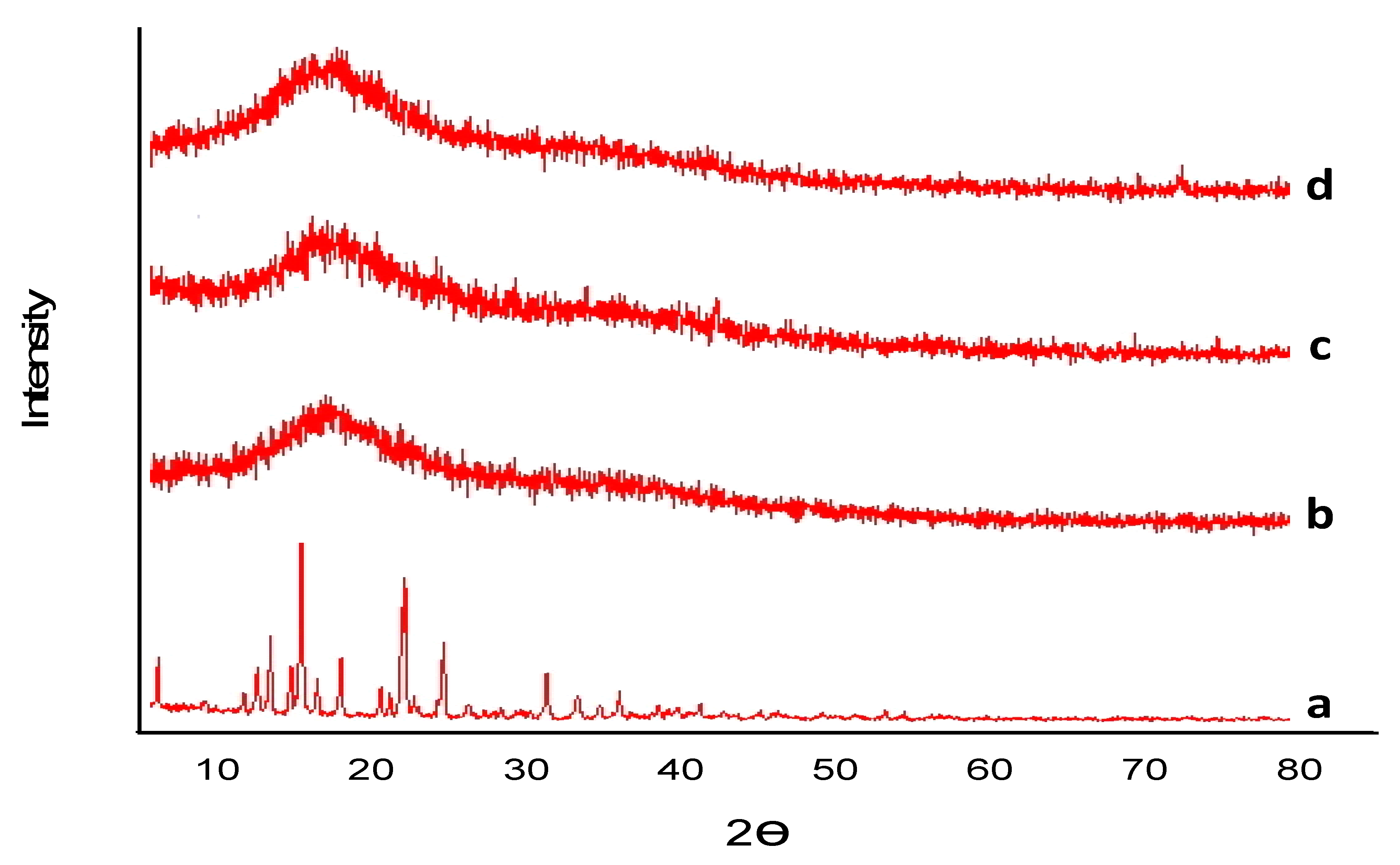
2.4.3. Powder X-ray Diffraction (PXRD)
2.4.4. Scanning Electron Microscopy (SEM)
2.4.5. Porosity Studies
2.4.6. Rheological Properties of BSG-co-poly (AA) Hydrogels
2.5. Determination of Gel%, Yield% and Gel Time
2.6. Swelling Studies
2.6.1. Dynamic Swelling Studies
2.6.2. Equilibrium Swelling Studies
2.7. Drug Loading
2.8. In Vitro Drug-Release Studies
2.9. Drug-Release Kinetics
2.10. Sol–Gel Analysis
2.11. Statistical Analysis
2.12. Acute Toxicology Study
3. Results and Discussion
3.1. Physicochemical Analysis of Prepared Hydrogels
3.1.1. Fourier Transformed Infrared (FTIR) Spectroscopic Studies
3.1.2. TGA (Thermo Gravimetric Analysis) and DSC (Differential Scanning Calorimetric)
3.1.3. Powder X-ray Diffraction Analysis
3.1.4. Scanning Electron Microscopy
3.1.5. Porosity Studies
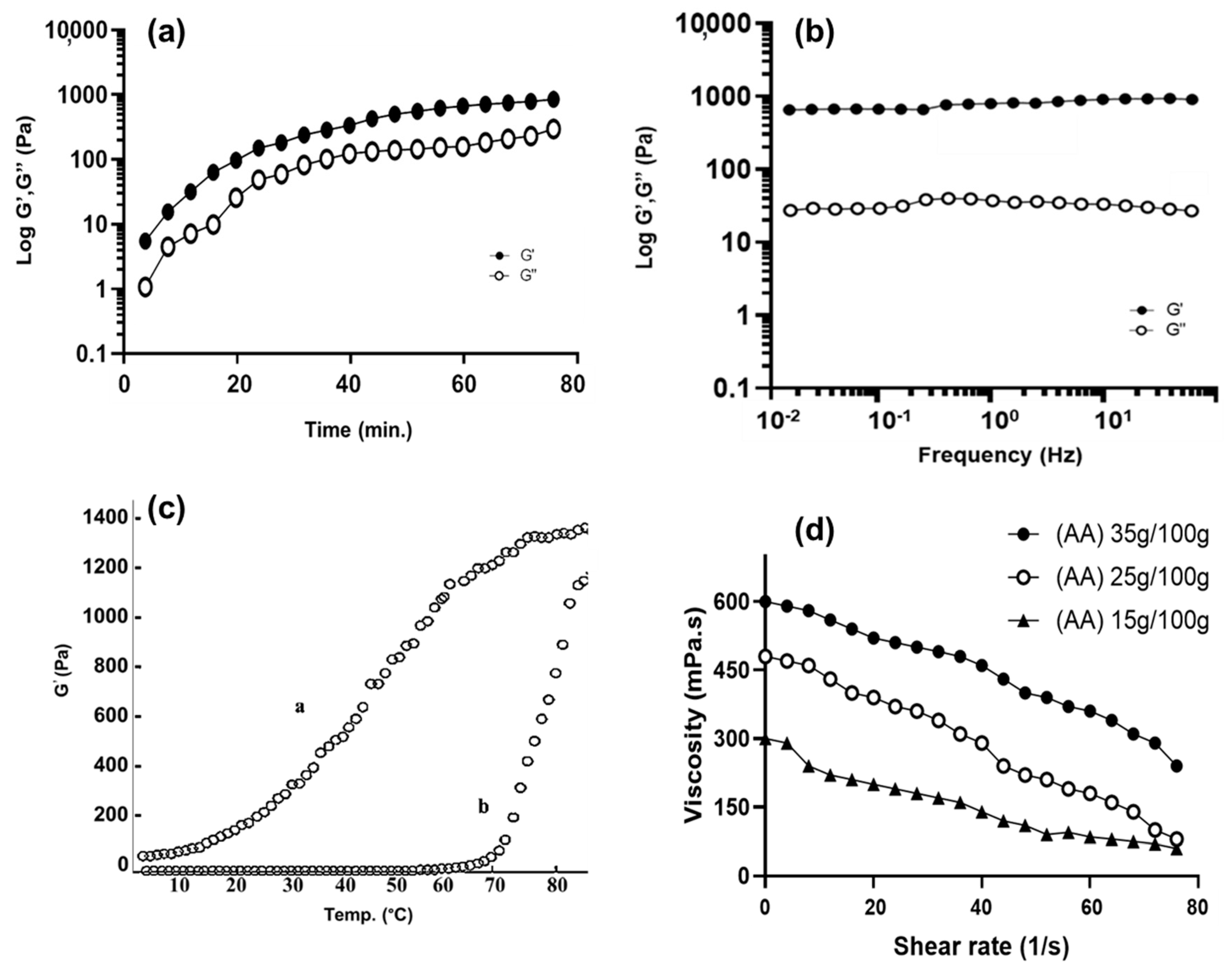
3.1.6. Rheological Properties of BSG-co-poly (AA) Hydrogels
3.2. Determination of Yield and Gel Percentage and Gelling Time
3.3. Swelling Behavior of Hydrogels
3.3.1. Effect of Medium pH on Hydrogel Swelling
3.3.2. Effect of BSG Content on Hydrogel Swelling
3.3.3. Effect of AA on Swelling of Prepared Hydrogel
3.3.4. Effect of MBA Content on Hydrogel Swelling
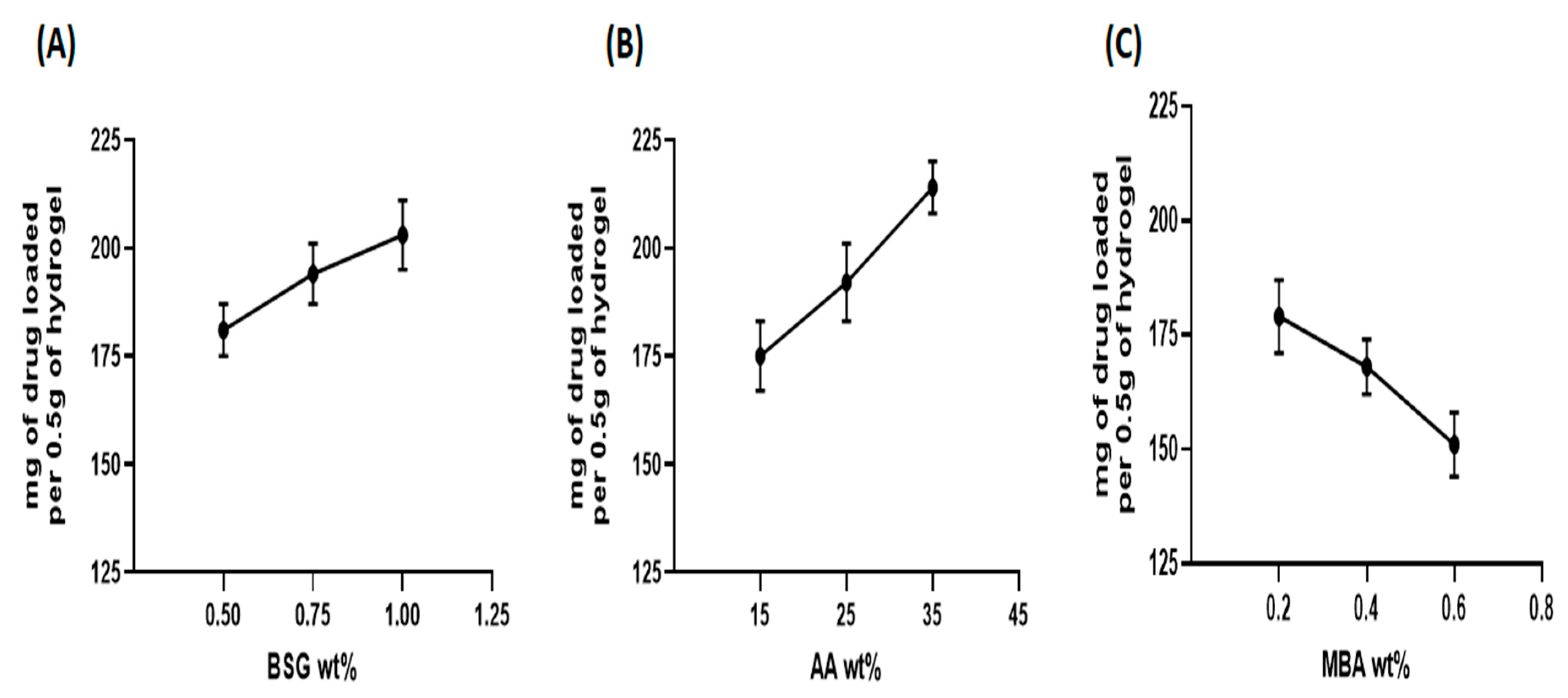
3.4. Drug Loading
3.5. In Vitro Drug-Release Studies
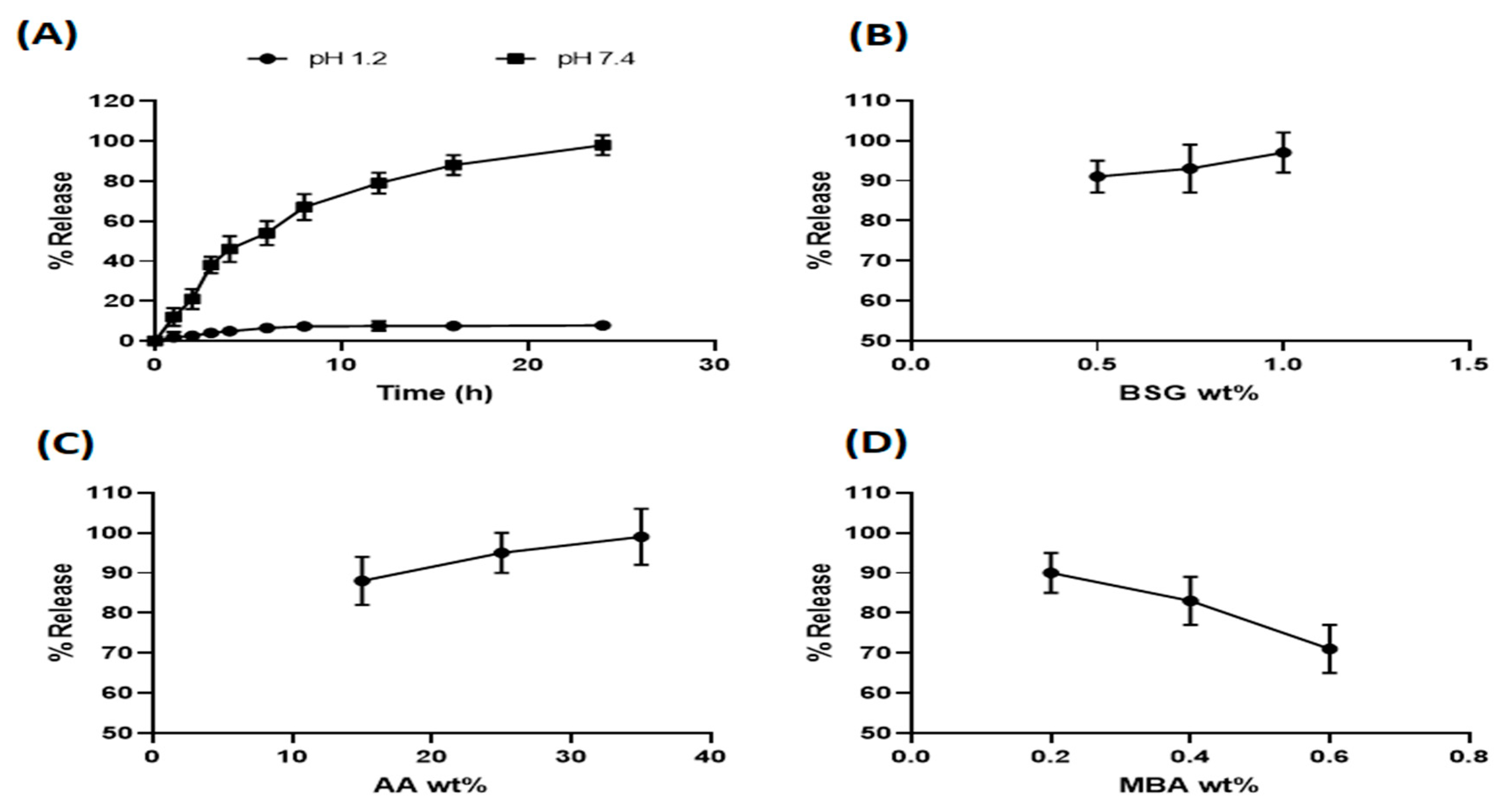
3.5.1. Effect of pH on Captopril Release from Hydrogels
3.5.2. Effect of Different Composition Parameters on Captopril Release from Hydrogels
3.6. Drug-Release Kinetic Models
3.7. Sol–Gel Analysis
3.8. General Signs of Acute Toxicology Study
3.8.1. Biochemical Plasma Analysis
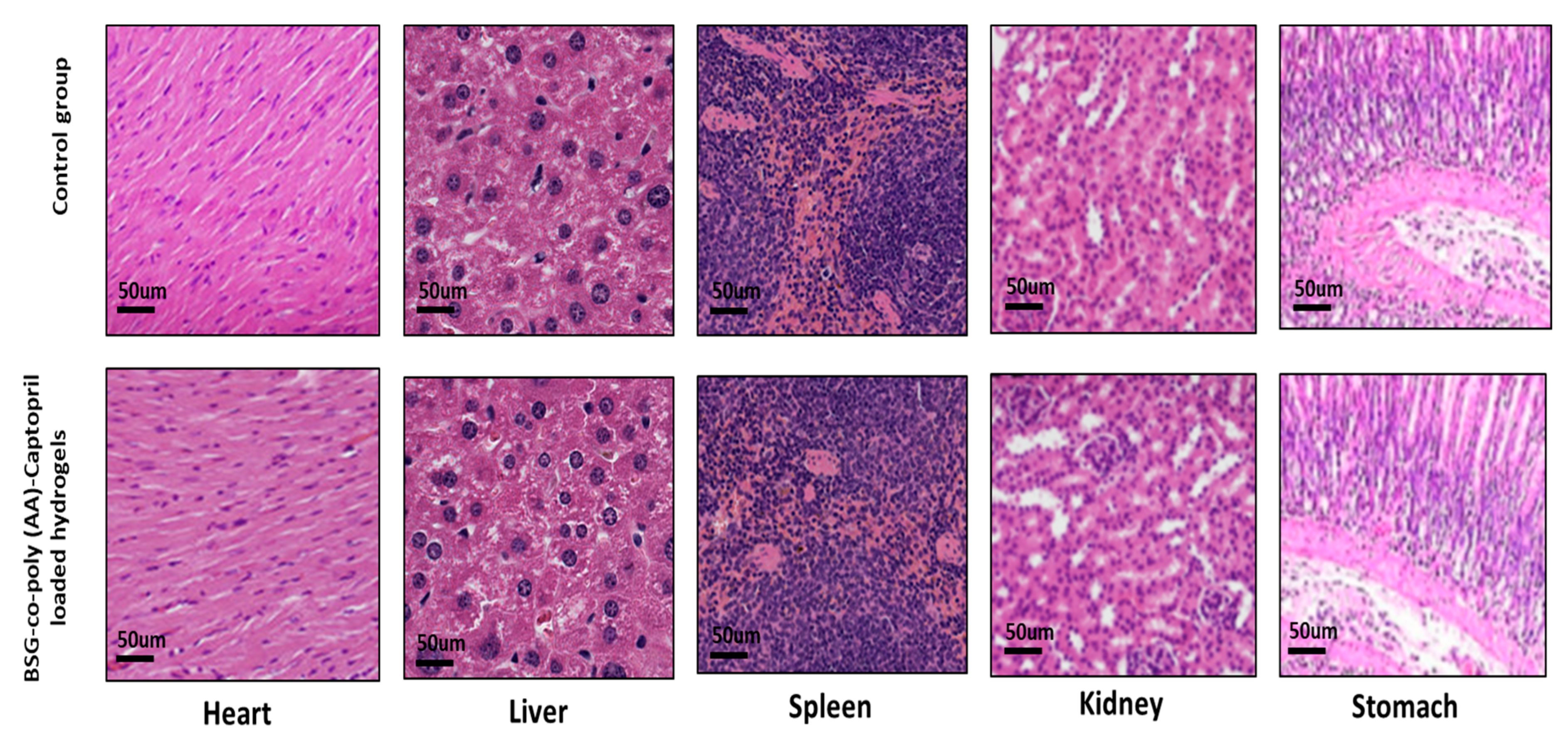
3.8.2. Histological Study of Vital Organs
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Vasir, J.K.; Tambwekar, K.; Garg, S. Bioadhesive microspheres as a controlled drug delivery system. Int. J. Pharm. 2003, 255, 13–32. [Google Scholar] [CrossRef]
- Bheemidi, V.S.; Tiruckovela, M.; Varanasi, P. An imperative note on novel drug delivery systems. J. Nanomed. Nanotech 2011, 2, 100125. [Google Scholar] [CrossRef]
- Peppas, N.A.; Hilt, J.Z.; Khademhosseini, A.; Langer, R. Hydrogels in biology and medicine: From molecular principles to bionanotechnology. Adv. Mater. 2006, 18, 1345–1360. [Google Scholar] [CrossRef]
- Hoffman, A.S. Hydrogels for biomedical applications. Adv. Drug Deliv. Rev. 2012, 64, 18–23. [Google Scholar] [CrossRef]
- Chandel, A.K.S.; Shimizu, A.; Hasegawa, K.; Ito, T. Advancement of Biomaterial-Based Postoperative Adhesion Barriers. Macromol. Biosci. 2021, 21, 2000395. [Google Scholar] [CrossRef]
- Paroha, S.; Chandel, A.K.S.; Dubey, R.D. Nanosystems for drug delivery of coenzyme Q10. Environ. Chem. Lett. 2018, 16, 71–77. [Google Scholar] [CrossRef]
- Chandel, A.K.S.; Kumar, C.U.; Jewrajka, S.K. Effect of polyethylene glycol on properties and drug encapsulation–release performance of biodegradable/cytocompatible agarose–polyethylene glycol–polycaprolactone amphiphilic co-network gels. ACS Appl. Mater. Interfaces 2016, 8, 3182–3192. [Google Scholar] [CrossRef]
