Abstract
Trametes lactinea is a macrofungus commonly found on broadleaf tree felling piles with rich bioactivity, and its main active substances are polysaccharides, which have a certain medicinal value. Few studies have been conducted on the simultaneous study of intracellular and extracellular polysaccharides of fermented mycelia in liquid culture. In this study, T. lactinea collected from Eucalyptus stumps was subjected to liquid fermentation culturing and the extraction of intracellular and extracellular polysaccharides from the mycelium was optimized using unidirectional and orthogonal test methods. The intracellular and extracellular crude polysaccharides were isolated and purified, and polysaccharide fractions were obtained and assayed for their chemical composition. The main findings of this study are as follows: (1) The optimal conditions for the extraction of intracellular polysaccharides from T. lactinea mycelium were a material–liquid ratio of 1:40, an extraction temperature of 100 °C, a time of 5 h, and four repeats, wherein the polysaccharide extraction rate reached 5.1%. The optimal extraction conditions for the extracellular polysaccharides were a concentration ratio of 5:1, a concentration temperature of 70 °C, an ethanol concentration of 100.0%, and an alcohol settling time of 12 h. The polysaccharides could thus be extracted up to 0.63 mg/mL. (2) The intracellular and extracellular crude polysaccharides of T. lactinea mycelium were successfully isolated and purified, resulting in three homogeneous fractions of intracellular polysaccharides (IP-1, IP-2, and IP-3) and two homogeneous fractions of extracellular polysaccharides (EP-1 and EP-2). (3) The polysaccharide chemical composition resulted in the highest total sugar content of EP-1. IP-3 had the highest uronic acid content. The results of the monosaccharide composition analysis showed that the mycelial intracellular polysaccharides IP-1, IP-2, and IP-3 were all highest in glucose. Both IP-2 and IP-3 contained guluronic acid and glucuronic acid. Mannose was the most abundant extracellular polysaccharide in both EP-1 and EP-2. This study provides theoretical and technical bases for the comprehensive development and utilization of T. lactinea polysaccharides.
1. Introduction
Polysaccharides are complex macromolecules formed by the polymerization of multiple monosaccharides. They can exist independently or form complexes with proteins and flavonoids [1]. These compounds have a high number of hydrophilic groups, making them soluble in water but difficult to dissolve in organic solvents. The intricate structure of polysaccharides, with various connections between the main chain and branched chain, makes them prone to forming complexes with other substances, limiting their in-depth study [2]. Fungal polysaccharides are naturally occurring compounds found in ascospores, mycorrhizae, hyphae, and fermentation broth [3], and have a wide range of biological activities. For example, the polysaccharides from Coriolus versicolor and Agaricus blazei can exert multifaceted regulatory effects on the immune system by promoting cytokine production and other pathways [4]. The polysaccharides from Wolfiporia cocos [5], Lentinus edodes [6], and Grifola frondosa [7] can be used as adjuvants in the treatment of tumors. Both Ganoderma lucidum and Ophiocordyceps sinensis polysaccharides have significant antioxidant effects [8]. L. edodes polysaccharides also have an inhibitory effect on adenovirus type XII and anti-arbor virus [9]. Stropharia rugosoannulata liquid deep culture of extracellular polysaccharides administered via gavage has shown significant hypoglycemic and hypolipidemic effects in mice with streptozotocin-induced diabetes [10]. Hericium erinaceus polysaccharides have a significant hypoglycemic effect and a beneficial effect on serum total cholesterol and triacylglycerol levels [11].
Fungal polysaccharides can be categorized into intracellular and extracellular polysaccharides, depending on the extraction site. Liquid fermentation allows for the extraction of both types, with intracellular polysaccharides being obtained from the mycelium and extracellular polysaccharides being obtained from the fermentation broth. Compared to polysaccharides extracted from substrates, mycelial liquid fermentation offers advantages such as a short growth cycle, simplicity, ease of operation, and suitability for large-scale production. This method can effectively address the challenges posed by high market demands and oversupply [12]. Polysaccharide extraction methods are crucial in polysaccharide research. Numerous reports have been published on various extraction processes [13]. Understanding the impact of different extraction methods on the extraction rate and functional properties of polysaccharides is essential for enhancing their biological activity. For mycelium extraction of polysaccharides, solvent extraction [14], enzyme extraction [15], microwave extraction [16], ultrasonic extraction [17], and other emerging methods [18] can be employed, taking into account the characteristics of fungi. To extract extracellular polysaccharides from fungal liquid fermentation, the ethanol precipitation method is commonly used. This involves concentrating the fungal liquid fermentation to a specific volume, adding a proportionate amount of anhydrous ethanol, and allowing it to sit overnight to obtain crude polysaccharide precipitation.
The polysaccharides obtained through hot water leaching and ethanol precipitation still contain high levels of proteins and small molecule impurities. To accurately determine their chemical composition, it is necessary to separate and purify them further. The purification process involves the removal of polysaccharide impurities, which can be categorized into small molecule removal, protein removal, and decolorization treatment. Small molecule impurities are typically eliminated through dialysis [19]. Protein removal methods consist of the Sevag method [20], trichloroacetic acid method [21], and proteolysis [22]. Decolorization techniques include the use of activated carbon, macroporous resin adsorption, and decolorization by hydrogen peroxide.
Trametes lactinea is a type of fungi that belongs to the Basidiomycota phylum, Agaricomycotina class, Agaricomycetes order, Polyporales family, and Trametes genus [23]. It is commonly found on the wood of broad-leaved trees in temperate regions and is known for its medicinal properties. T. lactinea is rich in nutrients such as brassicas, phenolics, glycoconjugates, polysaccharides, proteins, and total amino acids.
T. lactinea has high medicinal value. For example, 3-hydroxylation of B-sitosterol extracted from T. lactinea has anti-ulcer effects on gastric tissues. At low doses, it selectively inhibits the viability of gastric cancer cells in an acidic environment and reduces apoptosis in HGC-27 cells. The polysaccharides in the fruiting body of T. lactinea can promote apoptosis in human liver cancer Hep G-2 cells, indicating potential anti-cancer abilities. The ethanolic extract of T. lactinea has hypoglycemic effects, particularly in petroleum ether and other organic solvent extraction parts [24,25].
Polysaccharides, in particular, exhibit various pharmacological activities such as antitumor and hepatoprotective effects, making them important active substances in T. lactinea. The polysaccharides in T. lactinea exhibit a good scavenging activity against DPPH, ABTS, and superoxide anion radicals, as well as a good dose-dependent iron ion reduction ability [26]. Polysaccharides also significantly prolong the intoxication time and shorten the sobering time in mice, reduce the liver index of mice with alcohol-induced liver injury, significantly decrease the levels of alanine aminotransferase, aspartate aminotransferase, triglycerides, total cholesterol, and malondialdehyde in serum, and significantly increase the activity of superoxide dismutase and catalase in the liver.
Previous studies on T. lactinea have mainly focused on the physiological activities of ascospores and mycelial polysaccharides [26,27]. However, there is limited research on the production of intracellular and extracellular polysaccharides through liquid fermentation of mycelium, which is unfavorable for the utilization of polysaccharides from T. lactinea. Therefore, in this study, we isolated mycelium from T. lactinea fruit bodies and used the extraction rate (amount) of intracellular and extracellular polysaccharides as an index to optimize the extraction process. We employed a one-way test of variance combined with an orthogonal design to determine the optimal conditions for polysaccharide extraction and achieve a higher polysaccharide content. Additionally, we analyzed the chemical components of polysaccharide fractions, including the total sugars, proteins, uronic acid, sulfate groups, flavonoids, and total phenols. These findings provide a theoretical basis for the comprehensive development and utilization of T. lactinea polysaccharides.
2. Materials and Methods
2.1. Fruit Body Collection
Fungal fruit bodies were collected from the Guangxi State-owned Qipo Forest Farm (108°18′ E, 22°28′ N). It belongs to a typical subtropical monsoon climate, with an annual average temperature of 21.6 °C, an annual average rainfall of 1304.2 mm, an average relative humidity of 79.0%, and an elevation of 150–400 m. The slope of the land ranges from 25 to 30°. The main landform type is hills, and the soil is typical red soil.
The fresh fungal fruit bodies used in this study were collected in late April 2021 from the stumps of second-generation Eucalyptus plantations in this forest farm. A small portion of the stumps, along with the fruit bodies, was cut using a small handsaw, placed in sealed plastic bags, and brought back to the laboratory. The fungal mycelium pure culture was obtained after isolation and purification in the laboratory.
