Valorising Agro-industrial Wastes within the Circular Bioeconomy Concept: the Case of Defatted Rice Bran with Emphasis on Bioconversion Strategies
Abstract
:1. Introduction
Rice Production and Defatted Rice Bran (DRB). A New Framework Emerges in the Circular Bio-Economy
2. Composition of DRB
3. Valorisation Options of Defatted Rice Bran
3.1. Biotechnological Conversion of DRB in Value-added Products
3.1.1. Pretreatment
3.1.2. Production of Bioethanol
3.1.3. Production of Lactic Acid
3.1.4. Production of Biobutanol
3.1.5. Production of Bio-Hydrogen
3.1.6. Production of Enzymes
3.1.7. Production of Other Biotechnological Products
3.2. Use of Defatted Rice Bran in Foodstuffs
4. Discussion and Future Prospects
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Sanz-Hernández, A.; Esteban, E.; Garrido, P. Transition to a bioeconomy: Perspectives from social sciences. J. Clean. Prod. 2019, 224, 107–119. [Google Scholar] [CrossRef] [Green Version]
- Cherubini, F. The biorefinery concept: Using biomass instead of oil for producing energy and chemicals. Energy Convers. Manag. 2010, 51, 1412–1421. [Google Scholar] [CrossRef]
- Lokesh, K.; Ladu, L.; Summerton, L. Bridging the gaps for a “circular” bioeconomy: Selection criteria, bio-based value chain and stakeholder mapping. Sustainability 2018, 10. [Google Scholar] [CrossRef] [Green Version]
- Gul, K.; Yousuf, B.; Singh, A.K.; Singh, P.; Wani, A.A. Rice bran: Nutritional values and its emerging potential for development of functional food—A review. Bioact. Carbohydr. Diet. Fibre 2015, 6, 24–30. [Google Scholar] [CrossRef]
- OECD-FAO Agricultural Outlook 2018–2027. Available online: https://stats.oecd.org/Index.aspx?DataSetCode=HIGH_AGLINK_2018 (accessed on 15 July 2019).
- Moraes, C.A.M.; Fernandes, I.J.; Calheiro, D.; Kieling, A.G.; Brehm, F.A.; Rigon, M.R.; Berwanger Filho, J.A.; Schneider, I.A.H.; Osorio, E. Review of the rice production cycle: By-products and the main applications focusing on rice husk combustion and ash recycling. Waste Manag. Res. 2014, 32, 1034–1048. [Google Scholar] [CrossRef]
- Available online: http://www.knowledgebank.irri.org/step-by-step-production/postharvest/rice-by-products (accessed on 15 July 2019).
- Available online: http://www.fao.org/faostat/en/#data/GA (accessed on 15 July 2019).
- Abraham, A.; Mathew, A.K.; Sindhu, R.; Pandey, A.; Binod, P. Potential of rice straw for bio-refining: An overview. Bioresour. Technol. 2016, 215, 29–36. [Google Scholar] [CrossRef]
- Barana, D.; Salanti, A.; Orlandi, M.; Ali, D.S.; Zoia, L. Biorefinery process for the simultaneous recovery of lignin, hemicelluloses, cellulose nanocrystals and silica from rice husk and Arundo donax. Ind. Crop. Prod. 2016, 86, 31–39. [Google Scholar] [CrossRef]
- Santos Michel, R.J.; Canabarro, N.I.; Alesio, C.; Maleski, T.; Laber, T.; Sfalcin, P.; Foletto, E.L.; Mayer, F.D.; Kuhn, R.C.; Mazutti, M.A. Enzymatic saccharification and fermentation of rice processing residue for ethanol production at constant temperature. Biosyst. Eng. 2016, 142, 110–116. [Google Scholar] [CrossRef]
- Available online: https://www.globenewswire.com/news-release/2019/07/15/1882391/0/en/Rice-Bran-Oil-Market-was-1-23-Bn-in-2018-and-is-estimated-to-Grow-at-4-1-CAGR-by-2025-Analysis-by-Production-Outlook-Key-Trends-Value-Chain-Strategic-Framework-and-Opportunities-Ad.html (accessed on 31 March 2020).
- Sharif, M.K.; Butt, M.S.; Anjum, F.M.; Khan, S.H. Rice Bran: A Novel Functional Ingredient. Crit. Rev. Food Sci. Nutr. 2014, 54, 807–816. [Google Scholar] [CrossRef]
- Abaide, E.R.; Tres, M.V.; Zabot, G.L.; Mazutti, M.A. Reasons for processing of rice coproducts: Reality and expectations. Biomass Bioenergy 2019, 120, 240–256. [Google Scholar] [CrossRef]
- Available online: https://stats.oecd.org/Index.aspx?DataSetCode=HIGH_AGLINK_2018# (accessed on 31 March 2020).
- Available online: www.imarcgroup.com/rice-bran-oil-processing-plant (accessed on 31 March 2020).