- Chandel, A.K.S.; Kannan, D.; Nutan, B.; Singh, S.; Jewrajka, S.K. Dually crosslinked injectable hydrogels of poly (ethylene glycol) and poly [(2-dimethylamino) ethyl methacrylate]-b-poly (N-isopropyl acrylamide) as a wound healing promoter. J. Mater. Chem. B 2017, 5, 4955–4965. [Google Scholar] [CrossRef]
- Bera, A.; Chandel, A.K.S.; Kumar, C.U.; Jewrajka, S.K. Degradable/cytocompatible and pH responsive amphiphilic conetwork gels based on agarose-graft copolymers and polycaprolactone. J. Mater. Chem. B 2015, 3, 8548–8557. [Google Scholar] [CrossRef]
- Chandel, A.K.S.; Bera, A.; Nutan, B.; Jewrajka, S.K. Reactive compatibilizer mediated precise synthesis and application of stimuli responsive polysaccharides-polycaprolactone amphiphilic co-network gels. Polymer 2016, 99, 470–479. [Google Scholar] [CrossRef]
- Zhang, Z.; Chen, L.; Zhao, C.; Bai, Y.; Deng, M.; Shan, H.; Zhuang, X.; Chen, X.; Jing, X. Thermo-and pH-responsive HPC-g-AA/AA hydrogels for controlled drug delivery applications. Polymer 2011, 52, 676–682. [Google Scholar] [CrossRef]
- Omidian, H.; Rocca, J.G.; Park, K. Advances in superporous hydrogels. J. Control. Release 2005, 102, 3–12. [Google Scholar] [CrossRef] [PubMed]
- Schexnailder, P.; Schmidt, G. Nanocomposite polymer hydrogels. Colloid Polym. Sci. 2009, 287, 1–11. [Google Scholar] [CrossRef]
- Wu, W.; Wang, D.-S. A fast pH-responsive IPN hydrogel: Synthesis and controlled drug delivery. React. Funct. Polym. 2010, 70, 684–691. [Google Scholar] [CrossRef]
- Li, Y.; Yang, C.; Khan, M.; Liu, S.; Hedrick, J.L.; Yang, Y.-Y.; Ee, P.-L.R. Nanostructured PEG-based hydrogels with tunable physical properties for gene delivery to human mesenchymal stem cells. Biomaterials 2012, 33, 6533–6541. [Google Scholar] [CrossRef]
- Qian, C.; Zhang, T.; Gravesande, J.; Baysah, C.; Song, X.; Xing, J. Injectable and self-healing polysaccharide-based hydrogel for pH-responsive drug release. Int. J. Biol. Macromol. 2019, 123, 140–148. [Google Scholar] [CrossRef]
- Mahkam, M. Modified chitosan cross-linked starch polymers for oral insulin delivery. J. Bioact. Compat. Polym. 2010, 25, 406–418. [Google Scholar] [CrossRef]
- Guilherme, M.R.; Campese, G.M.; Radovanovic, E.; Rubira, A.F.; Feitosa, J.P.; Muniz, E.C. Morphology and water affinity of superabsorbent hydrogels composed of methacrylated cashew gum and acrylamide with good mechanical properties. Polymer 2005, 46, 7867–7873. [Google Scholar] [CrossRef]
- Ata, S.; Rasool, A.; Islam, A.; Bibi, I.; Rizwan, M.; Azeem, M.K.; Iqbal, M. Loading of Cefixime to pH sensitive chitosan based hydrogel and investigation of controlled release kinetics. Int. J. Biol. Macromol. 2019, 155, 1236–1244. [Google Scholar] [CrossRef]
- Teodorescu, M.; Morariu, S.; Bercea, M.; Săcărescu, L. Viscoelastic and structural properties of poly (vinyl alcohol)/poly (vinylpyrrolidone) hydrogels. RSC Adv. 2016, 6, 39718–39727. [Google Scholar] [CrossRef]