2.2. Fungal Isolation, Purification, and Identification
The fresh fungal fruiting bodies were rinsed with running water and the surface of the fruiting bodies was gently wiped with 75.0% alcohol cotton balls. They were placed on a super-clean workbench, then rinsed 2–3 times with sterile water, and then the remaining water was absorbed with sterile absorbent papers. A part of the fungal cap was cut using sterile scissors, and then a sterile dissecting knife was used to quickly cut small pieces of the exposed flesh and transfer them to the center of the culture medium (glucose 20 g/L, yeast powder 5 g/L, KH2PO4 1.5 g/L, MgSO4 1.0 g/L, vitamin B1 0.1 μg/L, agar 20 g/L, pH natural), for 5 replicates of this inoculation. The plates were inverted and placed in a 25 °C constant-temperature incubator for light-free cultivation, and observed and recorded daily. After the hyphae germinated, we collected a few hyphae from the edge of the colony with a sterile inoculation needle for further purification and cultivation to obtain a pure culture. Fungal DNA was extracted and the fungal ITS sequence was amplified using fungal universal primers ITS4 (5′-TCCTCCGCTTATTGATATATGC-3′) and ITS5 (5′-GGAAGTAAAAGTCGTAACAAGG-3′). The sequencing results were uploaded to the NCBI (http://www.ncbi.nlm.nih.gov, accessed on 10 May 2021) database for comparative analysis via BLAST. The pure culture was identified as T. lactinea based on the sequencing results.
2.3. Liquid Fermentation
A portion of the T. lactinea mycelium was taken from the ultra-clean workbench and inoculated into a 250 mL conical flask containing 100 mL of liquid fermentation medium (glucose 20 g/L, yeast powder 5 g/L, KH2PO4 1.5, MgSO4 1.0 g/L, vitamin B1 0.1 μg/L, pH natural). The flask was sealed with a sterile sealing film and placed in an oscillating incubator at 30 °C with a shaking frequency of 150 r/min for continuous cultivation for 3 days as a seed solution. The seed solution was homogenized using an internal cutting homogenizer to form a fungal suspension, which was then added to 100 mL liquid fermentation medium at a volume ratio of 1:20. Under the same fermentation conditions, the suspension was continuously cultivated in an oscillating incubator at 30 °C for 7 days with 5 replicates (Figure 1).

Figure 1.
Liquid fermentation of T. lactinea mycelium.
2.4. Polysaccharide Extraction Process
2.4.1. Intracellular Polysaccharide Extraction
The polysaccharides inside the mycelium were extracted using the hot water immersion method [28]. The mycelium and liquid were separated using gauze, and the mycelium was rinsed with deionized water 4–5 times, then freeze-dried and crushed. A suitable amount of mycelium powder was mixed with deionized water in a ratio of 1:30 and placed in a constant temperature water bath at 90 °C for 5 h. This extraction process was repeated 2 times, and the resulting mixture was centrifuged at 8000 rpm for 5 min; then, the centrifuged extraction liquid was combined. Ethanol without water was added to the extraction liquid in a volume ratio of 1:3 and mixed well. The mixture was left to stand at 4 °C for 12 h, then centrifuged at 8000 rpm for 5 min at 25 °C to collect the precipitate. After the ethanol evaporated, the precipitate was dissolved in deionized water to obtain a crude polysaccharide solution. The solution was then dialyzed using a dialysis bag (8000–12,000 Da) with running water for 24 h, followed by dialysis with deionized water for another 24 h. The solution was concentrated using a rotary evaporator and then dried using a freeze-drying machine to obtain the crude polysaccharide dry product (Figure 2).
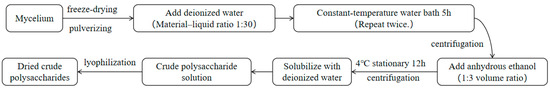
Figure 2.
Extraction process for the intracellular polysaccharides.
2.4.2. Extracellular Polysaccharide Extraction
The ethanol precipitation method was used to extract the extracellular polysaccharides from the mycelium [29]. The mycelial fermentation liquid was separated using gauze and then centrifuged at 8000 r/min for 5 min at 4 °C. The centrifuged mycelial liquid was then combined and concentrated at a 5:1 volume ratio using a rotary evaporator at 60 °C. The volume of 95.0% ethanol was added three times to the concentrated mycelial liquid and quickly stirred. The mixture was left to stand at 4 °C for 12 h, followed by the same steps as the intracellular polysaccharide extraction to obtain crude extracellular polysaccharide powder (Figure 3).
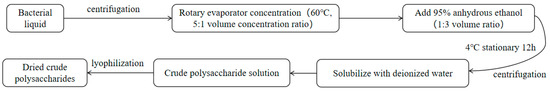
Figure 3.
Extraction process for the extracellular polysaccharides.
2.4.3. Determination of Extraction Rate (Amount)
The crude polysaccharide dry product was diluted with deionized water to a volume of 100.0 mL as the sample solution. A 1.0 mL portion of this sample solution was taken in a test tube, to which 1.0 mL of 5.0% phenol solution was added and mixed well. Then, 5.0 mL of concentrated sulfuric acid was quickly added and the mixture was allowed to stand at room temperature for 10 min. It was then placed in a 30 °C water bath and reacted for 20 min. The absorbance was measured at a wavelength of 490 nm. The polysaccharide extraction rate (amount) was calculated using the following formula. Each treatment was repeated 3 times.
In this formula, the glucose concentration (mg/mL) of the sample solution is C, the dilution factor is N, the total volume of the sample solution (mL) is V, and the amount of the raw material sample (mg or mL) is M.
2.4.4. One-Way Test of Variance
- (1)
- Intracellular polysaccharides
A one-way test was conducted to investigate the factors affecting the extraction rate of intracellular polysaccharides of T. lactinea, including the liquid-to-material ratio, extraction time, extraction temperature, and extraction frequency. The basic experimental conditions were a liquid-to-material ratio of 1:30, an extraction temperature of 90 °C, an extraction time of 5 h, and an extraction frequency of 2 times. The controlled-variable method was used to keep other conditions constant. The effects of different liquid-to-material ratios (1:10, 1:20, 1:30, 1:40, 1:50), extraction temperatures (60 °C, 70 °C, 80 °C, 90 °C, 100 °C), extraction times (1 h, 2 h, 3 h, 4 h, 5 h, 6 h), and extraction frequencies (1 time, 2 times, 3 times, 4 times) on the extraction rate of intracellular polysaccharides were compared.
- (2)
- Extracellular polysaccharides
Similar to the intracellular polysaccharides, a one-way test also was applied to investigate the factors that influence the extraction of extracellular polysaccharides, including the concentration ratio, concentration temperature, ethanol concentration, and alcohol precipitation time. The basic experimental conditions were a concentration ratio of 5:1, a concentration temperature of 60 °C, an ethanol concentration of 95.0%, and an alcohol precipitation time of 12 h. The method of controlling variables was used, keeping other conditions constant, in order to compare the effects of different concentration ratios (2:1, 3:1, 4:1, 5:1, 6:1), concentration temperatures (50 °C, 60 °C, 70 °C, 80 °C, 90 °C), ethanol concentrations (75.0%, 80.0%, 85.0%, 90.0%, 95.0%, 100.0%), and alcohol precipitation times (6 h, 12 h, 18 h, 24 h, 30 h) on the extraction of extracellular polysaccharides.
2.4.5. Orthogonal Test
- (1)
- Intracellular polysaccharides
Based on the one-way test, a four-factor three-level orthogonal experiment (L9 (34)) was conducted to investigate the effects of four factors, namely the liquid-to-solid ratio, extraction time, extraction temperature, and number of extractions, on the extraction rate of intracellular polysaccharides. This experiment aimed to determine the optimal extraction conditions. The orthogonal design and factor levels can be found in Table 1.

Table 1.
Orthogonal test of the intracellular polysaccharide extraction process.
- (2)
- Extracellular polysaccharides
Another four-factor three-level orthogonal experiment (L9 (34)) was also conducted to investigate the effects of the concentration ratio, concentration temperature, ethanol concentration, and alcohol precipitation time on the extraction yield of extracellular polysaccharides. The orthogonal design and factor levels for this are shown in Table 2.

Table 2.
Orthogonal test of the extracellular polysaccharide extraction process.
- (3)
- Verification test
Five replicates were performed according to the optimal extraction conditions for the intracellular and extracellular polysaccharides, as optimized by the orthogonal experiments, in order to verify the rationality and reliability of the optimization results.
2.5. Determination of Chemical Composition
The dried intracellular and extracellular polysaccharides were obtained following the method described by Hou et al. [30]. DEAE-52 cellulose was pretreated and loaded onto a column to initially separate the intracellular and extracellular polysaccharide fractions, based on the method developed by Zhang et al. [31]. The purified intracellular and extracellular polysaccharide fractions were obtained by loading dextran gel G-100 on the column, as described by Zhang et al. The total sugar content was determined using the phenol–sulfuric acid method [32]. Protein content was determined using the Coomassie Brilliant Blue method [33]. Uronic acid content was determined using the sulfuric acid–carbazole method [34]. Sulfuric acid-based content was determined using the barium sulfate–turbidimetry method [35]. Flavonoid content was determined using the rutin standard method [36]. Total phenols content was determined using the Folin–Ciocalteau method [37]. The monosaccharide composition was determined via ion chromatography [38].
