Exploring the Potential of Artificial Intelligence for Hydrogel Development—A Short Review
Abstract
:1. Introduction: The Concept of AI in Hydrogel Design
2. Physical and Chemical Methods for Designing Hydrogels
2.1. Physical Crosslinking
2.2. Chemical Crosslinking
3. Numerical and Analytical Methods in Hydrogel Design
3.1. Numerical Simulations
3.2. Analytical Methods
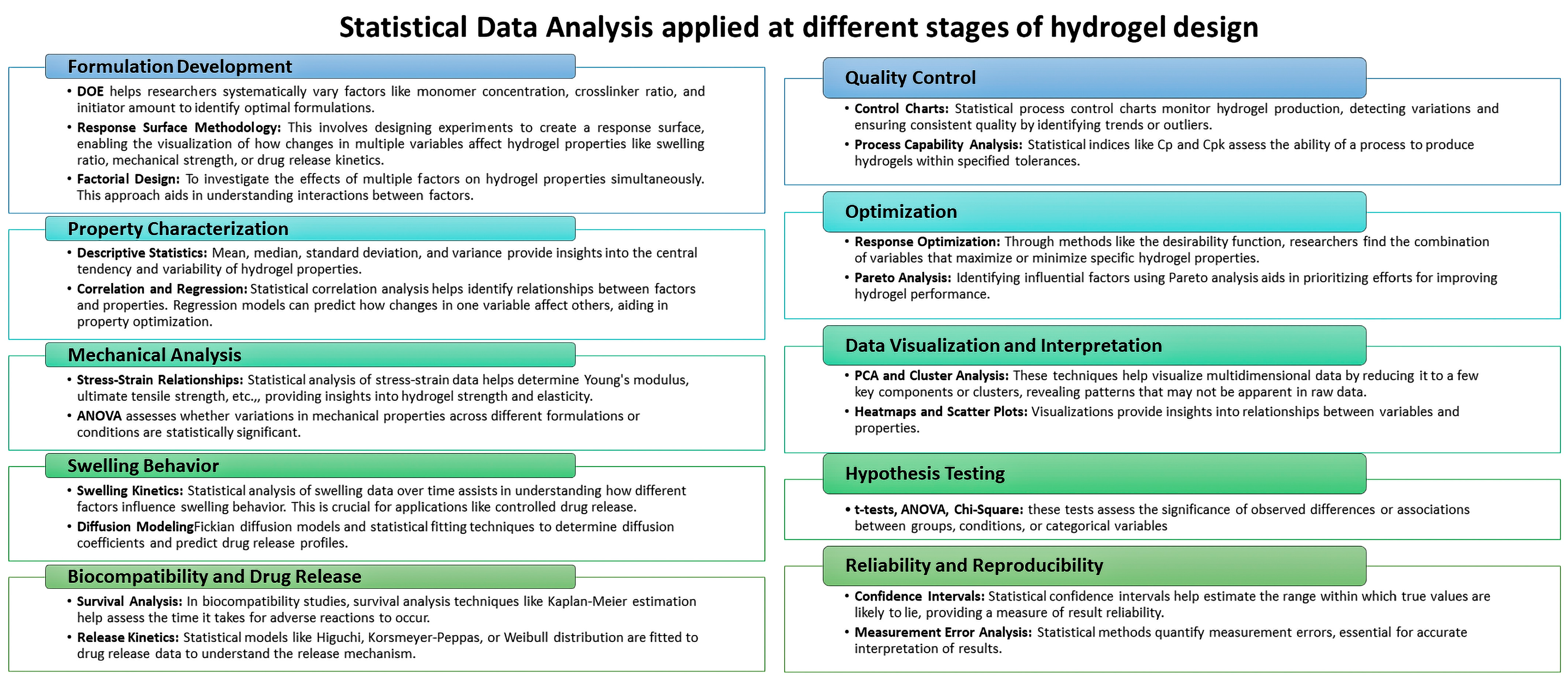
Statistical Data Analysis
4. Leveraging Artificial Intelligence in Hydrogel Design
5. Machine Learning Techniques in Hydrogel Development
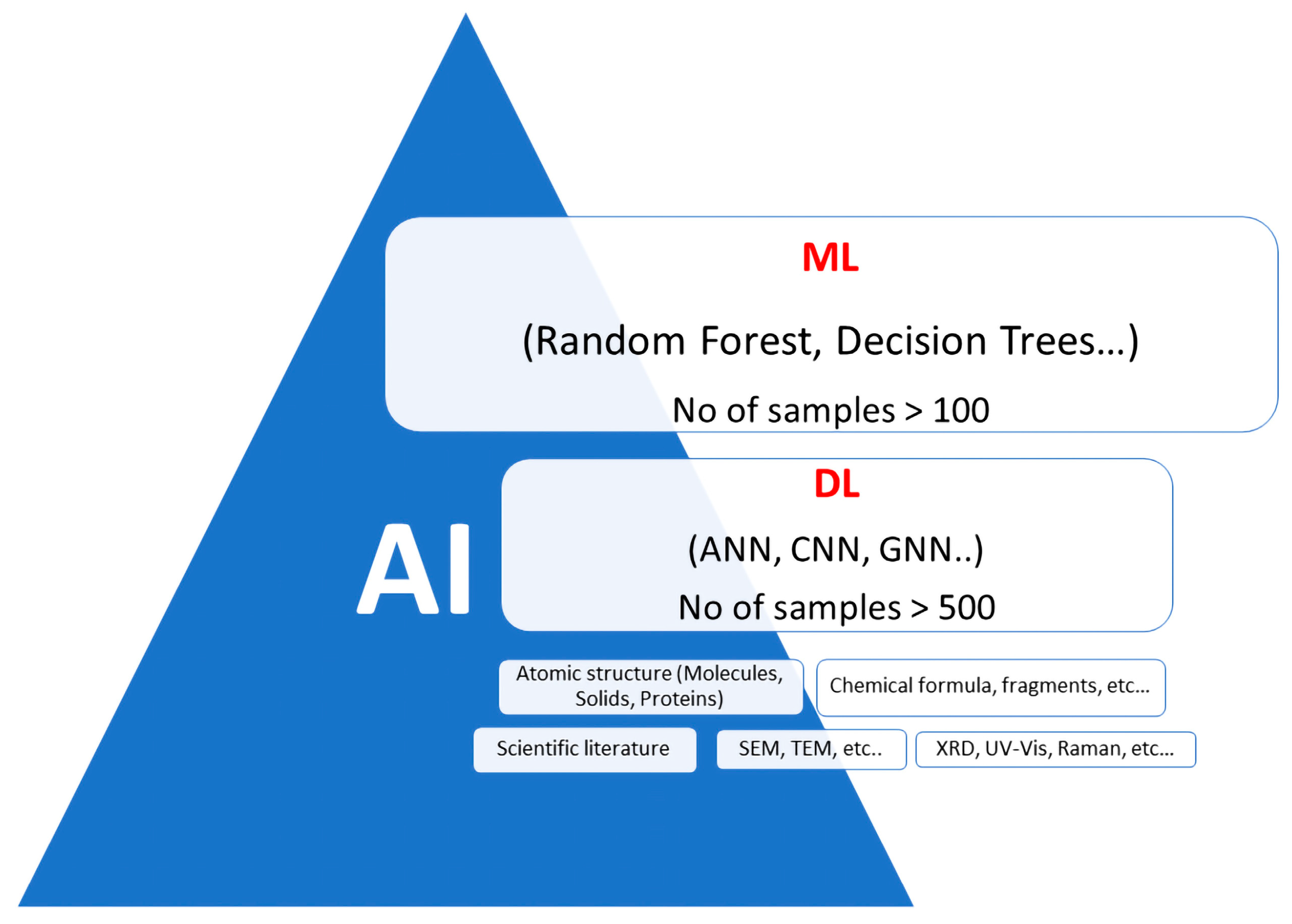
5.1. Machine Learning Subsets
5.2. Machine Learning Algorithms
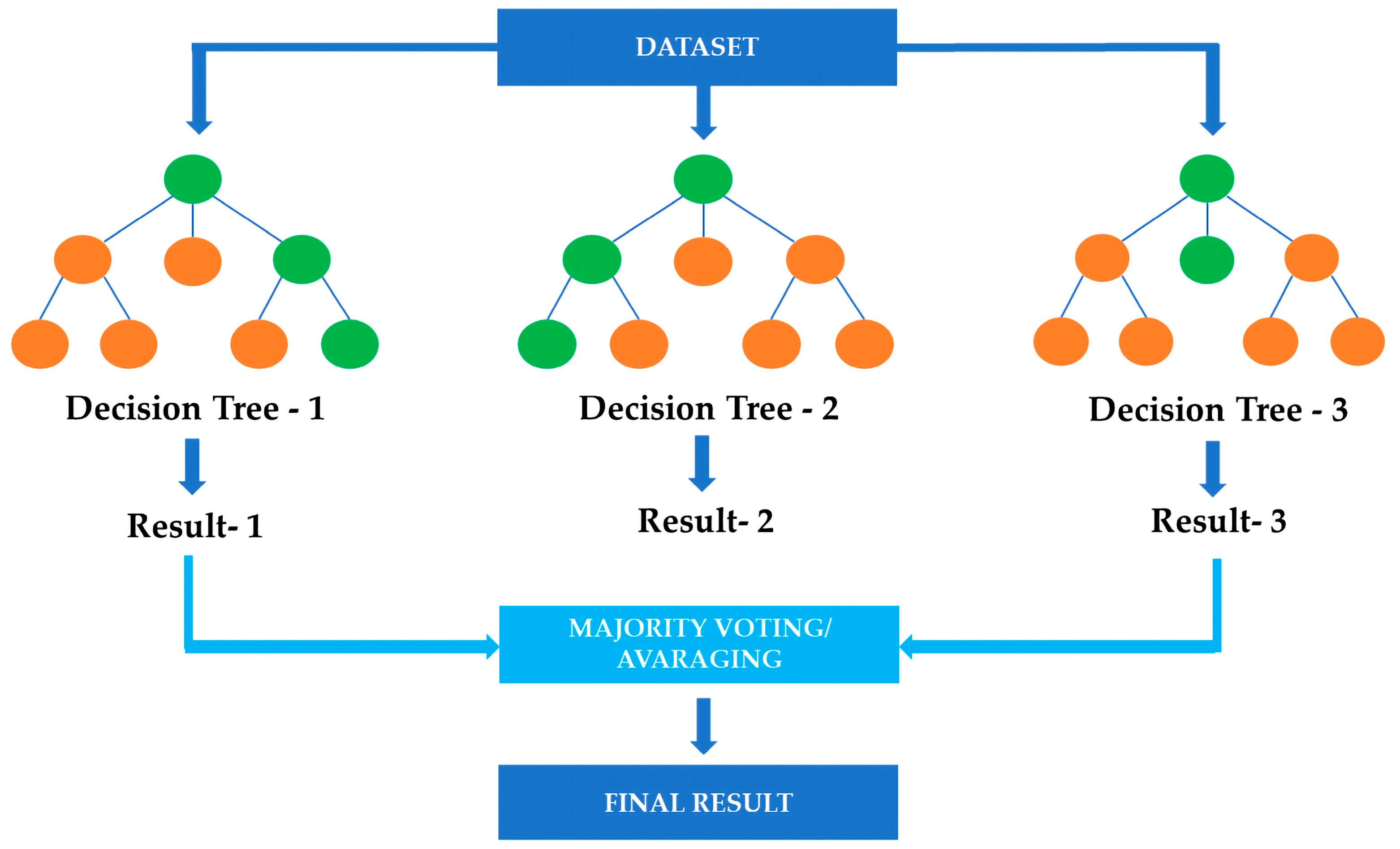
5.2.1. Random Forest
5.2.2. Artificial Neural Network
5.2.3. Support Vector Machines
5.2.4. Deep Neural Networks
5.2.5. Convolutional Neural Networks
6. Conclusions and Future Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Vermonden, T.; Klumperman, B. The past, present and future of hydrogels. Eur. Polym. J. 2015, 72, 341–343. [Google Scholar] [CrossRef]
- Buwalda, S.J.; Vermonden, T.; Hennink, W.E. Hydrogels for Therapeutic Delivery: Current Developments and Future Directions. Biomacromolecules 2017, 18, 316–330. [Google Scholar] [CrossRef] [PubMed]
- Liu, M.; Guo, R.; Ma, Y. Construction of a specific and efficient antibacterial agent against Pseudomonas aeruginosa based on polyethyleneimine cross-linked fucose. J. Mater. Sci. 2021, 56, 6083–6094. [Google Scholar] [CrossRef]
- Revete, A.; Aparicio, A.; Cisterna, B.A.; Revete, J.; Luis, L.; Ibarra, E.; Segura González, E.A.; Molino, J.; Reginensi, D. Advancements in the Use of Hydrogels for Regenerative Medicine: Properties and Biomedical Applications. Int. J. Biomater. 2022, 2022, e3606765. [Google Scholar] [CrossRef] [PubMed]
- Alka, A.; Verma, A.; Mishra, N.; Singh, N.; Singh, P.; Nisha, R.; Pal, R.R.; Saraf, S.A. Polymeric Gel Scaffolds and Biomimetic Environments for Wound Healing. Curr. Pharm. Des. 2023, 29, 1–19. [Google Scholar] [CrossRef] [PubMed]
- Ko, A.; Liao, C. Hydrogel wound dressings for diabetic foot ulcer treatment: Status-quo, challenges, and future perspectives. BMEMat 2023, 1, e12037. [Google Scholar] [CrossRef]
- Nie, L.; Li, Y.; Liu, Y.; Shi, L.; Chen, H. Recent Applications of Contact Lenses for Bacterial Corneal Keratitis Therapeutics: A Review. Pharmaceutics 2022, 14, 2635. [Google Scholar] [CrossRef]
- Barhoum, A.; Sadak, O.; Ramirez, I.A.; Iverson, N. Stimuli-bioresponsive hydrogels as new generation materials for implantable, wearable, and disposable biosensors for medical diagnostics: Principles, opportunities, and challenges. Adv. Colloid Interface Sci. 2023, 317, 102920. [Google Scholar] [CrossRef]
- Gerlach, G.; Arndt, K.-F. Hydrogel Sensors and Actuators: Engineering and Technology; Springer Science & Business Media: Berlin/Heidelberg, Germany, 2009. [Google Scholar]
- Wang, Y.; Wallmersperger, T.; Ehrenhofer, A. Application of back propagation neural networks and random forest algorithms in material research of hydrogels. PAMM 2023, 23, e202200278. [Google Scholar] [CrossRef]
- Vora, L.K.; Gholap, A.D.; Jetha, K.; Thakur, R.R.S.; Solanki, H.K.; Chavda, V.P. Artificial Intelligence in Pharmaceutical Technology and Drug Delivery Design. Pharmaceutics 2023, 15, 1916. [Google Scholar] [CrossRef]
