3.1. Preparation and Study of Fermented Beverage Bases
The processes of obtaining fermented tea beverages have their own technological features, which depend on cultural traditions, the raw materials used, the concentration of tea and sugar, and the duration of fermentation [
72]. However, the basic technological steps are common and consist of brewing the tea, adding sucrose or other carbohydrate sources, filtering and cooling to room temperature, adding SCOBY and/or starter fluid (fermented kombucha from a previous batch), and fermentation [
72,
73]. The fermentation process of sweetened tea begins after the addition of SCOBY and/or starter liquids. Under the action of enzymes formed in yeast cells, hydrolysis of sucrose to glucose and fructose occurs at room temperature, which are subsequently converted to ethanol and carbon dioxide as a result of alcoholic fermentation [
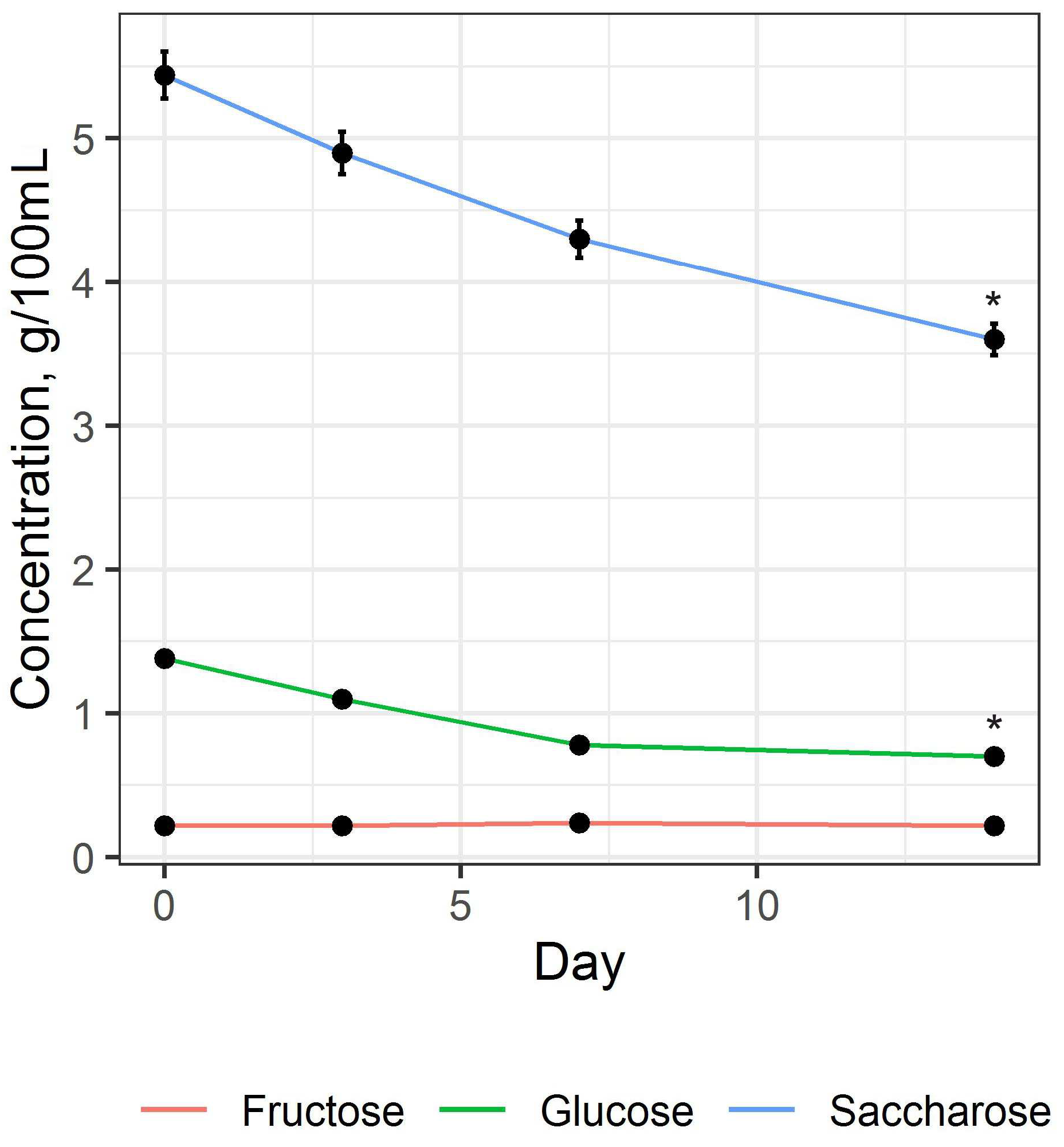
74]. The change in the profile of carbohydrates during fermentation is shown in
Figure 2.