- Alexandri, M.; Neu, A.K.; Schneider, R.; López-Gómez, J.P.; Venus, J. Evaluation of various Bacillus coagulans isolates for the production of high purity L-lactic acid using defatted rice bran hydrolysates. Int. J. Food Sci. Technol. 2019, 54, 1321–1329. [Google Scholar] [CrossRef]
- Hu, G.; Huang, S.; Cao, S.; Ma, Z. Effect of enrichment with hemicellulose from rice bran on chemical and functional properties of bread. Food Chem. 2009, 115, 839–842. [Google Scholar] [CrossRef]
- Wang, J.; Suo, G.; De Wit, M.; Boom, R.M.; Schutyser, M.A.I. Dietary fibre enrichment from defatted rice bran by dry fractionation. J. Food Eng. 2016, 186, 50–57. [Google Scholar] [CrossRef]
- Daou, C.; Zhang, H. Functional and physiological properties of total, soluble, and insoluble dietary fibres derived from defatted rice bran. J. Food Sci. Technol. 2014, 51, 3878–3885. [Google Scholar] [CrossRef] [Green Version]
- Nandi, I.; Ghosh, M. Studies on functional and antioxidant property of dietary fibre extracted from defatted sesame husk, rice bran and flaxseed. Bioact. Carbohydr. Diet. Fibre 2015, 5, 129–136. [Google Scholar] [CrossRef]
- Sairam, S.; Gopala Krishna, A.G.; Urooj, A. Physico-chemical characteristics of defatted rice bran and its utilization in a bakery product. J. Food Sci. Technol. 2011, 48, 478–483. [Google Scholar] [CrossRef] [Green Version]
- Yuwang, P.; Sulaeva, I.; Hell, J.; Henniges, U.; Böhmdorfer, S.; Rosenau, T.; Chitsomboon, B.; Tongta, S. Phenolic compounds and antioxidant properties of arabinoxylan hydrolysates from defatted rice bran. J. Sci. Food Agric. 2018, 98, 140–146. [Google Scholar] [CrossRef]
- Zhang, H.J.; Wang, J.; Zhang, B.H.; Zhang, H. Antioxidant activities of the fractionated protein hydrolysates from heat stable defatted rice bran. Int. J. Food Sci. Technol. 2014, 49, 1330–1336. [Google Scholar] [CrossRef]
- Wattanasiritham, L.; Theerakulkait, C.; Wickramasekara, S.; Maier, C.S.; Stevens, J.F. Isolation and identification of antioxidant peptides from enzymatically hydrolyzed rice bran protein. Food Chem. 2016, 192, 156–162. [Google Scholar] [CrossRef]
- Kaewjumpol, G.; Oruna-Concha, M.J.; Niranjan, K.; Thawornchinsombut, S. The production of hydrolysates from industrially defatted rice bran and its surface image changes during extraction. J. Sci. Food Agric. 2018, 98, 3290–3298. [Google Scholar] [CrossRef]
- Fabian, C.; Ayucitra, A.; Ismadji, S.; Ju, Y.H. Isolation and characterization of starch from defatted rice bran. J. Taiwan Inst. Chem. Eng. 2011, 42, 86–91. [Google Scholar] [CrossRef] [Green Version]
- Wang, Y.; Yang, Z.; Qin, P.; Tan, T. Fermentative l-(+)-lactic acid production from defatted rice bran. RSC Adv. 2014, 4, 8907. [Google Scholar] [CrossRef]
- Siepmann, F.B.; Canan, C.; de Jesus, M.M.M.; Pazuch, C.M.; Colla, E. Release optimization of fermentable sugars from defatted rice bran for bioethanol production. Acta Sci. Technol. 2018, 40, 1–10. [Google Scholar] [CrossRef]
- Tanaka, T.; Hoshina, M.; Tanabe, S.; Sakai, K.; Ohtsubo, S.; Taniguchi, M. Production of D-lactic acid from defatted rice bran by simultaneous saccharification and fermentation. Bioresour. Technol. 2006, 97, 211–217. [Google Scholar] [CrossRef]
- Yadav, D.N.; Singh, K.K.; Rehal, J. Studies on fortification of wheat flour with defatted rice bran for chapati making. J. Food Sci. Technol. 2012, 49, 96–102. [Google Scholar] [CrossRef] [Green Version]
- Adsul, M.G.; Singhvi, M.S.; Gaikaiwari, S.A.; Gokhale, D.V. Development of biocatalysts for production of commodity chemicals from lignocellulosic biomass. Bioresour. Technol. 2011, 102, 4304–4312. [Google Scholar] [CrossRef] [PubMed]
- Kumari, D.; Singh, R. Pretreatment of lignocellulosic wastes for biofuel production: A critical review. Renew. Sustain. Energy Rev. 2018, 90, 877–891. [Google Scholar] [CrossRef]
- Alexandri, M.; Schneider, R.; Papapostolou, H.; Ladakis, D.; Koutinas, A.; Venus, J. Restructuring the conventional sugar beet industry into a novel biorefinery: Fractionation and bioconversion of sugar beet pulp into succinic acid and value-added coproducts. ACS Sustain. Chem. Eng. 2019, 7, 6569–6579. [Google Scholar] [CrossRef]
- Pleissner, D.; Neu, A.K.; Mehlmann, K.; Schneider, R.; Puerta-Quintero, G.I.; Venus, J. Fermentative lactic acid production from coffee pulp hydrolysate using Bacillus coagulans at laboratory and pilot scales. Bioresour. Technol. 2016, 218, 167–173. [Google Scholar] [CrossRef] [Green Version]
