Synthesis and Characterization of a New Cryogel Matrix for Covalent Immobilization of Catalase
Abstract
:1. Introduction
2. Results and Discussion
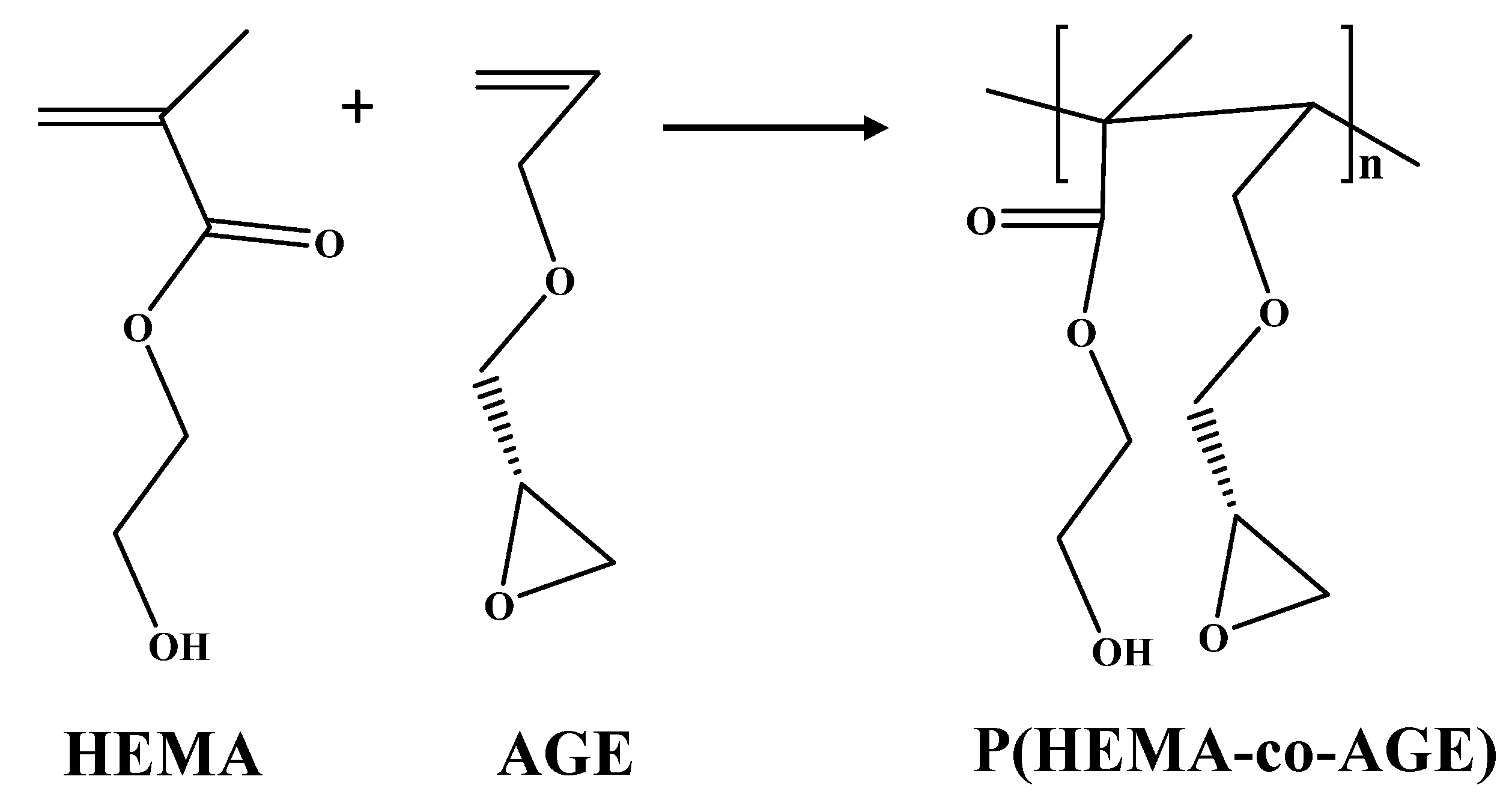
2.1. Characterization of p(HEMA-co-AGE) Cryogels
2.2. Catalase Binding Studies into p(HEMA-co-ADE) Cryogel from Aqueous Solutions
2.2.1. Effect of Flow Rate
2.2.2. Effects of pH and Initial Catalase Concentration
2.3. Kinetic Parameters
2.4. Effect of Temperature and pH on the Catalytic Activity
2.5. Reusability and Stability of Catalase-Immobilized p(HEMA-co-AGE) Cryogel
3. Conclusions
4. Materials and Methods
4.1. Materials
4.2. Synthesis and Characterization of p(HEMA-co-AGE) Cryogels
4.3. Catalase Immobilization onto p(HEMA-co-AGE) Cryogels and Desorption of Catalase from p(HEMA-co-AGE) Cryogels
4.4. Activity Assays of Free and Immobilized Catalase Enzymes
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Tan, Z.; Bilal, M.; Raza, A.; Cui, J.; Ashraf, S.S.; Iqbal, H.M.N. Expanding the Biocatalytic Scope of Enzyme-Loaded Polymeric Hydrogels. Gels 2021, 7, 194. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Lan, P.C.; Ma, S. Metal–Organic Frameworks for Enzyme Immobilization: Beyond Host Matrix Materials. ACS Cent. Sci. 2020, 6, 1497–1506. [Google Scholar] [CrossRef] [PubMed]
- Zucca, P.; Fernandez-Lafuente, R.; Sanjust, E. Agarose and Its Derivatives as Supports for Enzyme Immobilization. Molecules 2016, 21, 1577. [Google Scholar] [CrossRef] [PubMed]
- Ye, P.; Xu, Z.; Che, A.F.; Wu, J.; Seta, P. Chitosan-tethered poly(acrylonitrile--maleic acid) hollow fiber membrane for lipase immobilization. Biomaterials 2005, 26, 6394–6403. [Google Scholar] [CrossRef]
- Öztürk, N.; Tabak, A.; Akgöl, S.; Denizli, A. Reversible immobilization of catalase by using a novel bentonite–cysteine (Bent–Cys) microcomposite affinity sorbents. Colloids Surf. A Physicochem. Eng. Asp. 2008, 322, 148–154. [Google Scholar] [CrossRef]
- Akgöl, S.; Kaçar, Y.; Özkara, S.; Yavuz, H.; Denizli, A.; Arica, M.Y. Immobilization of catalase via adsorption onto l-histidine grafted functional pHEMA based membrane. J. Mol. Catal. B Enzym. 2001, 15, 197–206. [Google Scholar] [CrossRef]
- Lozinsky, V.I.; Galaev, I.Y.; Plieva, F.M.; Savina, I.N.; Jungvid, H.; Mattiasson, B. Polymeric cryogels as promising materials of biotechnological interest. Trends Biotechnol. 2003, 21, 445–451. [Google Scholar] [CrossRef]
- Kokufuta, E.; Jinbo, E. A hydrogel capable of facilitating polymer diffusion through the gel porosity and its application in enzyme immobilization. Macromolecules 1992, 25, 3549–3552. [Google Scholar] [CrossRef]
- Akkuş Çetinus, Ş.; Nursevin Öztop, H. Immobilization of catalase into chemically crosslinked chitosan beads. Enzym. Microb. Technol. 2003, 32, 889–894. [Google Scholar] [CrossRef]