- Yoo, S.-H.; Song, Y.-B.; Chang, P.-S.; Lee, H.G. Microencapsulation of α-tocopherol using sodium alginate and its controlled release properties. Int. J. Biol. Macromol. 2006, 38, 25–30. [Google Scholar] [CrossRef] [PubMed]
- Shantha, K.; Harding, D. Preparation and in-vitro evaluation of poly [N-vinyl-2-pyrrolidone-polyethylene glycol diacrylate]-chitosan interpolymeric pH-responsive hydrogels for oral drug delivery. Int. J. Pharm. 2000, 207, 65–70. [Google Scholar] [CrossRef]
- Sharma, K.; Kumar, V.; Chaudhary, B.; Kaith, B.; Kalia, S.; Swart, H. Application of biodegradable superabsorbent hydrogel composite based on Gum ghatti-co-poly (acrylic acid-aniline) for controlled drug delivery. Polym. Degrad. Stab. 2016, 124, 101–111. [Google Scholar] [CrossRef]
- Bashir, S.; Teo, Y.Y.; Ramesh, S.; Ramesh, K. Synthesis, characterization, properties of N-succinyl chitosan-g-poly (methacrylic acid) hydrogels and in vitro release of theophylline. Polymer 2016, 92, 36–49. [Google Scholar] [CrossRef]
- Wichterle, O.; Lim, D. Hydrophilic gels for biological use. Nature 1960, 185, 117–118. [Google Scholar] [CrossRef]
- Chaduc, I.; Crepet, A.s.; Boyron, O.; Charleux, B.; D’Agosto, F.; Lansalot, M. Effect of the pH on the RAFT polymerization of acrylic acid in water. Application to the synthesis of poly (acrylic acid)-stabilized polystyrene particles by RAFT emulsion polymerization. Macromolecules 2013, 46, 6013–6023. [Google Scholar] [CrossRef]
- Silverstein, M.S. PolyHIPEs: Recent advances in emulsion-templated porous polymers. Prog. Polym. Sci. 2014, 39, 199–234. [Google Scholar] [CrossRef]
- Hosseini-Parvar, S.; Matia-Merino, L.; Goh, K.; Razavi, S.M.A.; Mortazavi, S.A. Steady shear flow behavior of gum extracted from Ocimum basilicum L. seed: Effect of concentration and temperature. J. Food Eng. 2010, 101, 236–243. [Google Scholar] [CrossRef]
- BahramParvar, M.; Goff, H.D. Basil seed gum as a novel stabilizer for structure formation and reduction of ice recrystallization in ice cream. Dairy Sci. Technol. 2013, 93, 273–285. [Google Scholar] [CrossRef] [Green Version]
- Razavi, S.M.; Mortazavi, S.A.; Matia-Merino, L.; Hosseini-Parvar, S.H.; Motamedzadegan, A.; Khanipour, E. Optimisation study of gum extraction from Basil seeds (Ocimum basilicum L.). Int. J. Food Sci. Technol. 2009, 44, 1755–1762. [Google Scholar] [CrossRef]
- Osano, J.; Matia-Merino, L.; Hosseini-Parvar, S.; Golding, M.; Goh, K. Adsorption properties of basil (Ocimum basilicum L.) seed gum. USM R D 2010, 18, e117. [Google Scholar]
- Osano, J.P.; Hosseini-Parvar, S.H.; Matia-Merino, L.; Golding, M. Emulsifying properties of a novel polysaccharide extracted from basil seed (Ocimum bacilicum L.): Effect of polysaccharide and protein content. Food Hydrocoll. 2014, 37, 40–48. [Google Scholar] [CrossRef]
- Naji-Tabasi, S.; Razavi, S.M.A.; Mohebbi, M.; Malaekeh-Nikouei, B. New studies on basil (Ocimum bacilicum L.) seed gum: Part I–Fractionation, physicochemical and surface activity characterization. Food Hydrocoll. 2016, 52, 350–358. [Google Scholar] [CrossRef]