- Boztepe, C.; Künkül, A.; Yüceer, M. Application of artificial intelligence in modeling of the doxorubicin release behavior of pH and temperature responsive poly(NIPAAm-co-AAc)-PEG IPN hydrogel. J. Drug Deliv. Sci. Technol. 2020, 57, 101603. [Google Scholar] [CrossRef]
- Soleimani, S.; Heydari, A.; Fattahi, M. Swelling prediction of calcium alginate/cellulose nanocrystal hydrogels using response surface methodology and artificial neural network. Ind. Crops Prod. 2023, 192, 116094. [Google Scholar] [CrossRef]
- Mukherjee, S.; Kim, B.; Cheng, L.Y.; Doerfert, M.D.; Li, J.; Hernandez, A.; Liang, L.; Jarvis, M.I.; Rios, P.D.; Ghani, S.; et al. Screening hydrogels for antifibrotic properties by implanting cellularly barcoded alginates in mice and a non-human primate. Nat. Biomed. Eng. 2023, 7, 867–886. [Google Scholar] [CrossRef] [PubMed]
- de Lima, C.S.A.; Balogh, T.S.; Varca, J.P.R.O.; Varca, G.H.C.; Lugão, A.B.; Camacho-Cruz, L.A.; Bucio, E.; Kadlubowski, S.S. An Updated Review of Macro, Micro, and Nanostructured Hydrogels for Biomedical and Pharmaceutical Applications. Pharmaceutics 2020, 12, 970. [Google Scholar] [CrossRef]
- Lou, J.; Mooney, D.J. Chemical strategies to engineer hydrogels for cell culture. Nat. Rev. Chem. 2022, 6, 726–744. [Google Scholar] [CrossRef]
- El-Husseiny, H.M.; Mady, E.A.; Hamabe, L.; Abugomaa, A.; Shimada, K.; Yoshida, T.; Tanaka, T.; Yokoi, A.; Elbadawy, M.; Tanaka, R. Smart/stimuli-responsive hydrogels: Cutting-edge platforms for tissue engineering and other biomedical applications. Mater. Today Bio 2022, 13, 100186. [Google Scholar] [CrossRef]
- Yang, J.; Chen, Y.; Zhao, L.; Zhang, J.; Luo, H. Constructions and Properties of Physically Cross-Linked Hydrogels Based on Natural Polymers. Polym. Rev. 2023, 63, 574–612. [Google Scholar] [CrossRef]
- Cui, K.; Yu, C.; Ye, Y.N.; Li, X.; Gong, J.P. Mechanism of temperature-induced asymmetric swelling and shrinking kinetics in self-healing hydrogels. Proc. Natl. Acad. Sci. USA 2022, 119, e2207422119. [Google Scholar] [CrossRef]
- Gadziński, P.; Froelich, A.; Jadach, B.; Wojtyłko, M.; Tatarek, A.; Białek, A.; Krysztofiak, J.; Gackowski, M.; Otto, F.; Osmałek, T. Ionotropic Gelation and Chemical Crosslinking as Methods for Fabrication of Modified-Release Gellan Gum-Based Drug Delivery Systems. Pharmaceutics 2023, 15, 108. [Google Scholar] [CrossRef]
- Omar, J.; Ponsford, D.; Dreiss, C.A.; Lee, T.-C.; Loh, X.J. Supramolecular Hydrogels: Design Strategies and Contemporary Biomedical Applications. Chem.–Asian J. 2022, 17, e202200081. [Google Scholar] [CrossRef]
- Tripathi, M.; Sharma, R.; Hussain, A.; Kumar, I.; Sharma, A.K.; Sarkar, A. Chapter 17—Hydrogels and their combination with lipids and nucleotides. In Sustainable Hydrogels; Thomas, S., Sharma, B., Jain, P., Shekhar, S., Eds.; Elsevier: Amsterdam, The Netherlands, 2023; pp. 471–487. [Google Scholar] [CrossRef]
- Liu, J.; Su, C.; Chen, Y.; Tian, S.; Lu, C.; Huang, W.; Lv, Q. Current Understanding of the Applications of Photocrosslinked Hydrogels in Biomedical Engineering. Gels 2022, 8, 216. [Google Scholar] [CrossRef] [PubMed]
