Evaluation of the Growth Kinetics of Lactobacillus Plantarum ATCC 8014 on a Medium Based on Hydrolyzed Bovine Blood Plasma at Laboratory and Bench-Scale Levels and Its Application as a Starter Culture in a Meat Product
Abstract
1. Introduction
2. Materials and Methods
2.1. Bovine Blood Plasma
2.2. Enzymatic Hydrolysis of Bovine Blood Plasma
2.3. Strain Activation and Preservation
2.4. Lactic Acid Bacteria Count and Identification
2.5. Preparation of Culture Media
2.6. Fermentation Kinetics of L. plantarum at the Laboratory Level
2.7. Fermentation Kinetics of L. plantarum at Bench Scale
2.8. Mathematical Modeling of L. plantarum Culture
2.9. Validation of the Use of Biomass Obtained in the Elaboration of a Meat Product
2.10. Sensory Analysis of Fermented Products
3. Results and Discussion
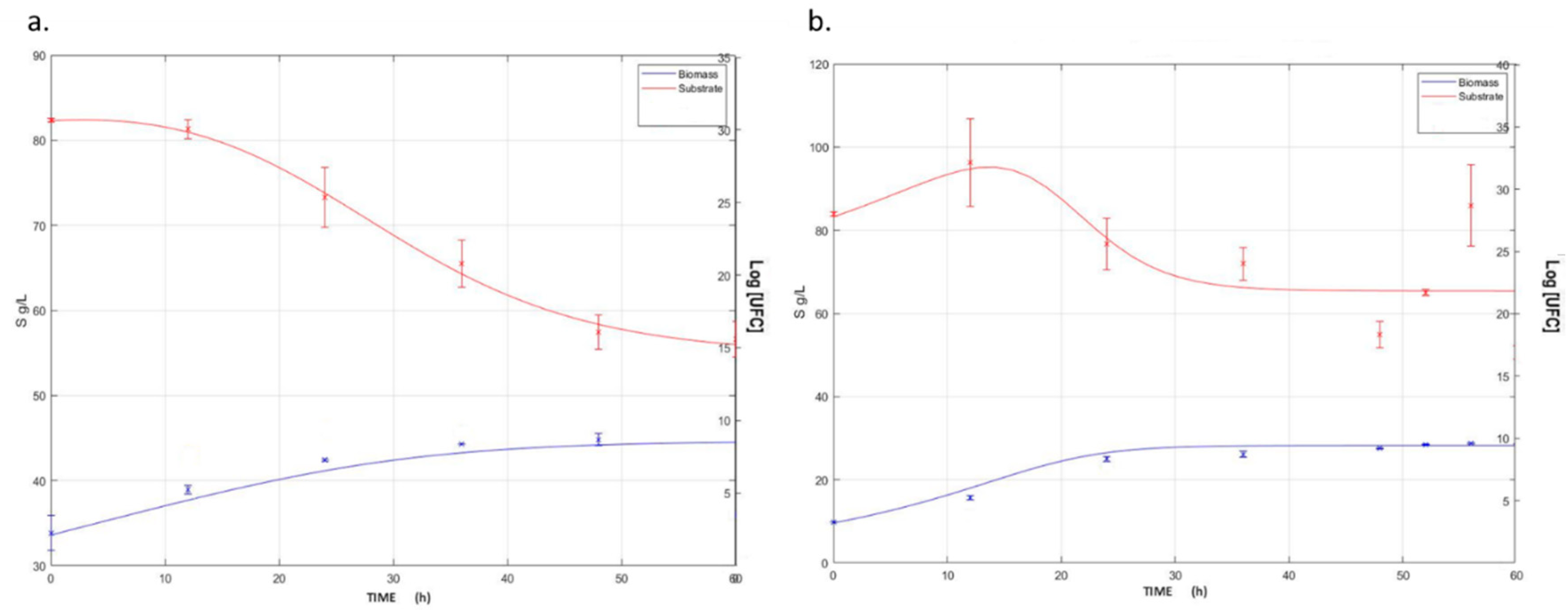
3.1. Kinetic Analysis of L. plantarum Culture on Plasma-Based Media at Laboratory and Bench Scale
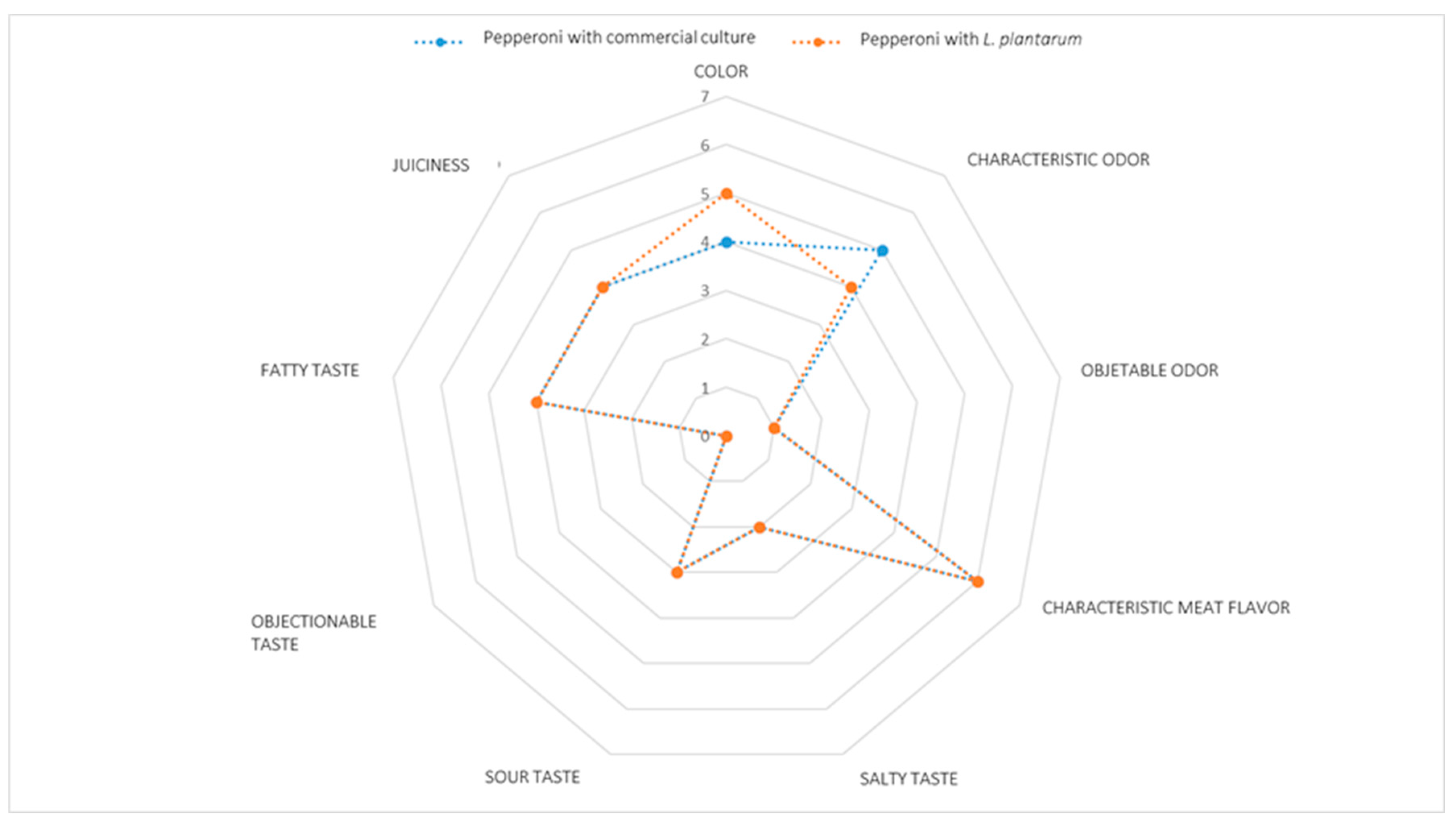
3.2. Validation of the Use of the Biomass Obtained for the Elaboration of a Meat Product
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Rul, F.; Zagorec, M.; Champomier-Vergès, M.-C. Lactic acid bacteria in fermented foods. In Proteomics in Foods; Springer: Boston, MA, USA, 2013; pp. 261–283. [Google Scholar]
- Bogsan, C.S.; Nero, L.A.; Todorov, S.D. From Traditional Knowledge to an Innovative Approach for Bio-preservation in Food by Using Lactic Acid Bacteria. In Beneficial Microorganisms in Food and Nutraceuticals; Springer: Boston, MA, USA, 2015; pp. 1–36. [Google Scholar]
- Siamansouri, M.; Mozaffari, S.; Alikhani, F.E. Bacteriocins and lactic acid bacteria. J. Biol. 2013, 2, 227–234. [Google Scholar]
- Concha-Meyer, A.; Schöbitz, R.; Brito, C.; Fuentes, R. Lactic acid bacteria in an alginate film inhibit Listeria monocytogenes growth on smoked salmon. Food Control 2011, 22, 485–489. [Google Scholar] [CrossRef]
- Crueger, W.; Crueger, A. Industrial Microbiology Manual; ACRIBIA: Zaragoza, Spain, 1989. [Google Scholar]
- Karoviéová, J.; Kohajdová, Z. Lactic acid fermented vegetable juices. Hortic. Sci. 2003, 30, 152–158. [Google Scholar] [CrossRef]
- Axelsson, L. Lactic acid bacteria: Classification and physiology. In Lactic Acid Bacteria. Microbiological and Functional Aspects, 3rd ed.; Salminen, S., von Wright, A., Eds.; Marcel Dekker: New York, NY, USA, 2004; Volume 139, pp. 1–66. [Google Scholar]