- Rayegan, A.; Allafchian, A.; Sarsari, I.A.; Kameli, P. Synthesis and characterization of basil seed mucilage coated Fe3O4 magnetic nanoparticles as a drug carrier for the controlled delivery of cephalexin. Int. J. Biol. Macromol. 2018, 113, 317–328. [Google Scholar] [CrossRef] [PubMed]
- Sabzevar, Z.S.M.; Mehrshad, M.; Naimipour, M. A biological magnetic nano-hydrogel based on basil seed mucilage: Study of swelling ratio and drug delivery. Iran. Polym. J. 2021, 30, 485–493. [Google Scholar] [CrossRef]
- Alvarez-Lorenzo, C.; Blanco-Fernandez, B.; Puga, A.M.; Concheiro, A. Crosslinked ionic polysaccharides for stimuli-sensitive drug delivery. Adv. Drug Deliv. Rev. 2013, 65, 1148–1171. [Google Scholar] [CrossRef]
- Tareq, A.Z.; Hussein, M.S.; Mustafa, A.M. Synthesis and characterization of PVA-gelatin hydrogel membranes for controlled delivery of captopril. Int. Res. J. Pure Appl. Chem. 2016, 12, 1–10. [Google Scholar] [CrossRef]
- Patil, B.S.; Sonawane, S.J.; Kulkarni, U.; Hariprasanna, R. Formulation and in-vitro evaluation of captopril floating matrix tablets using HPMC 50cps. J. Pharm. Sci. Biosci. Res. 2012, 2, 97–102. [Google Scholar]
- Naji-Tabasi, S.; Razavi, S.M.A.; Mehditabar, H. Fabrication of basil seed gum nanoparticles as a novel oral delivery system of glutathione. Carbohydr. Polym. 2017, 157, 1703–1713. [Google Scholar] [CrossRef]
- Bukhari, S.M.H.; Khan, S.; Rehanullah, M.; Ranjha, N.M. Synthesis and characterization of chemically cross-linked acrylic acid/gelatin hydrogels: Effect of pH and composition on swelling and drug release. Int. J. Polym. Sci. 2015, 2015, 187961. [Google Scholar] [CrossRef]
- Ranjha, N.M.; Madni, A.; Bakar, A.A.; Talib, N.; Ahmad, S.; Ahmad, H. Preparation and characterization of isosorbide mononitrate hydrogels obtained by free-radical polymerization for site-specific delivery. Trop. J. Pharm. Res. 2014, 13, 1979–1985. [Google Scholar] [CrossRef] [Green Version]
- Devi, N.; Narzary, A. Release dynamics of brufen from a drug-loaded polymer hydrogel containing polyvinyl alcohol, 2-acrylamide-2-methylpropane sulfonic acid and acrylamide. Int. J. Polym. Mater. 2012, 61, 821–833. [Google Scholar] [CrossRef]
- Ali, L.; Ahmad, M.; Usman, M. Evaluation of cross-linked hydroxypropyl methylcellulose graft-methacrylic acid copolymer as extended release oral drug carrier. Cell. Chem. Technol. 2015, 49, 143–151. [Google Scholar]
- Singh, B.; Sharma, V. Crosslinking of poly (vinylpyrrolidone)/acrylic acid with tragacanth gum for hydrogels formation for use in drug delivery applications. Carbohydr. Polym. 2017, 157, 185–195. [Google Scholar] [CrossRef] [PubMed]
- Calina, I.; Demeter, M.; Vancea, C.; Scarisoreanu, A.; Meltzer, V. E-beam radiation synthesis of xanthan-gum/carboxymethylcellulose superabsorbent hydrogels with incorporated graphene oxide. J. Macromol. Sci. Part A 2018, 55, 260–268. [Google Scholar] [CrossRef]