- Hao, M.; Wang, Y.; Li, L.; Liu, Y.; Bai, Y.; Zhou, W.; Lu, Q.; Sun, F.; Li, L.; Feng, S.; et al. Tough Engineering Hydrogels Based on Swelling–Freeze–Thaw Method for Artificial Cartilage. ACS Appl. Mater. Interfaces 2022, 14, 25093–25103. [Google Scholar] [CrossRef] [PubMed]
- Varshney, N.; Sahi, A.K.; Poddar, S.; Vishwakarma, N.K.; Kavimandan, G.; Prakash, A.; Mahto, S.K. Freeze–Thaw-Induced Physically Cross-linked Superabsorbent Polyvinyl Alcohol/Soy Protein Isolate Hydrogels for Skin Wound Dressing: In Vitro and In Vivo Characterization. ACS Appl. Mater. Interfaces 2022, 14, 14033–14048. [Google Scholar] [CrossRef]
- Xue, X.; Hu, Y.; Wang, S.; Chen, X.; Jiang, Y.; Su, J. Fabrication of physical and chemical crosslinked hydrogels for bone tissue engineering. Bioact. Mater. 2022, 12, 327–339. [Google Scholar] [CrossRef]
- Pei, X.; Wang, J.; Cong, Y.; Fu, J. Recent progress in polymer hydrogel bioadhesives. J. Polym. Sci. 2021, 59, 1312–1337. [Google Scholar] [CrossRef]
- García, J.M.; García, F.C.; Ruiz, J.A.R.; Vallejos, S.; Trigo-López, M. Smart Polymers: Principles and Applications; Walter de Gruyter GmbH & Co KG: Berlin, Germany, 2022. [Google Scholar]
- Yang, J.; Bai, R.; Chen, B.; Suo, Z. Hydrogel Adhesion: A Supramolecular Synergy of Chemistry, Topology, and Mechanics. Adv. Funct. Mater. 2020, 30, 1901693. [Google Scholar] [CrossRef]
- Seidi, F.; Zhao, W.; Xiao, H.; Jin, Y.; Reza Saeb, M.; Zhao, C. Radical polymerization as a versatile tool for surface grafting of thin hydrogel films. Polym. Chem. 2020, 11, 4355–4381. [Google Scholar] [CrossRef]
- Guaresti, O.; Basasoro, S.; González, K.; Eceiza, A.; Gabilondo, N. In situ cross–linked chitosan hydrogels via Michael addition reaction based on water–soluble thiol–maleimide precursors. Eur. Polym. J. 2019, 119, 376–384. [Google Scholar] [CrossRef]
- Khan, A.H.; Cook, J.K.; Wortmann III, W.J.; Kersker, N.D.; Rao, A.; Pojman, J.A.; Melvin, A.T. Synthesis and characterization of thiol-acrylate hydrogels using a base-catalyzed Michael addition for 3D cell culture applications. J. Biomed. Mater. Res. B Appl. Biomater. 2020, 108, 2294–2307. [Google Scholar] [CrossRef]
- Li, X.; Xiong, Y. Application of “Click” Chemistry in Biomedical Hydrogels. ACS Omega 2022, 7, 36918–36928. [Google Scholar] [CrossRef]
- Li, Y.; Wang, X.; Han, Y.; Sun, H.-Y.; Hilborn, J.; Shi, L. Click chemistry-based biopolymeric hydrogels for regenerative medicine. Biomed. Mater. 2021, 16, 022003. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Liu, Y.; Hsu, S. Hydrogels Based on Schiff Base Linkages for Biomedical Applications. Molecules 2019, 24, 3005. [Google Scholar] [CrossRef] [PubMed]
- He, Y.; Wang, F.; Wang, X.; Zhang, J.; Wang, D.; Huang, X. A photocurable hybrid chitosan/acrylamide bioink for DLP based 3D bioprinting. Mater. Des. 2021, 202, 109588. [Google Scholar] [CrossRef]
- Zhu, H.; Monavari, M.; Zheng, K.; Distler, T.; Ouyang, L.; Heid, S.; Jin, Z.; He, J.; Li, D.; Boccaccini, A.R. 3D Bioprinting of Multifunctional Dynamic Nanocomposite Bioinks Incorporating Cu-Doped Mesoporous Bioactive Glass Nanoparticles for Bone Tissue Engineering. Small 2022, 18, 2104996. [Google Scholar] [CrossRef]
- Yang, Z.; Yi, P.; Liu, Z.; Zhang, W.; Mei, L.; Feng, C.; Tu, C.; Li, Z. Stem Cell-Laden Hydrogel-Based 3D Bioprinting for Bone and Cartilage Tissue Engineering. Front. Bioeng. Biotechnol. 2022, 10, 865770. [Google Scholar] [CrossRef]