Figure 2 shows that during the fermentation process for 14 days, there is a decrease in saccharose content by an average of 45% (H = 8.897,
p = 0.045). The concentration of glucose also significantly decreased (H = 9.051,
p = 0.029), but the change in the fructose concentration had only an insignificant tendency (H = 0.723,
p = 0.868), indicating the different metabolism of these carbohydrates by SCOBY microorganisms. The change in the concentration of glucose and fructose during fermentation depends on the composition of the SCOBY [
5]. Thus, acetic acid bacteria assimilate fructose and glucose to produce various organic acids and form a cellulose film [
74], while the assimilation of glucose and fructose is not the same [
75]. Yeast mainly assimilates glucose with the formation of carbon dioxide and ethanol; however, yeast of the genus
Saccharomyces prefers glucose, whereas certain yeast of the genus
Zygosaccharomyces prefer fructose. In our study, the fermentation process was continued for 14 days, after which the fermented base contained residual sugars. However, there are alternative methods. For example, prolonged fermentation for more than 30 days allows the use of a fermented base with 0% sugar content, due to which its caloric value is lower than that of a base containing sugar. For the preparation of the beverages, the base is necessarily diluted with juices, herbal infusions, etc., while for sweetening, stevia extract or other sweeteners can be used. In our study, the criterion for the completion of the primary fermentation process was the pH value. Residual sugar in the studied base gave a sour-sweet taste and reduced the perception of the sour taste of the beverages. During fermentation, due to the accumulation of organic acids, mainly acetic acid, the pH value decreases, and the acidity increases [
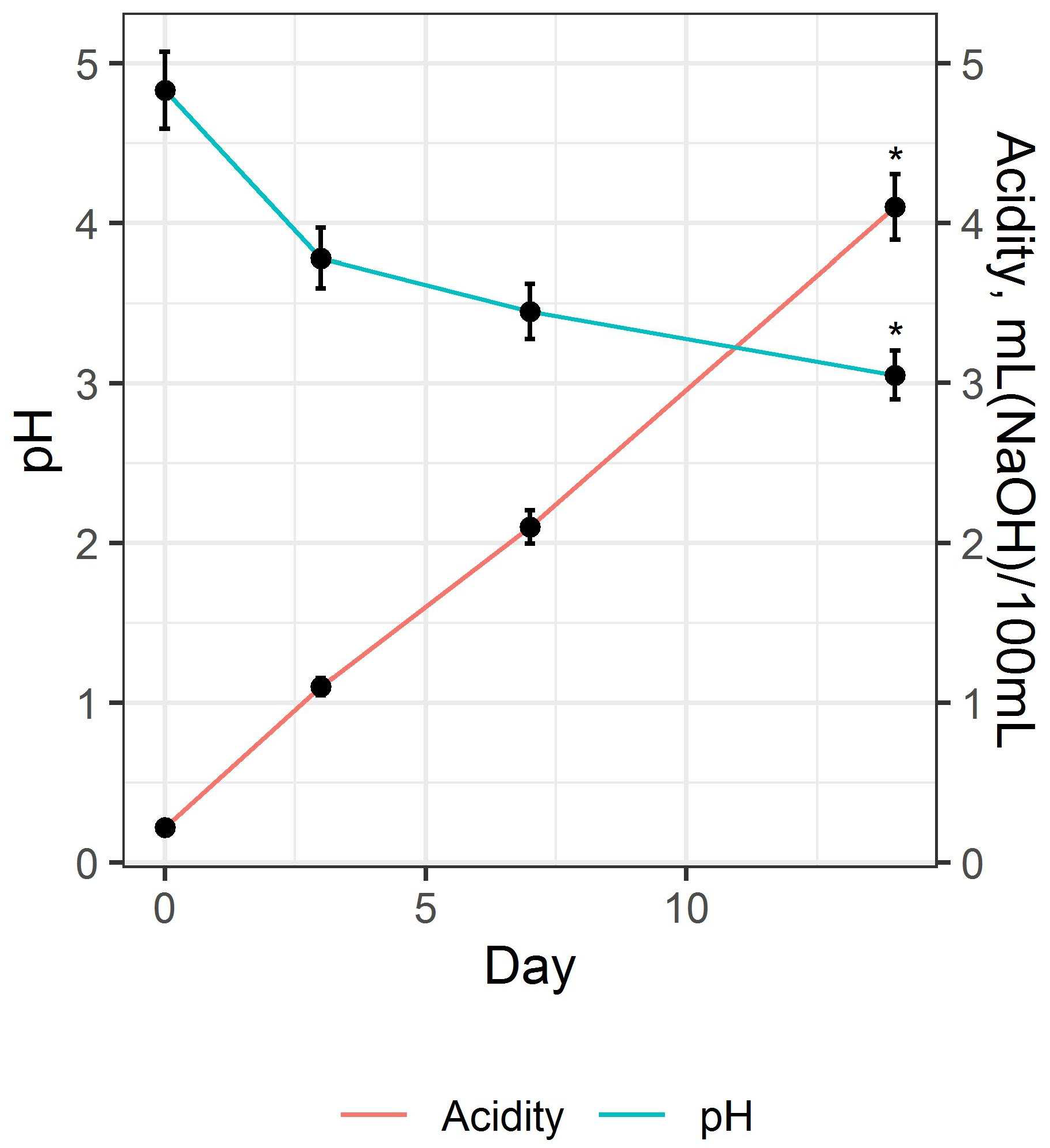
74]. Control of these indicators allows for the preparation of a beverage with the desired organoleptic characteristics. The results of changes in pH and acidity during the fermentation of the beverage base are shown in
Figure 3.
As shown in
Figure 3, the rate of change in the pH and acidity of the beverage base during fermentation is non-linear. At the beginning of the fermentation process (up to 3 days), there is an intense decrease in pH values with a subsequent decrease in rate (H = 13.5,
p = 0.0091). At the same time, the change in acidity is reversed: during the first 7 days, acidity increases slightly, then increases sharply, which is associated with the accumulation of organic acids in the substrate, in particular acetic acid (
Figure 4). It should be noted that the sharp increase in acidity (H = 13.5,
p = 0.0091) does not have a strong correlation (r = −0.864,
p < 0.001) correlate with the change in pH value, which may be due to the manifestation of buffering properties of the fermented beverages [
76]. Fermentation was continued until reaching pH values of 3.3 ± 0.3.
