A Review of Pyrene Bioremediation Using Mycobacterium Strains in a Different Matrix
Abstract
:1. Introduction
2. Degradation of Pyrene by Mycobacterium sp.
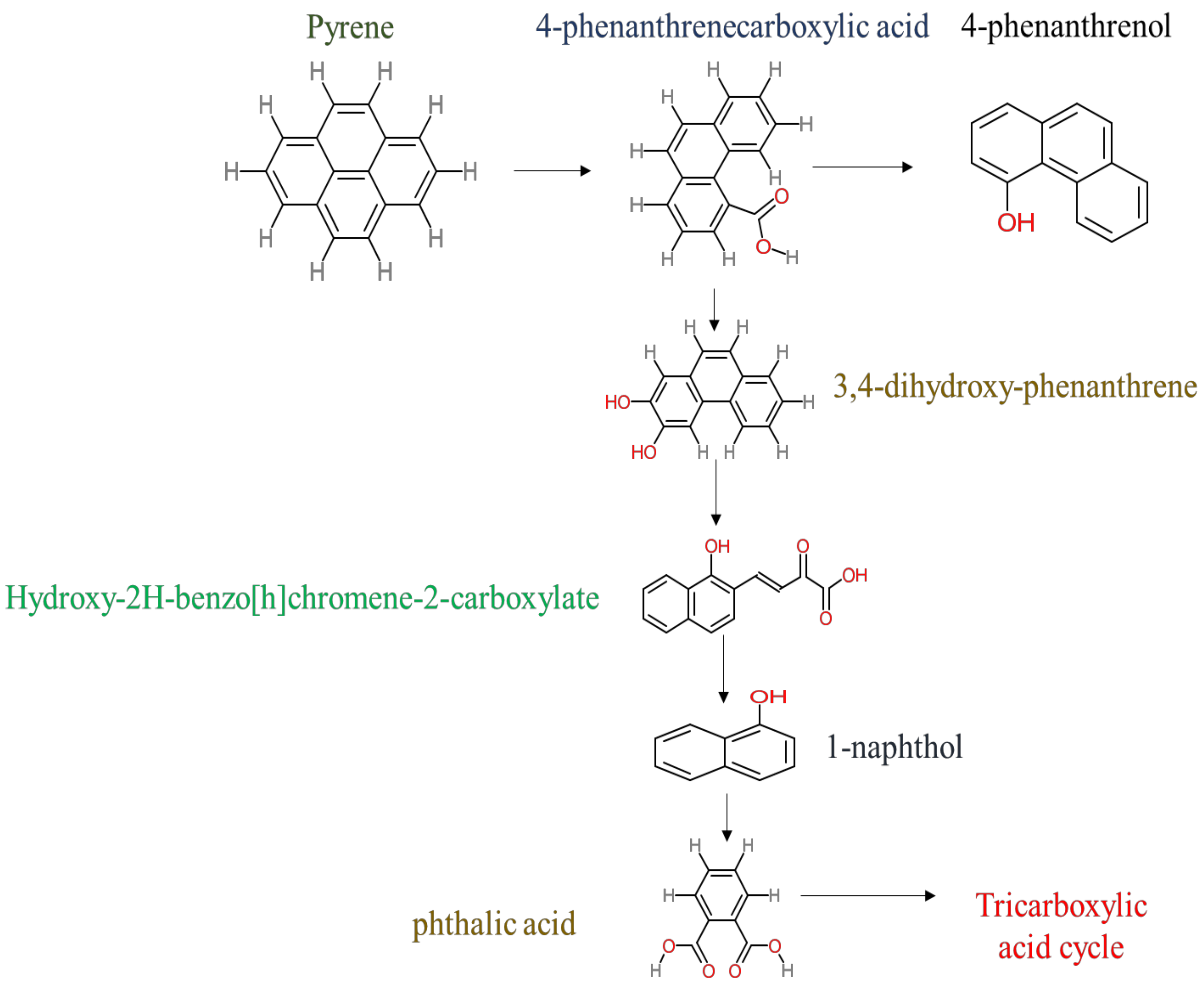
3. Identification of Pyrene Metabolites Degraded by Mycobacterium sp. and Their Biotoxicity
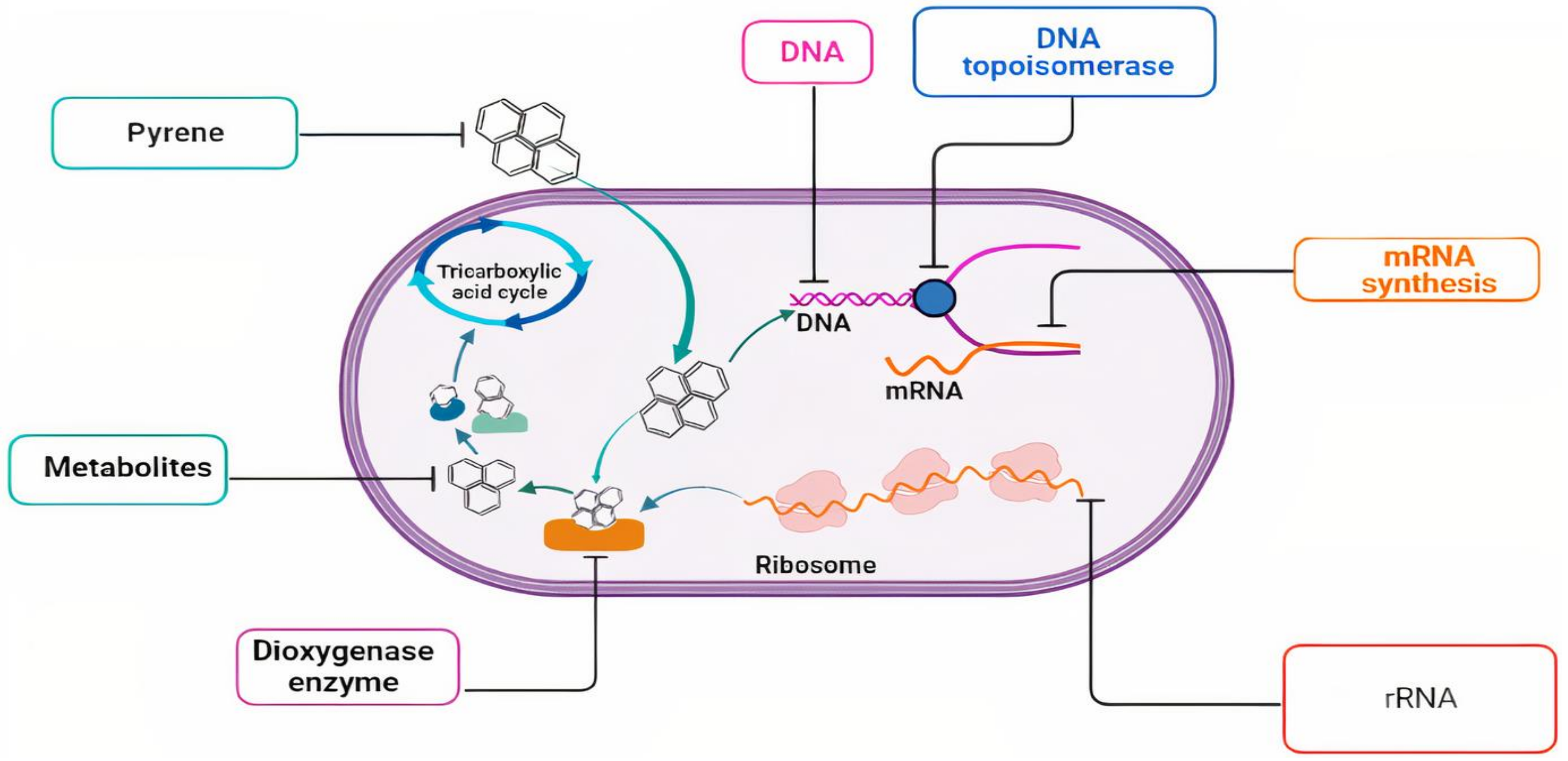
4. Proposed Biodegradation Pathways
5. Future Perspectives and Challenges
- A knowledge gap between pyrene oxidation at the field site compared to laboratory conditions needs to be addressed for each product seeking commercial success.
- The degradation of pyrene by Mycobacterium strains generates many metabolites. Some of the metabolites and their bio-toxicity have been identified, while most of them need bio-toxicity assessment.
- The main biodegradation drawback is the limitation of the bioavailability of the target pollutant. Therefore, it is highly recommended to add a biosurfactant to increase the bioavailability.
- The literature revealed that the biodegradation of pyrene via consortium microbial gives a better result than a single strain. That is referred to diverse enzymes capable of oxidizing pyrene and its metabolites.
- There are several studies that applied successful synergetic biodegradation systems for pyrene degradation, such as biofuel cells and coupling of the advanced oxidation process and biodegradation system.
6. Conclusions
- Mycobacterium strains are efficient biological agents to degrade pyrene, that is, referring to their ability to produce many functional enzymes able to metabolite pyrene and its transformation molecules.
- Phenantharene-4,5-dicarboxylic acid, dihydroxy pyrene, phenanthrene-4-carboxylic acid, phthalic acid, and pyrene-4,5-dihydrodiol were the most frequent metabolites that were detected when Mycobacterium sp. strains were used for pyrene degradation.