2.6. Data Analysis
The data processing in this study was carried out using Microsoft Excel 2019. The results of the orthogonal test were analyzed by one-way ANOVA using SPSS 26.0, and a graphical representation was created using Origin 2023. Each group of tests was repeated three times during the experiment, and the experimental data were presented as their mean ± standard deviation values.
3. Results
3.1. One-Way Test of Variance for the Intracellular Polysaccharide Extraction Process
The effect of the feed–liquid ratio on the extraction rate of intracellular polysaccharides was investigated at an extraction temperature of 90 °C, extraction time of 5 h, and with two repetitions of extraction. Figure 4a shows that the extraction rate of the intracellular polysaccharides initially increased from 3.8% and peaked at 4.3% with a feed–liquid ratio of 1:30. The highest extraction rate was observed at a feed–liquid ratio of 1:30, and the neighboring peaks at ratios 1:20 and 1:40 were also selected for subsequent optimization experiments.
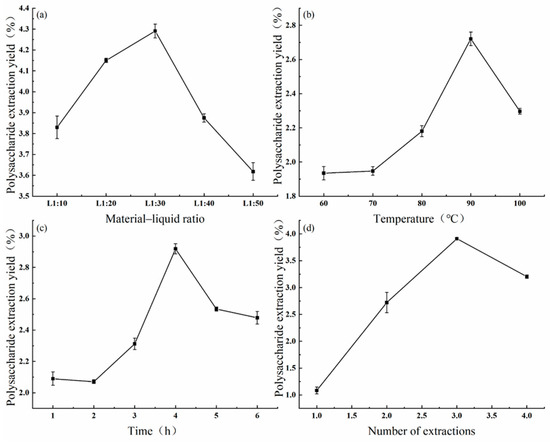
Figure 4.
Effects of the material–liquid ratio (a), extraction temperature (b), extraction time (c), and extraction number (d) on the extraction of intracellular polysaccharides.
Similarly, the effect of the extraction temperature on the extraction rate of intracellular polysaccharides was investigated at a feed–liquid ratio of 1:30 and extraction time of 5 h, with two repetitions of extraction. Figure 4b shows that the extraction rate increased from 1.9% to 2.7% with increases in the temperature from 60 °C to 90 °C, peaking at 90 °C. However, the extraction rate decreased significantly to 2.3% at temperatures above 90 °C. Therefore, the extraction temperature of 90 °C, as well as temperatures of 80 °C and 100 °C, were selected for subsequent optimization tests.
The effect of the extraction time on the extraction rate of intracellular polysaccharides was investigated at a feed–liquid ratio of 1:30 and extraction temperature of 90 °C, with two repetitions of extraction. Figure 4c shows that the extraction rate increased from 2.1% to 2.9% within 1–4 h, peaking at 4 h. However, the extraction rate decreased to 2.5% with extraction times longer than 4 h. Therefore, we selected the extraction time of 4 h, as well as the times of 3 h and 5 h, for subsequent optimization experiments.
Lastly, the effects of the number of extraction times on the extraction rate of intracellular polysaccharides were investigated at a feed–liquid ratio of 1:30, extraction temperature of 90 °C, and extraction time of 5 h. The results showed that the extraction rate increased linearly with the number of extractions. Figure 4d demonstrates that the extraction rate reached a peak value of 3.9% at three times of repeated extractions, after which it decreased to 3.2% with further extractions. Therefore, we selected an extraction repeat number of 3 times, as well as the repeat numbers of 2 and 4 times, for subsequent optimization tests.
3.2. Orthogonal Optimization of Intracellular Polysaccharide Extraction
The orthogonal method was employed to determine the optimal conditions for extracting intracellular polysaccharides. These test results are presented in Table 3. Based on the R-value, the factors influencing the extraction rate of intracellular polysaccharides were ranked as follows: extraction temperature > number of extractions > extraction time > material–liquid ratio. This indicates that extraction temperature has the greatest impact on the polysaccharide extraction rate. By analyzing the K-value, it was determined that the ideal combination for the best intracellular polysaccharide extraction rate was A3B3C3D3, which corresponds to a material–liquid ratio of 1:40, an extraction temperature of 100 °C, a duration of 5 h, and four repeats. The ANOVA results in Table 4 demonstrate that the material–liquid ratio, extraction temperature, extraction time, and number of extractions significantly affect the extraction rate of intracellular polysaccharides (p < 0.01). The extraction was repeated five times using the optimal conditions to validate the optimization results. The average intracellular polysaccharide extraction rate obtained was 5.1%, which was higher than the rates achieved with other combinations. This confirms the reliability of the optimization results.

Table 3.
Orthogonal test results for the extraction of intracellular polysaccharides.

Table 4.
Variance analysis results for the effects of extraction conditions on the extraction rate of intracellular polysaccharides.
3.3. One-Way Test of Variance for the Extracellular Polysaccharide Extraction Process
The effects of different concentration ratios (2:1, 3:1, 4:1, 5:1, 6:1) on the extraction of extracellular polysaccharides were investigated at a temperature of 60 °C, using ethanol with a concentration of 95.0%, and with an alcohol settling time of 12 h. The results were summarized as follows: Figure 5a shows that the extraction of extracellular polysaccharides increased linearly with increases in the concentration ratio of the bacterial broth within the range of 2:1 to 4:1. At a concentration ratio of 4:1, the extraction reached a peak value of 0.37 mg/mL. However, beyond this ratio, the extraction decreased to 0.20 mg/mL, which was approximately half of the value at the concentration ratio of 4:1. Therefore, the concentration ratio of 4:1 was chosen, along with the neighboring peaks on both sides at the ratios of 3:1 and 5:1, for further optimization tests.
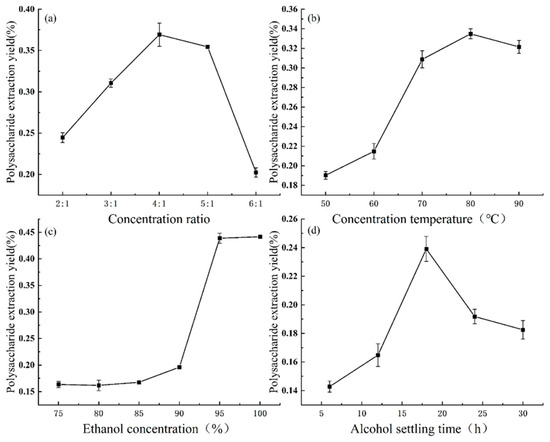
Figure 5.
Effects of the concentration ratio (a), concentration temperature (b), ethanol concentration (c), and alcohol settling time (d) on the extraction of extracellular polysaccharides.
The effects of different concentration temperatures (50 °C, 60 °C, 70 °C, 80 °C, 90 °C) on the extraction of extracellular polysaccharides were investigated at a concentration ratio of 5:1, using ethanol with a concentration of 95.0%, and with an alcohol settling time of 12 h. The results were summarized as follows: Figure 5b indicates that the amount of extracellular polysaccharides extracted increased gradually with increases in the concentration temperature. The extraction reached a peak value of 0.33 mg/mL at a temperature of 80 °C, after which it slightly decreased to 0.32 mg/mL. Therefore, the concentration temperature of 80 °C was selected, along with the neighboring peaks on both sides at temperatures 70 °C and 90 °C, for subsequent optimization tests.
The effect of different ethanol concentrations (75.0%, 80.0%, 85.0%, 90.0%, 95.0%, and 100.0%) on the extraction of extracellular polysaccharides was investigated at a concentration ratio of 5:1, a concentration temperature of 60 °C, and an alcohol settling time of 12 h. The results were summarized as follows: Figure 5c shows that the increase in the amount of extracted polysaccharides was slow within the range of 75.0% to 90.0% ethanol concentration, with only 0.20 mg/mL extracted at a concentration of 90.0%. However, the amount of extracted polysaccharides sharply increased to 0.44 mg/mL within the range of 90.0% to 95.0% ethanol concentration, and any further increase in the ethanol concentration did not significantly affect the extraction. Therefore, the ethanol concentrations of 95.0% and 100.0%, along with 90.0%, which was before the plateau was reached, were chosen for subsequent optimization tests.
The effect of different alcohol settling times (6 h, 12 h, 18 h, 24 h, and 30 h) on the extraction of extracellular polysaccharides was investigated at a concentration ratio of 5:1, a concentration temperature of 60 °C, and an ethanol concentration of 95.0%. As shown in Figure 5d, the amount of extracted polysaccharides continued to increase from 6 h to 18 h of settling, reaching a peak of 0.24 mg/mL at 18 h. After 18 h, the extraction gradually decreased. Therefore, we selected the alcohol settling time of 18 h, along with the times of the two neighboring peaks at 12 h and 24 h, for subsequent optimization tests.