- Alam, K.; Iqbal, M.; Hasan, A.; Al-Maskari, N. Rheological characterization of biological hydrogels in aqueous state. J. Appl. Biotechnol. Rep. 2020, 7, 171–175. [Google Scholar]
- Sánchez-Cid, P.; Jiménez-Rosado, M.; Alonso-González, M.; Romero, A.; Perez-Puyana, V. Applied Rheology as Tool for the Assessment of Chitosan Hydrogels for Regenerative Medicine. Polymers 2021, 13, 2189. [Google Scholar] [CrossRef]
- Dergunov, S.A.; Nam, I.K.; Mun, G.A.; Nurkeeva, Z.S.; Shaikhutdinov, E.M. Radiation synthesis and characterization of stimuli-sensitive chitosan–polyvinyl pyrrolidone hydrogels. Radiat. Phys. Chem. 2005, 72, 619–623. [Google Scholar] [CrossRef]
- Tulain, U.R.; Ahmad, M.; Rashid, A.; Malik, M.Z.; Iqbal, F.M. Fabrication of Ph-responsive hydrogel and its in vitro and in vivo evaluation. Adv. Polym. Technol. 2018, 37, 290–304. [Google Scholar] [CrossRef]
- George, D.J.; Shipp, A.M. Exposure assessment. Drugs Pharm. Sci. 2000, 103, 283–304. [Google Scholar] [CrossRef]
- Srivastava, A.; Khare, B.; Argal, R.; Patel, S. Microdetermination of anti-hypertensive drug captopril using 2, 6-dichlorophenol indophenol. Indian J. Chem. 2003, 42, 3036–3040. [Google Scholar]
- Tantiwatcharothai, S.; Prachayawarakorn, J. Characterization of an antibacterial wound dressing from basil seed (Ocimum basilicum L.) mucilage-ZnO nanocomposite. Int. J. Biol. Macromol. 2019, 135, 133–140. [Google Scholar] [CrossRef] [PubMed]
- Iqbal, F.M.; Iqbal, S.; Nasir, B.; Hassan, W.; Ahmed, H.; Iftikhar, S.Y. Formulation of captopril-loaded hydrogel by microwave-assisted free radical polymerization and its evaluation. Polym. Bull. 2021, 1–21. [Google Scholar] [CrossRef]
- Giri, T.; Choudhary, C.; Alexander, A. Sustained release of diltiazem hydrochloride from cross-linked biodegradable IPN hydrogel beads of pectin and modified xanthan gum. Indian J. Pharm. Sci. 2013, 75, 619. [Google Scholar]
- Huacai, G.; Wan, P.; Dengke, L. Graft copolymerization of chitosan with acrylic acid under microwave irradiation and its water absorbency. Carbohydr. Polym. 2006, 66, 372–378. [Google Scholar] [CrossRef]
- Brinker, C.J.; Sehgal, R.; Hietala, S.; Deshpande, R.; Smith, D.; Loy, D.; Ashley, C. Sol-gel strategies for controlled porosity inorganic materials. J. Membr. Sci. 1994, 94, 85–102. [Google Scholar] [CrossRef]
- Chen, X.Y.; Low, H.R.; Loi, X.Y.; Merel, L.; Mohd Cairul Iqbal, M.A. Fabrication and evaluation of bacterial nanocellulose/poly (acrylic acid)/graphene oxide composite hydrogel: Characterizations and biocompatibility studies for wound dressing. J. Biomed. Mater. Res. Part B Appl. Biomater. 2019, 107, 2140–2151. [Google Scholar] [CrossRef]
- Babaei, J.; Mohammadian, M.; Madadlou, A. Gelatin as texture modifier and porogen in egg white hydrogel. Food Chem. 2019, 270, 189–195. [Google Scholar] [CrossRef]
- Chavda, H.V.; Patel, R.D.; Modhia, I.P.; Patel, C.N. Preparation and characterization of superporous hydrogel based on different polymers. Int. J. Pharm. Investig. 2012, 2, 134. [Google Scholar] [CrossRef] [Green Version]