Due to alcoholic fermentation, ethanol was formed during the first stages of the fermentation process [
74]. It was found that the ethanol content on day 7 of fermentation was 0.12%, and on day 14, it did not exceed 0.22%. When selecting a tea substrate for kombucha, the dynamics of ethanol accumulation during fermentation should be taken into account [
77,
78].
The fermentation process begins with the hydrolysis of sucrose into fructose and glucose by enzymes produced in the yeast cells. Part of the glucose and fructose is then used by the yeast to produce ethanol and carbon dioxide [
11,
79]. Another part of the fructose and glucose and the resulting ethanol is used by acetic acid bacteria to produce cellulose and organic acids, of which acetic acid is the main one [
11,
79]. In addition to acetic acid, other organic acids, such as gluconic acid, lactic acid, malic acid, citric acid, tartaric acid, and others, are accumulated in much smaller amounts during fermentation [
80]. Regulation of the concentration and type of organic acids formed is achieved by varying the component composition of the tea base and carbohydrate sources [
81,
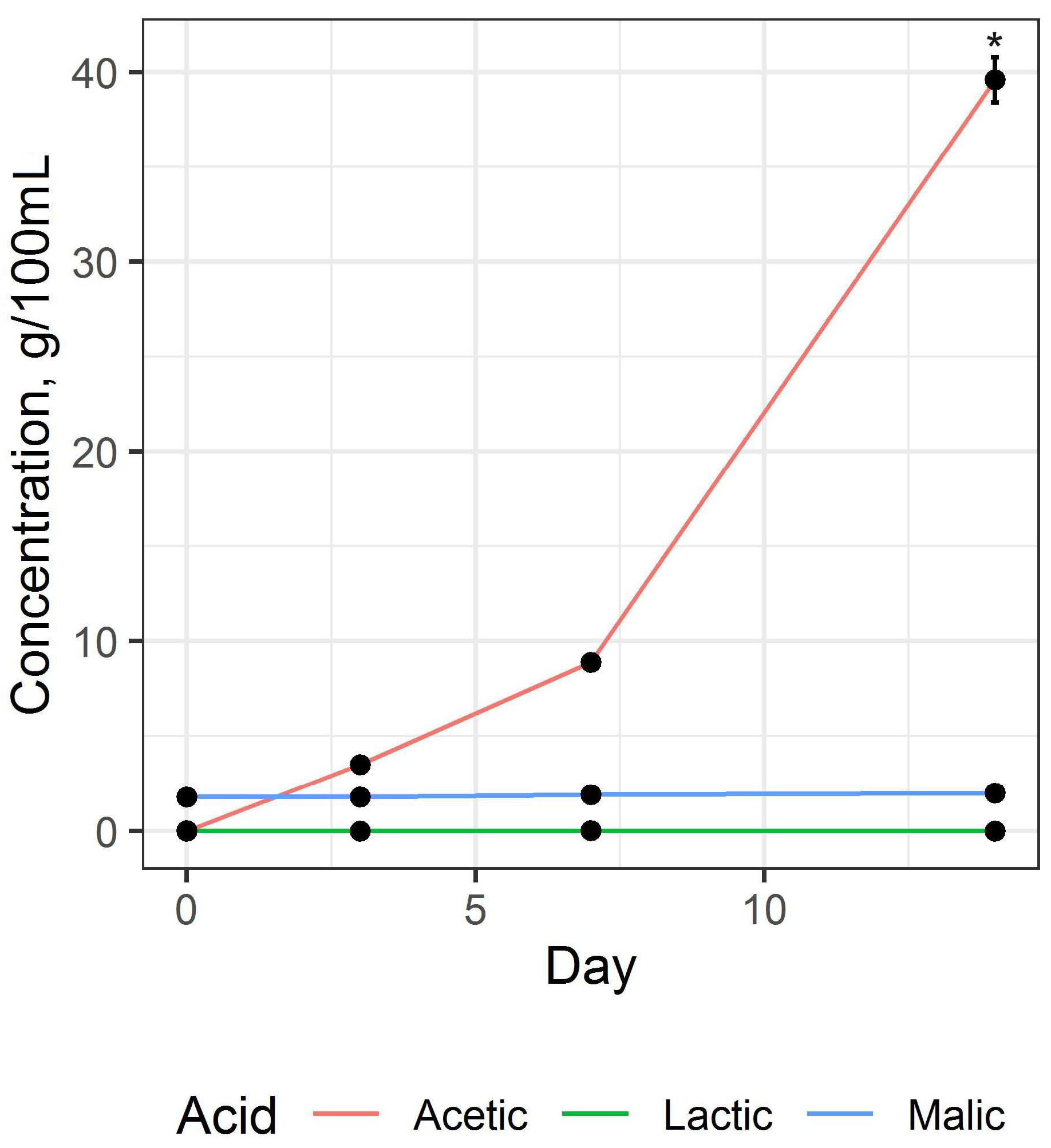
82]. In this work, to obtain a fermented beverage base, traditional raw materials were used: black tea and sugar; therefore, the main organic acid formed in the process of fermentation was acetic acid (
Figure 4).