3.4. Orthogonal Optimization of Extracellular Polysaccharide Extraction
The orthogonal method was utilized to determine the optimal conditions for extracting extracellular polysaccharides. These test results are presented in Table 5. Based on the R-value, it is evident that the concentration ratio had the greatest impact on polysaccharide extraction, followed by the ethanol concentration, concentration temperature, and alcohol settling time, in that order. K-value analysis revealed that the ideal combination for extracellular polysaccharide extraction was A3B1C3D1, which corresponds to a concentration ratio of 5:1, a concentration temperature of 70 °C, an ethanol concentration of 100.0%, and an alcoholic settling time of 12 h. The ANOVA results in Table 6 demonstrate that the concentration ratio, concentration temperature, ethanol concentration, and alcohol settling time significantly influenced the amount of extracellular polysaccharides extracted (p < 0.01). To validate these results, the optimal extraction conditions were repeated five times, yielding a mean value of 0.63 mg/mL for extracellular polysaccharide extraction. This value was higher than those obtained from other combinations, confirming the reliability of the optimization results.

Table 5.
Orthogonal test results for the extraction of extracellular polysaccharides.

Table 6.
Variance analysis results for the effects of extraction conditions on the extraction of extracellular polysaccharides.
3.5. Isolation and Purification of Intracellular Polysaccharides
3.5.1. Results of the Isolation and Purification of Intracellular Crude Polysaccharides
The extraction of intracellular polysaccharides was performed using optimized conditions, including a material–liquid ratio of 1:40, an extraction temperature of 100 °C, a time of 5 h, and with four repeats. The extracted crude polysaccharides were then deproteinized and separated using DEAE-52 cellulose anion-exchange chromatography columns. Elution was conducted using a concentration gradient of deionized water, 0.1 mol/L, 0.3 mol/L, and 0.5 mol/L of NaCl. The absorbance values at 490 nm of each eluate tube were determined using the phenol–sulfuric acid method to obtain the elution curve (Figure 6). The absorption peaks obtained in the water elution part (tubes 10–21), 0.1 mol/L NaCl elution part (tubes 42–49), and 0.3 mol/L NaCl elution part (tubes 69–75) exhibited well-defined peak shapes without any obvious trailing. Consequently, the eluents corresponding to the absorption peak tubes were collected, labeled as IP-1, IP-2, and IP-3, and then concentrated, dialyzed, freeze-dried, and further investigated.
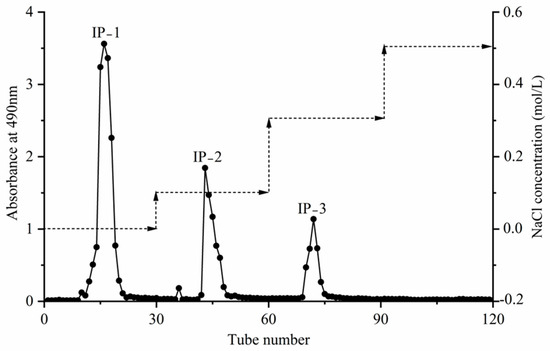
Figure 6.
DEAE-52 cellulose anion exchange elution profile of the intracellular polysaccharides.
3.5.2. Results of the Separation and Purification of Intracellular Polysaccharides
The intracellular polysaccharides IP-1, IP-2, and IP-3 were initially separated and purified. They were then dissolved in deionized water and further purified using a dextran gel G-100 column. Deionized water was used as the mobile phase, and the absorbance value of each eluate tube was measured at 490 nm using the phenol–sulfuric acid method. This allowed us to create an elution curve, as shown in Figure 7. IP-1, IP-2, and IP-3 exhibited single absorption peaks with well-defined shapes. The eluates corresponding to these peaks were collected, concentrated, dialyzed, and finally freeze-dried to obtain the purified IP-1, IP-2, and IP-3 (Figure 7).
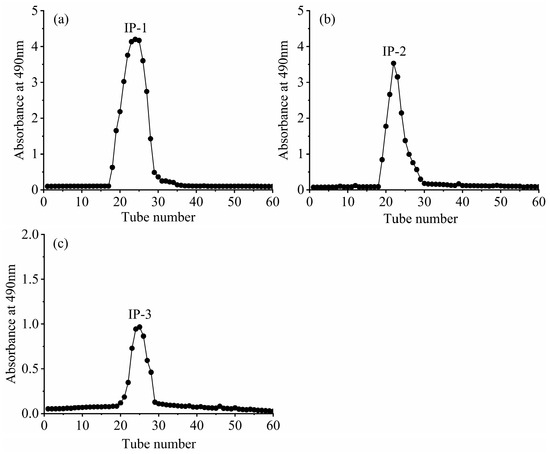
Figure 7.
Dextran gel elution of intracellular polysaccharides IP-1 (a), IP-2 (b), and IP-3 (c).
3.6. Isolation and Purification of Extracellular Polysaccharides
3.6.1. Results of the Isolation and Purification of Extracellular Crude Polysaccharides
Extracellular polysaccharide extraction was performed using optimal conditions: a concentration ratio of 5:1, a concentration temperature of 70 °C, a 100.0% ethanol concentration, and a 12 h alcohol settling time. The extracted crude polysaccharides were deproteinized and separated using DEAE-52 cellulose anion-exchange chromatography columns. Elution was carried out using a concentration gradient of deionized water, and NaCl at concentrations of 0.1 mol/L, 0.3 mol/L, and 0.5 mol/L. The absorbance values at 490 nm of each eluate tube were determined using the phenol–sulfuric acid method to obtain the elution curve (Figure 8). The absorption peaks obtained in the water elution section (tubes 11–21) and the 0.1 mol/L NaCl elution section (tubes 42–49) displayed good peak shapes and no obvious trailing. However, the absorption peaks obtained in the 0.2 mol/L NaCl elution section (tubes 71–77) were low and impure and therefore were not further studied. In summary, the eluents corresponding to the tubes with absorption peaks were collected, named EP-1 and EP-2, concentrated, dialyzed, freeze-dried, and further analyzed.
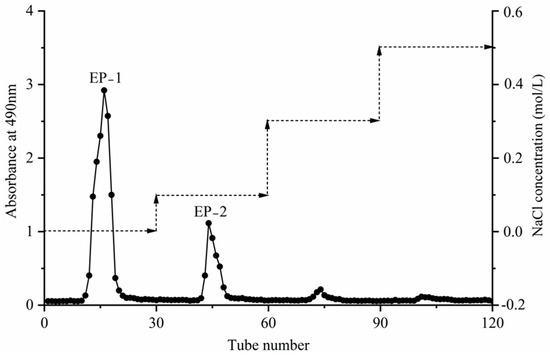
Figure 8.
DEAE-52 cellulose anion exchange elution profile of the extracellular polysaccharides.
3.6.2. Results of the Separation and Purification of Extracellular Polysaccharides
The EP-1 and EP-2 extracellular polysaccharides, obtained through preliminary isolation and purification, were dissolved in deionized water. They were further purified using a dextran gel G-100 chromatography column with deionized water as the mobile phase. The eluent from each tube was analyzed for absorbance at 490 nm using the phenol–sulfuric acid method to obtain the elution curve (Figure 9). Figure 9 shows that EP-1 and EP-2 exhibited a single absorption peak with a well-defined shape. The eluent corresponding to this absorption peak was collected, concentrated, dialyzed, and finally freeze-dried to obtain the purified EP-1 and EP-2.
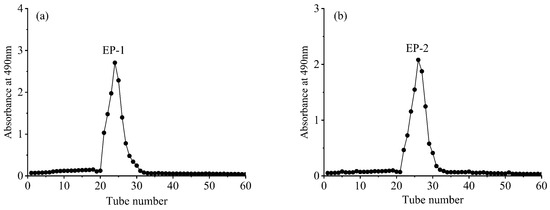
Figure 9.
Dextran gel elution of extracellular polysaccharides EP-1 (a) and EP-2 (b).
3.7. Chemical Compositional Analysis
3.7.1. Polysaccharide Chemical Composition
The chemical composition of the intracellular polysaccharide fractions (IP-1, IP-2, IP-3) and extracellular polysaccharide fractions (EP-1, EP-2) of T. lactinea mycelium was determined (Table 7). As shown in Table 8, EP-1 had the highest total sugar content of 55.7%, followed by IP-1 at 52.9%. The protein content of IP-1 was 0.4%, and the protein content of EP-1 was only 0.1%, which was negligible, and IP-2, IP-3, and EP-2 did not contain any proteins. The glucuronide contents of IP-2 and IP-3 were higher than that of IP-1, and EP-2’s glyoxalate content, as well as that of of IP-2 and IP-3, was higher than that of EP-1, with IP-3 having the highest uronic acid of 10.1%. The sulfate group contents in the five polysaccharide fractions were similar, with a mean value of 11.4%, with EP-1 having the highest sulfate group content of 12.8%. After isolation and purification, both the intracellular and extracellular polysaccharide fractions contained a few of flavonoids and total phenols, among which IP-3 contained higher levels of flavonoids and total phenols of 1.2% and 1.1%, respectively.