- Zhou, K.; Sun, Y.; Yang, J.; Mao, H.; Gu, Z. Hydrogels for 3D embedded bioprinting: A focused review on bioinks and support baths. J. Mater. Chem. B 2022, 10, 1897–1907. [Google Scholar] [CrossRef]
- Xie, M.; Su, J.; Zhou, S.; Li, J.; Zhang, K. Application of Hydrogels as Three-Dimensional Bioprinting Ink for Tissue Engineering. Gels 2023, 9, 88. [Google Scholar] [CrossRef]
- Ghandforoushan, P.; Alehosseini, M.; Golafshan, N.; Castilho, M.; Dolatshahi-Pirouz, A.; Hanaee, J.; Davaran, S.; Orive, G. Injectable hydrogels for cartilage and bone tissue regeneration: A review. Int. J. Biol. Macromol. 2023, 246, 125674. [Google Scholar] [CrossRef]
- Deptuła, M.; Zawrzykraj, M.; Sawicka, J.; Banach-Kopeć, A.; Tylingo, R.; Pikuła, M. Application of 3D- printed hydrogels in wound healing and regenerative medicine. Biomed. Pharmacother. 2023, 167, 115416. [Google Scholar] [CrossRef]
- Shahriari-Khalaji, M.; Sattar, M.; Cao, R.; Zhu, M. Angiogenesis, hemocompatibility and bactericidal effect of bioactive natural polymer-based bilayer adhesive skin substitute for infected burned wound healing. Bioact. Mater. 2023, 29, 177–195. [Google Scholar] [CrossRef]
- Qi, J.; Zheng, S.; Zhao, N.; Li, Y.; Zhang, G.; Yin, W. A 3D bioprinted hydrogel multilevel arc vascular channel combined with an isomaltol core sacrificial process. Mater. Today Commun. 2023, 36, 106492. [Google Scholar] [CrossRef]
- Zheng, F.; Xiao, Y.; Liu, H.; Fan, Y.; Dao, M. Patient-Specific Organoid and Organ-on-a-Chip: 3D Cell-Culture Meets 3D Printing and Numerical Simulation. Adv. Biol. 2021, 5, e2000024. [Google Scholar] [CrossRef] [PubMed]
- Mei, Q.; Rao, J.; Bei, H.P.; Liu, Y.; Zhao, X. 3D Bioprinting Photo-Crosslinkable Hydrogels for Bone and Cartilage Repair. Int. J. Bioprint. 2021, 7, 367. [Google Scholar] [CrossRef] [PubMed]
- Solanki, D.; Vinchhi, P.; Patel, M.M. Design Considerations, Formulation Approaches, and Strategic Advances of Hydrogel Dressings for Chronic Wound Management. ACS Omega 2023, 8, 8172–8189. [Google Scholar] [CrossRef] [PubMed]
- Dutta, S.D.; Ganguly, K.; Hexiu, J.; Randhawa, A.; Moniruzzaman, M.; Lim, K.-T. A 3D Bioprinted Nanoengineered Hydrogel with Photoactivated Drug Delivery for Tumor Apoptosis and Simultaneous Bone Regeneration via Macrophage Immunomodulation. Macromol. Biosci. 2023, 23, 2300096. [Google Scholar] [CrossRef]
- Jiang, H.; Hao, Z.; Zhang, J.; Tang, J.; Li, H. Bioinspired swelling enhanced hydrogels for underwater sensing. Colloids Surf. Physicochem. Eng. Asp. 2023, 664, 131197. [Google Scholar] [CrossRef]
- Shokrollahi, Y.; Dong, P.; Gamage, P.T.; Patrawalla, N.; Kishore, V.; Mozafari, H.; Gu, L. Finite Element-Based Machine Learning Model for Predicting the Mechanical Properties of Composite Hydrogels. Appl. Sci. 2022, 12, 10835. [Google Scholar] [CrossRef]
- Kakarla, A.B.; Kong, I.; Nukala, S.G.; Kong, W. Mechanical Behaviour Evaluation of Porous Scaffold for Tissue-Engineering Applications Using Finite Element Analysis. J. Compos. Sci. 2022, 6, 46. [Google Scholar] [CrossRef]