- Some metabolites showed positive results for the Ames mutagenicity prediction test, such as 1,2-phenanthrenedicarboxylic acid, 1-hydroxypyrene, 4,5-dihydropyrene, 4-phenanthrene-carboxylic acid, 3,4-Dihydroxyphenanthrene, Monohydroxy pyrene, and 9,10-phenanthrenequinone. However, 4-phenanthrol showed positive results for experimental and prediction tests.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Mekonnen, M.M.; Hoekstra, A.Y. Sustainability: Four Billion People Facing Severe Water Scarcity. Sci. Adv. 2016, 2, e1500323. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Qutob, M.; Rafatullah, M.; Qamar, M.; Alor, H.S.; Al Romaizan, A.N.; Hussein, M.A. A Review on Heterogeneous Oxidation of Acetaminophen Based on Micro and Nanoparticles Catalyzed by Di Ff Erent Activators. Nanotechnol. Rev. 2022, 11, 497–525. [Google Scholar] [CrossRef]
- Degefu, D.M.; Weijun, H.; Zaiyi, L.; Liang, Y.; Zhengwei, H.; Min, A. Mapping Monthly Water Scarcity in Global Transboundary Basins at Country-Basin Mesh Based Spatial Resolution. Sci. Rep. 2018, 8, 1–10. [Google Scholar] [CrossRef] [Green Version]
- Miraji, H.; Ripanda, A.; Moto, E. A Review on the Occurrences of Persistent Organic Pollutants in Corals, Sediments, Fish and Waters of the Western Indian Ocean. Egypt. J. Aquat. Res. 2021, 47, 373–379. [Google Scholar] [CrossRef]
- Gutierrez-Urbano, I.; Villen-Guzman, M.; Perez-Recuerda, R.; Rodriguez-Maroto, J.M. Removal of Polycyclic Aromatic Hydrocarbons (PAHs) in Conventional Drinking Water Treatment Processes. J. Contam. Hydrol. 2021, 243, 103888. [Google Scholar] [CrossRef]
- Dinç, B.; Çelebi, A.; Avaz, G.; Canlı, O.; Güzel, B.; Eren, B.; Yetis, U. Spatial Distribution and Source Identification of Persistent Organic Pollutants in the Sediments of the Yeşilırmak River and Coastal Area in the Black Sea. Mar. Pollut. Bull. 2021, 172, 112884. [Google Scholar] [CrossRef]
- IARC Monographs on the Evaluation of Carcinogenic Risks to Humans. IARC Monogr. Eval. Carcinog. Risks Hum. 2010, 93, 9–38. [CrossRef] [Green Version]
- Hussain, K.; Hoque, R.R.; Balachandran, S.; Medhi, S.; Idris, M.G.; Rahman, M.; Hussain, F.L. Monitoring and Risk Analysis of PAHs in the Environment. In Handbook of Environmental Materials Management; Springer: New York, NY, USA, 2019; pp. 973–1007. [Google Scholar] [CrossRef]
- Yang, Y.; Woodward, L.A.; Li, Q.X.; Wang, J. Concentrations, Source and Risk Assessment of Polycyclic Aromatic Hydrocarbons in Soils from Midway Atoll, North Pacific Ocean. PLoS ONE 2014, 9, e86441. [Google Scholar] [CrossRef]
- EPA. National Recommended Water Quality Criteria—Human Health Criteria Table. Available online: https://www.epa.gov/wqc/national-recommended-water-quality-criteria-human-health-criteria-table (accessed on 24 May 2022).
- Nikolić, V.M.; Karić, S.D.; Nikolić, Ž.M.; Tošić, M.S.; Tasić, G.S.; Milovanovic, D.M.; Kaninski, M.P.M. Novel Photochemical Advanced Oxidation Process for the Removal of Polycyclic Aromatic Hydrocarbons from Polluted Concrete. Chem. Eng. J. 2017, 312, 99–105. [Google Scholar] [CrossRef]
- Oller, I.; Malato, S.; Sánchez-Pérez, J.A. Combination of Advanced Oxidation Processes and Biological Treatments for Wastewater Decontamination-A Review. Sci. Total Environ. 2011, 409, 4141–4166. [Google Scholar] [CrossRef]
- Azubuike, C.C.; Chikere, C.B.; Okpokwasili, G.C. Bioremediation Techniques–Classification Based on Site of Application: Principles, Advantages, Limitations and Prospects. World J. Microbiol. Biotechnol. 2016, 32, 180. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Quintella, C.M.; Mata, A.M.T.; Lima, L.C.P. Overview of Bioremediation with Technology Assessment and Emphasis on Fungal Bioremediation of Oil Contaminated Soils. J. Environ. Manag. 2019, 241, 156–166. [Google Scholar] [CrossRef]
- Chen, S.; Peng, J.; Duan, G. Enrichment of Functional Microbes and Genes during Pyrene Degradation in Two Different Soils. J. Soils Sediments 2016, 16, 417–426. [Google Scholar] [CrossRef]
- Subashchandrabose, S.R.; Venkateswarlu, K.; Naidu, R.; Megharaj, M. Biodegradation of High-Molecular Weight PAHs by Rhodococcus Wratislaviensis Strain 9: Overexpression of Amidohydrolase Induced by Pyrene and BaP. Sci. Total Environ. 2019, 651, 813–821. [Google Scholar] [CrossRef] [PubMed]
- Peng, J.J.; Cai, C.; Qiao, M.; Li, H.; Zhu, Y.G. Dynamic Changes in Functional Gene Copy Numbers and Microbial Communities during Degradation of Pyrene in Soils. Environ. Pollut. 2010, 158, 2872–2879. [Google Scholar] [CrossRef]
- Kim, S.J.; Kweon, O.; Jones, R.C.; Freeman, J.P.; Edmondson, R.D.; Cerniglia, C.E. Complete and Integrated Pyrene Degradation Pathway in Mycobacterium Vanbaalenii PYR-1 Based on Systems Biology. J. Bacteriol. 2007, 189, 464–472. [Google Scholar] [CrossRef] [Green Version]
- Percival, S.L.; Williams, D.W. Mycobacterium, 2nd ed.; Elsevier: Amsterdam, The Netherlands, 2013. [Google Scholar] [CrossRef]
- Heitkamp, M.A.; Franklin, W.; Cerniglia, C.E. Microbial Metabolism of Polycyclic Aromatic Hydrocarbons: Isolation and Characterization of a Pyrene-Degrading Bacterium. Appl. Environ. Microbiol. 1988, 54, 2549–2555. [Google Scholar] [CrossRef] [Green Version]
- Rehmann, K.; Noll, H.P.; Steinberg, C.E.W.; Kettrup, A.A. Pyrene Degradation by Mycobacterium sp. Strain KR2. Chemosphere 1998, 36, 2977–2992. [Google Scholar] [CrossRef]
- Zada, S.; Zhou, H.; Xie, J.; Hu, Z.; Ali, S.; Sajjad, W.; Wang, H. Bacterial Degradation of Pyrene: Biochemical Reactions and Mechanisms. Int. Biodeterior. Biodegrad. 2021, 162, 105233. [Google Scholar] [CrossRef]
- Haritash, A.K.; Kaushik, C.P. Biodegradation Aspects of Polycyclic Aromatic Hydrocarbons (PAHs): A Review. J. Hazard. Mater. 2009, 169, 1–15. [Google Scholar] [CrossRef]
- Elyamine, A.M.; Kan, J.; Meng, S.; Tao, P.; Wang, H.; Hu, Z. Aerobic and Anaerobic Bacterial and Fungal Degradation of Pyrene: Mechanism Pathway Including Biochemical Reaction and Catabolic Genes. Int. J. Mol. Sci. 2021, 22, 8202. [Google Scholar] [CrossRef]
- Miller, C.D.; Hall, K.; Liang, Y.N.; Nieman, K.; Sorensen, D.; Issa, B.; Anderson, A.J.; Sims, R.C. Isolation and Characterization of Polycyclic Aromatic Hydrocarbon-Degrading Mycobacterium Isolates from Soil. Microb. Ecol. 2004, 48, 230–238. [Google Scholar] [CrossRef] [PubMed]
- Zeng, J.; Zhu, Q.; Wu, Y.; Chen, H.; Lin, X. Characterization of a Polycyclic Aromatic Ring-Hydroxylation Dioxygenase from Mycobacterium sp. NJS-P. Chemosphere 2017, 185, 67–74. [Google Scholar] [CrossRef] [PubMed]
- Costa, K.C.; Moskatel, L.S.; Meirelles, L.A.; Newman, D.K. PhdA Catalyzes the First Step of Phenazine-1-Carboxylic Acid Degradation in Mycobacterium Fortuitum. J. Bacteriol. 2018, 200, 10. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wanapaisan, P.; Laothamteep, N.; Vejarano, F.; Chakraborty, J.; Shintani, M.; Muangchinda, C.; Morita, T.; Suzuki-Minakuchi, C.; Inoue, K.; Nojiri, H.; et al. Synergistic Degradation of Pyrene by Five Culturable Bacteria in a Mangrove Sediment-Derived Bacterial Consortium. J. Hazard. Mater. 2018, 342, 561–570. [Google Scholar] [CrossRef] [PubMed]
- Yuan, K.; Xie, X.; Wang, X.; Lin, L.; Yang, L.; Luan, T.; Chen, B. Transcriptional Response of Mycobacterium sp. Strain A1-PYR to Multiple Polycyclic Aromatic Hydrocarbon Contaminations. Environ. Pollut. 2018, 243, 824–832. [Google Scholar] [CrossRef]
- Habe, H.; Kanemitsu, M.; Nomura, M.; Takemura, T.; Iwata, K.; Nojiri, H.; Yamane, H.; Omori, T. Isolation and Characterization of an Alkaliphilic Bacterium Utilizing Pyrene as a Carbon Source. J. Biosci. Bioeng. 2004, 98, 306–308. [Google Scholar] [CrossRef]