- Neu, A.K.; Pleissner, D.; Mehlmann, K.; Schneider, R.; Puerta-Quintero, G.I.; Venus, J. Fermentative utilization of coffee mucilage using Bacillus coagulans and investigation of down-stream processing of fermentation broth for optically pure l(+)-lactic acid production. Bioresour. Technol. 2016, 211, 398–405. [Google Scholar] [CrossRef]
- Chandel, A.K.; Narasu, M.L.; Rudravaram, R.; Pogaku, R.; Rao, L.V. Bioconversion of de-oiled rice bran (DORB) hemicellulosic hydrolysate into ethanol by Pichia stipitis NCM3499 under optimized conditions. Int. J. Food Eng. 2009, 5, 1–13. [Google Scholar] [CrossRef]
- Tsigie, Y.A.; Wang, C.Y.; Kasim, N.S.; Diem, Q.D.; Huynh, L.H.; Ho, Q.P.; Truong, C.T.; Ju, Y.H. Oil production from Yarrowia lipolytica Po1g using rice bran hydrolysate. J. Biomed. Biotechnol. 2012, 2012. [Google Scholar] [CrossRef] [PubMed]
- Sutanto, S.; Go, A.W.; Chen, K.H.; Ismadji, S.; Ju, Y.H. Maximized utilization of raw rice bran in microbial oils production and recovery of active compounds: A proof of concept. Waste Biomass Valorization 2017, 8, 1067–1080. [Google Scholar] [CrossRef]
- Al-Shorgani, N.K.N.; Al-Tabib, A.I.; Kadier, A.; Zanil, M.F.; Lee, K.M.; Kalil, M.S. Continuous butanol fermentation of dilute acid-pretreated de-oiled rice bran by Clostridium acetobutylicum YM1. Sci. Rep. 2019, 9, 1–13. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Al-Shorgani, N.K.N.; Al-Tabib, A.I.; Kalil, M.S. Production of butanol from acetyl chloride-treated deoiled rice bran by Clostridium acetobutylicum YM1. BioResources 2017, 12, 8505–8518. [Google Scholar] [CrossRef]
- Al-Shorgani, N.K.N.; Kalil, M.S.; Yusoff, W.M.W. Biobutanol production from rice bran and de-oiled rice bran by Clostridium saccharoperbutylacetonicum N1-4. Bioprocess. Biosyst. Eng. 2012, 35, 817–826. [Google Scholar] [CrossRef]
- Lee, J.; Seo, E.; Kweon, D.H.; Park, K.; Jin, Y.S. Fermentation of rice bran and defatted rice bran for butanol production using Clostridium beijerinckii NCIMB 8052. J. Microbiol. Biotechnol. 2009, 19, 482–490. [Google Scholar] [CrossRef] [Green Version]
- Liu, J.; Ma, Z.; Zhu, H.; Caiyin, Q.; Liang, D.; Wu, H.; Huang, X.; Qiao, J. Improving xylose utilization of defatted rice bran for nisin production by overexpression of a xylose transcriptional regulator in Lactococcus lactis. Bioresour. Technol. 2017, 238, 690–697. [Google Scholar] [CrossRef]
- Kong, H.; Yang, X.; Gu, Z.; Li, Z.; Cheng, L.; Hong, Y.; Li, C. Heat pretreatment improves the enzymatic hydrolysis of granular corn starch at high concentration. Process. Biochem. 2018, 64, 193–199. [Google Scholar] [CrossRef]
- Li, J.; Vasanthan, T.; Bressler, D.C. Improved cold starch hydrolysis with urea addition and heat treatment at subgelatinization temperature. Carbohydr. Polym. 2012, 87, 1649–1656. [Google Scholar] [CrossRef]
- Shariffa, Y.N.; Karim, A.A.; Fazilah, A.; Zaidul, I.S.M. Enzymatic hydrolysis of granular native and mildly heat-treated tapioca and sweet potato starches at sub-gelatinization temperature. Food Hydrocoll. 2009, 23, 434–440. [Google Scholar] [CrossRef]
- Azman, N.F.; Abdeshahian, P.; Kadier, A.; Nasser Al-Shorgani, N.K.; Salih, N.K.M.; Lananan, I.; Hamid, A.A.; Kalil, M.S. Biohydrogen production from de-oiled rice bran as sustainable feedstock in fermentative process. Int. J. Hydrog. Energy 2016, 41, 145–156. [Google Scholar] [CrossRef]
- Zheng, L.; Zheng, P.; Sun, Z.; Bai, Y.; Wang, J.; Guo, X. Production of vanillin from waste residue of rice bran oil by Aspergillus niger and Pycnoporus cinnabarinus. Bioresour. Technol. 2007, 98, 1115–1119. [Google Scholar] [CrossRef] [PubMed]
- Liu, Q.; Cao, X.; Zhuang, X.; Han, W.; Guo, W.; Xiong, J.; Zhang, X. Rice bran polysaccharides and oligosaccharides modified by Grifola frondosa fermentation: Antioxidant activities and effects on the production of NO. Food Chem. 2017, 223, 49–53. [Google Scholar] [CrossRef] [PubMed]
- Huzir, N.M.; Aziz, M.M.A.; Ismail, S.B.; Abdullah, B.; Mahmood, N.A.N.; Umor, N.A.; Syed Muhammad, S.A.F. ad Agro-industrial waste to biobutanol production: Eco-friendly biofuels for next generation. Renew. Sustain. Energy Rev. 2018, 94, 476–485. [Google Scholar] [CrossRef]
- Renewable Fuels Association. Available online: https://ethanolrfa.org/reports/ (accessed on 30 March 2020).