- Teoh, J.H.; Abdul Shakoor, F.T.; Wang, C.-H. 3D Printing Methyl Cellulose Hydrogel Wound Dressings with Parameter Exploration Via Computational Fluid Dynamics Simulation. Pharm. Res. 2022, 39, 281–294. [Google Scholar] [CrossRef]
- Li, S.; Liu, Y.; Li, Y.; Zhang, Y.; Hu, Q. Computational and experimental investigations of the mechanisms used by coaxial fluids to fabricate hollow hydrogel fibers. Chem. Eng. Process. Process Intensif. 2015, 95, 98–104. [Google Scholar] [CrossRef]
- Wei, Q.; Yang, R.; Sun, D.; Zhou, J.; Li, M.; Zhang, Y.; Wang, Y. Design and evaluation of sodium alginate/polyvinyl alcohol blend hydrogel for 3D bioprinting cartilage scaffold: Molecular dynamics simulation and experimental method. J. Mater. Res. Technol. 2022, 17, 66–78. [Google Scholar] [CrossRef]
- Shahshahani, S.; Shahgholi, M.; Karimipour, A. The thermal performance and mechanical stability of methacrylic acid porous hydrogels in an aqueous medium at different initial temperatures and hydrogel volume fraction using the molecular dynamics simulation. J. Mol. Liq. 2023, 382, 122001. [Google Scholar] [CrossRef]
- Salahshoori, I.; Ramezani, Z.; Cacciotti, I.; Yazdanbakhsh, A.; Hossain, M.K.; Hassanzadeganroudsari, M. Cisplatin uptake and release assessment from hydrogel synthesized in acidic and neutral medium: An experimental and molecular dynamics simulation study. J. Mol. Liq. 2021, 344, 117890. [Google Scholar] [CrossRef]
- Casalini, T.; Perale, G. From Microscale to Macroscale: Nine Orders of Magnitude for a Comprehensive Modeling of Hydrogels for Controlled Drug Delivery. Gels 2019, 5, 28. [Google Scholar] [CrossRef] [PubMed]
- Gharehnazifam, Z.; Dolatabadi, R.; Baniassadi, M.; Shahsavari, H.; Kajbafzadeh, A.-M.; Abrinia, K.; Baghani, M. Computational analysis of vincristine loaded silk fibroin hydrogel for sustained drug delivery applications: Multiphysics modeling and experiments. Int. J. Pharm. 2021, 609, 121184. [Google Scholar] [CrossRef]
- Gharehnazifam, Z.; Dolatabadi, R.; Baniassadi, M.; Shahsavari, H.; Kajbafzadeh, A.-M.; Abrinia, K.; Gharehnazifam, K.; Baghani, M. Multiphysics modeling and experiments on ultrasound-triggered drug delivery from silk fibroin hydrogel for Wilms tumor. Int. J. Pharm. 2022, 621, 121787. [Google Scholar] [CrossRef]
- Liu, D.; Ma, S.; Yuan, H.; Markert, B. Modelling and simulation of coupled fluid transport and time-dependent fracture in fibre-reinforced hydrogel composites. Comput. Methods Appl. Mech. Eng. 2022, 390, 114470. [Google Scholar] [CrossRef]
- Karvinen, J.; Kellomäki, M. Characterization of self-healing hydrogels for biomedical applications. Eur. Polym. J. 2022, 181, 111641. [Google Scholar] [CrossRef]
- Li, P.; Malveau, C.; Zhu, X.X.; Wuest, J.D. Using Nuclear Magnetic Resonance Spectroscopy to Probe Hydrogels Formed by Sodium Deoxycholate. Langmuir 2022, 38, 5111–5118. [Google Scholar] [CrossRef]
- Xing, W.; Tang, Y. On mechanical properties of nanocomposite hydrogels: Searching for superior properties. Nano Mater. Sci. 2022, 4, 83–96. [Google Scholar] [CrossRef]
- Asy-Syifa, N.; Kusjuriansah; Waresindo, W.X.; Edikresnha, D.; Suciati, T.; Khairurrijal, K. The Study of the Swelling Degree of the PVA Hydrogel with varying concentrations of PVA. J. Phys. Conf. Ser. 2022, 2243, 012053. [Google Scholar] [CrossRef]