It was found that the accumulation of acetic acid in the first three days of fermentation occurs slowly, and subsequently, the rate of accumulation increases (H = 10.531,
p = 0.015). The character of the curve of acetic acid accumulation correlates with the change in the acidity of the base during fermentation (
Figure 3). In the period from the 10th to the 14th day, there is an accumulation of acetic acid, which negatively affects the organoleptic properties of the base due to the appearance of a sharp smell and sour taste. Accumulation of other major organic acids was much lower and was statistically significant for lactic acid (H = 9.581,
p = 0.023) and insignificant for malic acid (H = 1.049,
p = 0.789).
Consumption of beverages with a high organic acid content is unsafe and poses potential risks to consumers. To eliminate the negative influence of the temperature factor on the technological process and the formation of undesirable fermentation products in fermenters, they were maintained at a temperature of 25 ± 1 °C.
The popularity of fermented tea beverages is due not only to their original taste and odor but also to their health-promoting properties, in particular, their antioxidant properties. The biological activity of SCOBY is primarily related to the chemical composition of the tea itself used in the preparation of the beverage [
6]. Flavonoids such as flavanols (flavan-3-ols), flavonols, flavones, flavanones, and anthocyanidins are the main components of tea leaves, which together account for up to 30% of the dry weight of tea leaves [
83]. The composition of polyphenolic compounds and their content depend on the type of tea, growing conditions, processing and storage technology, etc. [
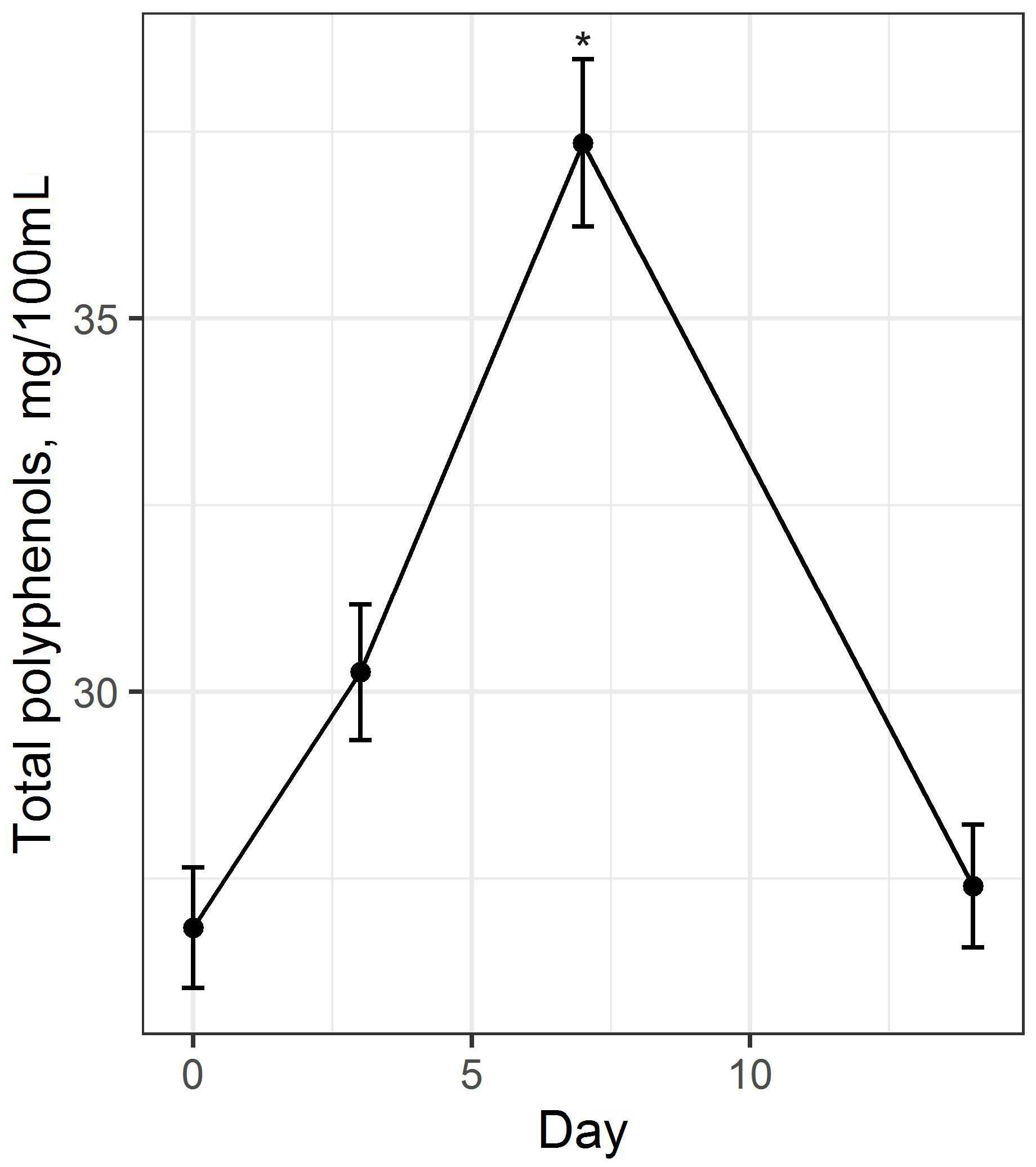
84]. The results of the evaluation of the total content of polyphenolic compounds in the beverage base and their change during fermentation are shown in
Figure 5.
