Improvement of L-asparaginase, an Anticancer Agent of Aspergillus arenarioides EAN603 in Submerged Fermentation Using a Radial Basis Function Neural Network with a Specific Genetic Algorithm (RBFNN-GA)
Abstract
:1. Introduction
2. Materials and Methods
2.1. Fungal Strains and Cultural Conditions
2.2. Semi-Quantitative Screening for L-asparaginase Production
2.3. Production of L-asparaginase in Submerged Fermentation
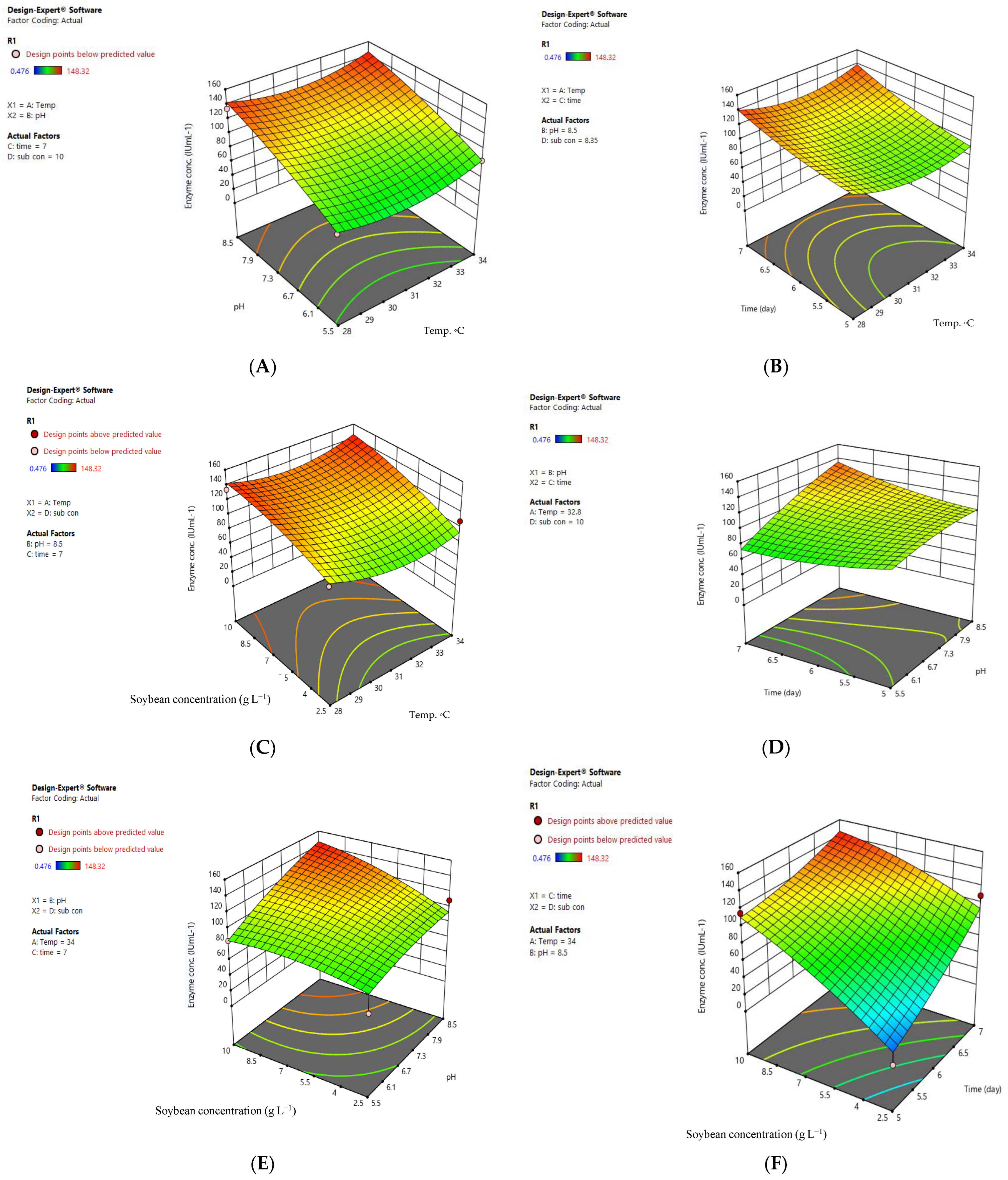
2.4. Optimizing L-asparaginase Production by A. arenarioides EAN603 Using a Box–Behnken Design
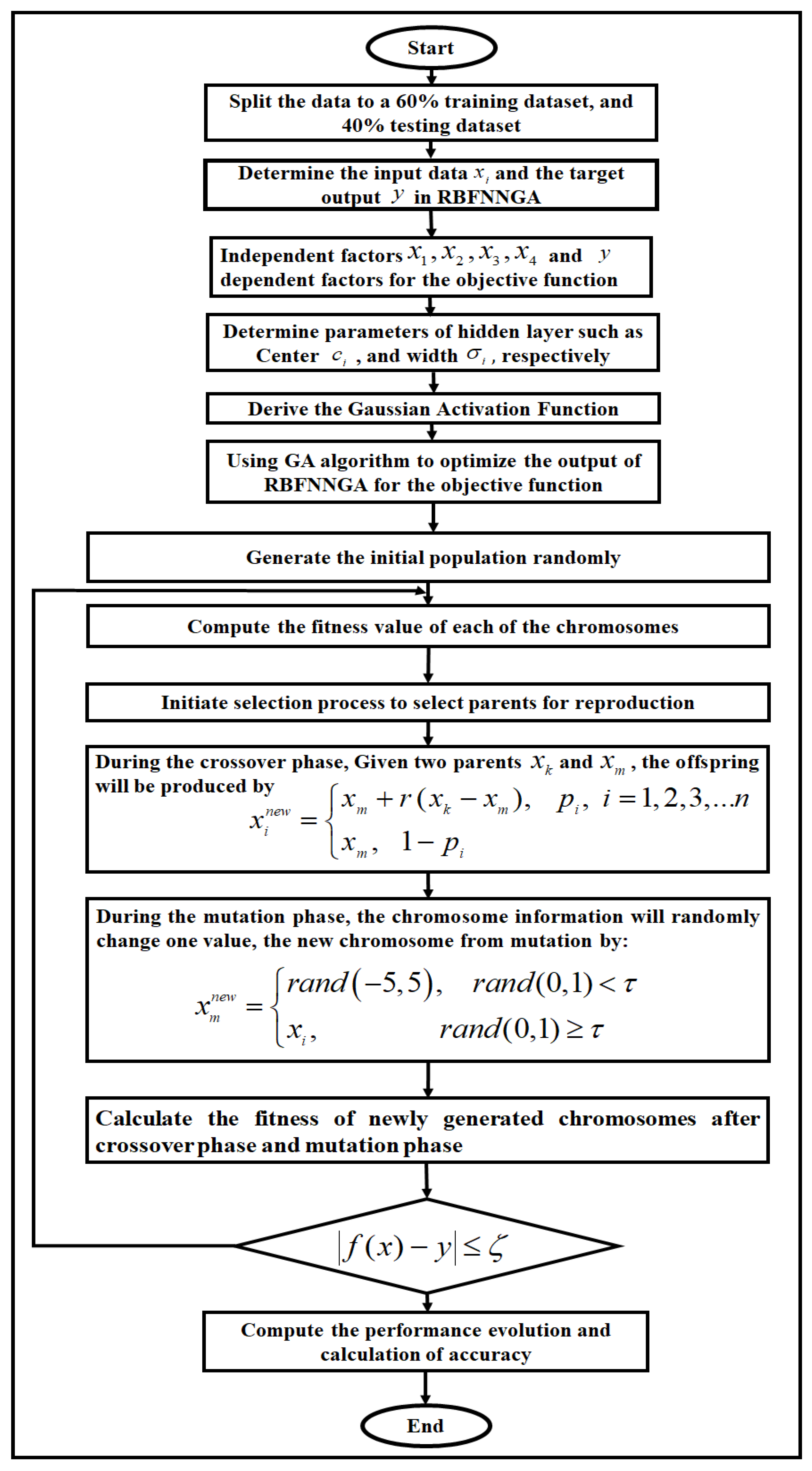
2.5. Radial Basis Function Neural Network with a Specific Genetic Algorithm (RBFNNGA)
2.6. Purification and Characterization
3. Results and Discussion
3.1. L-asparaginase Production by Fungal Strains
3.2. Optimal Conditions for Producing L-asparaginase by A. arenarioides EAN603
3.3. Purification and Stability
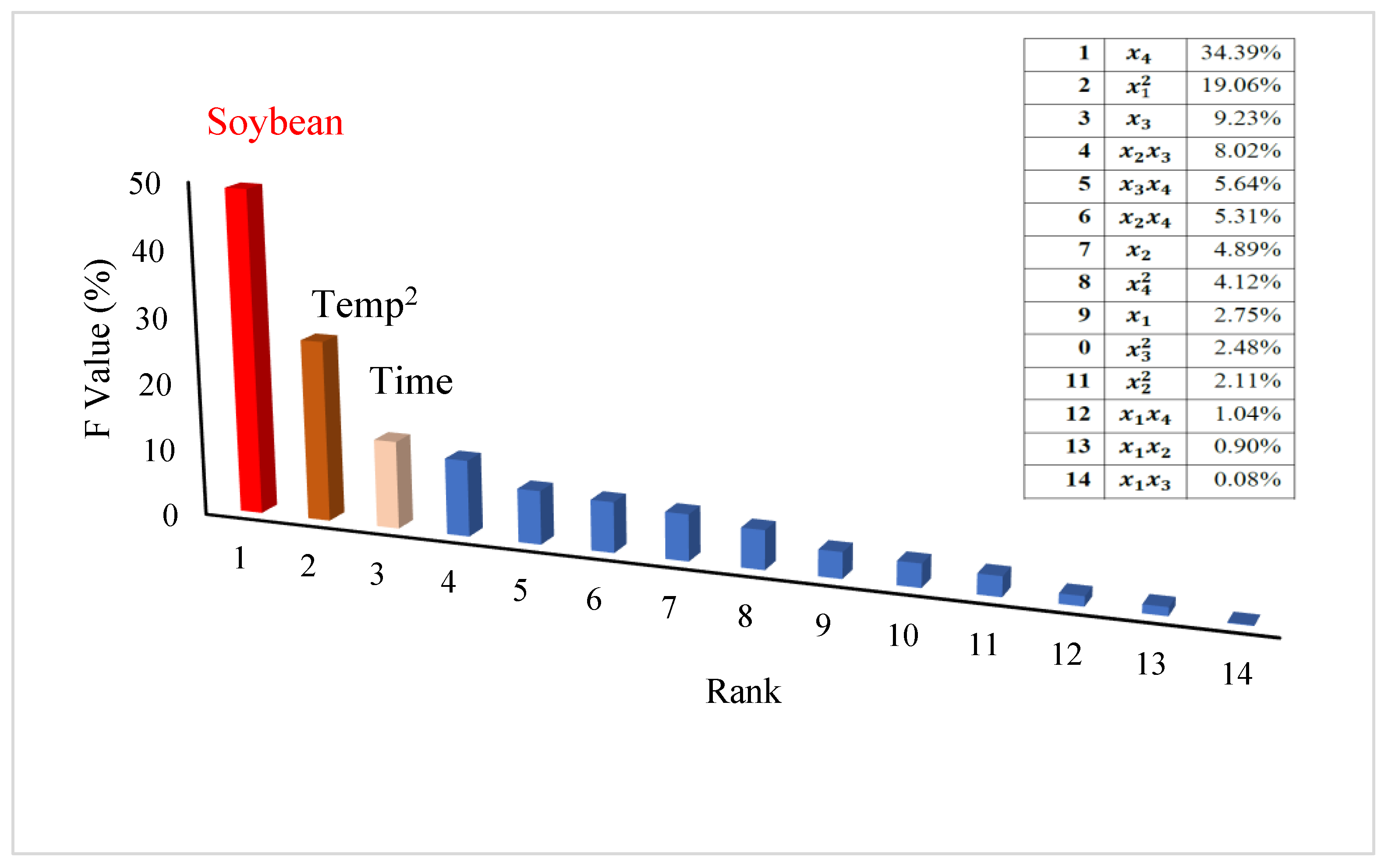
3.4. Prediction Models Using Machine Learning
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Qeshmi, F.I.; Homaei, A.; Fernandes, P.; Javadpour, S. Marine microbial L-asparaginase: Biochemistry, molecular approaches and applications in tumor therapy and in food industry. Microbiol. Res. 2018, 208, 99–112. [Google Scholar] [CrossRef] [PubMed]
- Cachumba, J.J.M.; Antunes, F.A.F.; Peres, G.F.D.; Brumano, L.P.; Santos, J.C.D.; Da Silva, S.S. Current applications and different approaches for microbial L-asparaginase production. Braz. J. Microbiol. 2016, 47, 77–85. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Baskar, G.; Renganathan, S. Production of L-asparaginase from natural substrates by Aspergillus terreus MTCC 1782: Effect of substrate, supplementary nitrogen source and L-asparagine. Int. J. Chem. React. Eng. 2009, 7. [Google Scholar] [CrossRef]
- El-Naggar, N.; Moawad, H.; Abdelwahed, N.A. Optimization of fermentation conditions for enhancing extracellular production of L-asparaginase, an anti-leukemic agent, by newly isolated Streptomyces brollosae NEAE-115 using solid state fermentation. Ann. Microbiol. 2017, 67, 1–15. [Google Scholar] [CrossRef]