- McCoy, M.; Kalghatgi, K.; Regnier, F.E.; Afeyan, N. Perfusion chromatography—Characterization of column packings for chromatography of proteins. J. Chromatogr. A 1996, 743, 221–229. [Google Scholar] [CrossRef]
- Altıntaş, E.B.; Denizli, A. Affinity adsorption of recombinant human interferon-α on monosize dye-affinity beads. J. Appl. Polym. Sci. 2007, 103, 975–981. [Google Scholar] [CrossRef]
- Demirci, S.; Sahiner, M.; Yilmaz, S.; Karadag, E.; Sahiner, N. Enhanced enzymatic activity and stability by in situ entrapment of α-Glucosidase within super porous p(HEMA) cryogels during synthesis. Biotechnol. Rep. 2020, 28, e00534. [Google Scholar] [CrossRef]
- Lou, X.; Jiang, Y.; Zhao, F.; Zhang, Y.; Qu, X.; Liu, T.; Zhang, W.; Zhu, L.; Yun, J. Preparation and characterization of semi-hydrophobic cryogels for culture of Lactobacillus strains and bioconversion towards phenyllactic acid bioproduction. Biochem. Eng. J. 2022, 179, 108312. [Google Scholar] [CrossRef]
- Demirci, S.; Sahiner, N. Urease-Immobilized PEI Cryogels for the Enzymatic Hydrolysis of Urea and Carbon Dioxide Uptake. Ind. Eng. Chem. Res. 2022, 61, 2771–2782. [Google Scholar] [CrossRef]
- Smagulova, I.; Tatykhanova, G.; Shakhvorostov, A.; Akbayeva, D.; Kudaibergenov, S. Oxidation of iso-propanol and n-butanol by catalase encapsulated within macroporous polyampholyte cryogel matrix. Polym. Adv. Technol. 2021, 32, 3817–3826. [Google Scholar] [CrossRef]
- Abdel-Halim, E.S.; Al-Deyab, S.S. One-step bleaching process for cotton fabrics using activated hydrogen peroxide. Carbohydr. Polym. 2013, 92, 1844–1849. [Google Scholar] [CrossRef] [PubMed]
- Xu, C.; Hinks, D.; Sun, C.; Wei, Q. Establishment of an activated peroxide system for low-temperature cotton bleaching using N-[4-(triethylammoniomethyl)benzoyl]butyrolactam chloride. Carbohydr. Polym. 2015, 119, 71–77. [Google Scholar] [CrossRef]
- Barczewski, M.; Matykiewicz, D.; Szostak, M. The effect of two-step surface treatment by hydrogen peroxide and silanization of flax/cotton fabrics on epoxy-based laminates thermomechanical properties and structure. J. Mater. Res. Technol. 2020, 9, 13813–13824. [Google Scholar] [CrossRef]
- Akgöl, S.; Öztürk, N.; Denizli, A. New generation polymeric nanospheres for catalase immobilization. J. Appl. Polym. Sci. 2009, 114, 962–970. [Google Scholar] [CrossRef]
- Horozova, E.; Dimcheva, N.; Jordanova, Z. Electrooxidation of phenol by catalase immobilized on graphite electrodes. Bioelectrochemistry 2001, 53, 11–16. [Google Scholar] [CrossRef]
- Vasudevan, P.T.; Thakur, D.S. Soluble and immobilized catalase. Appl. Biochem. Biotechnol. 1994, 49, 173–189. [Google Scholar] [CrossRef] [PubMed]