During the fermentation process under the influence of SCOBY during the first 7 days, there is an increase in the total content of polyphenolic compounds (H = 7.308,
p = 0.042). As the process continues, their content decreases. Data from other researchers support our conclusions [
85]. The transformation of phenolic compounds from their conjugated forms into their free forms, as well as the oxidation and dimerization of tea catechins with the increased catalytic ability of polyphenol oxidase, are all linked to changes in this indicator during fermentation [
85]. The conversion of flavonoids during fermentation, e.g., thearubigin to theaflavin, leads to a change in the color of the fermented base from dark to light [
86].
The type of tea plant, growing conditions, and a variety of other factors affect how the flavor profile of the finished product develops [
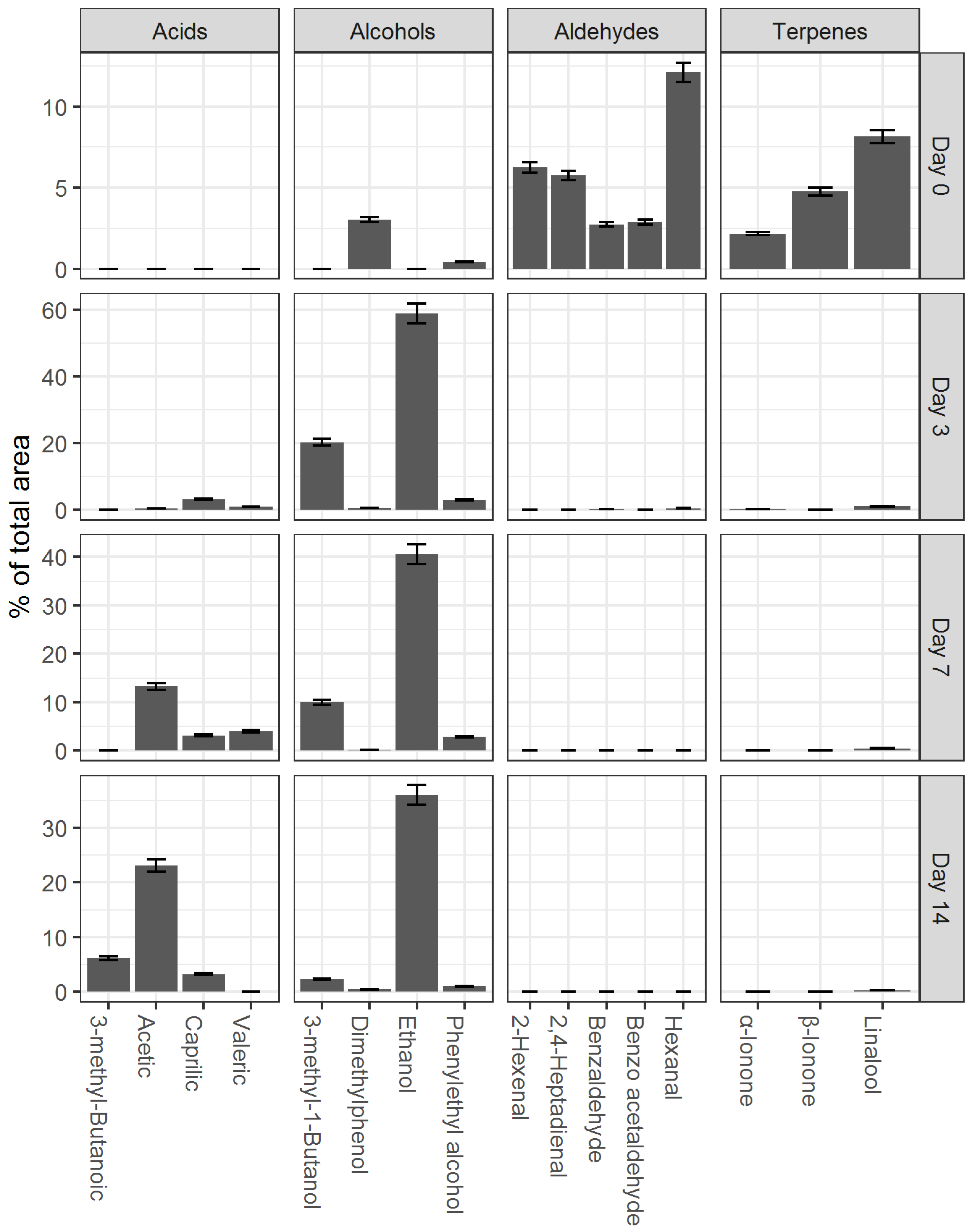
87]. In the process of fermentation, there is the formation of volatile compounds that form a specific category of beverages’ flavor profile. As a result of this research, 70 volatile compounds were identified in the initial infusion of black tea before the beginning of the fermentation process. On the 14th day of fermentation (the completion of the fermentation process), 55 volatile compounds were identified. From the total list of identified volatile organic compounds, substances with a high content in the aromatic profile were selected. These substances were subdivided into four main groups: aldehydes, acids, alcohols, and terpenes (
Figure 6).