- McLellan, S.L.; Warshawsky, D.; Shann, J.R. The Effect of Polycyclic Aromatic Hydrocarbons on the Degradation of Benzo[a]Pyrene by Mycobacterium sp. Strain RJGII-Environ. Toxicol. Chem. 2002, 21, 253–259. [Google Scholar] [CrossRef]
- Churchill, P.F.; Morgan, A.C.; Kitchens, E. Characterization of a Pyrene-Degrading Mycobacterium sp. Strain CH-2. J. Environ. Sci. Health Part B Pestic. Food Contam. Agric. Wastes 2008, 43, 698–706. [Google Scholar] [CrossRef]
- Kim, D.W.; Lee, K.; Lee, D.H.; Cha, C.J. Comparative Genomic Analysis of Pyrene-Degrading Mycobacterium Species: Genomic Islands and Ring-Hydroxylating Dioxygenases Involved in Pyrene Degradation. J. Microbiol. 2018, 56, 798–804. [Google Scholar] [CrossRef]
- Wang, Z.; Sheng, H.; Xiang, L.; Bian, Y.; Herzberger, A.; Cheng, H.; Jiang, Q.; Jiang, X.; Wang, F. Different Performance of Pyrene Biodegradation on Metal-Modified Montmorillonite: Role of Surface Metal Ions from a Bioelectrochemical Perspective. Sci. Total Environ. 2022, 805, 150324. [Google Scholar] [CrossRef]
- Zhang, Y.; Wang, F.; Zhu, X.; Zeng, J.; Zhao, Q.; Jiang, X. Extracellular Polymeric Substances Govern the Development of Biofilm and Mass Transfer of Polycyclic Aromatic Hydrocarbons for Improved Biodegradation. Bioresour. Technol. 2015, 193, 274–280. [Google Scholar] [CrossRef] [PubMed]
- Sun, S.; Wang, H.; Chen, Y.; Lou, J.; Wu, L.; Xu, J. Salicylate and Phthalate Pathways Contributed Differently on Phenanthrene and Pyrene Degradations in Mycobacterium sp. WY10. J. Hazard. Mater. 2019, 364, 509–518. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Liu, H.; Yang, W.; Sheng, H.; Wang, F.; Harindintwali, J.D.; Herath, H.M.S.K.; Zhang, Y. Humic Acid Enhanced Pyrene Degradation by Mycobacterium sp. NJS-1. Chemosphere 2021, 288, 132613. [Google Scholar] [CrossRef] [PubMed]
- Wu, F.; Guo, C.; Liu, S.; Liang, X.; Lu, G.; Dang, Z. Pyrene Degradation by Mycobacterium Gilvum: Metabolites and Proteins Involved. Water Air Soil Pollut. 2019, 230, 67. [Google Scholar] [CrossRef]
- Chen, S.C.; Duan, G.L.; Ding, K.; Huang, F.Y.; Zhu, Y.G. DNA Stable-Isotope Probing Identifies Uncultivated Members of Pseudonocardia Associated with Biodegradation of Pyrene in Agricultural Soil. FEMS Microbiol. Ecol. 2018, 94, 1–10. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Terzaghi, E.; Posada-Baquero, R.; Di Guardo, A.; Ortega-Calvo, J.J. Microbial Degradation of Pyrene in Holm Oak (Quercus Ilex) Phyllosphere: Role of Particulate Matter in Regulating Bioaccessibility. Sci. Total Environ. 2021, 786, 147431. [Google Scholar] [CrossRef]
- Sarma, S.J.; Pakshirajan, K. Surfactant Aided Biodegradation of Pyrene Using Immobilized Cells of Mycobacterium Frederiksbergense. Int. Biodeterior. Biodegrad. 2011, 65, 73–77. [Google Scholar] [CrossRef]
- Mahanty, B.; Pakshirajan, K.; Dasu, V.V. A Two Liquid Phase Partitioning Bioreactor System for the Biodegradation of Pyrene: Comparative Evaluation and Cost-Benefit Analysis. J. Chem. Technol. Biotechnol. 2010, 85, 349–355. [Google Scholar] [CrossRef]
- Dean-Ross, D.; Cerniglia, C.E. Degradation of Pyrene by Mycobacterium Flavescens. Appl. Microbiol. Biotechnol. 1996, 46, 307–312. [Google Scholar] [CrossRef]
- Vila, J.; López, Z.; Sabaté, J.; Minguillón, C.; Solanas, A.M.; Grifoll, M. Identification of a Novel Metabolite in the Degradation of Pyrene by Mycobacterium sp. Strain AP1: Actions of the Isolate on Two- and Three-Ring Polycyclic Aromatic Hydrocarbons. Appl. Environ. Microbiol. 2001, 67, 5497–5505. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhong, Y.; Luan, T.; Zhou, H.; Lan, C.; Tam, N.F.Y. Metabolite Production in Degradation of Pyrene Alone or in a Mixture with Another Polycyclic Aromatic Hydrocarbon by Mycobacterium sp. Environ. Toxicol. Chem. 2006, 25, 2853–2859. [Google Scholar] [CrossRef] [PubMed]
- Luo, S.; Chen, B.; Lin, L.; Wang, X.; Tam, N.F.Y.; Luan, T. Pyrene Degradation Accelerated by Constructed Consortium of Bacterium and Microalga: Effects of Degradation Products on the Microalgal Growth. Environ. Sci. Technol. 2014, 48, 13917–13924. [Google Scholar] [CrossRef]
- Zhong, Y.; Luan, T.; Lin, L.; Liu, H.; Tam, N.F.Y. Production of Metabolites in the Biodegradation of Phenanthrene, Fluoranthene and Pyrene by the Mixed Culture of Mycobacterium sp. and Sphingomonas sp. Bioresour. Technol. 2011, 102, 2965–2972. [Google Scholar] [CrossRef]
- Lease, C.W.M.; Bentham, R.H.; Gaskin, S.E.; Juhasz, A.L. Isolation and Identification of Pyrene Mineralizing Mycobacterium spp. from Contaminated and Uncontaminated Sources. Appl. Environ. Soil Sci. 2011, 2011, 1–11. [Google Scholar] [CrossRef] [Green Version]
- Schneider, J.; Grosser, R.; Jayasimhulu, K.; Xue, W.; Warshawsky, D. Degradation of Pyrene, Benz[a]Anthracene, and Benzo[a]Pyrene by Mycobacterium sp. Strain RJGII-135, Isolated from a Former Coal Gasification Site. Appl. Environ. Microbiol. 1996, 62, 13–19. [Google Scholar] [CrossRef] [Green Version]
- Fan, R.; Tian, H.; Wu, Q.; Yi, Y.; Yan, X.; Liu, B. Mechanism of Bio-Electrokinetic Remediation of Pyrene Contaminated Soil: Effects of an Electric Field on the Degradation Pathway and Microbial Metabolic Processes. J. Hazard. Mater. 2022, 422, 126959. [Google Scholar] [CrossRef]
- Lu, H.; Sun, J.; Zhu, L. The Role of Artificial Root Exudate Components in Facilitating the Degradation of Pyrene in Soil. Sci. Rep. 2017, 7, 1–10. [Google Scholar] [CrossRef] [Green Version]
- Wang, S.; Li, X.; Liu, W.; Li, P.; Kong, L.; Ren, W.; Wu, H.; Tu, Y. Degradation of Pyrene by Immobilized Microorganisms in Saline-Alkaline Soil. J. Environ. Sci. 2012, 24, 1662–1669. [Google Scholar] [CrossRef]
- Deng, F.; Zhang, Z.; Yang, C.; Guo, C.; Lu, G.; Dang, Z. Pyrene Biodegradation with Layer-by-Layer Assembly Bio-Microcapsules. Ecotoxicol. Environ. Saf. 2017, 138, 9–15. [Google Scholar] [CrossRef]
- Toyama, T.; Furukawa, T.; Maeda, N.; Inoue, D.; Sei, K.; Mori, K.; Kikuchi, S.; Ike, M. Accelerated Biodegradation of Pyrene and Benzo[a]Pyrene in the Phragmites Australis Rhizosphere by Bacteria-Root Exudate Interactions. Water Res. 2011, 45, 1629–1638. [Google Scholar] [CrossRef]
- Mahanty, B.; Pakshirajan, K.; Venkata Dasu, V. Biodegradation of Pyrene by Mycobacterium Frederiksbergense in a Two-Phase Partitioning Bioreactor System. Bioresour. Technol. 2008, 99, 2694–2698. [Google Scholar] [CrossRef] [PubMed]
- Ortega-Calvo, J.J.; Gschwend, P.M. Influence of Low Oxygen Tensions and Sorption to Sediment Black Carbon on Biodegradation of Pyrene. Appl. Environ. Microbiol. 2010, 76, 4430–4437. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Deng, F.; Liao, C.; Yang, C.; Guo, C.; Dang, Z. Enhanced Biodegradation of Pyrene by Immobilized Bacteria on Modified Biomass Materials. Int. Biodeterior. Biodegrad. 2016, 110, 46–52. [Google Scholar] [CrossRef]
- Chang, B.V.; Chang, I.T.; Yuan, S.Y. Biodegradation of Phenanthrene and Pyrene from Mangrove Sediment in Subtropical Taiwan. J. Environ. Sci. Health Part A Toxic/Hazardous Subst. Environ. Eng. 2008, 43, 233–238. [Google Scholar] [CrossRef]
- Zeng, J.; Lin, X.; Zhang, J.; Li, X. Isolation of Polycyclic Aromatic Hydrocarbons (PAHs)-Degrading Mycobacterium Spp. and the Degradation in Soil. J. Hazard. Mater. 2010, 183, 718–723. [Google Scholar] [CrossRef]
- Jacques, R.J.S.; Okeke, B.C.; Bento, F.M.; Teixeira, A.S.; Peralba, M.C.R.; Camargo, F.A.O. Microbial Consortium Bioaugmentation of a Polycyclic Aromatic Hydrocarbons Contaminated Soil. Bioresour. Technol. 2008, 99, 2637–2643. [Google Scholar] [CrossRef]