- Meghavarnam, A.K.; Janakiraman, S. Solid state fermentation: An effective fermentation strategy for the production of L-asparaginase by Fusarium culmorum (ASP-87). Biocatal. Agric. Biotechnol. 2017, 11, 124–130. [Google Scholar] [CrossRef]
- Vala, A.K.; Sachaniya, B.; Dudhagara, D.; Panseriya, H.Z.; Gosai, H.; Rawal, R.; Dave, B.P. Characterization of L-asparaginase from marine-derived Aspergillus niger AKV-MKBU, its antiproliferative activity and bench scale production using industrial waste. Int. J. Biol. Macromol. 2018, 108, 41–46. [Google Scholar] [CrossRef]
- Fan, B.; Dewapriya, P.; Li, F.; Grauso, L.; Blümel, M.; Mangoni, A.; Tasdemir, D. Pyrenosetin D, a new pentacyclic decalinoyltetramic acid derivative from the algicolous fungus pyrenochaetopsis sp. FVE-087. Mar. Drugs 2020, 18, 281. [Google Scholar] [CrossRef]
- El-Gendy, M.M.A.A.; Awad, M.F.; El-Shenawy, F.S.; El-Bondkly, A.M.A. Production, purification, characterization, antioxidant and antiproliferative activities of extracellular L-asparaginase produced by Fusarium equiseti AHMF4. Saudi J. Biol. Sci. 2021, 28, 2540–2548. [Google Scholar] [CrossRef]
- Krishnapura, P.R.; Belur, P.D. Partial purification and characterization of L-asparaginase from an endophytic Talaromyces pinophilus isolated from the rhizomes of Curcuma amada. J. Mol. Catal. B Enzym. 2016, 124, 83–91. [Google Scholar] [CrossRef]
- Baskar, G.; Sree, N.S. Synthesis, characterization and anticancer activity of β-cyclodextrin-Asparaginase nanobiocomposite on prostate and lymphoma cancer cells. J. Drug Deliv. Sci. Technol. 2020, 55, 101417. [Google Scholar] [CrossRef]
- Paul, V.; Tiwary, B.N. An investigation on the acrylamide mitigation potential of l-asparaginase from Aspergillus terreus BV-C strain. Biocatal. Agric. Biotechnol. 2020, 27, 101677. [Google Scholar] [CrossRef]