- Pragalaki, T.; Bloukas, J.G.; Kotzekidou, P. Inhibition of Listeria monocytogenes and Escherichia coli O157:H7 in liquid broth medium and during processing of fermented sausage using autochthonous starter cultures. Meat Sci. 2013, 95, 458–464. [Google Scholar] [CrossRef] [PubMed]
- Di Gioia, D.; Mazzola, G.; Nikodinoska, I.; Aloisio, I.; Langerholc, T.; Rossi, M.; Raimondi, S.; Melero, B.; Rovira, J. Lactic acid bacteria as protective cultures in fermented pork meat to prevent Clostridium spp. growth. Int. J. Food Microbiol. 2016, 235, 53–59. [Google Scholar] [CrossRef] [PubMed]
- Peh, K.; Pyar, H.; Liong, M.-T. Characteristics and Antibacterial Activity of Metabolites from Lactobacillus acidophilus Strains Produced from Novel Culture Media. Int. J. Pharmacol. 2013, 9, 92–97. [Google Scholar] [CrossRef]
- Davoodabadi, A.; Soltan Dallal, M.M.; Rahimi Foroushani, A.; Douraghi, M.; Sharifi Yazdi, M.k.; Amin Harati, F. Antibacterial activity of Lactobacillus spp. isolated from the feces of healthy infants against enteropathogenic bacteria. Anaerobe 2015, 34, 53–58. [Google Scholar] [CrossRef]
- Joerger, R. Alternatives to antibiotics: Bacteriocins, antimicrobial peptides and bacteriophages. Poult. Sci. 2003, 82, 640–647. [Google Scholar] [CrossRef]
- Papagianni, M. Effects of dissolved oxygen and pH levels on weissellin A production by Weissella paramesenteroides DX in fermentation. Bioprocess Biosyst. Eng. 2012, 35, 1035–1041. [Google Scholar] [CrossRef]
- Katikou, P.; Ambrosiadis, I.; Georgantelis, D.; Koidis, P.; Georgakis, S. Effect of Lactobacillus-protective cultures with bacteriocin-like inhibitory substances’ producing ability on microbiological, chemical and sensory changes during storage of refrigerated vacuum-packaged sliced beef. J. Appl. Microbiol. 2005, 99, 1303–1313. [Google Scholar] [CrossRef]
- Vallejo, M.; Ledesma, P.; Anselmino, L.; Marguet, E. Effect of growth conditions and culture medium composition on bacteriocin production by Enterococcus mundtii Tw56. Rev. Colomb. De Biotecnol. 2014, 16, 174–179. [Google Scholar] [CrossRef]
- Arnau, J. Problemas de los embutidos crudos curados. Eurocarne 2011, 194, 50–65. [Google Scholar]
- Barboza, Y.; Marquez, E.; Arias, B.; Faría, J.; Castejón, O. Utilización del plasma sanguíneo de bovino como fuente proteica en la formulación de un medio de cultivo para Lactobacilos. Rev. Cient. Fac. Cienc. Vet. Univ. Zulia 1994, 4, 55–59. [Google Scholar]
