Abstract
Antioxidant activity is an important feature for evaluating fruit quality and tolerance to biotic or abiotic stresses. Moreover, antioxidant activity is involved in chilling injury (CI) response and postharvest quality in fruit. Chemical elicitors can induce systemic acquired resistance in fruit against pathogens, which could partially replace synthetic fungicides. Recently, researchers have found that preharvest sprays with chemical elicitors can improve antioxidant activity, reduce CI, and maintain quality in harvested fruit. In this review, we summarize that preharvest elicitors spray improve antioxidant activity in harvested fruit by promoting antioxidant components biosynthesis as well as antioxidant ability in vitro. Moreover, preharvest elicitors spray alleviates CI in fruit by regulation of membrane lipid metabolism and reactive oxygen species metabolism. In addition, preharvest elicitors spray maintains fruit quality by modulation of respiration and ethylene release. Finally, this review points out the issues existing and proposes an outlook on preharvest elicitors spray to maintain postharvest fruit quality.
1. Introduction
Fresh fruits have aesthetic appearance and unique flavor as well as nutrition. However, postharvest loss in fruit is huge. The global average of postharvest loss in fresh fruit is estimated to be 40% [1]. Fungal infection is the main reason for postharvest loss of fruit, but over-ripening and senescence, and chilling injury (CI) caused by inappropriate low temperature are also considered to be important causes of the loss [2,3]. Various measures can effectively control quality deterioration and CI in fruit. However, most of them focus on postharvest treatments, which are inconvenient and inevitably increase postharvest procedures and costs [2,4,5].
Chemical elicitors are synthetic chemicals, mainly including salicylic acid (SA), jasmonic acid (JA) and chitosan and their analogs or derivatives. These elicitors can induce fruit resistance against pathogens by eliciting systemic acquired resistance (SAR), which could partially replace synthetic fungicides [6]. Recently, in addition to inhibiting fruit diseases by inducing resistance, chemical elicitor shave been found to contribute to enhancing antioxidant activity, reducing CI, and maintaining postharvest quality in fruit [7,8,9].
The periods of fruit development are critical for the formation of postharvest resistance and quality in fruit. During the development, continuous cell division and expansion lead to fruit enlargement and the formation of barriers that can contribute to fruit resistant, including epidermal wax, cut in, cell wall, and cell membrane [10]. Meanwhile, products such as sugars, organic acids, aromas, ascorbic acid, carotenoids, and phenolic compounds are continuously accumulated in cells, which can affect fruit quality and tolerance biotic or abiotic stresses [11,12]. This review focuses on the effect of preharvest elicitors spray on improving antioxidant activity, alleviating CI, and maintaining quality in harvested fruit, and analyzes the possible reasons.
2. Improvement of Antioxidant Capacity
Phenolics, ascorbic acid, and carotenoids are important antioxidant compounds in fruit, which are known to be beneficial to human health [13].
2.1. Enhancing Phenolics, Ascorbic Acid and Carotenoids Contents
Preharvest elicitors spray effectively enhances the accumulation of phenolic compounds in fruit. Similarly, 0.1 mM methyl jasmonate (MeJA) or 0.5 mM SA that was sprayed 4 times on ‘Fino’ lemon before harvest increased the contents of total phenolics and two main flavonoids (hesperidin and eriocitrin) [14]. ‘Xiaobai’ Apricot was sprayed twice with 0.05% chitosan oligochitosan or 1 mmol L−1 SA at 7 and 2 days before harvest, resulting in higher contents of total phenolics, total flavonoids and three main phenolic compounds (5-O-caffeoylquinic acid, 3-O-caffeoylquinic acid, quercetin-3-rutinoside) at harvest and during cold storage [7]. Preharvest with phenylalanine increased the total flavonoids and anthocyanins in ‘Kent’ and ‘Shelly’ mango fruits and ‘Pink Lady’ apples [15]. Spraying twice with 0.1%, 0.2%, or 0.4% prohydrojasmon on 4 and 2 weeks before harvest increased the contents of total anthocyanin and total flavonoid and the accumulation of anthocyanin monomers and flavonoids in ‘Kent’ mango fruit during cold storage, of which 0.4% treatment showed the best effect. On the 21st day of cold storage, the contents of total anthocyanin and total flavonoid in 0.4%-sprayed fruit showed 4 folds and 67% higher than those of the control. Moreover, the contents of anthocyanin monomers, including cyanidin, methyl-cyanidin, cyanidin-3-galactoside, 7-O-methylcyanidin-3-O-β-D-galactopyranoside, as well as quercetin and kaempferol content, were significantly higher than those of the control [16]. In addition, at harvest and during cold storage, contents of total phenolics, total anthocyanins, and anthocyanin monomers enhanced in ‘Mollar de Elche’ pomegranate sprayed by 1, 5, or 10 mmol L−1 MeJA at 94, 64, 34, and 4 days before harvest, of which 5 mmol L−1 MeJA spay showed the best effect [17].