- Available online: https://www.statista.com/statistics/281606/ethanol-production-in-selected-countries/_ (accessed on 3 July 2019).
- Watanabe, M.; Takahashi, M.; Sasano, K.; Kashiwamura, T.; Ozaki, Y.; Tsuiki, T.; Hidaka, H.; Kanemoto, S. Bioethanol production from rice washing drainage and rice bran. J. Biosci. Bioeng. 2009, 108, 524–526. [Google Scholar] [CrossRef]
- Gupta, A.; Verma, J.P. Sustainable bio-ethanol production from agro-residues: A review. Renew. Sustain. Energy Rev. 2015, 41, 550–567. [Google Scholar] [CrossRef]
- Zabed, H.; Sahu, J.N.; Suely, A.; Boyce, A.N.; Faruq, G. Bioethanol production from renewable sources: Current perspectives and technological progress. Renew. Sustain. Energy Rev. 2017, 71, 475–501. [Google Scholar] [CrossRef]
- Todhanakasem, T.; Sangsutthiseree, A.; Areerat, K.; Young, G.M.; Thanonkeo, P. Biofilm production by Zymomonas mobilis enhances ethanol production and tolerance to toxic inhibitors from rice bran hydrolysate. N. Biotechnol. 2014, 31, 451–459. [Google Scholar] [CrossRef]
- Beliya, E.; Tiwari, S.; Jadhav, S.K.; Tiwari, K.L. De-oiled rice bran as a source of bioethanol. Energy Explor. Exploit. 2013, 31, 771–782. [Google Scholar] [CrossRef]
- Djukić-Vuković, A.; Mladenović, D.; Ivanović, J.; Pejin, J.; Mojović, L. Towards sustainability of lactic acid and poly-lactic acid polymers production. Renew. Sustain. Energy Rev. 2019, 108, 238–252. [Google Scholar] [CrossRef]
- Alves de Oliveira, R.; Komesu, A.; Vaz Rossell, C.E.; Maciel Filho, R. Challenges and opportunities in lactic acid bioprocess design—From economic to production aspects. Biochem. Eng. J. 2018, 133, 219–239. [Google Scholar] [CrossRef]
- Becker, J.; Lange, A.; Fabarius, J.; Wittmann, C. Top value platform chemicals: Bio-based production of organic acids. Curr. Opin. Biotechnol. 2015, 36, 168–175. [Google Scholar] [CrossRef] [PubMed]
- López-Gómez, J.P.; Alexandri, M.; Schneider, R.; Latorre-Sánchez, M.; Coll Lozano, C.; Venus, J. Organic fraction of municipal solid waste for the production of L-lactic acid with high optical purity. J. Clean. Prod. 2020, 247. [Google Scholar] [CrossRef]
- Pleissner, D.; Demichelis, F.; Mariano, S.; Fiore, S.; Schneider, R.; Venus, J.; Michelle, I.; Guti, N. Direct production of lactic acid based on simultaneous sacchari fi cation and fermentation of mixed restaurant food waste. J. Clean. Prod. 2017, 143, 615–623. [Google Scholar] [CrossRef]
- Demichelis, F.; Pleissner, D.; Fiore, S.; Mariano, S.; Michelle, I.; Gutiérrez, N.; Schneider, R.; Venus, J. Investigation of food waste valorization through sequential lactic acid fermentative production and anaerobic digestion of fermentation residues. Bioresour. Technol. 2017, 241, 508–516. [Google Scholar] [CrossRef]
- Kwan, T.H.; Vlysidis, A.; Wu, Z.; Hu, Y.; Koutinas, A.; Lin, C.S.K. Lactic acid fermentation modelling of Streptococcus thermophilus YI-B1 and Lactobacillus casei Shirota using food waste derived media. Biochem. Eng. J. 2017, 127, 97–109. [Google Scholar] [CrossRef]
- Li, L.; Cai, D.; Wang, C.; Han, J.; Ren, W.; Zheng, J.; Wang, Z.; Tan, T. Continuous L-lactic acid production from defatted rice bran hydrolysate using corn stover bagasse immobilized carrier. RSC Adv. 2015, 5, 18511–18517. [Google Scholar] [CrossRef]
- Taniguchi, M.; Hoshina, M.; Tanabe, S.; Higuchi, Y.; Sakai, K.; Ohtsubo, S.; Hoshino, K.; Tanaka, T. Production of L-lactic acid by simultaneous saccharification and fermentation using unsterilized defatted rice bran as a carbon source and nutrient components. Food Sci. Technol. Res. 2005, 11, 400–406. [Google Scholar] [CrossRef] [Green Version]
- Kumar, M.; Gayen, K. Developments in biobutanol production: New insights. Appl. Energy 2011, 88, 1999–2012. [Google Scholar] [CrossRef]
- Yasmin, J.; Gupta, H.; Bansal, B.; Srivastava, O. Effect of combustion duration on the performance and emission characteristics of a spark ignition engine using hydrogen as a fuel. Int. J. Hydrog. Energy 2000, 25, 581–589. [Google Scholar] [CrossRef]
- Arreola-Vargas, J.; Razo-Flores, E.; Celis, L.; Alatriste-Mondragón, F. Sequential hydrolysis of oat straw and hydrogen production from hydrolysates: Role of hydrolysates constituents. Int. J. Hydrog. Energy 2015, 34, 10756–10765. [Google Scholar] [CrossRef]
- Chang, J.S.; Lee, K.S.; Lin, P.J. Biohydrogen production with fixed-bed bioreactors. Int. J. Hydrog. Energy 2002, 27, 1167–1174. [Google Scholar] [CrossRef]