- Cerniglia, C.E. Degradation Of A Mixture Of High-Molecular-Weight Polycyclic Aromatic Hydrocarbons By A Mycobacterium Strain Pyr-1. J. Soil Contam. 1995, 4, 77–91. [Google Scholar] [CrossRef]
- Sho, M.; Hamel, C.; Greer, C.W. Two Distinct Gene Clusters Encode Pyrene Degradation in Mycobacterium sp. Strain S65. FEMS Microbiol. Ecol. 2004, 48, 209–220. [Google Scholar] [CrossRef] [Green Version]
- López, Z.; Vila, J.; Ortega-Calvo, J.J.; Grifoll, M. Simultaneous Biodegradation of Creosote-Polycyclic Aromatic Hydrocarbons by a Pyrene-Degrading Mycobacterium. Appl. Microbiol. Biotechnol. 2008, 78, 165–172. [Google Scholar] [CrossRef] [Green Version]
- Child, R.; Miller, C.D.; Liang, Y.; Sims, R.C.; Anderson, A.J. Pyrene Mineralization by Mycobacterium sp. Strain KMS in a Barley Rhizosphere. J. Environ. Qual. 2007, 36, 1260–1265. [Google Scholar] [CrossRef] [PubMed]
- Badejo, A.C.; Badejo, A.O.; Shin, K.H.; Chai, Y.G. A Gene Expression Study of the Activities of Aromatic Ring-Cleavage Dioxygenases in Mycobacterium Gilvum PYR-GCK to Changes in Salinity and PH during Pyrene Degradation. PLoS ONE 2013, 8, e58066. [Google Scholar] [CrossRef] [Green Version]
- Ramirez, N.; Cutright, T.; Ju, L.K. Pyrene Biodegradatin in Aqueous Solutions and Soil Slurries by Mycobacteriurn PYR-1 and Enriched Consortium. Chemosphere 2001, 44, 1079–1086. [Google Scholar] [CrossRef]
- Vila, J.; Grifoll, M. Actions of Mycobacterium sp. Strain AP1 on the Saturated- and Aromatic-Hydrocarbon Fractions of Fuel Oil in a Marine Medium. Appl. Environ. Microbiol. 2009, 75, 6232–6239. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jimenez, I.Y.; Bartha, R. Solvent-Augmented Mineralization of Pyrene by a Mycobacterium sp. Appl. Environ. Microbiol. 1996, 62, 2311–2316. [Google Scholar] [CrossRef] [Green Version]
- Guo, C.; Dang, Z.; Wong, Y.; Tam, N.F. Biodegradation Ability and Dioxgenase Genes of PAH-Degrading Sphingomonas and Mycobacterium Strains Isolated from Mangrove Sediments. Int. Biodeterior. Biodegrad. 2010, 64, 419–426. [Google Scholar] [CrossRef]
- Seo, J.S.; Keum, Y.S.; Kim, K.; Li, Q.X. Degradation of Pyrene by Mycobacterium Aromativorans Strain JS19b1. J. Appl. Biol. Chem. 2010, 53, 323–329. [Google Scholar] [CrossRef]
- Kim, Y.H.; Freeman, J.P.; Moody, J.D.; Engesser, K.H.; Cerniglia, C.E. Effects of PH on the Degradation of Phenanthrene and Pyrene by Mycobacterium Vanbaalenii PYR-1. Appl. Microbiol. Biotechnol. 2005, 67, 275–285. [Google Scholar] [CrossRef]
- Liang, Y.; Gardner, D.R.; Miller, C.D.; Chen, D.; Anderson, A.J.; Weimer, B.C.; Sims, R.C. Study of Biochemical Pathways and Enzymes Involved in Pyrene Degradation by Mycobacterium sp. Strain KMS. Appl. Environ. Microbiol. 2006, 72, 7821–7828. [Google Scholar] [CrossRef] [Green Version]
- Sarma, P.M.; Duraja, P.; Deshpande, S.; Lal, B. Degradation of Pyrene by an Enteric Bacterium, Leclercia Adecarboxylata PS4040. Biodegradation 2010, 21, 59–69. [Google Scholar] [CrossRef]
- Zheng, T.; Liu, R.; Chen, J.; Gu, X.; Wang, J.; Li, L.; Hou, L.; Li, N.; Wang, Y. Fire Phoenix Plant Mediated Microbial Degradation of Pyrene: Increased Expression of Functional Genes and Diminishing of Degraded Products. Chem. Eng. J. 2021, 407, 126343. [Google Scholar] [CrossRef]
- Agrawal, N.; Shahi, S.K. Degradation of Polycyclic Aromatic Hydrocarbon (Pyrene) Using Novel Fungal Strain Coriolopsis Byrsina Strain APC5. Int. Biodeterior. Biodegrad. 2017, 122, 69–81. [Google Scholar] [CrossRef]
- Lu, J.; Guo, C.; Zhang, M.; Lu, G.; Dang, Z. Biodegradation of Single Pyrene and Mixtures of Pyrene by a Fusant Bacterial Strain F14. Int. Biodeterior. Biodegrad. 2014, 87, 75–80. [Google Scholar] [CrossRef]
- Al Farraj, D.A.; Hadibarata, T.; Yuniarto, A.; Syafiuddin, A.; Surtikanti, H.K.; Elshikh, M.S.; Al Khulaifi, M.M.; Al-Kufaidy, R. Characterization of Pyrene and Chrysene Degradation by Halophilic Hortaea sp. B15. Bioprocess Biosyst. Eng. 2019, 42, 963–969. [Google Scholar] [CrossRef]
- Ma, J.; Xu, L.; Jia, L. Characterization of Pyrene Degradation by Pseudomonas sp. Strain Jpyr-1 Isolated from Active Sewage Sludge. Bioresour. Technol. 2013, 140, 15–21. [Google Scholar] [CrossRef]
- Rathour, R.; Gupta, J.; Tyagi, B.; Kumari, T.; Thakur, I.S. Biodegradation of Pyrene in Soil Microcosm by Shewanella sp. ISTPL2, a Psychrophilic, Alkalophilic and Halophilic Bacterium. Bioresour. Technol. Rep. 2018, 4, 129–136. [Google Scholar] [CrossRef]
- Swati; Ghosh, P.; Thakur, I.S. Biodegradation of Pyrene by Pseudomonas sp. ISTPY2 Isolated from Landfill Soil: Process Optimisation Using Box-Behnken Design Model. Bioresour. Technol. Rep. 2019, 8, 100329. [Google Scholar] [CrossRef]
- Swati; Kumari, M.; Ghosh, P.; Thakur, I.S. Evaluation of a Biosurfactant Producing Bacterial Strain Pseudomonas sp. ISTPY2 for Efficient Pyrene Degradation and Landfill Soil Bioremediation through Soil Microcosm and Proteomic Studies. Bioresour. Technol. Rep. 2020, 12, 100607. [Google Scholar] [CrossRef]
- Jin, J.; Yao, J.; Zhang, Q.; Liu, J. Biodegradation of Pyrene by Pseudomonas sp. JPN2 and Its Initial Degrading Mechanism Study by Combining the Catabolic NahAc Gene and Structure-Based Analyses. Chemosphere 2016, 164, 379–386. [Google Scholar] [CrossRef]
- Fernández-López, C.; Posada-Baquero, R.; García, J.L.; Castilla-Alcantara, J.C.; Cantos, M.; Ortega-Calvo, J.J. Root-Mediated Bacterial Accessibility and Cometabolism of Pyrene in Soil. Sci. Total Environ. 2021, 760, 143408. [Google Scholar] [CrossRef]
- Kashyap, N.; Moholkar, V.S. Intensification of Pyrene Degradation by Native Candida Tropicalis MTCC 184 with Sonication: Kinetic and Mechanistic Investigation. Chem. Eng. Process. Process Intensif. 2021, 164, 108415. [Google Scholar] [CrossRef]
- Ghosh, I.; Jasmine, J.; Mukherji, S. Biodegradation of Pyrene by a Pseudomonas Aeruginosa Strain RS1 Isolated from Refinery Sludge. Bioresour. Technol. 2014, 166, 548–558. [Google Scholar] [CrossRef]
- Nzila, A.; Ramirez, C.O.; Musa, M.M.; Sankara, S.; Basheer, C.; Li, Q.X. Pyrene Biodegradation and Proteomic Analysis in Achromobacter Xylosoxidans, PY4 Strain. Int. Biodeterior. Biodegrad. 2018, 130, 40–47. [Google Scholar] [CrossRef]
- Umar, Z.D.; Nor Azwady, A.A.; Zulkifli, S.Z.; Muskhazli, M. Effective Phenanthrene and Pyrene Biodegradation Using Enterobacter sp. MM087 (KT933254) Isolated from Used Engine Oil Contaminated Soil. Egypt. J. Pet. 2018, 27, 349–359. [Google Scholar] [CrossRef]
- Ghosh, I.; Mukherji, S. Substrate Interaction Effects during Pyrene Biodegradation by Pseudomonas Aeruginosa RS1. J. Environ. Chem. Eng. 2017, 5, 1791–1800. [Google Scholar] [CrossRef]
- Gupta, B.; Puri, S.; Thakur, I.S.; Kaur, J. Enhanced Pyrene Degradation by a Biosurfactant Producing Acinetobacter Baumannii BJ5: Growth Kinetics, Toxicity and Substrate Inhibition Studies. Environ. Technol. Innov. 2020, 19, 100804. [Google Scholar] [CrossRef]
- Jiang, B.; Chen, Y.; Xing, Y.; Lian, L.; Shen, Y.; Zhang, B.; Zhang, H.; Sun, G.; Li, J.; Wang, X.; et al. Negative Correlations between Cultivable and Active-yet-Uncultivable Pyrene Degraders Explain the Postponed Bioaugmentation. J. Hazard. Mater. 2022, 423, 127189. [Google Scholar] [CrossRef]
- Yang, W.; Hadibarata, T.; Mahmoud, A.H.; Yuniarto, A. Biotransformation of Pyrene in Soil in the Presence of Earthworm Eisenia Fetida. Environ. Technol. Innov. 2020, 18, 100701. [Google Scholar] [CrossRef]
- Li, X.; Zhang, X.; Li, L.; Lin, C.; Dong, W.; Shen, W.; Yong, X.; Jia, H.; Wu, X.; Zhou, J. Anaerobic Biodegradation of Pyrene by Klebsiella sp. LZ6 and Its Proposed Metabolic Pathway. Environ. Technol. 2020, 41, 2130–2139. [Google Scholar] [CrossRef]