- Duarte de Sutherland, A.; Márquez, E.; Ferrer, A.; Páez de Fernández, G. Preparación de cultivos iniciadores homolácticos para ser utilizados en la fermentación de productos carnicos. Rev. Científica 1992, 2, 66–71. [Google Scholar]
- Hyun, C.-K.; Shin, H.-K. Utilization of bovine blood plasma obtained from a slaughterhouse for economic production of probiotics. J. Ferment. Bioeng. 1998, 86, 34–37. [Google Scholar] [CrossRef]
- Adler-Nissen, J. Enzymic Hydrolysis of Food Proteins; Elsevier Applied Science Publishers: London, UK, 1986. [Google Scholar]
- Figueroa, O.A.M.; Zapata, J.E.M.; Buelvas, L.M.; Ortiz, O. Hidrolizados proteicos y perspectivas del modelamiento en cinética enzimática de proteínas: Una revisión. Rev. Agunkuya 2015, 2, 64–78. [Google Scholar]
- Wokes, F.; Still, B.M. The estimation of protein by the biuret and Greenberg methods. Biochem. J. 1942, 36, 797. [Google Scholar] [CrossRef]
- Barragán, P.J.; Sánchez, Ó.J.; Montoya, S. Modeling of the hydrolysis kinetics of the proteins from bovine blood plasma using bacterial alkaline endoprotease. Vitae 2016, 23, S264–S268. [Google Scholar]
- AOAC. Official Methods of Analysis; Association of Official Analytical Chemists (AOAC): Washington, DC, USA, 1980. [Google Scholar]
- Ossa, J.A.; Vanegas, M.C.; Badillo, Á.M. Evaluación de la melaza de caña como sustrato para el crecimiento de Lactobacillus plantarum. Rev. Actual. Divulg. Científica Univ. Los Andes 2010, 13, 97–104. [Google Scholar]
- MacFaddin, J.F. Pruebas Bioquímicas Para la Identificación de Bacterias de Importancia Clínica; Médica Panamericana: Buenos Aires, Argentina, 2003. [Google Scholar]
- Koneman, E.; Allen, S.; Janda, W.; Schreckenberger, P.; Winn, W. Diagnóstico Microbiológico: Texto y Atlas Color; Editorial Médica Panamericana: Buenos Aires, Argentina, 2001; p. 1432. [Google Scholar]
- Man, J.C.; Rogosa, M.; Sharpe, M.E. A medium for the cultivation of lactobacilli. J. Appl. Bacteriol. 1960, 23, 130–135. [Google Scholar] [CrossRef]
- Dubois, M.; Gilles, K.A.; Hamilton, J.K.; Rebers, P.; Smith, F. Colorimetric method for determination of sugars and related substances. Anal. Chem. 1956, 28, 350–356. [Google Scholar] [CrossRef]
- Williams, D.A. Extra-binomial variation in logistic linear models. Appl. Stat. 1982, 144–148. [Google Scholar] [CrossRef]
- Montoya, S.; Sánchez, Ó.J.; Levin, L. Production of lignocellulolytic enzymes from white-rot fungi by solid-state fermentation and mathematical modeling. Afr. J. Biotechnol. 2015, 14, 1304–1317. [Google Scholar]