- Oh, S.E.; Zuo, Y.; Zhang, H.; Guiltiman, M.J.; Logan, B.E.; Regan, J.M. Hydrogen production by Clostridium acetobutylicum ATCC 824 and megaplasmid-deficient mutant M5 evaluated using a large headspace volume technique. Int. J. Hydrog. Energy 2009, 34, 9347–9353. [Google Scholar] [CrossRef]
- Srivastava, N.; Srivastava, M.; Kushwaha, D.; Gupta, V.K.; Manikanta, A.; Ramteke, P.W.; Mishra, P.K. Efficient dark fermentative hydrogen production from enzyme hydrolyzed rice straw by Clostridium pasteurianum (MTCC116). Bioresour. Technol. 2017, 238, 552–558. [Google Scholar] [CrossRef]
- Tandon, M.; Thakur, V.; Tiwari, K.L.; Jadhav, S.K. Enterobacter ludwigii strain IF2SW-B4 isolated for bio-hydrogen production from rice bran and de-oiled rice bran. Environ. Technol. Innov. 2018, 10, 345–354. [Google Scholar] [CrossRef]
- Manera, A.P.; Kamimura, E.S.; Brites, L.M.; Kalil, S.J. Adsorption of amyloglucosidase from Aspergillus niger NRRL 3122 using ion exchange resin. Braz. Arch. Biol. Technol. 2008, 51, 1015–1024. [Google Scholar] [CrossRef]
- Chugh, P.; Soni, R.; Soni, S.K. Deoiled Rice Bran: A substrate for co-production of a consortium of hydrolytic enzymes by Aspergillus niger P-19. Waste Biomass Valorization 2016, 7, 513–525. [Google Scholar] [CrossRef]
- Colla, E.; Santos, L.O.; Deamici, K.; Magagnin, G.; Vendruscolo, M.; Costa, J.A.V. Simultaneous production of amyloglucosidase and exo-polygalacturonase by Aspergillus niger in a rotating drum reactor. Appl. Biochem. Biotechnol. 2017, 181, 627–637. [Google Scholar] [CrossRef]
- Linde, G.A.; Magagnin, G.; Costa, J.A.V.; Bertolin, T.E.; Colauto, N.B. Column bioreactor use for optimization of pectinase production in solid substrate cultivation. Braz. J. Microbiol. 2007, 38, 557–562. [Google Scholar] [CrossRef] [Green Version]
- Bhavsar, K.; Shah, P.; Soni, S.; Khire, J. Influence of pretreatment of agriculture residues on phytase production by Aspergillus niger NCIM 563 under submerged fermentation conditions. Afr. J. Bitecnol. 2008, 7. [Google Scholar]
- Pogaku, R.; Rudravaram, R.; Chandel, A.K.; Linga, V.R.; Yim, Z.H. The effect of de-oiled rice bran for single cell protein production using fungal cultures under solid state fermentation. Int. J. Food Eng. 2009, 5. [Google Scholar] [CrossRef]
- Ranjan, A.; Sahu, N.P.; Deo, A.D.; Kumar, S. Solid state fermentation of de-oiled rice bran: Effect on in vitro protein digestibility, fatty acid profile and anti-nutritional factors. Food Res. Int. 2019, 119, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Uraji, M.; Kimura, M.; Inoue, Y.; Kawakami, K.; Kumagai, Y.; Harazono, K.; Hatanaka, T. Enzymatic production of ferulic acid from defatted rice bran by using a combination of bacterial enzymes. Appl. Biochem. Biotechnol. 2013, 171, 1085–1093. [Google Scholar] [CrossRef]
- Webber, D.M.; Hettiarachchy, N.S.; Li, R.; Horax, R.; Theivendran, S. Phenolic Profile and Antioxidant activity of extracts prepared from fermented heat-stabilized defatted rice bran. J. Food Sci. 2014, 79, H2383–H2391. [Google Scholar] [CrossRef] [PubMed]
- Tuan, V.N.; Phung, L.T.K.; Dat, L.Q. Purification of gamma-amino butyric acid (GABA) from fermentation of defatted rice bran extract by using ion exchange resin. In AIP Conference Proceedings; AIP Publishing LLC: Melville, NY, USA, 2017; Volume 1878, p. 20007. [Google Scholar]
- Demirci, A.S. Xanthan gum production from hydrolyzed rice bran as a carbon source by Xanthomonas spp. Korean J. Microbiol. Biotechnol. 2012, 40, 356–363. [Google Scholar] [CrossRef]
- Prestes, D.N.; Spessato, A.; Talhamento, A.; Gularte, M.A.; Schirmer, M.A.; Vanier, N.L.; Rombaldi, C.V. The addition of defatted rice bran to malted rice improves the quality of rice beer. Lwt 2019, 112. [Google Scholar] [CrossRef]
- Foong, L.C.; Imam, M.U.; Ismail, M. Iron-binding capacity of defatted rice bran hydrolysate and bioavailability of iron in Caco-2 cells. J. Agric. Food Chem. 2015, 63, 9029–9036. [Google Scholar] [CrossRef]
- Kurdi, P.; Hansawasdi, C. Assessment of the prebiotic potential of oligosaccharide mixtures from rice bran and cassava pulp. Lwt Food Sci. Technol. 2015, 63, 1288–1293. [Google Scholar] [CrossRef]
- Available online: https://www.grandviewresearch.com/industry-analysis/enzymes-industry (accessed on 3 April 2020).