- Martinez-Garcia, F.D.; van Dongen, J.A.; Burgess, J.K.; Harmsen, M.C. Matrix Metalloproteases from Adipose Tissue-Derived Stromal Cells Are Spatiotemporally Regulated by Hydrogel Mechanics in a 3D Microenvironment. Bioengineering 2022, 9, 340. [Google Scholar] [CrossRef] [PubMed]
- Joshi, J.; Homburg, S.V.; Ehrmann, A. Atomic Force Microscopy (AFM) on Biopolymers and Hydrogels for Biotechnological Applications—Possibilities and Limits. Polymers 2022, 14, 1267. [Google Scholar] [CrossRef] [PubMed]
- Jayawardena, I.; Turunen, P.; Cambraia Garms, B.; Rowan, A.; Corrie, S.; Grøndahl, L. Evaluation of techniques used for visualisation of hydrogel morphology and determination of pore size distributions. Mater. Adv. 2023, 4, 669–682. [Google Scholar] [CrossRef]
- Sulaeman, A.S.; Putro, P.A.; Nikmatin, S. Thermal studies of hydrogels based on poly(acrylic acid) and its copolymers by differential scanning calorimetry: A systematic literature review. Polym. Polym. Compos. 2022, 30, 09673911221094022. [Google Scholar] [CrossRef]
- Reguieg, F.; Ricci, L.; Bouyacoub, N.; Belbachir, M.; Bertoldo, M. Thermal characterization by DSC and TGA analyses of PVA hydrogels with organic and sodium MMT. Polym. Bull. 2020, 77, 929–948. [Google Scholar] [CrossRef]
- Zhao, Y.; Li, H.; Wang, Y.; Zhang, Z.; Wang, Q. Preparation, characterization and release kinetics of a multilayer encapsulated Perilla frutescens L. essential oil hydrogel bead. Int. J. Biol. Macromol. 2023, 249, 124776. [Google Scholar] [CrossRef]
- Vildanova, R.R.; Petrova, S.F.; Kolesov, S.V.; Khutoryanskiy, V.V. Biodegradable Hydrogels Based on Chitosan and Pectin for Cisplatin Delivery. Gels 2023, 9, 342. [Google Scholar] [CrossRef]
- Wang, R.; Xin, J.; Ji, Z.; Zhu, M.; Yu, Y.; Xu, M. Spin-Space-Encoding Magnetic Resonance Imaging: A New Application for Rapid and Sensitive Monitoring of Dynamic Swelling of Confined Hydrogels. Molecules 2023, 28, 3116. [Google Scholar] [CrossRef]
- Takayama, G.; Kondo, T. Quantitative evaluation of fiber network structure–property relationships in bacterial cellulose hydrogels. Carbohydr. Polym. 2023, 321, 121311. [Google Scholar] [CrossRef]
- Xu, T.; Wang, J.; Zhao, S.; Chen, D.; Zhang, H.; Fang, Y.; Kong, N.; Zhou, Z.; Li, W.; Wang, H. Accelerating the prediction and discovery of peptide hydrogels with human-in-the-loop. Nat. Commun. 2023, 14, 3880. [Google Scholar] [CrossRef] [PubMed]
- Shu, J.; Wang, C.; Tao, Y.; Wang, S.; Cheng, F.; Zhang, Y.; Shi, K.; Xia, K.; Wang, R.; Wang, J.; et al. Thermosensitive hydrogel-based GPR124 delivery strategy for rebuilding blood-spinal cord barrier. Bioeng. Transl. Med. 2023, 8, e10561. [Google Scholar] [CrossRef] [PubMed]
- Pannala, R.K.P.K.; Juyal, U.; Kodavaty, J. Optimization of hydrogel composition for effective release of drug. Chem. Prod. Process Model. 2023. [Google Scholar] [CrossRef]
- Taaca, K.L.M.; Nakajima, H.; Thumanu, K.; Prieto, E.I.; Vasquez, M.R. Network formation and differentiation of chitosan–acrylic acid hydrogels using X-ray absorption spectroscopy and multivariate analysis of Fourier transform infrared spectra. J. Electron Spectrosc. Relat. Phenom. 2023, 267, 147372. [Google Scholar] [CrossRef]