- Giménez, B.N.; Conte, L.O.; Alfano, O.M.; Schenone, A.V. Paracetamol Removal by Photo-Fenton Processes at near-Neutral PH Using a Solar Simulator: Optimization by D-Optimal Experimental Design and Toxicity Evaluation. J. Photochem. Photobiol. A Chem. 2020, 397, 112584. [Google Scholar] [CrossRef]
- Dabrowska, D.; Kot-Wasik, A.; Namieśnik, J. Pathways and Analytical Tools in Degradation Studies of Organic Pollutants. Crit. Rev. Anal. Chem. 2005, 35, 155–176. [Google Scholar] [CrossRef]
- Krivobok, S.; Kuony, S.; Meyer, C.; Louwagie, M.; Willison, J.C.; Jouanneau, Y. Identification of Pyrene-Induced Proteins in Mycobacterium sp. Strain 6PY1: Evidence for Two Ring-Hydroxylating Dioxygenases. J. Bacteriol. 2003, 185, 3828–3841. [Google Scholar] [CrossRef] [Green Version]
- Maruyama, T.; Ishikura, M.; Taki, H.; Shindo, K.; Kasai, H.; Haga, M.; Inomata, Y.; Misawa, N. Isolation and Characterization of O-Xylene Oxygenase Genes from Rhodococcus Opacus TKN14. Appl. Environ. Microbiol. 2005, 71, 7705–7715. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pagnout, C.; Frache, G.; Poupin, P.; Maunit, B.; Muller, J.F.; Férard, J.F. Isolation and Characterization of a Gene Cluster Involved in PAH Degradation in Mycobacterium sp. Strain SNP11: Expression in Mycobacterium Smegmatis Mc2155. Res. Microbiol. 2007, 158, 175–186. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.J.; Kweon, O.; Freeman, J.P.; Jones, R.C.; Adjei, M.D.; Jhoo, J.W.; Edmondson, R.D.; Cerniglia, C.E. Molecular Cloning and Expression of Genes Encoding a Novel Dioxygenase Involved in Low- and High-Molecular-Weight Polycyclic Aromatic Hydrocarbon Degradation in Mycobacterium Vanbaalenii PYR-1. Appl. Environ. Microbiol. 2006, 72, 1045–1054. [Google Scholar] [CrossRef] [Green Version]
- Adamou, J.E.; Heinrichs, J.H.; Erwin, A.L.; Walsh, W.; Gayle, T.; Dormitzer, M.; Dagan, R.; Brewah, Y.A.; Barren, P.; Lathigra, R.; et al. Identification and Characterization of a Novel Family of Pneumococcal Proteins That Are Protective against Sepsis. Infect. Immun. 2001, 69, 949–958. [Google Scholar] [CrossRef] [Green Version]
- Wu, Y.; Xu, Y.; Zhou, N. A Newly Defined Dioxygenase System from Mycobacterium Vanbaalenii PYR-1 Endowed with an Enhanced Activity of Dihydroxylation of High-Molecular-Weight Polyaromatic Hydrocarbons. Front. Environ. Sci. Eng. 2020, 14, 1–11. [Google Scholar] [CrossRef]
- Levieux, J.A.; Medellin, B.; Johnson, W.H.; Erwin, K.; Li, W.; Johnson, I.A.; Zhang, Y.J.; Whitman, C.P. Structural Characterization of the Hydratase-Aldolases, NahE and PhdJ: Implications for the Specificity, Catalysis, and N-Acetylneuraminate Lyase Subgroup of the Aldolase Superfamily. Biochemistry 2018, 57, 3524–3536. [Google Scholar] [CrossRef]
- Kumari, S.; Regar, R.K.; Bajaj, A.; Ch, R.; Satyanarayana, G.N.V.; Mudiam, M.K.R.; Manickam, N. Simultaneous Biodegradation of Polyaromatic Hydrocarbons by a Stenotrophomonas Sp: Characterization of Nid Genes and Effect of Surfactants on Degradation. Indian J. Microbiol. 2017, 57, 60–67. [Google Scholar] [CrossRef] [Green Version]
- Saito, A.; Iwabuchi, T.; Harayama, S. A Novel Phenanthrene Dioxygenase from Nocardioides sp. Strain KP7: Expression in Escherichia Coli. J. Bacteriol. 2000, 182, 2134–2141. [Google Scholar] [CrossRef] [Green Version]
- Walter, U.; Beyer, M.; Klein, J.; Rehm, H. Appeal Microbiology Biote Nology Degradation of Pyrene by Rhodococcus sp. UW1. Power 1991, 671–676. [Google Scholar]
- Sakshi; Singh, S.K.; Haritash, A.K. Catabolic Enzyme Activity and Kinetics of Pyrene Degradation by Novel Bacterial Strains Isolated from Contaminated Soil. Environ. Technol. Innov. 2021, 23, 101744. [Google Scholar] [CrossRef]
- Zhou, H.; Zhang, S.; Xie, J.; Wei, H.; Hu, Z.; Wang, H. Pyrene Biodegradation and Its Potential Pathway Involving Roseobacter Clade Bacteria. Int. Biodeterior. Biodegrad. 2020, 150, 104961. [Google Scholar] [CrossRef]
- Khanna, G.P.; Goyal, D.; Khanna, S. Pyrene Biodegradation by Bacillus Spp. Isolated from Coal Tar-Contaminated Soil. Bioremediat. J. 2011, 15, 12–25. [Google Scholar] [CrossRef]
- Vasconcelos, M.R.S.; Vieira, G.A.L.; Otero, I.V.R.; Bonugli-Santos, R.C.; Rodrigues, M.V.N.; Rehder, V.L.G.; Ferro, M.; Boaventura, S.; Bacci, M.; Sette, L.D. Pyrene Degradation by Marine-Derived Ascomycete: Process Optimization, Toxicity, and Metabolic Analyses. Environ. Sci. Pollut. Res. 2019, 26, 12412–12424. [Google Scholar] [CrossRef]


| Strain | Functional Genes | Reference |
|---|---|---|
| Mycobacterium sp. | NidA, NidA3 | [15] |
| Mycobacterium sp. 6PY1 | PdoB2, PdoA1, PdoA2 | [24] |
| Mycobacterium sp. JLS | NidB and NidA | [25] |
| Mycobacterium sp. MCS | NidA and NidB | |
| Mycobacterium sp. NJS-P | PdoAB | [26] |
| Mycobacterium sp. S65 | PdoAB | |
| Mycobacterium fortuitum | PhdA and PhdB | [27] |
| Mycobacterium sp. PO1 and PO2 | NidA, PhdA, and NidA3 | [28] |
| Mycobacterium sp. AP1-PYR | NidAB and PdoA2B2 | [29] |
| Mycobacterium sp. MHP-1 | NidAB | [30] |
| Mycobacterium sp. RJGII-135 | NahAc, BphA1, OrtolC1C2 | [31] |
| Mycobacterium sp. KMS | PdoF | [32] |
| Mycobacterium vanbaalenii PYR-1 | NidB, NidA, NidB2, PhdF, PhdG, NidD, PhdJ, PhtAb, PhtAc, PhtAd, PhtB, NidA3, NidB3 | |
| Mycobacterium gilvum PYR-GCK | AraC | |
| Mycobacterium sp. gilvum PYR10 | NidAB and NidA3B3 | [33] |
| Mycobacterium sp. Pallens PYR15 | NidAB and NidA3B3 |
| Strains | Accession No./or Reference No. | Biodegradation Matrix | Degradation % | Incubation Time | pH | Temperature (°C) | Concentration of Pyrene | References | |
|---|---|---|---|---|---|---|---|---|---|
| Mycobacterium strains | * | Pyrene-containing soil | 80 | 35 days | Around 8 | 25 | 60 mg/kg | Ranging from 8.9 × 109 to 1.9 × 1010 copies/g | [15] |
| Myco66F/Myco600R | FN690762 and FN690936 | Pyrene-spiked soils | 81 | 60 days | 5.84 | 25 | 50 mg/kg | * | [17] |
| Mycobacterium vanbaalenii PYR-1 | NR_074572.1 | Phosphate-based minimal medium | 100 | 24 h | * | * | 25 µM | OD600 = 1.0 | [18] |
| Mycobacterium sp. KR2 | * | Mineral salts medium | 60 | 8 days | 7 | 20 | 0.5 mg/mL | OD578 = 0.5–0.6 | [21] |
| Mycobacterium sp. PO1 and PO2 | PO1 (NZ_BLTG00000000.1) PO2 (NZ_BLTH00000000.1) | Carbon-free mineral medium (CFMM) culture | 100 | 6 days | * | 30 | 100 mg/L | 108 CFU/mL | [28] |
| Novosphingobium pentaromativorans PY1 | |||||||||
| 3-Ochrobactrum sp. PW1 | |||||||||
| 4-Bacillus sp. FW1 | |||||||||
| Mycobacterium sp. NJS-1 | AB548662 | Metal-modified montmorillonite | 93.6 | 3 days | 7 | 28 | 15 mg/L | 1.6 × 107 CFU/mL | [34] |
| Micrococcus sp. PHE9 | AB548663 | Biofilms extracellular polymeric substances-extracted bacteria | 58 | 18 days | * | 28 | 100 mg/L | 1.6 × 108 CFU/mL | [35] |
| Mycobacterium sp. NJS-P | |||||||||
| Mycobacterium sp. WY10 | NZ_CP018043.1 | Mineral salts medium | 83 | 72 h | * | 28 | 50 mg/L | OD600 = 1.0 3 × 108 CFU/mL | [36] |
| Mycobacterium sp. NJS-1 | AB548662.1 | Mineral medium | 90 | 7 days | Acidic condition | 28 | 200 mg/L | 1.6 × 107 CFU/mL | [37] |
| Mycobacterium gilvum CP13 | KF378755 | Mineral salts medium | 95 | 7 days | Alkaline environment | 35 | 50 mg/L | OD600 = 0.5 | [38] |
| 2-Mycobacterium sp. denovo930873 | * | Agricultural soil | 80 | 35 days | * | 25 | 100 mg/kg | * | [39] |
| Mycobacterium gilvum VM552 | ATCC 43909 | Pyrene present on the leaf surface of holm oak (Quercus ilex) | 17 | 2 weeks | * | 23± 2 | * | 104 cells/g | [40] |
| Mycobacterium frederiksbergense | Taxonomy ID: 117567 | Batch shake flask experiments | 100 | 200 h | 7 | 28 | 1000 mg/L | * | [41] |
| Mycobacterium frederiksbergense | Taxonomy ID: 117567 | Slurry phase and surfactant-aided systems | 100 | 6 days | 7 | 28 | 400 mg/L | * | [42] |