- Bio-Based Industries Consortium Mapping European Biorefineries|Bio-Based Industries Consortium. Available online: http://biconsortium.eu/news/mapping-european-biorefineries (accessed on 16 April 2020).
- D’Amato, D.; Droste, N.; Allen, B.; Kettunen, M.; Lähtinen, K.; Korhonen, J.; Leskinen, P.; Matthies, B.D.; Toppinen, A. Green, circular, bio economy: A comparative analysis of sustainability avenues. J. Clean. Prod. 2017, 168, 716–734. [Google Scholar] [CrossRef]
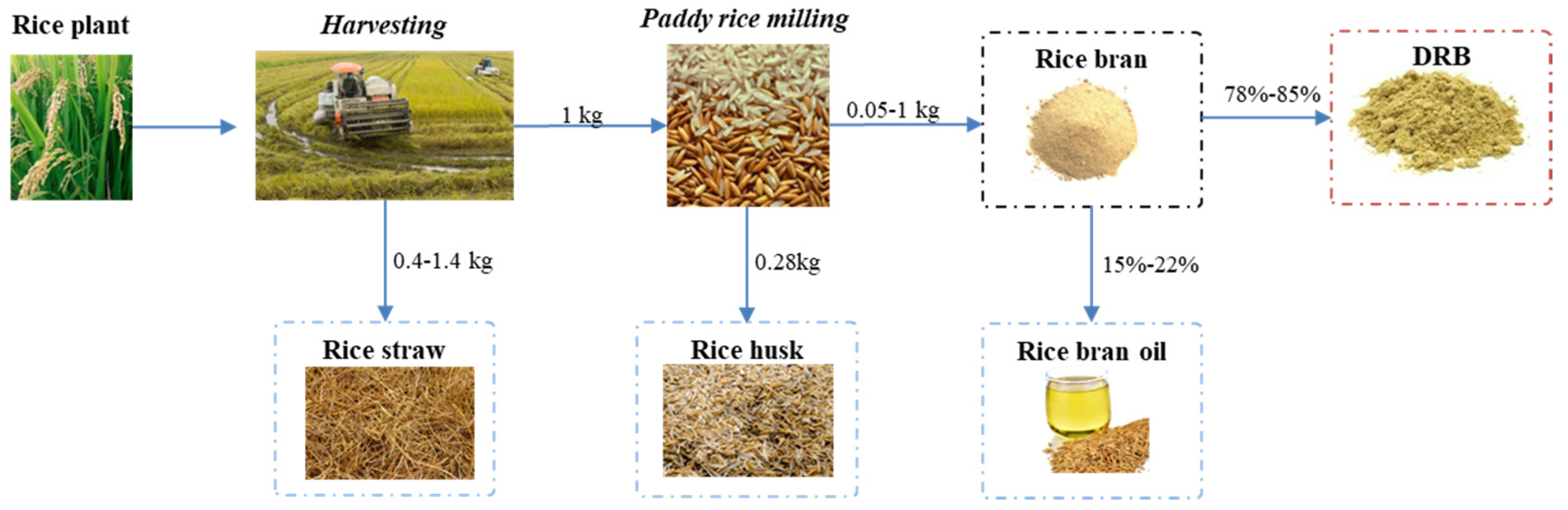

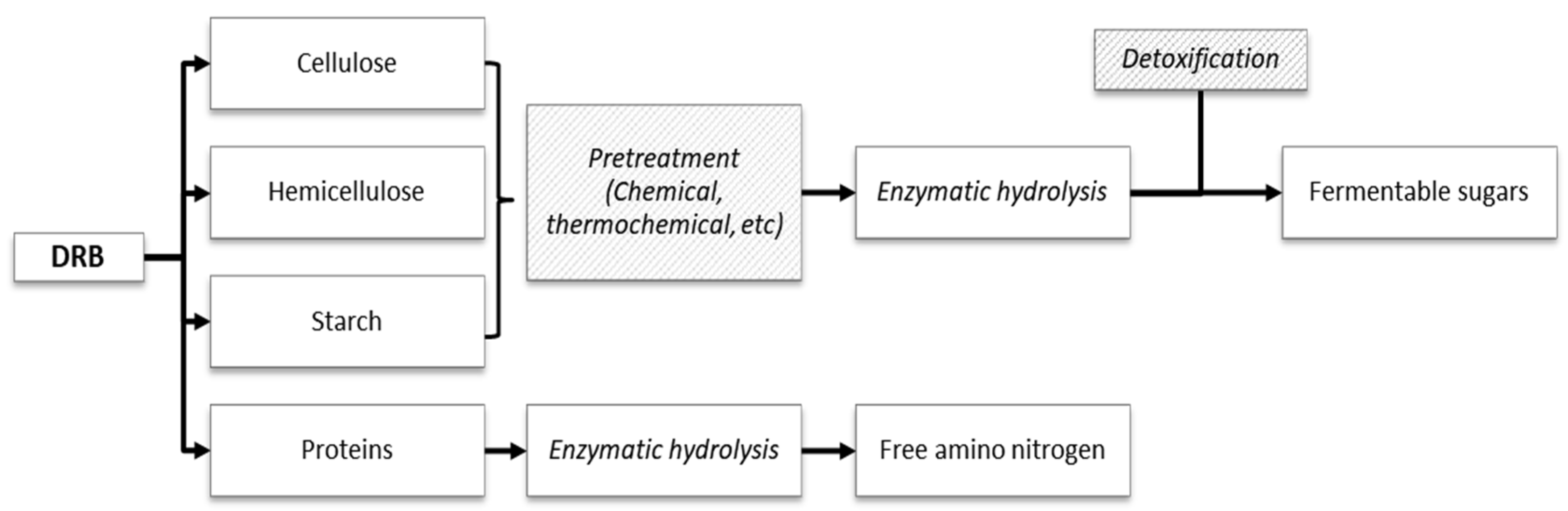

| Component | Wang et al. [28] | Siepmann et al. [29] | Alexandri et al. [17] | Tanaka et al. [30] | Sairam et al. [22] | Yadav et al. [31] | Daou and Zhang [20] |
|---|---|---|---|---|---|---|---|
| Protein | 15.61 | 14.89 | 17.3 | 18.4 | 17.2 | 14.9 | 16.2 |
| Lipid | 0.54 | 1.67 | 3.0 | 1.4 | 0.66 | 2.7 | 2.8 |
| Starch | 47.5 | - | 35.3 | 46.7 * | - | - | |
| Ash | 12.44 | 11.31 | - | 10.4 | 14.65 | 8.2 | 10.7 |
| Moisture | 8.46 | 11.09 | 10.1 | - | 11.1 | 12.7 | 8.7 |
| Total dietary fibre | - | - | - | - | 11.44 | 12.7 | 32.9 |
| Carbohydrates | - | - | - | - | - | 48.8 | - |
| Crude fibre | 8.42 | - | - | - | 9.19 | - | 30.2 |
| Cellulose | - | - | 9.8 | - | - | - | - |
| Hemicellulose | - | - | 20.6 | - | - | - | - |
| Lignin | - | - | 3.9 | - | - | - | - |
| Pretreatment and Hydrolysis Conditions | Sugars Produced (g/L) | Reference |
|---|---|---|
| Solid-to-liquid ratio 1:3.5 (w/w); 20 μL amylase/kg DRB Protein hydrolysis with 12% residue from starch hydrolysis; 40 mg protease/kg, 1:3 (w/w) solid-to-liquid ratio, 50 °C for 10 h | 150.0 | [28] |
| Starch and protein hydrolysis with 20% solids Liquefaction: 0.7 mL/kg Termamyl SC at 85 °C, 500 rpm for 2 h; Saccharification: 1 mL/kg Dextrozyme DX 1.5 and 0.5 mL/kg Fermgen, 50 °C for 19 h. | 82.3 | [17] |
| Enzymatic hydrolysis with α-amylase (30 μL/g DRB for 2 h), amyloglucosidase (40 μL/g DRB, for 3 h) and protease (15 μL/g DRB) at 200 g/L DRB concentration | 68.8 | [29] |
| Acid hydrolysis with 3% H2SO4, solid-to-liquid ratio 1:8 (g/mL), 90 °C, 6 h; detoxification by neutralization with Ca(OH)2 | 50.2 | [38] |
| Solid-to-liquid ratio 15:120 (g/mL), 3% H2SO4 at 90 °C for 6 h in a waterbath; detoxification with activated carbon or by overliming with Ca(OH)2 | 50.1 | [39] |