- Zuidema, J.M.; Rivet, C.J.; Gilbert, R.J.; Morrison, F.A. A protocol for rheological characterization of hydrogels for tissue engineering strategies. J. Biomed. Mater. Res. Part B Appl. Biomater. 2014, 102, 1063–1073. [Google Scholar] [CrossRef]
- Lin-Gibson, S.; Walls, H.J.; Kennedy, S.B.; Welsh, E.R. Reaction kinetics and gel properties of blocked diisocyinate crosslinked chitosan hydrogels. Carbohydr. Polym. 2003, 54, 193–199. [Google Scholar] [CrossRef]
- Li, R.; Liu, Q.; Wu, H.; Wang, K.; Li, L.; Zhou, C.; Ao, N. Preparation and characterization of in-situ formable liposome/chitosan composite hydrogels. Mater. Lett. 2018, 220, 289–292. [Google Scholar] [CrossRef]
- Draget, K.; Skjåk-Bræk, G.; Christensen, B.a.; Gåserød, O.; Smidsrød, O. Swelling and partial solubilization of alginic acid gel beads in acidic buffer. Carbohydr. Polym. 1996, 29, 209–215. [Google Scholar] [CrossRef]
- Feng, D.; Bai, B.; Ding, C.; Wang, H.; Suo, Y. Synthesis and swelling behaviors of yeast-g-poly (acrylic acid) superabsorbent co-polymer. Ind. Eng. Chem. Res. 2014, 53, 12760–12769. [Google Scholar] [CrossRef]
- Şolpan, D.L.; Kölge, Z.; Torun, M. Preparation and Characterization of Poly (N-Vinylpyrrolidone-co-Methacrylic Acid) Hydrogels. J. Macromol. Sci. Part A Pure Appl. Chem. 2005, 42, 705–721. [Google Scholar] [CrossRef]
- Li, X.; Wu, W.; Wang, J.; Duan, Y. The swelling behavior and network parameters of guar gum/poly (acrylic acid) semi-interpenetrating polymer network hydrogels. Carbohydr. Polym. 2006, 66, 473–479. [Google Scholar] [CrossRef]
- Tally, M.; Atassi, Y. Synthesis and characterization of pH-sensitive superabsorbent hydrogels based on sodium alginate-g-poly (acrylic acid-co-acrylamide) obtained via an anionic surfactant micelle templating under microwave irradiation. Polym. Bull. 2016, 73, 3183–3208. [Google Scholar] [CrossRef]
- Naji-Tabasi, S.; Razavi, S.M.A. Functional properties and applications of basil seed gum: An overview. Food Hydrocoll. 2017, 73, 313–325. [Google Scholar] [CrossRef]
- Gad, Y.; Aly, R.; Abdel-Aal, S. Synthesis and characterization of Na-alginate/acrylamide hydrogel and its application in dye removal. J. Appl. Polym. Sci. 2011, 120, 1899–1906. [Google Scholar] [CrossRef]
- Davidson, G.R., III; Peppas, N.A. Solute and penetrant diffusion in swellable polymers: V. Relaxation-controlled transport in P (HEMA-co-MMA) copolymers. J. Control. Release 1986, 3, 243–258. [Google Scholar] [CrossRef]
- Garcıa, D.; Escobar, J.; Bada, N.; Casquero, J.; Hernáez, E.; Katime, I. Synthesis and characterization of poly (methacrylic acid) hydrogels for metoclopramide delivery. Eur. Polym. J. 2004, 40, 1637–1643. [Google Scholar] [CrossRef]