| Mycobacterium sp. flavescens PYR-1 | * | Mineral salts medium | 38.8 | 2 weeks | Natural and 4 | 24 | 50 µg/ml | 2.2 × 107 cells/mL | [43] |
| Mycobacterium sp. AP1 | JX239754 | Pyrene-mineral salts medium | Decreased from 180 to 50 µg/mL around 72 | 6 days | * | 25 | 180 µg/ml | * | [44] |
| Mycobacterium sp. A1-PYR | X93183 | PYR in liquid medium | 33 | 7 days | * | 30 | 10 mg/L | OD600 = 1.0 | [45] |
| Selenastrum capricornutum | X93183 | Soil extract (SE) medium | 100 | 14 days | 7 | * | 10 mg/L | 1.0 × 107 CFU/mL | [46] |
| Mycobacterium sp. A1-PYR | |||||||||
| Mycobacterium sp. A1-PYR | X93183 | Pyrene-mineral salts medium | 50 | 7 days | * | 30 | 10 mg/L | OD600 = 1.0 | [47] |
| Sphingomonas sp. PheB4 | |||||||||
| 1-Mycobacterium monacense B9-21-178 | AF107039.2 | Liquid Culture | 100 | 20 days | * | 30 | 250 mg/L | 105–106 cells/mL | [48] |
| 2-Mycobacterium sp. KMS | AY083217 | ||||||||
| 3-Mycobacterium sp. JLS | AF387804 | ||||||||
| 4-Mycobacterium gilvum VM0442 | AF544636.1 | ||||||||
| 5-Mycobacterium gilvum VM0552 | AF544635 | ||||||||
| 6-Mycobacterium gilvum VM0504 | AF544634 | ||||||||
| 7-Mycobacterium gilvum VM0505 | AF544633 | ||||||||
| 8-Mycobacterium sp. PYR GCK | AY694989 | ||||||||
| 9-Mycobacterium petroleiphilum | UEGS01000001.1 | ||||||||
| 10-Mycobacterium chlorophenolicus PCP-1 | X79094 | ||||||||
| 1-Mycobacterium sp. PYR GCK | AY694989 | 94.3 | |||||||
| 2-Mycobacterium gilvum VM0583 | AF544637.1 | ||||||||
| 3-Mycobacterium gilvum VM0442 | AF544636.1 | ||||||||
| 4-Mycobacterium gilvum VM0552 | AF544635 | ||||||||
| 5-Mycobacterium gilvum iVM0504 | AF544634 | ||||||||
| 6-Mycobacterium gilvum VM0505 | AF544633 | ||||||||
| 7-Mycobacterium sp. BB1 | X81891 | ||||||||
| 8-Mycobacterium sp. HE5 | AJ012738 | ||||||||
| 9-Mycobacterium mucogenicum | AY457073.1 | ||||||||
| 1-Mycobacterium sp. JLS | AF387804 | 95.5 | |||||||
| 2-Mycobacterium monacense B9-21-178 | AF107039.2 | ||||||||
| 3-Mycobacterium vaccae VM0588 | AF544639.1 | ||||||||
| 4-Mycobacterium vaccae VM0587 | AF544638.1 | ||||||||
| 5-Mycobacterium sp. KMS | AY083217 | ||||||||
| 6-Mycobacterium sp. MCS | AF387803.1 | ||||||||
| 7-Mycobacterium doricum DSM 44339 | AF547917.1 | ||||||||
| 8-Mycobacterium doricum | AF264700.1 | ||||||||
| 9-Mycobacterium duvalii | NR_026073.1 | ||||||||
| 10-Mycobacterium duvalii CIP 104539 | AF547918.1 | ||||||||
| Mycobacterium sp. RJGII-135 | AY216464.1 | Minimal basal salts medium | 50 | 4–8 h | * | * | 0.5 µg/mL | * | [49] |
| Mycobacterium sp. MHP-1 | AB180481 | Carbon-free minimal medium | 50 | 7 days | 9 | 30 | Final concentration at 0.1% [w/v] | 3.9 × 109 CFU/mL | [30] |
| 1-Mycobacterium sp. gilvum PYR10 | * | Minimal media containing pyrene | 95 | 6 days | * | * | 100 mg/L | * | [33] |
| 2-Mycobacterium sp. pallens PYR15 | |||||||||
| 1-Mycobacterium | * | Bio-electrokinetic remediation | 54.3 | 91 days | 8 | Room temperature | 286 mg/kg | 107–108 CFU/g soil | [50] |
| 2-Aeromicrobium | |||||||||
| 3-Arenimonas | |||||||||
| 4-Bacillus | |||||||||
| 5-Hydrogenophaga | |||||||||
| 6-Azoarcus | |||||||||
| 7-Luteimonas | |||||||||
| Mycobacterium sp. | * | Soil placed into culture dishes | 54.3 ± 1.7 | 21 days | Soil pH 6.6 | * | 120.2 ± 1.76 mg/kg | * | [51] |
| Mycobacterium sp. B2 | * | Saline alkaline soils | 83.2 | 30 days | Soil pH 8.75 | 28 | 100 mg/L | * | [52] |
| Mycobacterium gilvum CP13 | KF378755.1 | Mineral salts medium by LBL bio-microcapsules | 95 | 3 days | 7 | * | 10 mg/L | OD600 = 2.0 | [53] |
| Mycobacterium gilvum IPF | AB491971 | Pyrene-basal salts medium | 100 | 3 days | 7.0–7.3 | 28 | 100 mg/L | OD600 = 0.02 | [54] |
| Mycobacterium frederiksbergense | Taxonomy ID: 117567 | Slurry phase system | 100 | 200 h | 7 | 28 | 50 mg/L | * | [55] |
| Mycobacterium gilvum VM552 | NR_118915.1 | Aqueous medium | 100 | 20 min | 5.8 | 23 | 8.4 ng/ml | OD600 = 0.019 | [56] |
| Mycobacterium gilvum CP13 | KF378755 | Aqueous solution + modified peanut hull powder | 98 | 7 days | 7 | 30 | 10 mg/L | OD600 = 2.0 × 107 CFU/mL | [57] |
| 1-Mycobacterium barrassi | * | Aqueous solution + sediments | 92 | 25 days | 7 | 30 | 50 µg/kg Pyrene + phenanthrene | * | [58] |
| 2-Dyella ginsengisoli | |||||||||
| 3-Rhodococcus equi | |||||||||
| 4-Bacillus pumilus | |||||||||
| 5-Bacillus weihenstephanensis | |||||||||
| 6-Labrys sp. | |||||||||
| Mycobacterium strains (NJS-1 and NJS-P) | (AB548662) for NJS-1 and (AB548663) for NJS-P | Liquid culture minimal medium | 87.9 and 92 for NJS-1 and NJS-P, respectively. | 2 weeks | 6.5–7 | 30 | 100 mg/L | 106 cells/g | [59] |
| 1-Mycobacterium fortuitum | U92089.1 | Pyrene-containing soil | 96.3 | 70 days | 7 | 30 | 962.7 mg/kg | 2.0 × 108 CFU/g | [60] |
| 2-Bacillus cereus | |||||||||
| 3-Microbacterium sp. | |||||||||
| 4-Gordonia polyisoprenivorans | |||||||||
| 5-Microbacteriaceae bacterium | |||||||||
| Mycobacterium sp. PYR-1 | * | Experimental Microcosms | 74 mixture of PAHs including pyrene | 6 days | * | 24 | 916.7 µg/400 µl | 4.5 × 107 cells/mL | [61] |
| Mycobacterium sp. S65 | AF544230 | Mineral salts medium | 60 | 96 h | * | 30 | 1 mg/L | 1.0 × 107 CFU/mL | [62] |
| Mycobacterium sp. AP1 | JX239754 | Mineral medium | 11.5 | 30 days | * | 26 | 0.20 nmol/mL | * | [63] |
| Mycobacterium sp. KMS | AY083217 | Microcosm system | Little to no pyrene mineralization | 10 days | * | 20 | 20 mg/155 µL | 1.0 × 108 CFU/mL | [64] |
| Mycobacterium gilvum PYR-GCK | NCBI Taxonomy ID 350054 | Fluctuating environmental conditions | 70 | 48 h | 6.5 | * | 1.0 M | OD545 = 2.95 | [65] |
| Mycobacterium sp. PYR-1 | ATCC 2676 | Aqueous pyrene solution | 50 | 25 h | 6.6 | 24 | 120 µg/L | * | [66] |
| Mycobacterium sp. AP1 | JX239754 | Marine medium | 75 | 60 days | * | 26 | 200 mg/L | 2.0 × 107 CFU/mL | [67] |
| Mycobacterium sp. | * | Mineral salts solution, | 50 | 2 to 3 days | 7 | 30 | 250 µg/mL | * | [68] |
| 1-Sphingomonas | * | Mineral salt medium | 100 | 14 days | 7.2 | 20 ± 2 | 10 mg/L for each phenanthrene, fluoranthene, and pyrene | OD600 = 3.0 | [69] |
| 2-Mycobacterium | |||||||||
| 3-Rhodococcus | |||||||||
| 4-Paracoccus | |||||||||
| 5-Pseudomonas |
| Scheme 4040 | Metabolites | References |
|---|---|---|
| Leclercia adecarboxylata PS4040 | 1,2-phenanthrenedicarboxylic acid 2-carboxybenzaldehyde Ortho-phthalic acid 1-hydroxypyrene | [73] |
| Fire Phoenix plant (Festuca spp.) mediated microbial | Phthalic acid dehydroxylated pyrene 1-hydroxypyrene 1-hydroxy-2-naphthoic acid Salicylic acid Benzoic acid | [74] |
| Coriolopsis byrsina strain APC5 | Pyruvic acid Benzoic acid Benzoic acid 2-hydroxy pentyl ester Phenanthrene Pthalic acid diisopropylester 4,5-dihydroxy pyrene | [75] |
| Fusant bacterial strain F14 fusion between Sphingomonas sp. GY2B and Pseudomonas sp. GP3A | 4,5-dihydropyrene | [76] |
| Hortaea sp. B15 | Phthalic acid 1-Hydroxy-2-naphthoic acid | [77] |
| Pseudomonas sp. strain Jpyr-1 | Phthalate 3,4-dihydrodiol Phthalate 1-hydroxy-2-naphthalene carboxylic acid 4-phenanthrene-carboxylic acid | [78] |
| Shewanella sp. ISTPL2 | 4,5-dihydroxypyrene 2-carboxybenzalpyruvate Phthalic acid Salicylic acid | [79] |
| Pseudomonas sp. ISTPY2 | Pyrene 4,5-Dihydroxypyren. 1,2-dihydroxynaphthalene 2,3-dihydroxybenzoate Phthalate Catechol | [80] |
| Pseudomonas sp. ISTPY2 | Phthalate 4,5-dioxygenase Aldehyde dehydrogenase | [81] |
| Pseudomonas sp. JPN2 | 4,5-dihydroxy-4,5-dihydropyrene 4-phenanthrol 1-hydroxy-2-naphthoic acid Phthalate | [82] |
| Pseudomonas putida G7. | 1-hydroxypyrene Phthalic acid Benzoic acid Silylated derivatives | [83] |