| Dilute acid pretreatment with 100 g/L DRB, 3.5% H2SO4 at 120 °C for 1 h and overliming | 38.5 | [37] |
| Dilute acid pretreatment with 1% (v/v) H2SO4 at 121 °C for 1 h with 12% (w/v) DRB; detoxification with activated charcoal | 40.2 | [40] |
| Dilute H2SO4 1% (v/v), 121 °C, 15 psi, 1 h with 100 g/L DRB | 32.9 | [48] |
| 1% (v/v) HCl at 80 °C for 3 h; enzymatic hydrolysis with α-amylase (500 μL, 30 °C for 4 h), β-amylase (15 mg) and amyloglucosidase (1 mL) at 37 °C for 4 h, 100 g/L DRB | 40.0 | [43] |
| Acid pretreatment with 1% (v/v) H2SO4 at 121 °C for 1h using 100 g/L DRB; enzymatic hydrolysis with Celluclast 1.5 L, Novozyme 188, Termamyl 120 L; detoxification by overliming and Amberlite XAD-4 resin | 33.4 | [42] |
| Pretreatment with 1% acetyl chloride at 121 °C, 15 psi for 1 h with 10% (w/v) DRB; detoxification with charcoal | 28.0 | [41] |
| Thermal treatment at 120 °C for 1 h of 100 g/L DRB; enzymatic hydrolysis for 5 h with cellulase (30 FPU/g), xylanase (100 XU/g) and glucoamylase (250 IU/g) | 45.6 | [44] |
| Thermal treatment at 135 °C for 5 h with ethanol at pH 8 | - | [49] |
| Microwave treatment at 400 W for 2 min; heating at 100 °C for 20 min at a solid-to-liquid ratio 1:10 (w/v); enzymatic hydrolysis with α-amylase (30 U/mL) and glucose-amylase (200 U/mL) at 60 °C for 4 h | 0.95 * | [50] |
| Product | Strain | Conditions | Titer (g/L) | Yield (gP/gDRB) | P (g/L/h) | Ref. |
|---|---|---|---|---|---|---|
| Bioethanol | P. stipitis NCIM3499 | Batch | 12.47 | 0.12 | 0.173 | [37] |
| P. kudriavzevii RCEF4907 | Batch | 11.4 a | - | 1.58 | [58] | |
| S. cerevisiae | Batch | 35.5 | 0.18 | 1.5 | [29] | |
| L-Lactic acid | B. coagulans | Batch | 66.3 | 0.33 | 2.82 | [17] |
| B. coagulans | Batch | 71.2 | 0.35 | 2.97 | ||
| L. rhamnosus | Single-stage continuous | 88 | - | 5.2 | [66] | |
| Two-stage continuous | 86 | - | 6.20/2.18 | |||
| L. rhamnosus LA-04-1 | Batch | 142 | 0.50 | 3.63 | [28] | |
| fed-batch | 21 | - | 2.56 | |||
| Different LAB isolates | SSF | 31.1 | 0.31 | - | [67] | |
| D-Lactic acid | L. delbrueckii subsp. delbrueckii | SSF | 28 | 0.28 | 0.78 | [30] |
| Biobutanol | C. beijerinckii NCIMB | Batch | 12.24 | 0.12 | 0.26 | [43] |
| C.saccharoperbutylaceticum N1-4 | Batch | 7.1 | 0.07 | 0.059 | [42] | |