- Nudell, V.; Wang, Y.; Pang, Z.; Lal, N.K.; Huang, M.; Shaabani, N.; Kanim, W.; Teijaro, J.; Maximov, A.; Ye, L. HYBRiD: Hydrogel-reinforced DISCO for clearing mammalian bodies. Nat. Methods 2022, 19, 479–485. [Google Scholar] [CrossRef] [PubMed]
- Reindel, W.; Steffen, R.; Mosehauer, G.; Schafer, J.; Rah, M. Assessment of a novel silicone hydrogel daily disposable lens among physically active subjects. Contact Lens Anterior Eye 2022, 45, 101611. [Google Scholar] [CrossRef]
- Choudhary, K.; DeCost, B.; Chen, C.; Jain, A.; Tavazza, F.; Cohn, R.; Park, C.W.; Choudhary, A.; Agrawal, A.; Billinge, S.J.L.; et al. Recent advances and applications of deep learning methods in materials science. Npj Comput. Mater. 2022, 8, 59. [Google Scholar] [CrossRef]
- Islamkulov, M.; Karakuş, S.; Özeroğlu, C. Design artificial intelligence-based optimization and swelling behavior of novel crosslinked polymeric network hydrogels based on acrylamide-2-hydroxyethyl methacrylate and acrylamide-N-isopropylacrylamide. Colloid Polym. Sci. 2023, 301, 259–272. [Google Scholar] [CrossRef]
- Martineau, R.L.; Bayles, A.V.; Hung, C.-S.; Reyes, K.G.; Helgeson, M.E.; Gupta, M.K. Engineering Gelation Kinetics in Living Silk Hydrogels by Differential Dynamic Microscopy Microrheology and Machine Learning. Adv. Biol. 2022, 6, 2101070. [Google Scholar] [CrossRef]
- Wei, J.; Chu, X.; Sun, X.-Y.; Xu, K.; Deng, H.-X.; Chen, J.; Wei, Z.; Lei, M. Machine learning in materials science. InfoMat 2019, 1, 338–358. [Google Scholar] [CrossRef]
- Zhang, J.; Liu, Y.; Durga, C.S.P.; Singh, M.; Tong, Y.; Kucukdeger, E.; Yoon, H.Y.; Haring, A.P.; Roman, M.; Kong, Z.; et al. Rapid, autonomous high-throughput characterization of hydrogel rheological properties via automated sensing and physics-guided machine learning. Appl. Mater. Today 2023, 30, 101720. [Google Scholar] [CrossRef]
- Younes, K.; Kharboutly, Y.; Antar, M.; Chaouk, H.; Obeid, E.; Mouhtady, O.; Abu-samha, M.; Halwani, J.; Murshid, N. Application of Unsupervised Learning for the Evaluation of Aerogels’ Efficiency towards Dye Removal—A Principal Component Analysis (PCA) Approach. Gels 2023, 9, 327. [Google Scholar] [CrossRef] [PubMed]
- Tseng, B.-Y.; Cai, Y.-C.; Conan Guo, C.-W.; Zhao, E.; Yu, C.-H. Reinforcement learning design framework for nacre-like structures optimized for pre-existing crack resistance. J. Mater. Res. Technol. 2023, 24, 3502–3512. [Google Scholar] [CrossRef]
- Lin, H.W.; Tegmark, M.; Rolnick, D. Why Does Deep and Cheap Learning Work So Well? J. Stat. Phys. 2017, 168, 1223–1247. [Google Scholar] [CrossRef]
- Owh, C.; Ho, D.; Loh, X.J.; Xue, K. Towards machine learning for hydrogel drug delivery systems. Trends Biotechnol. 2023, 41, 476–479. [Google Scholar] [CrossRef]
- Menon, D.; Ranganathan, R. A Generative Approach to Materials Discovery, Design, and Optimization. ACS Omega 2022, 7, 25958–25973. [Google Scholar] [CrossRef]
- Huljanah, M.; Rustam, Z.; Utama, S.; Siswantining, T. Feature Selection using Random Forest Classifier for Predicting Prostate Cancer. IOP Conf. Ser. Mater. Sci. Eng. 2019, 546, 052031. [Google Scholar] [CrossRef]
- Richter, R.; Kamal, M.A.M.; García-Rivera, M.A.; Kaspar, J.; Junk, M.; Elgaher, W.A.M.; Srikakulam, S.K.; Gress, A.; Beckmann, A.; Grißmer, A.; et al. A hydrogel-based in vitro assay for the fast prediction of antibiotic accumulation in Gram-negative bacteria. Mater. Today Bio 2020, 8, 100084. [Google Scholar] [CrossRef]