- Doriya, K.; Kumar, D.S. Solid state fermentation of mixed substrate for l-asparaginase production using tray and in-house designed rotary bioreactor. Biochem. Eng. J. 2018, 138, 188–196. [Google Scholar] [CrossRef]
- Moubasher, H.A.; Balbool, B.A.; Helmy, Y.A.; Alsuhaibani, A.M.; Atta, A.A.; Sheir, D.H.; Abdel-Azeem, A.M. Insights into Asparaginase from Endophytic Fungus Lasiodiplodia theobromae: Purification, Characterization and Antileukemic Activity. Int. J. Environ. Res. Public Health 2022, 19, 680. [Google Scholar] [CrossRef] [PubMed]
- Da Cunha, M.C.; Aguilar, J.G.D.S.; Orrillo Lindo, S.M.D.R.; de Castro, R.J.S.; Sato, H.H. L-asparaginase from Aspergillus oryzae spp.: Effects of production process and biochemical parameters. Prep. Biochem. Biotechnol. 2022, 52, 253–263. [Google Scholar] [CrossRef]
- Noman, E.; Al-Gheethi, T.; Mohamed, B.R.; Almoheer, R.; Al-Shaorgani, N. First Report of Six New Environmental Fungal Strains in Malaysia on a New Culture Medium (EVA Medium): Morpho-logical, Molecular, and Microstructure Characteristics. J. Sustain. 2022. revised. [Google Scholar]
- Al-Gheethi, A.; Noman, E.; Mohamed, R.M.S.R.; Talip, B.; Vo, D.V.N.; Algaifi, H.A. Cephalexin removal by a novel Cu–Zn bionanocomposite biosynthesized in secondary metabolic products of Aspergillus arenarioides EAN603 with pumpkin peels medium: Optimization, kinetic and artificial neural network models. J. Hazard. Mater. 2021, 419, 126500. [Google Scholar] [CrossRef]
- Sharma, D.; Mishra, A. L-asparaginase production in solid-state fermentation using Aspergillus niger: Process modeling by artificial neural network approach. Prep. Biochem. Biotechnol. 2022, 52, 549–560. [Google Scholar] [CrossRef]
- Alzaeemi, S.A.; Sathasivam, S. Artificial immune system in doing 2-satisfiability based reverse analysis method via a radial basis function neural network. Processes 2020, 8, 1295. [Google Scholar] [CrossRef]