Table 7.
Chemical composition contents of the intracellular and extracellular polysaccharides.

Table 8.
Monosaccharide compositions and molar ratios of the intracellular and extracellular polysaccharides.
3.7.2. Monosaccharide Composition
As seen in Figure 10a and Table 8, the rapid isolation of 16 standard monosaccharides was carried out within 60 min, with the intracellular polysaccharide IP-1 consisting of fucose (Fuc), by matching its retention time with that of the monosaccharide standard, galactose (Gal), glucose (Glc), and mannose (Man) at a molar ratio of 1.3:3.5:90.2:5.0. Both IP-2 and IP-3 consisted of Fuc, glucosamine hydrochloride (GlcN), Gal, Glc, Man, gulose amino acid (GulA), and glucuronic acid (GlcA) with molar ratios of 1.3:0.9:3.7:74.9:11.3:1.8:6.1 and 2.9:0.5:7.1:63.3:3.1:4.7:18.5, respectively. The highest content of Glc was found in IP-1, IP-2, and IP-3, while IP-2 and IP-3 both contained GulA and GlcA, and IP-3 had a higher content of GulA and GlcA. As shown in Figure 10b and Table 8, the extracellular polysaccharide EP-1 consisted of Fuc, aminogalactose hydrochloride (GalN), arabinose (Ara), GlcN, Gal, Glc, and Man, with a molar ratio of 11.5:0.2:2.0:1.6:22.7:2.0:55.9. EP-2 consisted of Fuc, GalN, Ara, GlcN, Gal, Glc, Man, and Rha, with a molar ratio of 7.0:0.2:2.3:8.0:1.8:25.4:3.7:51.6. Both EP-1 and EP-2 had the highest content of Man, and none of them contained the uronic acid component.
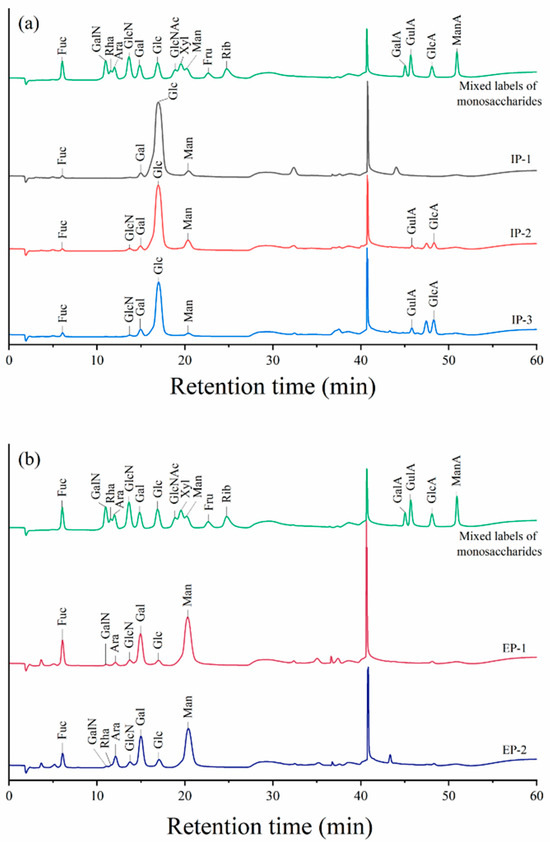
Figure 10.
Ion chromatograms of the monosaccharide compositions of intracellular (a) and extracellular (b) polysaccharides. Fuc, fucose; GalN, aminogalactose hydrochloride; Rha, rhamnose; Ara, arabinose; GlcN, glucosamine hydrochloride; Gal, galactose; Glc, glucose; GlcNAc, N-acetyl-D-glucosamine; Xyl, xylose; Man, mannose; Fru, fructose; Rib, ribose; GalA, galacturonic acid; GulA, gulo-glucuronide; GlcA, glucuronide; ManA, mannose aldehyde.
4. Discussion
4.1. Polysaccharide Extraction
Polysaccharide extraction is commonly executed using traditional methods such as hot water and acid-based extraction, as well as newer techniques like ultrasonic and microwave extraction. The traditional hot water leaching method involves using high temperatures to separate polysaccharide cells from the plasmic wall, followed by precipitation of the polysaccharides using anhydrous ethanol [39,40]. In this study, hot water immersion was used to extract intracellular polysaccharides from T. lactinea mycelium, while ethanol alcohol precipitation was used for extracellular polysaccharides. Wang et al. [41] extracted polysaccharides from Morchella sextelata using hot water leaching, with optimal extraction conditions of 89.94 °C, a liquid-to-material ratio of 31.07 mL/g, and a time of 162.86 min, resulting in a polysaccharide extraction rate of 24.0%. Qiao et al. [42] optimized the process of extracting extracellular polysaccharides from L. edodes using response surface design, finding that the optimal conditions were a 78.7% ethanol concentration, a pH of 7.93, and an alcohol settling time of 17.71 h. Despite the availability of new techniques, the traditional hot water leaching and ethanol alcohol precipitation methods are still widely used due to their low cost, simplicity, and mild conditions.
In this study, the process of extracting intracellular and extracellular polysaccharides from T. lactinea mycelium was investigated using a one-way test of variance method. The extraction rate of intracellular polysaccharides showed an increasing and then decreasing trend with increases in the material–liquid ratio, reaching a peak of 4.3% at a ratio of 1:30. This may be because increasing the solvent gradually solubilizes the polysaccharides and accelerates their diffusion, leading to an increased extraction rate. However, when the material–liquid ratio is too high, other substances in the mycelia start to dissolve, resulting in a decrease in the extraction rate [43]. The extraction rate of intracellular polysaccharides also showed a slow increase followed by a gradual decrease with increases in the extraction temperature, reaching a peak of 2.7% at 90 °C. Higher temperatures accelerate the molecular movement and polysaccharide solubilization, but temperatures above 90 °C can lead to polysaccharide degradation and a decrease in the extraction rate [44]. Similarly, the extraction rate of intracellular polysaccharides increased and then decreased with increases in the extraction time, reaching a peak of 2.9% at 4 h. Prolonged high temperatures can lead to polysaccharide degradation and a decrease in the extraction rate [45]. The extraction rate of the intracellular polysaccharides increased linearly and then decreased with the number of extraction repetitions, reaching a peak of 3.9% at three repetitions of extraction. Too many repetitions can lead to polysaccharide structure destruction and the dissolution of impurities, resulting in a decrease in the extraction rate [44]. The extraction rate of the extracellular polysaccharides from mycelium showed a linear increase followed by a sharp decrease with increases in the concentration ratio, reaching a peak of 0.37 mg/mL at a ratio of 4:1. High concentration ratios decrease solubility and cause precipitation of the polysaccharides, reducing the amount extracted. The extracted amount of extracellular polysaccharides also showed a trend of first increasing and then slowly decreasing with increases in the concentration temperature, reaching a peak of 0.33 mg/mL at 80 °C. Low concentration temperatures result in low solubility, while high temperatures can lead to polysaccharide cleavage and a decreased extraction amount. The amount of extracted extracellular polysaccharides increased slowly followed by a sharp decrease, before then levelling off, with increases in the ethanol concentration. Higher ethanol concentrations promote polysaccharide precipitation. Prolonging the alcoholic settling time resulted in an increasing and then decreasing trend, reaching a peak of 0.24 mg/mL at 18 h. Excessive settling time increases impurities and reduces polysaccharide extraction.
The one-way test of variance combined with an orthogonal approach is commonly used for optimizing fungal polysaccharide extraction processes [46]. In this study, the optimal extraction process for intracellular polysaccharides from T. lactinea mycelium was determined to be a material–liquid ratio of 1:40, an extraction temperature of 100 °C, and a duration of 5 h, with four repeats. Xiao et al. [47] used an orthogonal design to optimize the extraction process of Boletus spp. polysaccharides and found that the optimal conditions were a material–water ratio of 1:20 g/mL, two extractions, an extraction time of 1.5 h, and an ethanol concentration of 80.0%. The extraction temperature had the greatest effect on the extraction rate of intracellular polysaccharides from T. lactinea mycelium. Hao et al. [48] used a response surface design to optimize the extraction process of polysaccharides from T. lactinea mycelium and concluded that extraction temperature is an important factor affecting polysaccharide yield. The optimized temperature conditions for Ganoderma applanatum polysaccharide extraction, when studied by Du et al. [49], were also significantly higher than other factors, highlighting the importance of temperature control in intracellular polysaccharide extraction. The optimal extraction process for extracellular polysaccharides from T. lactinea mycelium was determined to be a concentration ratio of 5:1, a temperature of 70 °C, a 100.0% ethanol concentration, and 12 h of alcoholic settling time, which aligns with the optimal conditions for extracellular polysaccharide extraction from Pleurotus nebrodensis determined by Ma et al. [50] using orthogonal testing. The concentration ratio had the greatest effect on the amount of extracellular polysaccharides extracted from T. lactinea mycelium, consistent with other previous findings for G. applanatum [51] and Pleurotus citrinopileatus [52]. After orthogonal optimization, the intracellular polysaccharide extraction rate of T. lactinea mycelium reached 5.1%, higher than the polysaccharide extraction rate optimized by Hao et al. [48] but lower than the polysaccharide extraction rate of Perenniporia fraxinea mycelium optimized by Lu et al. [53]. Our extracellular polysaccharide extraction from T. lactinea mycelium reached 0.63 mg/mL after orthogonal optimization, higher than the optimized extraction of Tricholoma matsutake [54], but lower than the optimized extraction of Cordyceps militaris [55].