- Leshchinskaya, A.P.; Ezhova, N.M.; Pisarev, O.A. Synthesis and characterization of 2-hydroxyethyl methacrylate-ethylene glycol dimethacrylate polymeric granules intended for selective removal of uric acid. React. Funct. Polym. 2016, 102, 101–109. [Google Scholar] [CrossRef]
- Mohd Hidzir, N.; Mohd Radzali, N.A.; Abdul Rahman, I.; Shamsudin, S.A. Gamma irradiation-induced grafting of 2-hydroxyethyl methacrylate (HEMA) onto ePTFE for implant applications. Nucl. Eng. Technol. 2020, 52, 2320–2327. [Google Scholar] [CrossRef]
- Tomić, S.L.; Mićić, M.M.; Filipović, J.M.; Suljovrujić, E.H. Swelling and drug release behavior of poly(2-hydroxyethyl methacrylate/itaconic acid) copolymeric hydrogels obtained by gamma irradiation. Radiat. Phys. Chem. 2007, 76, 801–810. [Google Scholar] [CrossRef]
- Yildirim, Y. The Synthesis of Allyl Glycidyl Ether Copolymers and Their Thermokinetic Analysis. Anadolu Univ. J. Sci. Technol. Appl. Sci. Eng. 2016, 17, 377–389. [Google Scholar] [CrossRef]
- Nechaeva, A.; Artyukhov, A.; Luss, A.; Shtilman, M.; Gritskova, I.; Shulgin, A.; Motyakin, M.; Levina, I.; Krivoborodov, E.; Toropygin, I.; et al. Synthesis of Amphiphilic Copolymers of N-Vinyl-2-pyrrolidone and Allyl Glycidyl Ether for Co-Delivery of Doxorubicin and Paclitaxel. Polymers 2022, 14, 1727. [Google Scholar] [CrossRef]
- Tüzmen, N.; Kalburcu, T.; Denizli, A. Immobilization of catalase via adsorption onto metal-chelated affinity cryogels. Process Biochem. 2012, 47, 26–33. [Google Scholar] [CrossRef]
- Soomro, R.; Perçin, I.; Memon, N.; Iqbal Bhanger, M.; Denizli, A. Gelatin-loaded p(HEMA-GMA) cryogel for high-capacity immobilization of horseradish peroxidase. Artif. Cells Nanomed. Biotechnol. 2016, 44, 1708–1713. [Google Scholar] [CrossRef]
- Bayramoğlu, G.; Arıca, M.Y. Immobilization of laccase onto poly(glycidylmethacrylate) brush grafted poly(hydroxyethylmethacrylate) films: Enzymatic oxidation of phenolic compounds. Mater. Sci. Eng. C 2009, 29, 1990–1997. [Google Scholar] [CrossRef]
- Stevens, J.S.; de Luca, A.C.; Pelendritis, M.; Terenghi, G.; Downes, S.; Schroeder, S.L.M. Quantitative analysis of complex amino acids and RGD peptides by X-ray photoelectron spectroscopy (XPS). Surf. Interface Anal. 2013, 45, 1238–1246. [Google Scholar] [CrossRef]
- Jagst, E. Surface Functional Group Characterization Using Chemical Derivatization X-ray Photoelectron Spectroscopy (CD-XPS). Ph.D. Thesis, Freie Universität Berlin, Berlin, Germany, 2011. [Google Scholar]
- Akgöl, S.; Denizli, A. Novel metal-chelate affinity sorbents for reversible use in catalase adsorption. J. Mol. Catal. B Enzym. 2004, 28, 7–14. [Google Scholar] [CrossRef]
- Rojas, E.E.G.; dos Reis Coimbra, J.S.; Minim, L.A.; Saraiva, S.H.; da Silva, C.A.S. Hydrophobic interaction adsorption of hen egg white proteins albumin, conalbumin, and lysozyme. J. Chromatogr. B 2006, 840, 85–93. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.-Y.; Lee, J.-F.; Wu, C.-F.; Tsao, H.-K. Microcalorimetric Studies of the Interactions of Lysozyme with Immobilized Cu(II): Effects of pH Value and Salt Concentration. J. Colloid Interface Sci. 1997, 190, 49–54. [Google Scholar] [CrossRef]