| Duran bottles | 7.22 | 0.08 | 0.060 | |||
| C. acetobutylicum YM1 | Batch | 6.48 | - | 0.09 | [41] | |
| Batch (supplemented with TYA b) | 5.64 | - | 0.08 | |||
| Continuous | 5.89 | - | 0.118 | [40] | ||
| 6.87 | - | 0.136 | ||||
| Bio-hydrogen | E. ludwigii IF2SW-B4 | Batch | 295 c | - | 1.82 e | [74] |
| C. acetobutylicum YM1 | Batch (Supplemented with TYA) | 572.5 d | - | - | [48] | |
| Amyloglucosidase | A.niger NRRL 3122 | SF (supplemented with urea) | - | - | - | [75] |
| A.niger NRRL 3122 and t0005/007-2 | SF, rotating drum, pilot scale | Enzyme activity of 84 U per gram of dried medium | - | - | [77] | |
| Exo-polygalacturonase | A.niger NRRL 3122 and t0005/007-2 | SF, rotating drum, pilot scale | Enzyme activity of 84 U per gram of dried medium | - | - | [77] |
| Xantan gum | X. campestris NRRL B-1459 | Batch | 21.87 | 0.43 | - | [85] |
| X. campestris pv. campestris | 17.10 | 0.34 | - | |||
| Nisin | Engineered L. lactis | Batch | 3824.53 IU/mL | - | - | [44] |
| Vanillin | A.niger and P.cinnabarinus | Batch | 2.8 | - | - | [49] |
| Microbial oil | Y. lipolytica | Batch | 5.16 | 0.04 | - | [38] |
| 3.80 | 0.03 | 1.52 g/L/day | [39] | |||
| Phenolic-rich extracts | B.subtilis subsp. subtilis | Batch | 67.64 mg/100g fermented extract | 0.68 f | - | [83] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Alexandri, M.; López-Gómez, J.P.; Olszewska-Widdrat, A.; Venus, J. Valorising Agro-industrial Wastes within the Circular Bioeconomy Concept: the Case of Defatted Rice Bran with Emphasis on Bioconversion Strategies. Fermentation 2020, 6, 42. https://doi.org/10.3390/fermentation6020042
Alexandri M, López-Gómez JP, Olszewska-Widdrat A, Venus J. Valorising Agro-industrial Wastes within the Circular Bioeconomy Concept: the Case of Defatted Rice Bran with Emphasis on Bioconversion Strategies. Fermentation. 2020; 6(2):42. https://doi.org/10.3390/fermentation6020042
Chicago/Turabian StyleAlexandri, Maria, José Pablo López-Gómez, Agata Olszewska-Widdrat, and Joachim Venus. 2020. "Valorising Agro-industrial Wastes within the Circular Bioeconomy Concept: the Case of Defatted Rice Bran with Emphasis on Bioconversion Strategies" Fermentation 6, no. 2: 42. https://doi.org/10.3390/fermentation6020042