- Sathasivam, S.; Alzaeemi, S.A.; Ismail, M.T.; VaniPachala, V. Palm oil price forecasting in Malaysia using 2 satisfiability based reverse analysis method via radial basis function neural network. Solid State Technol. 2020, 63, 334–339. [Google Scholar]
- Li, T.; Liu, X.; Lin, Z.; Morrison, R. Ensemble offshore Wind Turbine Power Curve modelling–An integration of Isolation Forest, fast Radial Basis Function Neural Network, and metaheuristic algorithm. Energy 2022, 239, 122340. [Google Scholar] [CrossRef]
- Katoch, S.; Chauhan, S.S.; Kumar, V. A review on genetic algorithm: Past, present, and future. Multimed. Tools Appl. 2021, 80, 8091–8126. [Google Scholar] [CrossRef]
- Zhang, Q.; Abdullah, A.R.; Chong, C.W.; Ali, M.H. A study on regional gdp forecasting analysis based on radial basis function neural network with genetic algorithm (RBFNN-GA) for shandong economy. Comput. Intell. Neurosci. 2022, 2022, 8235308. [Google Scholar] [CrossRef]
- Gulati, R.; Saxena, R.K.; Gupta, R. A rapid plate assay for screening l-asparaginase producing micro-organisms. Lett. Appl. Microbiol. 1997, 24, 23–26. [Google Scholar] [CrossRef] [PubMed]
- Bradford, M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef] [PubMed]
- Mahajan, R.V.; Saran, S.; Saxena, R.K.; Srivastava, A.K. A rapid, efficient and sensitive plate assay for detection and screening of l-asparaginase-producing microorganisms. FEMS Microbiol. Lett. 2013, 341, 122–126. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hatamzadeh, S.; Rahnama, K.; Nasrollahnejad, S.; Fotouhifar, K.B.; Hemmati, K.; White, J.F.; Taliei, F. Isolation and identification of L-asparaginase-producing endophytic fungi from the Asteraceae family plant species of Iran. PeerJ 2020, 8, e8309. [Google Scholar] [CrossRef] [PubMed]
- Noman, E.; Al-Gheethi, A.A.; Talip, B.A.; Mohamed, R.; Kassim, A.H. Oxidative enzymes from newly local strain Aspergillus iizukae EAN605 using pumpkin peels as a production substrate: Optimized production, characterization, application and techno-economic analysis. J. Hazard. Mater. 2020, 386, 121954. [Google Scholar] [CrossRef]