4.2. Polysaccharide Purification
Fungal polysaccharides consist of various components with different charges and molecular weights. To analyze the structure of intracellular and extracellular polysaccharides of T. lactinea mycelium, it is necessary to isolate and purify crude polysaccharides to obtain homogeneous fractions [56]. Before isolation and purification, deproteinization is performed to remove solubilized proteins that are extracted along with the polysaccharides. Common methods for protein removal include the Sevag method, trichloroacetic acid method, and enzymatic digestion [56]. In this study, the Sevag method was used for deproteinization due to its mild and effective denaturation of proteins. Huang et al. [57] compared different deproteinization methods for Armillariella tabescens polysaccharides and found that the Sevag method had the highest polysaccharide yield.
Currently, polysaccharide isolation and purification methods focus on ion exchange chromatography and gel chromatography [58]. DEAE-52 cellulose anion exchange chromatography is commonly used to separate polysaccharides with different charges. Different concentrations of NaCl solutions are used to elute the neutral and acidic sugar fractions [59]. Gel chromatography, on the other hand, separates and purifies polysaccharides based on their molecular weight differences in the mobile phase. In this study, the T. lactinea mycelium intracellular crude polysaccharides were separated into three fractions (one neutral—IP-1; two acidic—IP-2 and IP-3), and the extracellular crude polysaccharides were separated into two fractions (one neutral—EP-1; one acidic—EP-2) using dextran gel chromatography. Hao et al. [48], He et al. [60], Bao et al. [61], and Shi et al. [62] also used similar methods to isolate and purify polysaccharides from different sources.
4.3. Chemical Composition of Polysaccharide Components
Chemical analysis revealed significant differences in the polysaccharide fractions after isolation and purification [63]. In this study, only IP-1 and EP-1 contained trace amounts of proteins, suggesting the formation of glycoproteins through the combination of proteins with polysaccharides. However, none of the other polysaccharide fractions contained proteins, indicating successful deproteinization, which aligns with the study of Zhang et al. [64]. The polysaccharides were isolated and purified with minimal protein content. Additionally, all polysaccharide fractions contained a few flavonoids and total phenols, except IP-1. It is possible that these compounds formed stable complexes with the polysaccharides through hydrogen bonding and covalent bonding, which may enhance their functional properties. The polysaccharide fractions eluted by different concentrations of NaCl had higher glyoxalate contents, indicating that the fractions eluted by deionized water were neutral sugars, while the fractions eluted by NaCl were acidic sugars. Among the intracellular polysaccharide fractions, IP-3 had the highest glyoxalate content, possibly due to its higher concentration of eluting salts [65]. The crude polysaccharides were effectively deproteinized and isolated to obtain homogeneous fractions free of unwanted impurities for further structural and biological analyses.
In this study, intracellular and extracellular polysaccharides were determined via ion chromatography, and significant differences were found in the major monosaccharide compositions of the intracellular and extracellular polysaccharides, with glucose accounting for the highest percentage of the intracellular polysaccharides IP-1, IP-2, and IP-3, which is similar to the monosaccharide composition of the polysaccharides extracted by Inara et al. [66] from Fomitopsis betulina, with glucose as the major component. The highest percentages in the extracellular polysaccharides EP-1 and EP-2 both consisted of mannose, whereas the extracellular polysaccharides extracted and isolated from Coprinus comatus by Cao et al. [67] consisted of galactose and xylose in larger proportions; this discrepancy may originate from the differences in strain origin, medium selection, cultivation conditions, and extraction conditions.
5. Conclusions
The extraction process of intracellular and extracellular polysaccharides of T. lactinea liquid fermentation culture was optimized, and the chemical composition of these polysaccharides was explored in this study. The optimal extraction conditions for extracting intracellular polysaccharides from T. lactinea mycelium were found to be a material–liquid ratio of 1:40, an extraction temperature of 100 °C, a duration of 5 h, and four repeats. Under these conditions, the polysaccharide extraction rate reached 5.1%. The best conditions for extracellular polysaccharide extraction were determined to be a concentration ratio of 5:1, a concentration temperature of 70 °C, an ethanol concentration of 100.0%, and an alcoholic settling time of 12 h. Three fractions of intracellular polysaccharides (IP-1, IP-2, and IP-3) and two fractions of extracellular polysaccharides (EP-1 and EP-2) were obtained from the crude polysaccharides. EP-1 had the highest total sugar content. IP-1 and EP-1 contained a few proteins, while the other fractions were protein-free. IP-2 and IP-3 had higher uronic acid contents than IP-1, and EP-2 had a higher uronic acid content than EP-1. IP-1 and EP-1 were neutral sugars, while IP-2, IP-3, and EP-2 were acidic sugars. All five polysaccharide fractions had similar sulfate group contents and contained a few flavonoids and total phenols. IP-3 had higher amounts of flavonoids and total phenols compared to the other fractions. IP-1, IP-2, and IP-3 all had the highest levels of glucose. Both IP-2 and IP-3 contained guluronic acid and glucuronic acid. Mannose was the most abundant extracellular polysaccharide in both EP-1 and EP-2. This study provides a theoretical and technical basis for the comprehensive development and utilization of T. lactinea polysaccharides.
Author Contributions
Conceptualization, Y.L., L.S. and F.C.; methodology, Y.L., L.S. and M.Y.; software, Y.L., L.S. and K.Y.; validation, Y.L., L.S. and F.C.; formal analysis, F.C.; investigation, Y.L., L.S. and F.C.; resources, M.Y. and K.Y.; data curation, Y.L. and L.S.; writing—original draft preparation, Y.L. and L.S.; writing—review and editing, M.Y., K.Y. and F.C. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by the Guangxi Key Research and Development Program “Collection, Preservation, and Ecologically Efficient Cultivation and Utilization of Understory Economic Characteristic Germplasm Resources in Guangxi”, grant number: Guike AB21238014.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Acknowledgments
The authors would like to thank all staff involved in the field sampling and experiments for their technical assistance.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Liao, Q.; Pang, L.; Li, J.; Zhang, C.; Li, J.; Zhang, X.; Mao, T.; Wu, D.; Ma, X.; Geng, F.; et al. Characterization and diabetic wound healing benefits of protein-polysaccharide complexes isolated from an animal ethno-medicine Periplaneta americana L. Int. J. Biol. Macromol. 2022, 195, 466–474. [Google Scholar] [CrossRef] [PubMed]
- Bian, C.; Wang, Z.; Shi, J. Extraction optimization, structural characterization, and anticoagulant activity of acidic polysaccharides from Auricularia auricula—judae. Molecules 2020, 25, 710. [Google Scholar] [CrossRef]