| Candida tropicalis MTCC 184 | Menthyl salicylate (methyl ester of salicyclic acid) | [84] |
| Pseudomonas aeruginosa strain RS1 | Phenanthrene 4,5-dicarboxylate 4-oxa-Pyrene-5-one Dihydroxypyrene 4-Phenanthroic acid 4,5-Dihydroxyphthalate 2,2-Dicarboxy-6,6-dihydroxybiphenyl 4-Phenanthroic acid 3,4-Dihydroxyphenanthrene | [85] |
| Achromobacter xylosoxidans PY4 strain | Monohydroxy pyrene 1-methoxyl-2-H-benzo[h]chromene-2-carboxylic acid 9,10-phenanthrenequinone 1-methoxyl-trans-2′-carboxybenzalpyruvate Dibutyl-phthalate | [86] |
| Enterobacter sp. MM087 (KT933254) | Pyrene cis-4,5-dihydrodiol. 3,4-dihydroxyphenathrene Phthalate Pyruvic acid Acetic acid Formic acid | [87] |
| Pseudomonas aeruginosa RS1 | Maphthalene 1-methylnaphthalen. | [88] |
| Acinetobacter baumannii BJ5 | Benzyl benzoate Butyl octyl phthalate Phenol −2,4-bis(1,1-dimethylethyl) Phenol, 2,4-di-tert-butyl-Ethyl benzoate n-Propyl acetate | [89] |
| Sphingomonas sp. YT1005 | 4-phenanthrenol Protocatechuic acid Phthalic acid 1-hydroxy-2-naphthoic acid 2-methylnaphthalene 2-hydroxy-2-H-benzo[h]chromene-2-carboxylic acid Dihydroxyphenanthrene cis-4,5-pyrene dihydrodiol Salicylic acid trans-2′-carboxybenzalpyruvate | [90] |
| Earthworm Eisenia fetida | Pyrene-4,5-dione Phenanthrene-4-carboxylic acid Phenanthrene-4,5-dicarboxylic acid Phenanthrene-4-carboxylic acid Protocatechuic acid | [91] |
| Klebsiella sp. LZ6 | 4,5-dihydro-phenanthrene Dibenzo-p-dioxin 4-hydroxycinnamate acid | [92] |
| Metabolites | Fathead Minnow LC50 (96 h) | Ames Mutagenicity | |||
|---|---|---|---|---|---|
| Prediction Value: −log (mol/L) | Prediction Value: (mg/L) | Prediction Value: Log10 (mol/L) | Experimental Result | Prediction Result | |
| 1,2-phenanthrenedicarboxylic acid | * | * | 0.86 | * | Mutagenicity Positive |
| 2-carboxybenzaldehyde | 4.30 | 7.49 | 0.29 | * | Mutagenicity Negative |
| 1-hydroxypyrene | 5.45 | 0.77 | 0.76 | * | Mutagenicity Positive |
| Phthalic acid | 3.69 | 34.15 | 0.14 | Mutagenicity Negative | Mutagenicity Negative |
| Benzoic acid | 3.21 | 75.43 | −0.05 | Mutagenicity Negative | Mutagenicity Negative |
| Salicylic acid | 3.34 | 63.62 | −0.08 | * | Mutagenicity Negative |
| 1-hydroxy-2-naphthoic acid | 3.77 | 31.97 | 0.17 | * | Mutagenicity Negative |
| Pyruvic acid | 2.08 | 734.13 | 0.41 | * | Mutagenicity Negative |
| 4,5-dihydroxy pyrene | 5.13 | 1.73 | 0.50 | * | Mutagenicity Negative |
| 4,5-dihydropyrene | 6.33 | 9.50 × 10−2 | 0.98 | * | Mutagenicity Positive |
| n-Propyl acetate | 3.06 | 89.38 | 0.19 | * | Mutagenicity Negative |
| 4-phenanthrene-carboxylic acid | 4.52 | 6.65 | 0.71 | * | Mutagenicity Positive |
| Protocatechuic acid | 3.73 | 28.53 | 0.30 | * | Mutagenicity Negative |
| 1,2-dihydroxynaphthalene | 4.55 | 4.49 | 0.28 | * | Mutagenicity Negative |
| 2,3-dihydroxybenzoate | 3.71 | 29.86 | −0.04 | * | Mutagenicity Negative |
| Dibenzo-p-dioxin | 4.53 | 5.40 | 0.23 | Mutagenicity Negative | Mutagenicity Negative |
| Catechol | 3.81 | 17.19 | 0.29 | Mutagenicity Negative | Mutagenicity Negative |
| 4,5-dihydroxy-4,5-dihydropyrene | 5.03 | 2.21 | 0.15 | Mutagenicity Negative | Mutagenicity Negative |
| 4-phenanthrol | 5.80 | 0.31 | 0.76 | Mutagenicity Positive | Mutagenicity Positive |
| Phenanthrene 4,5-dicarboxylate | * | * | 0.22 | * | Mutagenicity Negative |
| 4-oxa-Pyrene-5-one | 5.01 | 2.18 | 0.22 | Mutagenicity Negative | Mutagenicity Negative |
| 4,5-Dihydroxyphthalate | 3.58 | 51.61 | 0.47 | * | Mutagenicity Negative |
| 3,4-Dihydroxyphenanthrene | 6.05 | 0.19 | 0.60 | * | Mutagenicity Positive |
| Monohydroxy pyrene | 5.45 | 0.77 | 0.76 | * | Mutagenicity Positive |
| 9,10-phenanthrenequinone | 4.30 | 10.47 | 0.52 | Mutagenicity Negative | Mutagenicity Positive |
| Dibutyl-phthalate | 5.30 | 1.40 | 0.18 | Mutagenicity Negative | Mutagenicity Negative |
| Naphthalene | 4.20 | 8.15 | * | Mutagenicity Negative | * |
| 1-methylnaphthalen | 4.32 | 6.74 | * | Mutagenicity Negative | * |
| Benzyl benzoate | 4.86 | 2.92 | −0.05 | * | Mutagenicity Negative |
| Butyl octyl phthalate | 4.70 | 6.68 | 0.03 | * | Mutagenicity Negative |
| Phenanthrene-4,5-dicarboxylic acid | * | * | 0.22 | * | Mutagenicity Negative |
| Naphthalene-1,2-dicarboxylic acid | 3.93 | 25.69 | 0.10 | * | Mutagenicity Negative |
| Primers | Sequences | Probable Functions | References |
|---|---|---|---|
| NidA3 | Forward 5′-CCTGATGCGACGACAATG-3′ | Fluoranthene/pyrene ring-hydroxylating oxygenase, α subunit | [15] |
| Reverse 5′-GCAACCCTAGCCGACTCTT-3′ | |||
| NidA | Forward 5′-TTCCCGAGTACGAGGGATAC-3′ | α Subunit pyrene dioxygenase | [17] |
| Reverse 5′-TCACGTTGATGAACGACAAA-3′ | |||
| NidB2 | * | Pyrene/phenanthrene ring-hydroxylating oxygenase, β subunit | [18] |
| NidB3 | Reverse 5′-GCCGAGCTCGAATTCGGATCCTTAGATCCAGAATGACAG-3′ | Fluoranthene/pyrene ring-hydroxylating oxygenase,β subunit | |
| PdoAB | Forward 5′-GTATCCATGGGCAACGCGGTCGCGGTGGAC-3′ | α Subunit pyrene dioxygenase | [26] |
| Reverse 5′-ACGGATCCTCATCGAGCACCGCCGCGGAACTG-3′ | |||
| PdoA1 | Forward 5′-GGCATATGCAAACGGAAACGACCGA-3′ | α Subunit pyrene dioxygenase | [95] |
| Reverse 5′-GGGATATCTCAAGCACGCCCGCCGAATG-3′ | |||
| PdoA2B2 | Forward 5′-GGCATATGTCTACTGTCGGTAAGAA-3′ | α Subunit pyrene dioxygenase | |
| Reverse 5′-GGAGATCTTAGAAGAAGTTAGCCAG-3′ | |||
| PdoB1 | Forward 5′-GGCATATGAACGCCGTTGCCGTGGA-3′ | Pyrene/phenanthrene ring-hydroxylating oxygenase, β subunit | |
| Reverse 5′-GGGGATCCTACAGGACTACCGACAG-3′ | |||
| PdoA2B2 | A2-Forward 5′-GGCATATGTCTACTGTCGGTAAGAA-3′ | Catalysis of hydroxylation of HMW and LMW polyaromatic hydrocarbons including pyrene. | |
| B2-Reverse 5′-GGAGATCTTAGAAGAAGTTAGCCAG-3′ | |||
| TolC1C2 | Forward 5′-TGAATCAGACCGACACATCAC-3′ | Small subunits of toluene dioxygenase | [62] |
| Reverse 5′-TGTTACGGCGCAACGTATC-3′ | |||
| NahAc | Forward 5′-GCCAAAAGCACCTGA-3′ | Naphthalene dioxygenase | |
| Reverse 5′-TCTTCGTAAGTTCAGTATGCC-3′ | |||
| BphA1 | Forward 5′-GTGCAGGGAGCCCCTGTGAAG-3′ | Large subunit of biphenyl dioxygenase | |
| Reverse 5′-CAGGGCTTGAGCGTGGCCCAGC-3′ | |||
| PdoB2 | Forward 5′-CCGCTGCGAGATGGAGAAC-3′ | β Subunit dioxygenase | [63] |
| Reverse 5′-CGTGAGGGCGGATCTTCTG-3 | |||
| PdoF | Forward 5′-GCACCACCTTCTGACCGTAA-3′ | Putative extradiol dioxygenase | [65] |
| Reverse 5′-TTGGGTTTGAGGTGGGAACC-3′ | |||
| PhdI | Forward 5′-TGACGAAGTGATGGGTGCTC-3′ | 1-Hydroxy-2-naphthoate dioxygenase | |
| Reverse 5′-AGTGCCGTGTATTTCGTCGT-3′ | |||
| NidAB | Forward 5′-CGCGGATCCATGCTGAGCAACGAACTCCGGCAGACCCTCC-3′ | α Subunit pyrene dioxygenase | [96] |
| Reverse 5′-AAAACTGCAGATTCACATGATCAGGGCGAGGTTGTGTGTCATT-3′ | |||
| NidB | Forward 5′-TCGTCACCAACTTCAAGTC-3′ | β Subunit of arene dioxygenase | |
| Reverse 5′-GGTCTGATCAAGCAGCACAA-3′ | |||
| AraC | Forward 5′-GGACTACCTCGGCGATATGA-3′ | Transcriptional regulatory protein, AraC family | |
| Reverse 5′-TGTGGACGTGCTCTCCATAG-3′ | |||
| PdoA2 | Forward 5′-ACGCAGAACTCCACAAGCTC-3′ | Phenanthrene ring-hydroxylating oxygenase, subunit | [97] |
| Reverse 5′-ACTTCCATCGTGTCGTGTGA-3′ | |||
| NidA3B3 | Forward 5′-ACATATGGCGCCTGATGCGACGACAATG-3′ | Fluoranthene/pyrene ring-hydroxylating oxygenase | [98] |
| Reverse 5′-CAAGCTTTTAGATCCAGAATGACAGGTT-3′ | |||
| PhtAb | Forward 5′-ATCGGATCCTTCTTACGAGTTGGGACTGTATCAAGC-3′ | Oxygenase reductase component | [99] |
| Reverse 5′-AGGTCGAGAAAGCTTTTACTTACTCTCCTTTAATAAAGCCAATAG-3′ | |||
| PhtB | Forward 5′-TGCCCTAAGTGTTTGTCCCGGGTCCTATGAGCT-3′ | Phthalate 3,4-dihydrodiol dehydrogenase | |
| Reverse 5′-AGGTCGAGAAAGCTTTTACTTACTCTCCTTTAATAAAGCCAATAG-3′ | |||
| PhtAd | Forward 5′-ATCGGATCCTTCTTACGAGTTGGGACTGTATCAAGC-3′ | Oxygenase reductase component | |