Preharvest elicitors spray can significantly enhance the accumulation of ascorbic acid and carotenoids in fruit. At harvest, a higher content of ascorbic acid was observed in ‘El-Bayadi’ table grapefruit sprayed with 4.0 mM SA 4 times from the pea stage to the version stage [18]. A higher total carotenoid content was shown in ‘Black Splendor’ plum fruit that was sprayed 3 times with 0.5 mmol L−1 SA, 1 mmol L−1 acetyl salicylic acid (ASA), or 0.5 mmol L−1 Methyl salicylate (MeSA) at 61, 76, and 94 days after flowering. At harvest, the total carotenoid content in the three treated plum fruit increased by 17%, 25%, and 25% compared with the control, which was 18%, 55%, and 36% higher on the 40th day of cold storage, respectively [19]. Similarly, three sprays of 1 mM oxalic acid at 63, 77 and 98 after flowering increased total carotenoid content in ‘Black Splendor’ plum fruit, which increased by 20% compared with the control on the 35th of cold storage [20]. In addition, the total carotenoid content in ‘Neelum’, ‘Bangalore’, ‘Banganapalli’, and ‘Alphonso’ mango fruit sprayed twice with 2% hexanal at 30 and 15 days before harvest was 6.7%, 61%, 27%, and 32% higher than that of the control on the 9th day of cold storage [21]. Three sprays of 250 mmol L−1 MeJA at flowering, 24 days of turning green, and 7 days of turning red enhanced ascorbic acid content in ‘Camarosa’ strawberry, which increased by 20% and 63% compared with the control at harvest and 2 days of storage [22]. Further, two sprays of 1.2 mM hexanal4 and 2 weeks before harvest increased ascorbic acid content in ‘Allahabad Safeda’ guava fruit at harvest and during cold storage [23]. Preharvest elicitors spray on improving phenolics, ascorbic acid and carotenoids accumulation in harvested fruit are shown in Table 1.
Phenolics and flavonoids in fruit are mainly synthesized from phenylpropanoid metabolism pathway. L-phenylalanine is the substrate of phenylpropanoids metabolism and it can generate phenolics and flavonoids under the continuous activity of phenylalanine ammonialyase (PAL), cinnamate 4-hydroxylase (C4H), and 4-coumarate: coenzyme A ligase (4CL), and cinnamyl alcohol dehydrogenase (CAD) [24,25]. Preharvest phenylalanine spray improved the contents of total flavonoids and total anthocyanins in mango fruit [15]. Similarly, preharvest phenylalanine spray triggered PAL, C4H, 4CL, and CAD activity, increasing the accumulation of total phenolic and flavonoids in muskmelon fruit [26]. Preharvest melatonin up-regulated the levels of SlPAL, SlC4H, and Sl4CL expression in cherry tomato fruit during storage [27]. Moreover, preharvest prohydrojasmon spray up-regulated the expression of a key transcription factor (MYB114) that regulated key genes for anthocyanin biosynthesis (PAL, CHS, CHI, F3H, ANS, UFGT, FLS, and LAR) in pear fruit at harvest [28]. In addition, preharvest MeJA improved PAL activity, increasing flavonoid accumulation in raspberries at harvest [29]. Preharvest harpin spray increased PAL activity in muskmelon fruit during storage, improving total phenolics and flavonoid content [30]. Preharvest chitosan spray enhanced the activity of PAL, 4CL, CAD, and C4H, promoting the biosynthesis of phenolics and flavonoids in muskmelon fruit during storage [31,32]. Although the effects of preharvest elicitors spray on the biosynthesis of phenolics in harvested fruit have been studied at the biochemical level, the relevant molecular mechanisms remain to be further unlocked.