- Brahima, S.; Boztepe, C.; Kunkul, A.; Yuceer, M. Modeling of drug release behavior of pH and temperature sensitive poly(NIPAAm-co-AAc) IPN hydrogels using response surface methodology and artificial neural networks. Mater. Sci. Eng. C 2017, 75, 425–432. [Google Scholar] [CrossRef]
- Suykens, J.A.K. Support Vector Machines: A Nonlinear Modelling and Control Perspective. Eur. J. Control 2001, 7, 311–327. [Google Scholar] [CrossRef]
- Zhu, J.-A.; Jia, Y.; Lei, J.; Liu, Z. Deep Learning Approach to Mechanical Property Prediction of Single-Network Hydrogel. Mathematics 2021, 9, 2804. [Google Scholar] [CrossRef]
- Ning, H.; Zhou, T.; Joo, S.W. Machine learning boosts three-dimensional bioprinting. Int. J. Bioprint. 2023, 9, 739. [Google Scholar] [CrossRef] [PubMed]
- Allencherry, J.; Pradeep, N.; Shrivastava, R.; Joy, L.; Imbriacco, F.; Özel, T. Investigation of Hydrogel and Gelatin Bath Formulations for Extrusion-Based 3D Bioprinting using Deep Learning. Procedia CIRP 2022, 110, 360–365. [Google Scholar] [CrossRef]
- Ng, W.L.; Chan, A.; Ong, Y.S.; Chua, C.K. Deep learning for fabrication and maturation of 3D bioprinted tissues and organs. Virtual Phys. Prototyp. 2020, 15, 340–358. [Google Scholar] [CrossRef]
- Garcia-del Rio, L.; Diaz-Rodriguez, P.; Landin, M. New tools to design smart thermosensitive hydrogels for protein rectal delivery in IBD. Mater. Sci. Eng. C 2020, 106, 110252. [Google Scholar] [CrossRef]
- Mohammadi, S.; Hashemi, M.; Finklea, F.; Williams, B.; Lipke, E.; Cremaschi, S. Classification of cardiac differentiation outcome, percentage of cardiomyocytes on day 10 of differentiation, for hydrogel-encapsulated hiPSCs. J. Adv. Manuf. Process. 2023, 5, e10148. [Google Scholar] [CrossRef]
- Li, F.; Han, J.; Cao, T.; Li, L. Design of self-assembly dipeptide hydrogels and machine learning via their chemical features. Proc. Natl. Acad. Sci. USA 2019, 116, 11259–11264. [Google Scholar] [CrossRef]
- Nosrati, H.; Nosrati, M. Artificial Intelligence in Regenerative Medicine: Applications and Implications. Biomimetics 2023, 8, 442. [Google Scholar] [CrossRef]
- Zicari, R.V.; Ahmed, S.; Amann, J.; Braun, S.A.; Brodersen, J.; Bruneault, F.; Brusseau, J.; Campano, E.; Coffee, M.; Dengel, A.; et al. Co-Design of a Trustworthy AI System in Healthcare: Deep Learning Based Skin Lesion Classifier. Front. Hum. Dyn. 2021, 3, 688152. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Negut, I.; Bita, B. Exploring the Potential of Artificial Intelligence for Hydrogel Development—A Short Review. Gels 2023, 9, 845. https://doi.org/10.3390/gels9110845
Negut I, Bita B. Exploring the Potential of Artificial Intelligence for Hydrogel Development—A Short Review. Gels. 2023; 9(11):845. https://doi.org/10.3390/gels9110845
Chicago/Turabian StyleNegut, Irina, and Bogdan Bita. 2023. "Exploring the Potential of Artificial Intelligence for Hydrogel Development—A Short Review" Gels 9, no. 11: 845. https://doi.org/10.3390/gels9110845
APA StyleNegut, I., & Bita, B. (2023). Exploring the Potential of Artificial Intelligence for Hydrogel Development—A Short Review. Gels, 9(11), 845. https://doi.org/10.3390/gels9110845