- Wasser, S.P. Current findings, future trends, and unsolved problems in studies of medicinal mushrooms. Appl. Microbiol. Biotechnol. 2011, 89, 1323–1332. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Sun, T.; Wang, F.; Zhang, J.; Li, C.; Chen, X.; Li, Q.; Sun, S. A polysaccharide from the fungi of Huaier exhibits anti-tumor potential and immunomodulatory effects. Carbohyd. Polym. 2013, 92, 577–582. [Google Scholar] [CrossRef] [PubMed]
- Cao, Y. Studies on the pharmacological effects of Poria cocos polysaccharides. Chin. J. Mod. Drug Appl. 2013, 7, 217–218. [Google Scholar]
- Ma, Y.; Zhao, X. Clinical observation of Lentinan injection combined with chemotherapy in treatment of colorectal cancer. Chin. Arch. Tradit. Chin. Med. 2013, 31, 691–694. [Google Scholar]
- Zhang, B.; Wu, Y.; Liu, W.; Qiu, H.; Sun, P.; Zhang, A. Research progress on preparation and pharmacological activities of polysaccharide from Grifola frondosa. Edible Med. Mushrooms 2019, 27, 99–105. [Google Scholar]
- Kozarski, M.; Klaus, A.; Nikšić, M.; Vrvić, M.M.; Todorović, N.; Jakovljević, D.; Van Griensven, L.J.L.D. Antioxidative activities and chemical characterization of polysaccharide extracts from the widely used mushrooms Ganoderma applanatum, Ganoderma lucidum, Lentinus edodes and Trametes versicolor. J. Food Compos. Anal. 2012, 26, 144–153. [Google Scholar] [CrossRef]
- Shi, M.; Yang, Y.; Guan, D.; Zhang, Y.; Zhang, Z. Bioactivity of the crude polysaccharides from fermented soybean curd residue by Flammulina velutipes. Carbohyd. Polym. 2012, 89, 1268–1276. [Google Scholar] [CrossRef]
- Zhai, X.; Zhao, A.; Geng, L.; Xu, C. Fermentation characteristics and hypoglycemic activity of an exopolysaccharide produced by submerged culture of Stropharia rugosoannulata #2. Ann. Microbiol. 2013, 63, 1013–1020. [Google Scholar]
- Wang, J.C.; Hu, S.H.; Wang, J.T.; Chen, K.S.; Chia, Y.C. Hypoglycemic effect of extract of Hericium erinaceus. J. Sci. Food Agric. 2005, 85, 641–646. [Google Scholar] [CrossRef]
- Mao, Y.; Mao, J.; Meng, X. Extraction optimization and bioactivity of exopolysaccharides from Agaricus bisporus. Carbohyd. Polym. 2013, 92, 1602–1607. [Google Scholar] [CrossRef] [PubMed]
- Mohan, K.; Muralisankar, T.; Uthayakumar, V.; Chandirasekar, R.; Revathi, N.; Ramu Ganesan, A.; Velmurugan, K.; Sathishkumar, P.; Jayakumar, R.; Seedevi, P. Trends in the extraction, purification, characterisation and biological activities of polysaccharides from tropical and sub-tropical fruits—A comprehensive review. Carbohyd. Polym. 2020, 238, 116185. [Google Scholar] [CrossRef] [PubMed]
- Khoo, L.T.; Abas, F.; Abdullah, J.O.; Mohd Tohit, E.R.; Hamid, M.; Shi-Biao, W.; Wu, S. Anticoagulant activity of polyphenolic-polysaccharides isolated from Melastoma malabathricum L. Evid.-Based Complement. Altern. Med. 2014, 2014, 614273. [Google Scholar] [CrossRef] [PubMed]
- Nadar, S.S.; Rao, P.; Rathod, V.K. Enzyme assisted extraction of biomolecules as an approach to novel extraction technology: A review. Food Res. Int. 2018, 108, 309–330. [Google Scholar] [CrossRef] [PubMed]
- Carrero-Carralero, C.; Mansukhani, D.; Ruiz-Matute, A.I.; Martínez-Castro, I.; Ramos, L.; Sanz, M.L. Extraction and characterization of low molecular weight bioactive carbohydrates from mung bean (Vigna radiata). Food Chem. 2018, 266, 146–154. [Google Scholar] [CrossRef]
- Li, C.; Wang, C.; Wang, S.; Guoying, Q.; Qiuhua, Z.; Yuting, L.; Wei, W. Optimization of ultrasonic-assisted extraction technology of Sargassum fusiforme polysaccharides and evaluation of their antioxidant activity. Food Sci. Technol. Int. Tokyo 2013, 19, 157–162. [Google Scholar] [CrossRef][Green Version]
- Wang, T.; Li, X.; Qiu, L.; Nie, F.; Qiao, B.; Liang, C.; Zhao, C.; Li, C. Study on ultrasonic-boiling water extraction of polysaccharide Fromficus carica L. leaves. Heilongjiang Agric. Sci. 2021, 73–76. [Google Scholar] [CrossRef]
- Song, X.; Liu, Z.; Zhang, J.; Yang, Q.; Ren, Z.; Zhang, C.; Liu, M.; Gao, Z.; Zhao, H.; Jia, L. Anti-inflammatory and hepatoprotective effects of exopolysaccharides isolated from Pleurotus geesteranus on alcohol-induced liver injury. Sci. Rep. 2018, 8, 10413–10493. [Google Scholar] [CrossRef]
- Zhang, H.; Nie, S.; Cui, S.W.; Xu, M.; Ding, H.; Xie, M. Characterization of a bioactive polysaccharide from Ganoderma atrum: Re-elucidation of the fine structure. Carbohyd. Polym. 2017, 158, 58–67. [Google Scholar] [CrossRef]
- Zeng, X.; Li, P.; Chen, X.; Kang, Y.; Xie, Y.; Li, X.; Xie, T.; Zhang, Y. Effects of deproteinization methods on primary structure and antioxidant activity of Ganoderma lucidum polysaccharides. Int. J. Biol. Macromol. 2019, 126, 867–876. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Yuan, M.; Zou, S.; Guo, L. Extraction, separation, purification and structure identification of polysaccharides from Truffles. Food Ferment. Ind. 2020, 46, 196–200. [Google Scholar]
- Cui, B.; Li, H.; Ji, X.; Zhou, J.; Song, J.; Si, J.; Yang, Z.; Dai, Y. Species diversity, taxonomy and phylogeny of polyporaceae (Basidiomycota) in china. Fungal Divers. 2019, 97, 137–392. [Google Scholar] [CrossRef]
- Zhang, Q.; Huang, N.; Wang, J.; Luo, H.; He, H.; Ding, M.; Deng, W.; Zou, K. The h+/k+-atpase inhibitory activities of trametenolic acid b from Trametes lactinea pat, and its effects on gastric cancer cells. Fitoterapia 2013, 89, 210. [Google Scholar] [CrossRef]
- Zhang, Q.; Wang, J.; He, H.; Liu, H.; Yan, X.; Zou, K. Trametenolic acid b reverses multidrug resistance in breast cancer cells through regulating the expression level of p-glycoprotein: Trametenolic acid b reverses multidrug resistance through regulating p-glycoprotein. Phytother. Res. 2013, 28, 1037–1044. [Google Scholar] [CrossRef] [PubMed]
- Hao, J.; Meng, G.; Yang, J.; Wu, L.; Ma, S.; Ye, L.; Liu, G.; Fu, J.; Wu, X. Physicochemical properties, antioxidant activities and liver protective effects of polysaccharides from fruiting bodies of Trametes lactinea. Mycosystema 2020, 39, 2355–2368. [Google Scholar]
- Hao, J.; Ye, L.; Meng, G.; Song, Y.; Fu, J.; Wu, X. The protective effect and crucial biological pathways analysis of Trametes lactinea mycelium polysaccharides on acute alcoholic liver injury in mice based on transcriptomics and metabonomics. Food Sci. Hum. Wellness 2021, 10, 480–489. [Google Scholar] [CrossRef]
- Yan, A. Advancement in extraction methods of fungal intracellular polysaccharides. J. Microbiol. 2011, 31, 82–86. [Google Scholar]
- Li, M.; Sun, Y.; Zeng, Y.; Lei, Y.; Bao, M. Extraction process of polysaccharide content of mycelium and extracellular polysaccharide from Morchella crassipes. J. Fungal Res. 2017, 15, 195–200. [Google Scholar]
- Hou, X.; Lin, F.; Chang, M.; Liu, J.; Meng, J.; Wei, Y. Effects of ultrasound and deproteinization treatments on the extraction of polysaccharides from fruit-bodies of Hericium erinaceus. Mycosystema 2019, 38, 895–906. [Google Scholar]
- Zhang, C.; Yu, Y.; Liang, Y.; Chen, X. Purification, partial characterization and antioxidant activity of polysaccharides from Glycyrrhiza uralensis. Int. J. Biol. Macromol. 2015, 79, 681–686. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Xu, Q.; Xu, J. Determination of momordica charantia polysaccharide by improved phenol-sulfuric acid method. Food Res. Dev. 2015, 36, 82–85. [Google Scholar]
- Liu, Y.; Qian, T.; Jiiang, D.; He, Y.; Shen, X.; Jiang, S. The comparison with kjeldah and coomassie brilliant blue method on testing protein content of the polysaccharide from Sipunculus nudus. Chin. J. Exp. Tradit. Med. Formulae 2013, 19, 96–98. [Google Scholar]
- Wang, W.; Guo, S.; Li, L.; Wang, M.; Liang, J. Determination of uronic acids in polysaccarides from Stanuntonia chinensis. Food Sci. Technol. 2007, 32, 84–86. [Google Scholar]