Ascorbic acid is mainly synthesized through the L-galactose pathway in fruit [33]. However, there is no report on the regulation of the L-galactose pathway in fruit by preharvest elicitors spray. Carotenoids are mainly synthesized through 2-c-methyl-d-erythritol 4-phosphate and/or mevalonate pathway in fruit [34]. The transcriptome results showed that preharvest of benzothiadiazole (BTH) and chitosan spray up-regulated the expression of important transcription factors as WRKYs and NACs, involved in regulating the metabolism of carotenoid compounds in fruit, thereby promoting the synthesis of carotenoids in harvested ‘Alba’ strawberry fruit [35]. There are few reports on the effect of preharvest elicitors spray on carotenoid biosynthesis in harvested fruit, and the related mechanism needs to be further studied.


Table 1.
Preharvest elicitors spray improves antioxidant compounds and freeradicalscavengingactivity in fruit.
Table 1.
Preharvest elicitors spray improves antioxidant compounds and freeradicalscavengingactivity in fruit.
| Elicitor | Manufacturer | Fruit | Cultivar | Spraying Concentration | Spraying Times | Spraying Stage | Antioxidant Compounds | Free Radical Scavenging Activity | References | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Phenols | Flavonoids | Anthocyanins | VC | Carotenoids | DPPH | ABTS+ | FRAP | ||||||||
| SA | Sigma-Aldrich | Plum | Black Splendor | 0.5 mmol L−1 | 3 | 61, 76 and 94 days after full blossom | √ a | — b | √ | — | √ | — | — | — | [19,36] |
| Oxford Laboratory Reagents | Apricot | Canino | 2 or 4 mM | 2 | 30 and 15 days before harvest | — | — | — | — | √ | — | — | — | [37] | |
| — | Xiaobai | 1 mmol L−1 | 2 | 7 and 2 days before harvest | √ | √ | — | √ | — | √ | √ | √ | [7] | ||
| — | Palm | Khesab | 3% | 3 | 5 and 15 weeks from pollination, and two weeks before harvest | √ | √ | — | — | — | — | — | — | [38] | |
| ASA | Sigma-Aldrich | Plum | Black Splendor | 1 mmol L−1 | 3 | 61,76 and 94days after full blossom | √ | — | √ | — | √ | — | — | — | [19,36] |
| MeSA | Sigma-Aldrich | Plum | Black Splendor | 0.5 mmol L−1 | 3 | 61,76 and 94days after full blossom | √ | — | √ | — | √ | — | — | — | [19,36] |
| BTH | Novartis Crop Protection | Muskmelon | Yindi | 100 mg L−1 | 3 | flowering and 14, 28, 42 days after flowering | √ | √ | — | — | — | — | — | — | [39] |
| Prohydrojasmon | — | Pear | Nanhong | 50 or 100 mg L−1 | 2 | 100 and 103 days after blooming | √ | √ | √ | — | — | — | — | — | [28] |
| — | Mango | Kent, Shelly and Maya | 0.1, 0.2 or 0.4% | 2 | 4 and 2 weeks before harvest | — | √ | √ | — | — | — | — | — | [16] | |
| Chitosan | — | Strawberry | Seascape | 2, 4, or 6 g L−1 | 1 | just turning red | — | — | √ | — | — | — | — | — | [40] |
| — | Grape | Jingxiu | 1 g L−1 | 1 | 10 days before harvest | √ | — | — | — | — | — | — | — | [40] | |
| — | Yaghouti | 2% or 3% | 3 | fruit set, 25 and 50 days after fruit set | — | — | √ | √ | — | √ | — | — | [41] | ||
| WN Group of Publishers | Muskmelon | Manao | 0.001 | 4 | 14, 21, 28, 40 days after flowering | √ | √ | — | — | — | — | — | — | [31] | |
| Cornell Lab | Apricot | Canino | 1.5% or 2.5% | 2 | 30 and 15 days before harvest | — | — | — | — | √ | — | — | — | [37] | |
| — | Palm | Khesab | 1% | 3 | 5 and 15 weeks from pollination and two weeks before harvest | √ | √ | — | — | — | — | — | — | [38] | |