During the fermentation, the profile of volatile substances changes: some identified in the initial tea infusion disappeared while the concentration of others increased. As can be seen from
Figure 6, at the starting point, aldehydes and terpenes have the highest concentrations in the tea base, with hexanal (12.1%) and linalool (8.2%) predominating in these groups, respectively. The presence of hexanal and linalool imparts an herbaceous and floral aroma to the beverages, which disappears during fermentation. On the third day of fermentation, due to the alcoholic fermentation, the content of ethanol in the vapor phase increases rapidly (59%). On the 7th day, the content of organic acids increases significantly, mainly due to acetic (13.5%), pentanoic (4.3%), and octanoic (3.1%) acids. On the 7th day, the concentration of alcohols (phenylethyl, ethanol, dimethylphenol, and 3-methyl-1-butanol) decreases naturally. The maximum content was observed for ethanol (40%). On the 14th day of the study, the alcohol content decreased to below 3%, except for ethanol, where the concentration in the vapor phase above the fermented beverage sample was 35.2%. However, the ethanol content in the fermented base was 0.22%. A low alcohol content of up to 1% may not be felt by the consumer because the threshold of taste for ethanol ranges from 1 to 2% [
88], while a concentration of more than 1% may result in a bitter taste. The content of acetic acid on the 14th day increased to 23.4%, and the content of 3-methylbutanoic acid also increased significantly. The results obtained are generally characteristic of the process of tea fermentation by SCOBY [
89] and indicate that aldehydes and terpenes are the main volatile organic compounds that are metabolized in the fermentation process. This may be an indication of the loss of the traditional black tea aroma in the final product.
The kinetics of the accumulation and degradation of volatile substances in kombucha were analyzed. For substances whose content was measured throughout the experiment and varied at selected measurement intervals, information on the order and constants of reactions as well as on the time of semi-transformation of substances was obtained (
Table 2). The table shows information on the models with the highest values of linear correlation coefficients (r
2).
Only the accumulation of acetic acid fitted well (r2 = 0.960) with the zero-order reaction kinetic model. This indicates that the process of vinegar synthesis from ethanol under the action of acetic acid bacteria does not depend on ethanol concentration. Thus, we can say that up to the 14th day of fermentation, ethanol was in excess for the synthesis of acetic acid. This may be due, in particular, to the low rate of metabolization of ethanol, which has a half-life time of 18.05 days. The decrease in ethanol (after the third day) proceeds as a second-order reaction with the lowest reaction constant among those studied. In contrast to ethanol, the change in the content of 3-methyl-1-butanol proceeds faster as a first-order reaction with a half-life of 1.15 days.
The decrease in linalool and hexanal, which were initially present in the tea, also corresponds to the kinetics of the second-order reaction. They are characterized by a high rate of degradation: 0.42 days for linalool and 0.06 days for hexanal. The rate constants of these reactions are 3–4 orders of magnitude higher than the rate constant of the ethanol metabolization reaction. Nevertheless, the order of these reactions indicates that the decrease in the concentration of linalool and hexanal is not a simple process but is caused by a number of factors, such as the action of aldehyde dehydrogenase in acetic acid bacteria [
90].
3.2. Preparation and Study of the Fermented Beverage
A kombucha base was used as a matrix for the beverages. The use of fresh berries, fruits, and vegetables in the composition of beverages is due to their content of vitamins, minerals, polyphenolic compounds, phenolic acids, tannins, etc. [
91,
92]. Lime leaves give a special flavor to the product due to the essential oils. Crushed strawberries and lime leaves were introduced into the prepared fermented base and incubated at 23 ± 2 °C for 24 h for infusion and secondary fermentation, followed by filtration. The composition of the fermented strawberry and lime beverages is shown in
Table 3.
The addition of flavoring ingredients leads to secondary fermentation and the extraction of biologically active substances. The effect of the addition of flavoring ingredients (strawberry fruit and lime leaves) and secondary fermentation on the physicochemical parameters of the fermented base is presented in
Table 4.
Based on the data obtained (
Table 4), it was found that the addition of ingredients leads to secondary fermentation, as evidenced by the decrease in sucrose content and increase in alcohol and organic acids. Changing the profile of organic acids is associated not only with the process of secondary fermentation but also with the extraction of acids from strawberry fruits. According to [
93,
94], strawberry fruits mainly contain citric and malic acids and, in small amounts, tartaric, oxalic, and fumaric acids. The increase in the content of citric, malic, and tartaric acids in the fermented beverages after the second fermentation relative to the fermented base (
Table 4) indicates their extraction from strawberry fruits. The increase in alcohol content (from 0.22 to 0.43%) and decrease in sugar content (from 3.6 g/100 mL to 0.18 g/100 mL) indicate the progress of alcoholic fermentation. There is also a dramatic increase in acetic acid content after secondary fermentation. At the same time, the pH value increased insignificantly. As noted earlier, the change in pH is not always associated with a change in the content of organic acids because the fermented base may have buffering properties [
76].
In order to increase the nutritional value of the beverages, they were enriched with B vitamins and inulin, nutrients with clinically proven health benefits. Inulin was pre-dissolved in boiled water at 60–70 °C, and then the solution was cooled to room temperature. We added vitamin premix and stirred until it completely dissolved. When calculating the number of enriching ingredients, we proceeded from the recommendation to ensure the content in a beverage portion (220 g) of inulin was in the amount corresponding to the RDI for vitamins, from 15 to 50% of the RDI. The composition of the enriched fermented beverages is shown in
Table 5.