- Villegas de Gante, A. Tecnología de Alimentos de Origen Animal: Manual de Prácticas; Trillas: Mexico City, Mexico, 2009; pp. 114–115. [Google Scholar]
- ICONTEC. Norma Técnica Colombiana NTC 1325 Quinta Actualización: Productos Cárnicos Procesados no Enlatados; ICONTEC: Bogotá, Colombia, 2008; pp. 1–38. [Google Scholar]
- Larmond, E. Laboratory Methods for Sensory Evaluation of Food; Research Branch, Canada Dept. of Agriculture: Ottawa, ON, Canada, 1977. [Google Scholar]
- Criollo, J.; Criollo, D.; Aldana, A.S. Fermentación de la almendra de copoazú (Theobroma grandiflorum [Willd. ex Spreng.] Schum.): Evaluación y optimización del proceso. Cienc. Tecnol. Agropecu. 2010, 11, 107–115. [Google Scholar] [CrossRef]
- Díaz, A. Diseño Estadístico de Experimentos, 1st ed.; Universidad de Antioquia: Medellín, CO, USA, 1999. [Google Scholar]
- Pham, P.; Dupont, I.; Roy, D.; Lapointe, G.; Cerning, J. Production of exopolysaccharide by Lactobacillus rhamnosus R and analysis of its enzymatic degradation during prolonged fermentation. Appl. Environ. Microbiol. 2000, 66, 2302–2310. [Google Scholar] [CrossRef]
- Zamudio, K.L. Lactobacilos Nativos Productores de Exopolisacáridos de Intéres Biotecnológico. Bachelor’s Thesis, Universidad Nacional Mayor de San Marcos, Lima, Peru, 2005. [Google Scholar]
- Champagne, C.P.; Gardner, N.J. The growth and recovery of an exopolysaccharide producing Lactobacillus rhamnosus culture on growth media containing apple juice or molasses. J. Gen. Appl. Microbiol. 2008, 54, 237–241. [Google Scholar] [CrossRef]


| Components | Quantity |
|---|---|
| BBP with hydrolyzed proteins (corresponding to an AMN content of 825 mg/L) | 750 mL/L |
| Sucrose | 20.00 g/L (lab scale) 80.00 g/L (bench scale) |
| Potassium diacid phosphate | 2.00 g/L |
| Sodium acetate | 5.00 g/L |
| Ammonium citrate | 2.00 g/L |
| Magnesium sulfate | 0.20 g/L |
| Manganese sulfate | 0.05 g/L |
| pH: 6.4 ± 0.2 at 25 °C | |
| Ingredient | Percentage (%) | Quantity (kg) |
|---|---|---|
| Lean pork 90/10 | 72.80 | 3.640 |
| Bacon | 23.80 | 1.190 |
| Polyphosphate mix (801) | 0.36 | 0.018 |
| Garlic powder | 0.07 | 0.004 |
| Refined salt | 1.40 | 0.070 |
| Nitral-curing salt (5700) | 0.33 | 0.017 |
| Ascorbic acid | 0.14 | 0.007 |
| Commercial sucrose | 0.50 | 0.025 |
| White pepper | 0.28 | 0.014 |
| Black pepper | 0.28 | 0.014 |
| Starter culture (L. plantarum ATCC 8014 and/or Lyocarni SHI-59 commercial culture) | 0.03 | 0.002 |
| Total | 100.00 | 5.000 |