| Huarun Bioengineering | Kiwifruit | guichang | 28.6% | 3 | budding phase, fruit setting phase and expanding final phase | √ | √ | — | √ | — | — | — | — | [42] | |
| Chitosan oligosaccharide | Dalian GlycoBio | strawberry | qingxiang | 50 mg·L−1 | 4 | seedling stage, before flowering, fruit coloring and full bloom | √ | √ | √ | √ | — | √ | — | — | [43] |
| Chitosan oligochitosan | — | Apricot | Xiaobai | 0.05% | 2 | 7 and 2 days before harvest | √ | √ | — | √ | — | √ | √ | √ | [7] |
| Oligochitosan | Jinan Haidebei Marine Bioengineering | Navel orange | Osbeck | 15 g L−1 | 4 | 30, 60, and 90days after physiological fruit drop and 10 days before harvest | √ | — | — | — | — | — | — | — | [44] |
| β-aminobutyric acid | Sigma-Aldrich | Blueberry | Bluecrop | 20 mM | 1 | 7 days before harvest | — | — | √ | — | — | — | — | — | [45] |
| Oxalic acid | — | Peach | Anjirymaleki | 1, 3 or 5 mmol L−1 | 1 | 15 days before harvest | √ | √ | — | — | — | √ | — | √ | [46] |
| — | Kiwifruit | Bruno | 5 mM | 3 | 130days after full blossom and 2 times at 7 days intervals | √ | √ | — | √ | — | — | — | — | [47] | |
| Sigma-Aldrich | Sweet cherry | Sweet Heart and Sweet Late | 0.5, 1.0 or 2.0 mM | 3 | 98, 112, and 126 days after full blossom | √ | — | √ | — | — | — | — | — | [48] | |
| — | Plum | Black Splendor | 1 mM | 3 | 63,77 and 98 days after fullblossom | √ | — | — | — | √ | — | √ | — | [20] | |
| — | Kiwifruit | Bruno | 5 mmol L−1 | 3 | 130, 137 and 144 days after the flowering | — | — | — | √ | — | — | — | — | [49] | |
| Sigma-Aldrich | Pomegranate | Mollar deElche | 1, 5 or 10 mM | 3 | 80, 110, 140, and 170 days after full blossom | √ | — | √ | — | — | — | — | — | [50] | |
| — | Apricot | Red Flesh | 0.5, 1 or 2 mM | 1 | fruit set stage | √ | — | — | √ | — | — | — | — | [51] | |
| Sigma-Aldrich | Lemon | Fino | 0.1, 0.5 or 1.0 mM | 5 | from physiological fruit dropto 3 days before harvest | √ | — | — | — | — | — | — | — | [52] | |
| Harpin | Eden Bioscience | Muskmelon | Huanghemi | 50 mg L −1 | 3 | flowering and 14, 28, 42 days after flowering | √ | — | — | √ | — | — | — | — | [53] |
| Hexanal | Sigma-Aldrich | Mango | Neelum, Bangalora, Banganapalli and Alphonso | 0.02% | 1; 1; 2 | 15 days before harvest; 30 days before harvest; 15 and 30 days before harvest | — | — | — | √ | √ | — | — | — | [21] |
| — | Strawberry | Jewel, Kent, Mira and St. Pierre | 0.01 | 3 | once per week before harvest | √ | √ | √ | — | — | — | — | — | [54] | |
| — | Guava | Allahabad Safeda | 0.8, 1.2 or 1.6 mM | 2 | 4 and 2 weeks before harvest | √ | — | — | √ | — | — | — | — | [23] | |
| SNP | Sigma-Aldrich | Muskmelon | Manao | 0.5 mM | 4 | 14, 21, 28, 40 days after flowering | √ | √ | — | — | — | — | — | — | [55] |
| Sigma-Aldrich | Manao | 0.5 mM | 4 | young fruit stage, early stage of en largement, late stage of enlargement and mature stage | √ | √ | — | — | — | — | — | — | [56] | ||
| Sigma-Aldrich | Peach | G.H. Hill | 0, 25, 50 or 100 mol L−1 | 1 | 14 days before harvest | — | — | — | √ | — | — | — | — | [57] | |
| Putrescine and spermidine | — | Grape | Olhoghi and Rishbaba | 1 or 2 mM/1 or 2 mM | 2 | 40 and 20 days before harvest | √ | — | √ | — | — | — | — | — | [58] |
| Arginine | — | Pomegranate | MalaseSaveh | 0, 0.5, 1 or 2 mM | 3 | 20 days interval before commercial harvest | √ | √ | — | √ | — | √ | — | — | [59] |