- Lim, L.S.; Rosli, N.A.; Ahmad, I.; Mat Lazim, A.; Mohd Amin, M.C.I. Synthesis and swelling behavior of pH-sensitive semi-IPN superabsorbent hydrogels based on poly (acrylic acid) reinforced with cellulose nanocrystals. Nanomaterials 2017, 7, 399. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Murthy, P.K.; Mohan, Y.M.; Sreeramulu, J.; Raju, K.M. Semi-IPNs of starch and poly (acrylamide-co-sodium methacrylate): Preparation, swelling and diffusion characteristics evaluation. React. Funct. Polym. 2006, 66, 1482–1493. [Google Scholar] [CrossRef]
- Samanta, H.S.; Ray, S.K. Synthesis, characterization, swelling and drug release behavior of semi-interpenetrating network hydrogels of sodium alginate and polyacrylamide. Carbohydr. Polym. 2014, 99, 666–678. [Google Scholar] [CrossRef] [PubMed]
- Pandey, M.; Mohd Amin, M.C.I.; Ahmad, N.; Abeer, M.M. Rapid synthesis of superabsorbent smart-swelling bacterial cellulose/acrylamide-based hydrogels for drug delivery. Int. J. Polym. Sci. 2013, 2013, 905471. [Google Scholar] [CrossRef]
- Matricardi, P.; Di Meo, C.; Coviello, T.; Hennink, W.E.; Alhaique, F. Interpenetrating polymer networks polysaccharide hydrogels for drug delivery and tissue engineering. Adv. Drug Deliv. Rev. 2013, 65, 1172–1187. [Google Scholar] [CrossRef]
- Wang, Q.; Hu, X.; Du, Y.; Kennedy, J.F. Alginate/starch blend fibers and their properties for drug controlled release. Carbohydr. Polym. 2010, 82, 842–847. [Google Scholar] [CrossRef]
- Tan, L.; Xu, X.; Song, J.; Luo, F.; Qian, Z. Synthesis, characterization, and acute oral toxicity evaluation of pH-sensitive hydrogel based on MPEG, poly (ε-caprolactone), and itaconic acid. BioMed Res. Int. 2013, 2013, 239838. [Google Scholar] [CrossRef] [Green Version]


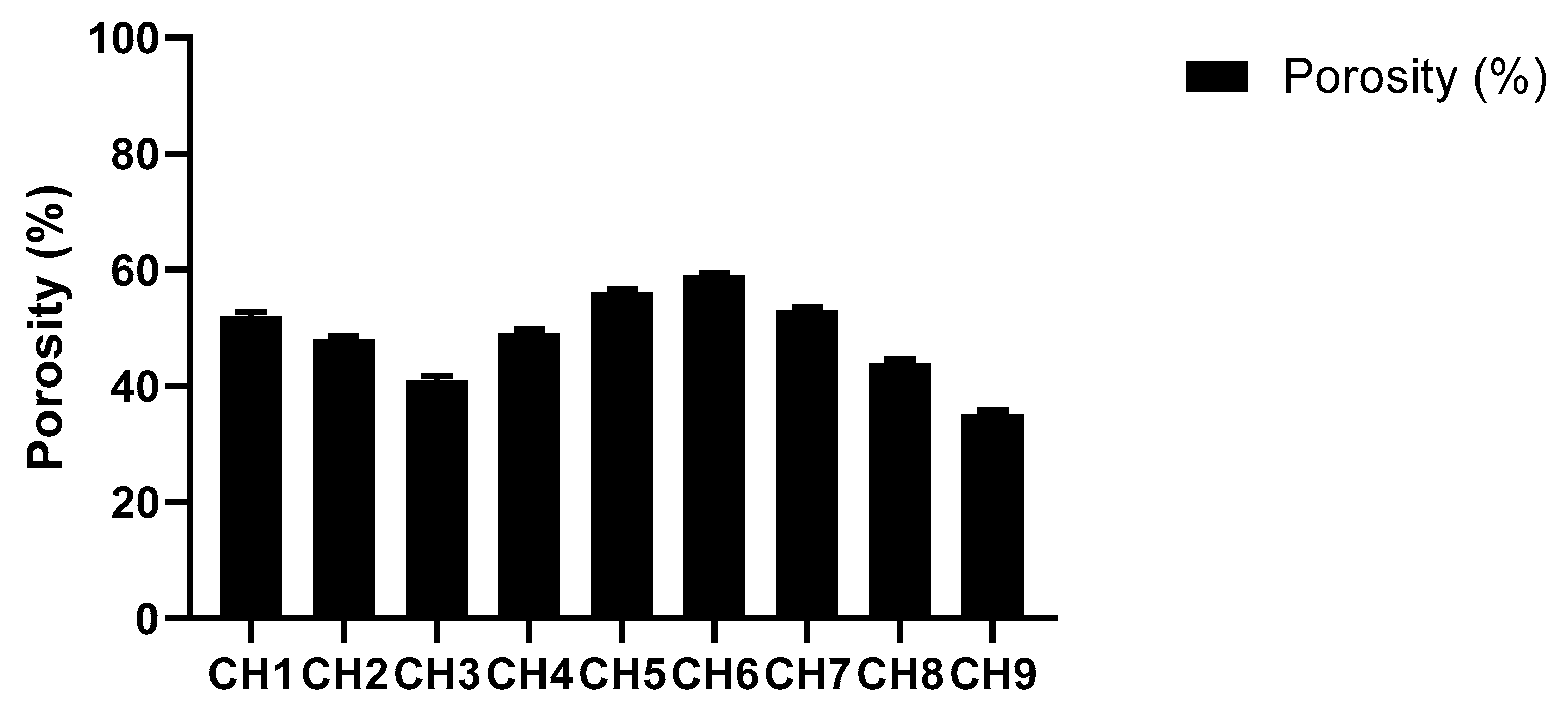



| Formulations | Polymer (BSG) % | Monomer (AA) % | Comonomer (MBA) % | Initiator KPS % |
|---|---|---|---|---|
| CH1 | 0.5 | 20 | 0.25 | 0.15 |
| CH2 | 0.75 | 20 | 0.25 | 0.15 |
| CH3 | 1 | 20 | 0.25 | 0.15 |
| CH4 | 0.5 | 15 | 0.25 | 0.15 |
| CH5 | 0.5 | 25 | 0.25 | 0.15 |
| CH6 | 0.5 | 35 | 0.25 | 0.15 |
| CH7 | 0.5 | 20 | 0.2 | 0.15 |
| CH8 | 0.5 | 20 | 0.4 | 0.15 |
| CH9 | 0.5 | 20 | 0.6 | 0.15 |
| Formulations | Zero Order R2 | First Order R2 | Higuchi R2 | Korsmeyer-Peppas | |
|---|---|---|---|---|---|
| R2 | n | ||||
| CH1 | 0.913 | 0.964 | 0.864 | 0.966 | 0.770 |
| CH2 | 0.906 | 0.960 | 0.861 | 0.954 | 0.766 |
| CH3 | 0.880 | 0.952 | 0.857 | 0.944 | 0.746 |
| CH4 | 0.936 | 0.974 | 0.878 | 0.978 | 0.779 |
| CH5 | 0.901 | 0.975 | 0.891 | 0.969 | 0.733 |
| CH6 | 0.802 | 0.969 | 0.868 | 0.933 | 0.658 |
| CH7 | 0.884 | 0.962 | 0.863 | 0.945 | 0.743 |
| CH8 | 0.950 | 0.954 | 0.810 | 0.960 | 0.883 |
| CH9 | 0.969 | 0.968 | 0.807 | 0.974 | 0.918 |
| Parameters to Observe | Control Group (A) | Treated Group (B) | p-Values |
|---|---|---|---|
| Mortality | Nil | Nil | |
| Sign of illness | Nil | Nil | |
| Alertness | + | + | |
| Dermal toxicity/Dermal irritation | Nil | Nil | |
| Ocular toxicity/Eye Irritation & Lacrimation | Nil | Nil | |
| Light Reflex | + | + | |
| Food intake | |||
| Pre-treatment | 3.2 ± 1.6 | 3.4 ± 1.6 | p > 0.05 |
| Day 1 | 3.7 ± 1.8 | 2.9 ± 1.4 | p > 0.05 |