- Qiu, F.; Zhang, L.; Yu, J. Determination of sulfate groups in antler velvet pilose antler polysaccharides by barium sulfate turbidimetric method. J. Chang. Univ. Techonol. (Nat. Sci. Ed.) 2005, 268–270. [Google Scholar] [CrossRef]
- Li, J.; Chen, X.; Deng, J.; Wu, Y.; Liu, L.; Tu, Y.; Zhou, Y. Extraction and antioxidant activity in vitro of okra flavonoids. Food Sci. 2014, 35, 121–125. [Google Scholar]
- Jayaprakasha, G.K.; Singh, R.P.; Sakariah, K.K. Antioxidant activity of grape seed (Vitis vinifera) extracts on peroxidation models in vitro. Food Chem. 2001, 73, 285–290. [Google Scholar] [CrossRef]
- Yu, Q.; Shang, S.; Feng, Y.; Wang, Y. Determination of monosaccharide composition of polysaccharide in Ganoderma lucidum spore by ion chromatography. Chin. Pharm. J. 2014, 49, 344–347. [Google Scholar]
- Alliouche Kerboua, K.; Benosmane, L.; Namoune, S.; Ouled-Diaf, K.; Ghaliaoui, N.; Bendjeddou, D. Anti-inflammatory and antioxidant activity of the hot water-soluble polysaccharides from Anacyclus pyrethrum (L.) Lag. Roots. J. Ethnopharmacol. 2021, 281, 114491. [Google Scholar] [CrossRef]
- Chen, J.; Zhou, J.; Zhang, L.; Nakamura, Y.; Norisuye, T. Chemical structure of the water-insoluble polysaccharide isolated from the fruiting body of Ganoderma lucidum. Polym. J. 1998, 30, 838–842. [Google Scholar] [CrossRef]
- Wang, Z.; Guan, Y.; Liu, Y.; Liu, S. Extraction process optimization, structural characterization and antioxidant activities of polysaccharide from Morchella sextelata. Mycosystema 2019, 38, 1548–1558. [Google Scholar]
- Qiao, Y.; Chen, W.; Xie, X.; Deng, B.; Peng, H.; Wang, Y. Optimization of selected parameters affecting extracellular polysaccharide extraction yields from Lentinula edodes spent culture fluid using response surface methodology. Acta Edulis Fungi 2015, 22, 69–73. [Google Scholar]
- Samavati, V. Polysaccharide extraction from Abelmoschus esculentus: Optimization by response surface methodology. Carbohyd. Polym. 2013, 95, 588–597. [Google Scholar] [CrossRef] [PubMed]
- Shu, X.; Zhang, Y.; Jia, J.; Ren, X.; Wang, Y. Extraction, purification and properties of water-soluble polysaccharides from mushroom Lepista nuda. Int. J. Biol. Macromol. 2019, 128, 858–869. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Wang, A.; Liu, L.; Tian, G.; Xu, F. Extraction of polysaccharides under vacuum condition from Lentinus edodes stipe and their antioxidant activities in vitro. Food Sci. Biotechnol. 2019, 28, 759–767. [Google Scholar] [CrossRef] [PubMed]
- Chun, H.; Tang, X.; Xu, L.; Liu, J. Optimization of the extraction process for polysaccharide from Terminalia chebula retz. by orthogonal test combined with response surface method. J. Mol. Sci. 2017, 33, 291–296. [Google Scholar]
- Xiao, Y.; Zhou, X.; Xu, Q.; Wang, R.; Zhao, C. Optimization of the extraction process of polysaccharides from wild boletus edulis by orthogonal method. J. Chengde Med. Coll. 2018, 35, 147–149. [Google Scholar]
- Hao, J.; Fu, M.; Wang, L.; Wu, X. Optimization of extraction technology, structure characterization and antioxidant activities of polysaccharides from mycelium of Trametes lactinea. Mycosystema 2021, 40, 2461–2479. [Google Scholar]
- Du, P.; Wang, L.; Liu, D.; Zhang, Y. Response surface methodology for optimizing extraction of polysaccharides from Ganoderma applanatum. J. Anhui Agric. Sci. 2018, 46, 148–151, 168. [Google Scholar]
- Ma, S.; Yu, N.; Liu, C.; Zhang, L. Research on extracting technique of excellularpolysaccharide from Pleurotus nebrodensis. J. Shenyang Agric. Univ. 2008, 39, 374–376. [Google Scholar]
- Li, Z.; Wang, S.; Sheng, W. Extraction conditions optimization of extracellular polysaccharides of Ganoderma applanatum. J. Anhui Sci. Technol. Univ. 2013, 27, 22–25. [Google Scholar]
- Zhang, G.; Xin, X.; Liang, J.; Liu, X.; Wang, S.; Kong, D. Selection of the method for determining extracelluar polysaccharides and optimization of extracting technology in Pleurotus citrinopileatus. Jiangsu J. Agric. Sci. 2005, 21, 230–233. [Google Scholar]
- Lu, T.; Li, D.; Xie, W.; Yu, C.; Chen, H.; Shang, H.; Yang, L.; Liu, Y.; Xie, Z. Optimization of extraction process of Perenniporia fraxinea polysaccharide and its antioxidant activity. China Brew. 2021, 40, 188–194. [Google Scholar]
- Chen, C.; Liu, J. Extraction conditions optimization of extracellular polysaccharides in tricholoma matsutake. Food Nutr. China 2012, 18, 60–62. [Google Scholar]
- Wei, Y.; Hou, X.; Du, X.; Chang, M.; Meng, J.; Liu, J. Optimization of extraction process of exopolysaccharides from Cordyceps militaris and its anti-colon cancer activity. J. Shanxi Agric. Univ. (Nat. Sci. Ed.) 2020, 40, 121–128. [Google Scholar]
- Sichert, A.; Le Gall, S.; Klau, L.J.; Laillet, B.; Rogniaux, H.; Aachmann, F.L.; Hehemann, J. Ion-exchange purification and structural characterization of five sulfated fucoidans from brown algae. Glycobiology 2021, 31, 352–357. [Google Scholar] [CrossRef] [PubMed]
- Huang, C.; Ma, C.; Song, H.; Li, T. Study on deproteinization and decoloration in extraction of Armillariella polysaccharide. Pharm. Clin. Res. 2007, 45–46. [Google Scholar] [CrossRef]
- Ji, X.; Peng, B.; Ding, H.; Cui, B.; Nie, H.; Yan, Y. Purification, structure and biological activity of pumpkin polysaccharides: A review. Food Rev. Int. 2023, 39, 307–319. [Google Scholar] [CrossRef]
- Huang, S.; Huang, G. Extraction, structural analysis, and activities of rice bran polysaccharide. Chem. Biol. Drug Des. 2021, 98, 631–638. [Google Scholar] [CrossRef]
- He, N.; Tian, L.; Zhai, X.; Zhang, X.; Zhao, Y. Composition characterization, antioxidant capacities and anti-proliferative effects of the polysaccharides isolated from Trametes lactinea (berk.) Pat. Int. J. Biol. Macromol. 2018, 115, 114–123. [Google Scholar] [CrossRef]
- Bao, Z.; Yao, L.; Zhang, X.; Lin, S. Isolation, purification, characterization, and immunomodulatory effects of polysaccharide from Auricularia auricula on RAW264.7 macrophages. J. Food Biochem. 2020, 44, e13516. [Google Scholar] [CrossRef]
- Shi, K.; Yang, G.; He, L.; Yang, B.; Li, Q.; Yi, S. Purification, characterization, antioxidant, and antitumor activity of polysaccharides isolated from silkworm cordyceps. J. Food Biochem. 2020, 44, e13482. [Google Scholar] [CrossRef]
- Shi, Z.; Liu, L.; Hong, H.; Gou, D.; Liu, D. Isolation, purification, structure analysis and comparison of antioxidant activity of the polysaccharides from Ostericum sieboldii. Mod. Food Sci. Technol. 2022, 38, 124–132. [Google Scholar]
- Zhang, H.; Li, H.; Netala, V.R.; Hou, T.; Zhang, Z. Optimization of complex enzyme-ultrasonic synergistic extraction of water-soluble polysaccharides from Perilla frutescens seed meal: Purification, characterization and in vitro antioxidant activity. J. Food Process. Preserv. 2022, 46, e16201. [Google Scholar] [CrossRef]
- Xu, T.; Zuo, Y.; Lin, W.; Liu, P. Isolation, purification, chemical composition and antioxidant activities of polysaccharides from Dictyosphaerium sp. 1A10. Food Res. Dev. 2023, 44, 57–65. [Google Scholar]
- De Jesus, L.I.; Smiderle, F.R.; Cordeiro, L.M.C.; de Freitas, R.A.; Van Griensven, L.J.L.D.; Iacomini, M. Simple and effective purification approach to dissociate mixed water-insoluble α- and β-d-glucans and its application on the medicinal mushroom Fomitopsis betulina. Carbohyd. Polym. 2018, 200, 353–360. [Google Scholar] [CrossRef] [PubMed]
- Cao, H.; Ma, S.; Guo, H.; Cui, X.; Wang, S.; Zhong, X.; Wu, Y.; Zheng, W.; Wang, H.; Yu, J.; et al. Comparative study on the monosaccharide compositions, antioxidant and hypoglycemic activities in vitro of intracellular and extracellular polysaccharides of liquid fermented Coprinus comatus. Int. J. Biol. Macromol. 2019, 139, 543–549. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).