- Burgess, R.R. Protein precipitation techniques. Methods Enzymol. 2009, 463, 331–342. [Google Scholar]
- Zhang, X.N.; Cheng, Q.; Chen, J.; Lam, A.T.; Lu, Y.; Dai, Z.; Pei, H.; Evdokimov, N.M.; Louie, S.G.; Zhang, Y. A ribose-functionalized NAD+ with unexpected high activity and selectivity for protein poly-ADP-ribosylation. Nat. Commun. 2019, 10, 4196. [Google Scholar] [CrossRef] [Green Version]
- Hamed, M.; Osman, A.A.; Ateş, M. Semi-quantitative detection for L-asparaginase producing fungi and the impact of carbon and nitrogen sources on enzyme activity. Biorxiv 2021. [Google Scholar] [CrossRef]
- Shanthipriya, A.; Rao, V.K.; Girisham, S.; Reddy, S.M. Factors influencing L-asparaginase production by three thermophilic caprophilous fungi. Res. Pharm. Sci. 2015, 5, 1242–1248. [Google Scholar]
- Vimal, A.; Kumar, A. Optimized Production of Medically Significant Enzyme L-Asparaginase Under Submerged and Solid-State Fermentation from Agricultural Wastes. Curr. Microbiol. 2022, 79, 394. [Google Scholar] [CrossRef] [PubMed]

| Name of Fungi | L-asparaginase Activity Diameter (mm) | Colony Diameter (mm) | Zone Index |
|---|---|---|---|
| P. lilacinum EAN601 | 24.0 | 22 | 1.09 |
| P. album EAN602 | ND | 18 | ND |
| A. arenarioides EAN603 | 37.2 | 18 | 2.1 |
| P. pedernalense EAN604 | 21.4 | 16.2 | 1.3 |
| A. iizukae EAN605 | 33.5 | 18 | 1.86 |
| P. brasiliense EAN202 | 30.1 | 19 | 1.58 |
| Fungal Strain | Average O.D | Concentration of Ammonium in the Final Solution (mM) | Crude Enzyme (IU mL−1) |
|---|---|---|---|
| P. lilacinum EAN601 | 0.307 | 0.091 | 45.5 |
| A. arenarioides EAN603 | 1.084 | 0.336 | 168.2 |
| P. pedernalense EAN604 | 0.150 | 0.042 | 21 |
| A. iizukae EAN605 | 0.158 | 0.045 | 22.5 |
| P. brasiliense EAN202 | 0.199 | 0.056 | 28 |
| Positive control | 0.580 | 0.181 | 90.6 |
| Supernatant | Precipitation | |||
|---|---|---|---|---|
| (NH₄)₂SO₄% | Mean O.D | Crude Enzyme (IU mL−1) | Mean O.D | Crude Enzyme (IU mL−1) |