The content of B vitamins in the enriched fermented beverages was as follows: vitamin B1—0.27 ± 0.02 mg/100 g, vitamin B2—0.25 ± 0.02 mg/100 g, vitamin B6—0.34 ± 0.03 mg/100 g, folic acid—0.04 ± 0.01 mg/100 g, and vitamin PP—2.76 ± 0.22 mg/100 g. Inulin content was 1.21 ± 0.12%. In the manufacturing process of enriched food products, vitamins are subjected to physical and chemical treatment, which may adversely affect their safety. The results of analytical studies have stated that the technological processes of beverage production, in particular carbonization and pasteurization, have a negative impact on the stability of folic acid (12% loss).
The antioxidant properties of tea and tea beverages are influenced by their high polyphenolic compound content [
6]. The DPPH inhibitory activity of the kombucha base was 89.9%. According to the proposed technology (
Section 2.4), crushed strawberry fruits and lime leaves were added to the base, which can lead to the extraction of substances that have antioxidant properties. At the same time, according to the recipe (
Table 3), dissolved inulin was added to the kombucha base, which could lead to dilution of the base and a slight decrease in antioxidant activity. Moreover, the technology of obtaining enriched fermented beverages involves a pasteurization stage (72 ± 2 °C, 40 min), in which the destruction of biologically active substances that have antioxidant properties may occur. Analysis of the enriched fermented beverages showed that the DPPH inhibitory activity was 82.0%.
Consumer properties of beverages are decisive for successful market promotion (or sale). In the finished product, the ethanol content did not exceed 0.43%, which classifies the developed beverages as non-alcoholic [
95]. Organoleptic properties of the developed beverages were formed as a result of fermentation processes as well as the extraction of flavor and aromatic substances from introduced strawberries and lime leaves. Characteristics of the organoleptic and the characteristics of the developed enriched beverages are presented in
Table 6.
The results of the study of organoleptic properties, shown in
Table 6, show a high estimate of the developed beverages by the indicators of “odor”, “consistency”, and “taste”.
At the stage of secondary fermentation, there is an extraction from the introduced ingredients of flavors, aromatics, sugars, vitamins, minerals, polyphenols, and other biologically active substances. This technological stage is very important for the formation of the organoleptic properties of the beverages. The resulting enriched fermented beverages have a pleasant color and exquisite aroma, not characteristic of traditional beverages. However, the dominant flavor and aroma of lime were noted by some tasters as a drawback.
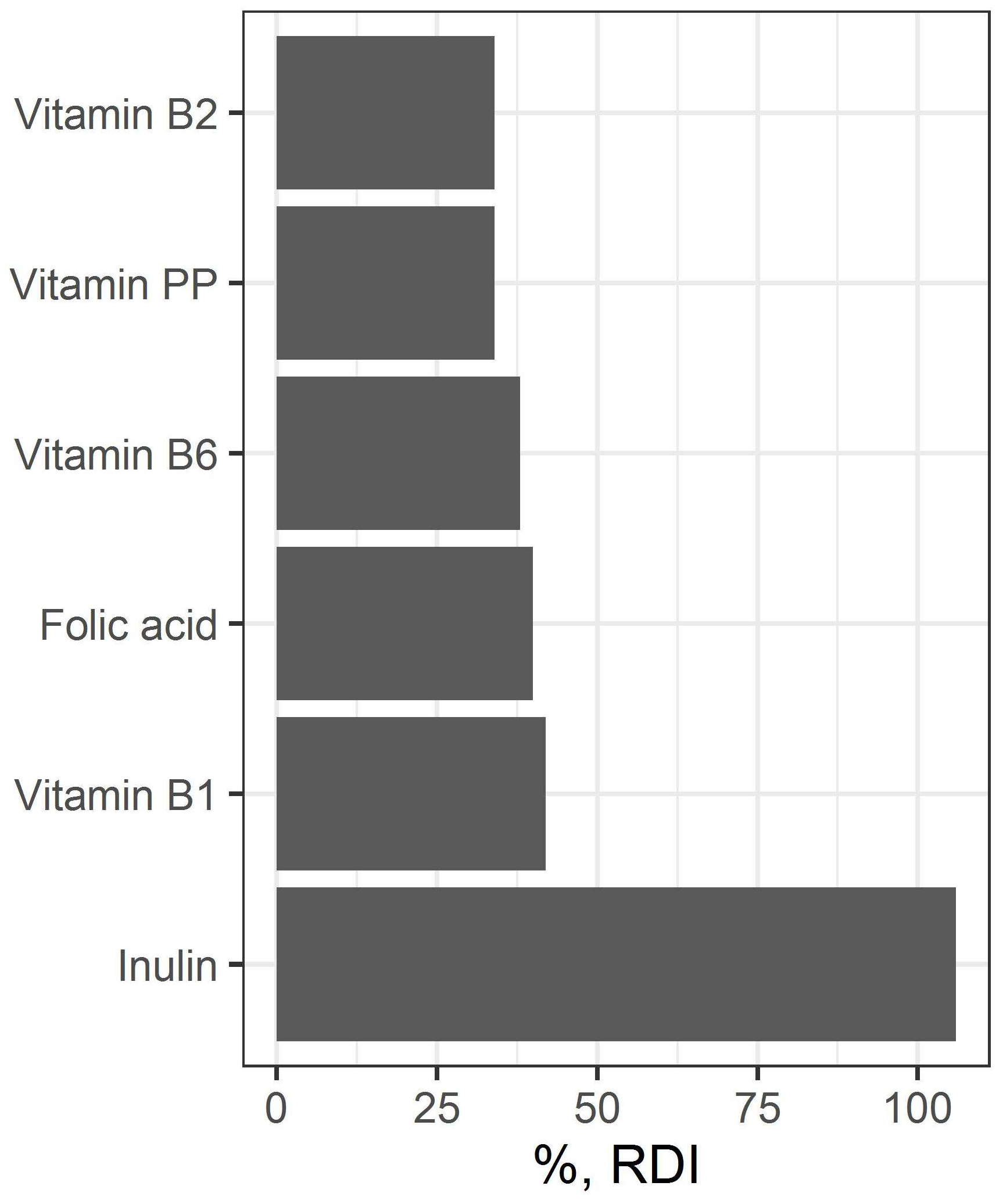
The content of vitamins in a portion (220 g) of a developed beverage refers to enriched products. Inulin content does not exceed the upper allowable level of consumption (
Figure 7).