- Nash, D.C.; McCreath, G.E.; Chase, H.A. Modification of polystyrenic matrices for the purification of proteins Effect of the adsorption of poly(vinyl alcohol) on the characteristics of poly(styrene-divinylbenzene) beads for use in affinity chromatography. J. Chromatogr. A 1997, 758, 53–64. [Google Scholar] [CrossRef]
- Roper, D.K.; Lightfoot, E.N. Separation of biomolecules using adsorptive membranes. J. Chromatogr. A 1995, 702, 3–26. [Google Scholar] [CrossRef]
- Inanan, T. Chitosan Co-polymeric nanostructures for catalase immobilization. React. Funct. Polym. 2019, 135, 94–102. [Google Scholar] [CrossRef]
- Çetinus, Ş.A.; Öztop, H.N.; Saraydın, D. Immobilization of catalase onto chitosan and cibacron blue F3GA attached chitosan beads. Enzym. Microb. Technol. 2007, 41, 447–454. [Google Scholar] [CrossRef]
- Erol, K.; Cebeci, B.K.; Köse, K.; Köse, D.A. Effect of immobilization on the activity of catalase carried by poly(HEMA-GMA) cryogels. Int. J. Biol. Macromol. 2019, 123, 738–743. [Google Scholar] [CrossRef]
- Sel, E.; Ulu, A.; Ateş, B.; Köytepe, S. Comparative study of catalase immobilization via adsorption on P(MMA-co-PEG500MA) structures as an effective polymer support. Polym. Bull. 2021, 78, 2663–2684. [Google Scholar] [CrossRef]
- Marcuz, C.; Alves Mourão, C.; Haupt, K.; Alves Bueno, S.M. Performance of phospho-L-tyrosine immobilized onto alginate/polyacrylamide-based cryogels: Effect of ligand coupling on human IgG adsorption and Fab fragments separation. J. Chromatogr. B 2021, 1165, 122530. [Google Scholar] [CrossRef]







| Scanning Area | Bond Type | Energy (eV) |
|---|---|---|
| A | C-C, C-H | 284.75 |
| B | C-O or C-N | 287 |
| C | C=O | 288.5 |
| D | C-N | 290.5 |
| E | COOH | 295 |
| p(HEMA) | p(HEMA-co-AGE) |
|---|---|
| 399.08 | 402.64 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Altunbaş, C.; Aslan, A.; Kuşat, K.; Sahiner, M.; Akgöl, S.; Sahiner, N. Synthesis and Characterization of a New Cryogel Matrix for Covalent Immobilization of Catalase. Gels 2022, 8, 501. https://doi.org/10.3390/gels8080501
Altunbaş C, Aslan A, Kuşat K, Sahiner M, Akgöl S, Sahiner N. Synthesis and Characterization of a New Cryogel Matrix for Covalent Immobilization of Catalase. Gels. 2022; 8(8):501. https://doi.org/10.3390/gels8080501
Chicago/Turabian StyleAltunbaş, Canan, Ahmet Aslan, Kevser Kuşat, Mehtap Sahiner, Sinan Akgöl, and Nurettin Sahiner. 2022. "Synthesis and Characterization of a New Cryogel Matrix for Covalent Immobilization of Catalase" Gels 8, no. 8: 501. https://doi.org/10.3390/gels8080501