| Parameter | Bench-Scale Fermentation | Lab-Scale Fermentation | Units |
|---|---|---|---|
| µmax | 0.0545 | 0.0111 | h−1 |
| Xmax | 9.4136 | 7.4410 | log CFU/mL |
| qp | −102.1780 | −478.7656 | g × log CFU−1 × h−1 |
| n | 4.6357 | 9.002 | Dimensionless |
| Physical-Chemical Analysis | |
|---|---|
| pH | 5.8–6.2 |
| Acidity | 1.745% |
| Moisture | 72.30% |
| Microbiological analysis | |
| Coagulase-positive staphylococci count, CFU/g | <100 |
| Sulfite-reducing clostridia spore count, CFU/g | <100 |
| Detection of Salmonella spp., /25 g | Absence |
| Escherichia coli count, /g | <3 bacteria/g |
| Physical-Chemical Analysis | |||
|---|---|---|---|
| Pepperoni with L. plantarum | Pepperoni with commercial culture | ||
| pH | 5.56 | pH | 5.28 |
| Acidity | 0.3843% | Acidity | 0.4240% |
| Moisture | 23% | Moisture | 22.46% |
| Microbiological Analysis | |||
| Pepperoni with L. plantarum | Pepperoni with commercial culture | ||
| Coliform count /g | <10 | Coliform count /g | <10 |
| Coagulase-positive Staphylococcus aureus count, CFU/g | <100 | Coagulase-positive Staphylococcus aureus count, CFU/g | <100 |
| Sulfite-reducing clostridia spore count, CFU/g | <10 | Sulfite-reducing Clostridium spore count, CFU/g | <10 |
| Detection of Salmonella spp, /25 g | Absence | Detection of Salmonella spp, /25 g | Absence |
| Detection of Listeria monocytogenes, /25 g | Absence | Detection of Listeria monocytogenes, /25 g | Absence |
| E. coli/g count | <10 | E. coli/g count | <10 |
| Triangle Test: (Pepperoni Control with Commercial Culture (622, 768) and Pepperoni with L. Plantarum (577) | ||||||
|---|---|---|---|---|---|---|
| Panel | Sample Code: 577 | Sample Code: 622, 768 | ||||
| Tester | Slight Difference | Moderate Difference | Large Difference | Slight Difference | Moderate Difference | Large Difference |
| Tester 1 | 1 | |||||
| Tester 2 | 1 | |||||
| Tester 3 | 1 | |||||
| Tester 4 | 1 | |||||
| Tester 5 | 1 | |||||
| Tester 6 | 2 | |||||
| Tester 7 | 2 | |||||
| Tester 8 | 1 | |||||
| Tester 9 | 2 | |||||
| Tester 10 | 1 | |||||
| Pepperoni Type with | Descriptor | Mean | Standard Deviation | Coefficient of Variation | p-Value Kruskal-Wallis |
|---|---|---|---|---|---|
| Commercial culture | Color | 4.1 | ±1.37032 | 33.42% | 0.310933 |
| L. plantarum | Color | 4.7 | ±1.25167 | 26.63% | |
| Commercial culture | Characteristic odor/aroma | 4.6 | ±1.26491 | 27.50% | 0.785341 |
| L. plantarum | Characteristic odor/aroma | 4.4 | ±1.3499 | 30.68% | |