| Reverse 5′-AAGCTTTTACTATATAGGAGCCGGTTGACT-3′ | |||
| PhtAc | Forward 5′-TCATCACCACAGCCAGGATCCGATGGGCGGAGTTATAAA-3′ | Oxygenase ferredoxin component | [100] |
| Reverse 5′-GCATTATGCGGCCGCAAGCTTTCATTCGTCTACGACTTC-3′ | |||
| PhdJ | Forward 5′-5′-CGAGAGAGCATATGGTGCACGT-3′ | trans-2-Carboxylbenzalpyruvate hydratase-aldolase | [101] |
| Reverse 5′-TCCTCAGGATCCGTGGTTCGAGAC-3’ | |||
| NidD | Forward 5′-ATGATCAGCAACCTGA-3′ | Aldehyde dehydrogenase | [102] |
| PhdG | * | Hydratase-aldolase | |
| PhdA | Forward 5′-GGGAATTCCATATGTCGGTAGTCAGCGGGGAT-3′ | α and β subunits of other ring-hydroxylating dioxygenases | [103] |
| Reverse 5′-CCGGAATTCGGTCGCAACTCATAAGACAGC-3′ | |||
| PhdB | Forward 5′-CCGGAAT TCAAGGAGATATACATATGC TGAC TAC TG T TGACGAGAATC-3′ | ||
| Reverse 5′-CGCGGATCCAGATCTGCCTGCGGGCTAGAAG AAGAACGC-3′ | |||
| MT1743 | * | Catechol O-methyltransferase |
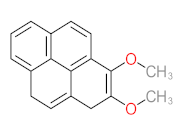
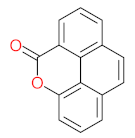
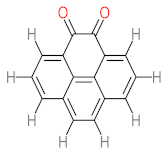
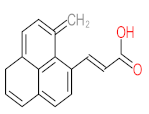
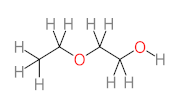 |  |  | 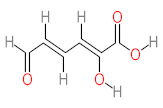 |  | |||||
| P1 | MW = 90.12 | P2 | MW = 142.15 | P3 | MW = 142.11 | P4 | MW = 141.10 | P5 | MW = 250.29 |
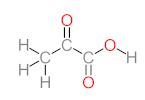 |  | 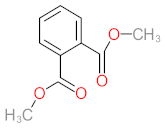 | 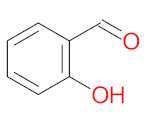 | 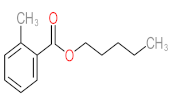 | |||||
| P6 | MW = 88.06 | P7 | MW = 166.13 | P8 | MW = 194.18 | P9 | MW = 122.12 | P10 | MW = 206.28 |
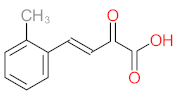 | 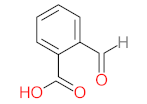 | 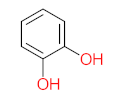 | 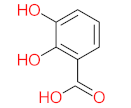 | 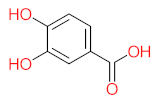 | |||||
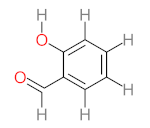
| P11 | MW = 190.19 | P12 | MW = 150.13 | P13 | MW = 110.11 | P14 | MW = 154.12 | P15 | MW = 154.12 |
 | 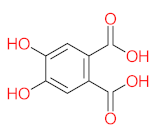 | 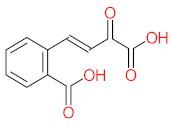 | 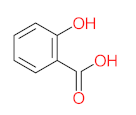 |  | |||||
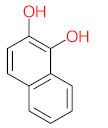
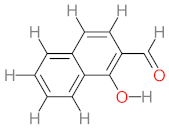
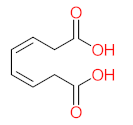
| P16 | MW = 232.23 | P17 | MW = 198.13 | P18 | MW = 220.18 | P19 | MW = 138.12 | P20 | MW = 122.12 |
 |  |  |  |  | |||||
| P21 | MW = 170.25 | P22 | MW = 160.17 | P23 | MW = 172.18 | P24 | MW = 200.15 | P25 | MW = 170.16 |
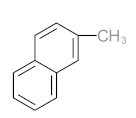 | 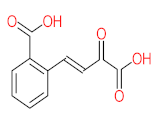 |  | 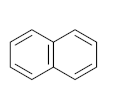 |  | |||||
| P26 | MW = 142.20 | P27 | MW = 220.18 | P28 | MW= 178.23 | P29 | MW = 128.17 | P30 | MW = 302.24 |
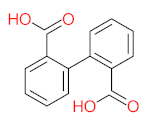 | 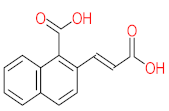 | 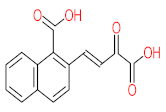 | 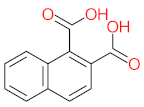 | 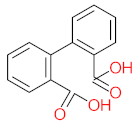 | |||||
| P31 | MW = 242.23 | P32 | MW = 242.23 | P33 | MW = 270.24 | P34 | MW = 216.19 | P35 | MW = 242.23 |
 |  | 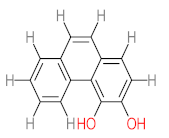 | 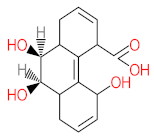 | 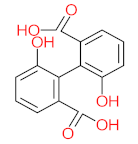 | |||||
| P36 | MW = 244.24 | P37 | MW = 188.18 | P38 | MW = 210.23 | P39 | MW = 278.30 | P40 | MW = 274.23 |
 | 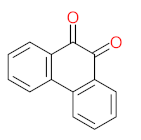 |  | 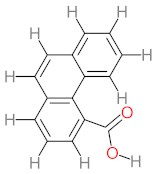 | 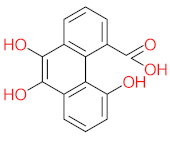 | |||||
| P41 | MW = 216.19 | P42 | MW = 208.21 | P43 | MW = 214.26 | P44 | MW = 222.24 | P45 | MW = 270.24 |
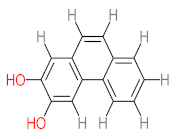 |  | 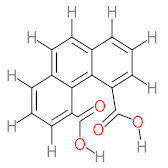 | 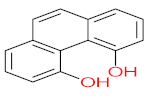 | 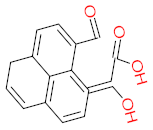 | |||||
| P46 | MW = 210.23 | P47 | MW = 266.25 | P48 | MW = 266.25 | P49 | MW = 210.23 | P50 | MW = 267.27 |
 |  | 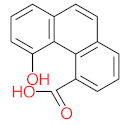 | 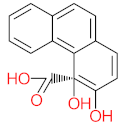 | 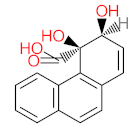 | |||||
| P51 | MW = 178.23 | P52 | MW = 194.23 | P53 | MW = 238.24 | P54 | MW = 255.25 | P55 | MW = 256.25 |
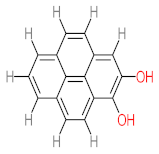 |  | 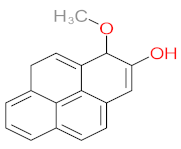 |  | 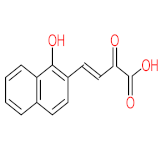 | |||||
| P56 | MW = 234.2 | P57 | MW = 220.27 | P58 | MW = 250.2 | P59 | MW = 241.22 | P60 | MW = 242.23 |
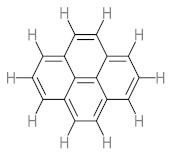 | 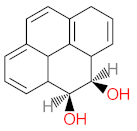 | 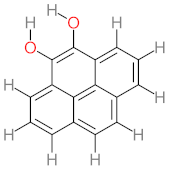 | 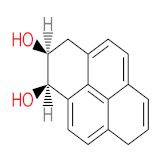 |  | |||||
| P61 | MW = 202 | P62 | MW = 238.2 | P63 | MW = 234.2 | P64 | MW = 238.2 | P65 | MW = 270.28 |
 |  |  |  |  | |||||
| P66 | MW = 264.32 | P67 | MW = 220.22 | P68 | MW = 236.26 | P69 | MW = 218.25 | P70 | MW = 249.28 |
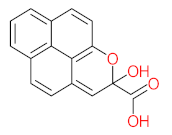 |  |  | 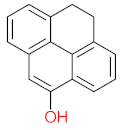 | 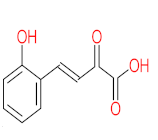 | |||||
| P71 | MW = 266.25 | P72 | MW = 208.25 | P73 | MW = 122.12 | P74 | MW = 218.25 | P75 | MW = 192.17 |
| Organism | Active Enzyme/Gene | Proposed Pathways | References |
|---|---|---|---|
| Mycobacterium vanbaalenii PYR-1 | * | [18] | |
| Mycobacterium vanbaalenii PYR-1 | PhdF, PhdG, PhdI, PhdJ, NidA3, NidAB, NidD, PhtAa, PhtB, and PhtAc | [22] | |
| Mycobacterium vanbaalenii PYR-1 | NidAB, PhdE, PhtC, PdoA2B2, PhdF, PhdG, NidD, PhdJ, PhtAaB, PhtB, and PcaGH | [24] | |
| Mycobacterium sp. KMS | |||
| Coriolopis byrsina APC5 | * | ||
| Mycobacterium spp. PO1 and PO2 | NidA, PhdA, and NidA3 | [28] | |
| Mycobacterium sp. flavescens PYR-1 | * | [43] | |
| Mycobacterium sp. A1-PYR | Monooxygenases and Dioxygenases | [45] | |
| Many bacterial strains including Mycobacterium | * | [50] | |
| Mycobacterium aromativorans Strain JS19b1 | * | [70] | |
| Mycobacterium vanbaalenii PYR-1 | * | [71] | |
| Leclerciaadecarboxylata PS4040 | * | [73] | |
| Coriolopsis byrsina strain APC5 | laccase, LiP and MnP | [75] | |
| Halophilic Hortaea sp. B15 | Dioxygenase | [77] | |
| Pseudomonas sp. ISTPY2 | * | [80] | |
| Achromobacterxylosoxidans PY4 strain | 4-hydroxyphenylpyruvate dioxygenase and homogentisate 1,2-dioxygenase | [86] | |
| Enterobacter sp. MM087 (KT933254) | * | [87] | |
| Consortium microbes | NidA, NidA3, and PAH-RHDα-GP) | [90] | |
| Earthworm (Eisenia fetida) | Peroxidase, Laccase, Invertase, Glucosidase, Phosphatase, Phytase, Urease, Hydrolase, Chitinase, Nitrogenase, Aminopeptidase, and Arylsulfatase. | [91] | |
| Rhodococcus sp. UW1 | * | [104] | |
| Kocuria flava and Rhodococcus pyridinivorans | catechol 2,3-dioxygenase, dehydrogenase, and peroxidase | [105] | |
| Ruegeria pomeroyi DSS-3, Dinoroseobacter shibae DFL 12, and Pelagibaca bermudensis HTCC2601 | PahE | [106] | |
| Rhodococcus sp. | MboI, RsaI, and AluI. | [107] | |
| Tolypocladium sp. strain CBMAI 1346and Xylaria sp. CBMAI 1464 | Many enzymes for example (phenol 2-monooxygenase, epoxide hydrolases, and some dioxygenases) | [108] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Qutob, M.; Rafatullah, M.; Muhammad, S.A.; Alosaimi, A.M.; Alorfi, H.S.; Hussein, M.A. A Review of Pyrene Bioremediation Using Mycobacterium Strains in a Different Matrix. Fermentation 2022, 8, 260. https://doi.org/10.3390/fermentation8060260
Qutob M, Rafatullah M, Muhammad SA, Alosaimi AM, Alorfi HS, Hussein MA. A Review of Pyrene Bioremediation Using Mycobacterium Strains in a Different Matrix. Fermentation. 2022; 8(6):260. https://doi.org/10.3390/fermentation8060260
Chicago/Turabian StyleQutob, Mohammad, Mohd Rafatullah, Syahidah Akmal Muhammad, Abeer M. Alosaimi, Hajer S. Alorfi, and Mahmoud A. Hussein. 2022. "A Review of Pyrene Bioremediation Using Mycobacterium Strains in a Different Matrix" Fermentation 8, no. 6: 260. https://doi.org/10.3390/fermentation8060260