| 20 | 0.845 | 134.2 | 0.254 | 37.6 |
| 40 | 0.751 | 119.3 | 0.457 | 70.6 |
| 60 | 0.359 | 55.44 | 0.873 | 138.7 |
| 80 | 0.596 | 93 | 0.684 | 106.7 |
| Supernatant | 0.914 | 142.5 | N.D | N.D |
| Positive control | 0.899 | 140.18 | N.D | N.D |
| Purification Procedure | Enzyme Activity (IU) | Protein (mg) | Specific Activity (IU mg−1) | Fold Purification | Yield (%) |
|---|---|---|---|---|---|
| Cell-Free Extract | 249 | 117 | 11.29 | 1.0 | 100 |
| Crude enzyme | 172 | 79 | 20.9 | 2.08 | 74 |
| The Final State after the RBFNNGA | Accuracy% | R |
|---|---|---|
| 91.67 | 0.94 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Alzaeemi, S.A.; Noman, E.A.; Al-shaibani, M.M.; Al-Gheethi, A.; Mohamed, R.M.S.R.; Almoheer, R.; Seif, M.; Tay, K.G.; Zin, N.M.; El Enshasy, H.A. Improvement of L-asparaginase, an Anticancer Agent of Aspergillus arenarioides EAN603 in Submerged Fermentation Using a Radial Basis Function Neural Network with a Specific Genetic Algorithm (RBFNN-GA). Fermentation 2023, 9, 200. https://doi.org/10.3390/fermentation9030200
Alzaeemi SA, Noman EA, Al-shaibani MM, Al-Gheethi A, Mohamed RMSR, Almoheer R, Seif M, Tay KG, Zin NM, El Enshasy HA. Improvement of L-asparaginase, an Anticancer Agent of Aspergillus arenarioides EAN603 in Submerged Fermentation Using a Radial Basis Function Neural Network with a Specific Genetic Algorithm (RBFNN-GA). Fermentation. 2023; 9(3):200. https://doi.org/10.3390/fermentation9030200
Chicago/Turabian StyleAlzaeemi, Shehab Abdulhabib, Efaq Ali Noman, Muhanna Mohammed Al-shaibani, Adel Al-Gheethi, Radin Maya Saphira Radin Mohamed, Reyad Almoheer, Mubarak Seif, Kim Gaik Tay, Noraziah Mohamad Zin, and Hesham Ali El Enshasy. 2023. "Improvement of L-asparaginase, an Anticancer Agent of Aspergillus arenarioides EAN603 in Submerged Fermentation Using a Radial Basis Function Neural Network with a Specific Genetic Algorithm (RBFNN-GA)" Fermentation 9, no. 3: 200. https://doi.org/10.3390/fermentation9030200