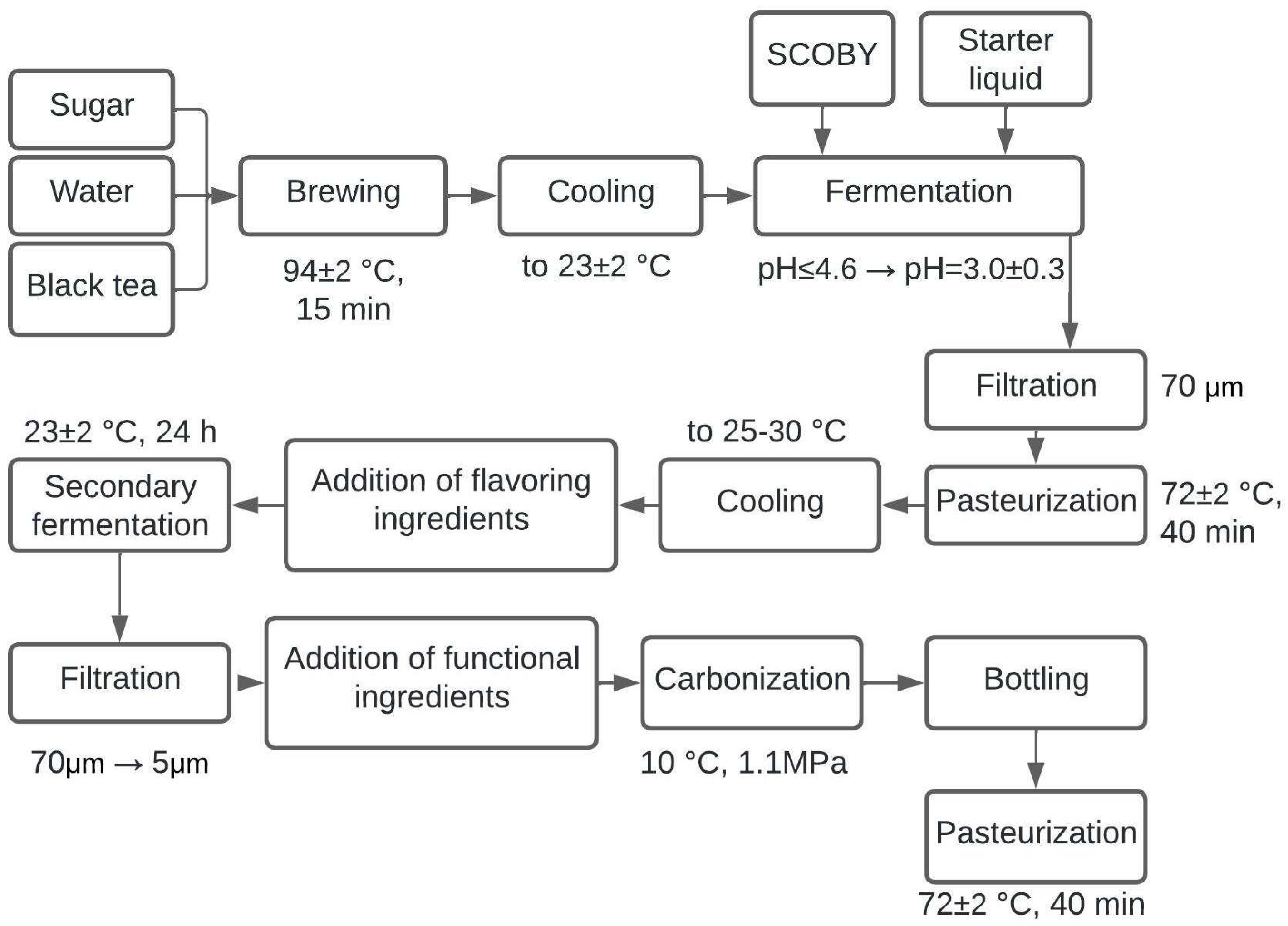
The technological scheme for producing kombucha enriched with inulin and B vitamins was developed (
Figure 8).
Basic and auxiliary processes in fermented beverage technology can be distinguished [
73]. The developed technology of obtaining fermented beverage kombucha includes the main stages: brewing tea, cooling, fermentation, and filtration, i.e., stages that allow obtaining classic unpasteurized fermented beverage without the additional introduction of other ingredients. The auxiliary stages of the process (pasteurization, addition of flavors and functional ingredients, secondary fermentation, and carbonation) allow for a broader range of beverages and additional health benefits. A clinical study of the developed beverage enriched with inulin and vitamins presented in [
96] showed a decrease in the intensity of the complaints significant for irritable bowel syndrome with constipation due to the normalization of stool frequency and consistency.