| Day 2 | 3.9 ± 2.9 | 3.4 ± 2.2 | p > 0.05 |
| Day 5 | 4.1 ± 1.0 | 3.8 ± 1.8 | p > 0.05 |
| Day 7 | 4.2 ± 1.4 | 4.1 ± 1.2 | p > 0.05 |
| Day 14 | 4.8 ± 1.2 | 4.4 ± 2.6 | p > 0.05 |
| Water intake | |||
| Pre-treatment | 7.8 ± 2.4 | 7.4 ± 2.0 | p > 0.05 |
| Day 1 | 8.4 ± 1.4 | 8.2 ± 1.0 | p > 0.05 |
| Day 2 | 9.2 ± 1.2 | 8.8 ± 1.6 | p > 0.05 |
| Day 5 | 11.2 ± 0.9 | 10.4 ± 1.8 | p > 0.05 |
| Day 7 | 11.6 ± 1.2 | 10.9 ± 2.1 | p > 0.05 |
| Day 14 | 13.2 ± 1.4 | 12.8 ± 1.8 | p > 0.05 |
| Body weight | |||
| Pre-treatment | 26.5 ± 2.4 | 27.5 ± 1.2 | p > 0.05 |
| Day 1 | 26.8 ± 1.2 | 27.9 ± 2.4 | p > 0.05 |
| Day 2 | 27.5 ± 1.6 | 28.3 ± 1.2 | p > 0.05 |
| Day 5 | 28.5 ± 2.2 | 28.8 ± 3.2 | p > 0.05 |
| Day 7 | 29.8 ± 1.4 | 30.4 ± 2.2 | p > 0.05 |
| Day 14 | 30.5 ± 1.6 | 31.2 ± 1.4 | p > 0.05 |
| Plasma Analysis | Control Group (A) | Treated Group (B) | p-Value |
|---|---|---|---|
| ALT (IU/L) | 58 ± 2.9 | 55 ± 4.3 | p > 0.05 |
| AST (IU/L) | 156 ± 8.1 | 144 ± 7.3 | p > 0.05 |
| ALP (IU/L) | 123.59 ± 1.9 | 119.95 ± 2.4 | p > 0.05 |
| Cholesterol (mg/dL) | 137 ± 4.3 | 126 ± 6.2 | p > 0.05 |
| Triglyceride (mg/dL) | 122 ± 2.8 | 110 ± 4.4 | p > 0.05 |
| Creatinine (mg/dL) | 0.43 ± 1.2 | 0.38 ± 0.9 | p > 0.05 |
| Glucose (mg/dL) | 112.5 ± 0.2 | 116.1 ± 0.5 | p > 0.05 |
| Urea (mg/dL) | 58 ± 3.2 | 62 ± 2.3 | p > 0.05 |
| Uric acid (mg/dL) | 4.4 ± 2.6 | 5.6 ± 1.2 | p > 0.05 |
| Hemoglobin (g/dL) | 13.9 ± 0.7 | 14.2 ± 0.5 | p > 0.05 |
| Hematocrit (%) | 40.8 ± 3.0 | 42.4 ± 2.1 | p > 0.05 |
| RBCs (106/uL) | 8.4 ± 1.8 | 8.9 ± 1.4 | p > 0.05 |
| Platelets (103/uL) | 998 ± 2.1 | 992 ± 4.2 | p > 0.05 |
| White blood cells (103/uL) | 4.2 ± 0.11 | 4.5 ± 0.17 | p > 0.05 |
| Groups | Heart | Liver | Spleen | Kidney | Stomach |
|---|---|---|---|---|---|
| Control (A) | 0.68 ± 0.03 | 6.25 ± 0.12 | 0.62 ± 0.01 | 0.96 ± 0.04 | 1.42 ± 0.24 |
| Hydrogels treated (B) | 0.57 ± 0.05 | 5.96 ± 0.16 | 0.56 ± 0.02 | 0.89 ± 0.02 | 1.36 ± 0.44 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ghumman, S.A.; Noreen, S.; Hameed, H.; Elsherif, M.A.; Shabbir, R.; Rana, M.; Junaid, K.; Bukhari, S.N.A. Synthesis of pH-Sensitive Cross-Linked Basil Seed Gum/Acrylic Acid Hydrogels by Free Radical Copolymerization Technique for Sustained Delivery of Captopril. Gels 2022, 8, 291. https://doi.org/10.3390/gels8050291
Ghumman SA, Noreen S, Hameed H, Elsherif MA, Shabbir R, Rana M, Junaid K, Bukhari SNA. Synthesis of pH-Sensitive Cross-Linked Basil Seed Gum/Acrylic Acid Hydrogels by Free Radical Copolymerization Technique for Sustained Delivery of Captopril. Gels. 2022; 8(5):291. https://doi.org/10.3390/gels8050291
Chicago/Turabian StyleGhumman, Shazia Akram, Sobia Noreen, Huma Hameed, Mervat A. Elsherif, Ramla Shabbir, Mavra Rana, Kashaf Junaid, and Syed Nasir Abbas Bukhari. 2022. "Synthesis of pH-Sensitive Cross-Linked Basil Seed Gum/Acrylic Acid Hydrogels by Free Radical Copolymerization Technique for Sustained Delivery of Captopril" Gels 8, no. 5: 291. https://doi.org/10.3390/gels8050291