| Commercial culture | Objectionable odor/aroma | 0.9 | ±1.52388 | 169.32% | 0.966559 |
| L. plantarum | Objectionable odor/aroma | 0.6 | ±0.699206 | 116.53% | |
| Commercial culture | Characteristic meat flavor | 5.8 | ±0.918937 | 15.84% | 1 |
| L. plantarum | Characteristic meat flavor | 5.8 | ±1.0328 | 17.81% | |
| Commercial culture | Salty taste | 2.4 | ±2.1187 | 88.28% | 0.843768 |
| L. plantarum | Salty taste | 2.4 | ±1.7127 | 71.36% | |
| Commercial culture | Sour taste | 2.8 | ±1.75119 | 62.54% | 0.938339 |
| L. plantarum | Sour taste | 2.7 | ±1.56702 | 58.04% | |
| Commercial culture | Objectionable taste | 0.3 | ±0.483046 | 161.02% | 0.887537 |
| L. plantarum | Objectionable taste | 0.4 | ±0.699206 | 174.80% | |
| Commercial culture | Fatty taste | 3.7 | ±1.49443 | 40.39% | 0.815117 |
| L. plantarum | Fatty taste | 3.8 | ±1.54919 | 40.77% | |
| Commercial culture | Juiciness | 4.4 | ±1.07497 | 24.43% | 0.874162 |
| L. plantarum | Juiciness | 4.3 | ±0.948683 | 22.06% |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Barragán, P.J.; Sánchez, Ó.J.; Henao-Rojas, J.C. Evaluation of the Growth Kinetics of Lactobacillus Plantarum ATCC 8014 on a Medium Based on Hydrolyzed Bovine Blood Plasma at Laboratory and Bench-Scale Levels and Its Application as a Starter Culture in a Meat Product. Fermentation 2020, 6, 45. https://doi.org/10.3390/fermentation6020045
Barragán PJ, Sánchez ÓJ, Henao-Rojas JC. Evaluation of the Growth Kinetics of Lactobacillus Plantarum ATCC 8014 on a Medium Based on Hydrolyzed Bovine Blood Plasma at Laboratory and Bench-Scale Levels and Its Application as a Starter Culture in a Meat Product. Fermentation. 2020; 6(2):45. https://doi.org/10.3390/fermentation6020045
Chicago/Turabian StyleBarragán, Pedro José, Óscar J. Sánchez, and Juan C. Henao-Rojas. 2020. "Evaluation of the Growth Kinetics of Lactobacillus Plantarum ATCC 8014 on a Medium Based on Hydrolyzed Bovine Blood Plasma at Laboratory and Bench-Scale Levels and Its Application as a Starter Culture in a Meat Product" Fermentation 6, no. 2: 45. https://doi.org/10.3390/fermentation6020045
APA StyleBarragán, P. J., Sánchez, Ó. J., & Henao-Rojas, J. C. (2020). Evaluation of the Growth Kinetics of Lactobacillus Plantarum ATCC 8014 on a Medium Based on Hydrolyzed Bovine Blood Plasma at Laboratory and Bench-Scale Levels and Its Application as a Starter Culture in a Meat Product. Fermentation, 6(2), 45. https://doi.org/10.3390/fermentation6020045