| L-phenylalanine | HunanShaofeng | Muskmelon | Manao | 8 mM | 4 | young fruit stage, early expansion stage, late expansion stage and one week before harvest | √ | √ | — | — | — | — | — | — | [26] |
| Melatonin | — | Apricot | Colorado and Mikado | 0.1 mM | 3 | pit hardening, final fruit growth, and 4 days before harvest | √ | — | — | — | — | — | — | — | [60] |
| Sigma-Aldrich | Sweet cherry | Ferrovia | 0.5 mM | 2 | 2 and 1 weeks prior to harvest | √ | — | — | — | — | — | — | — | [61] | |
| — | Pear | Nanhong | 50 or 200 μmol L−1 | 2 | dripping and 3 days after | √ | √ | — | — | — | — | — | — | [62] | |
a √ indicates detected. b — indicates no assessment.
2.2. Promoting the Antioxidant Activity
Preharvest elicitors spray also improves in vitro antioxidant ability in fruit (Table 1). For example, spraying 5 mmol L−1 oxalic acids 15 days before harvest had no significant effect on the values of DPPH and FRAP in ‘Anjiry Maleki’ peach fruit at harvest. However, the spray increased the values of DPPH and FRAP in the fruit by 25% and 20% compared with the control on the 4th of storage [46]. Another example is that four sprays of 4.0 mM SA at the ‘pea stage’ to the version stage increased the values of DPPH and ABTS+ in ‘El-Bayadi’ table grapes, which was 63% and 25% higher than that of the control at harvest [18].
DPPH, ABTS+, and FRAP are important indicators to evaluate antioxidant ability. DPPH mainly reflects the antioxidant ability of phenols, flavonoids, and terpenoids in fruits [63]. ABTS+ and FRAP reflect the antioxidant ability of ascorbic acid and carotenoids in fruit, respectively [64]. Due to the increase of the biosynthesis of phenolics and flavonoids as well as ascorbic acid and carotenoids, preharvest elicitors spray improves antioxidant ability in fruit. The current studies are mainly focus on the antioxidant ability in vitro of harvested fruit, while further research is needed on the antioxidant ability of human cells or in vivo.
3. Preharvest Elicitors Spray Alleviates CI in Fruit
3.1. Reducing the Occurrence of CI
Many fruits are sensitive to low temperatures, which makes them prone to CI during storage at suboptimal temperatures, resulting in surface pitting, water-soaked spots, epidermal or internal browning, abnormal ripening, and other CI symptoms. Cold-sensitive fruits mainly grow in tropical or subtropical regions, including citrus, bananas, avocados, mangos, pineapples, peaches, apricots, plums, papayas, pomegranates, melons, etc. [65]. Preharvest elicitors spray can effectively alleviate CI in fruit. For example, ‘Lane Late’ sweet orange fruit sprayed with 2, 4, 6, or 8 mM SA 10 days before harvest reduced CI index of fruit stored for 93 days at 5 °C. Compared with the control, the CI index decreased by 76.15% in the fruit sprayed with 8 mM SA on the 93rd day of cold storage, before harvest [8]. Another example is that preharvest spray at 7 and 2 days with 0.05% chitosan, oligochitosan, or 1 mmol L−1 SA reduce CI in ‘Xiaobai’ apricot that was stored for 14 days at 2 °C [7]. In addition, 0.02% hexanal spray at 15 and 10 days before harvest decreased the CI index in ‘Fantasia’ nectarine fruit by 55% compared to the control on the 45th day of storage at 2 °C [66]. Preharvest elicitors spray on CI alleviation in fruit during cold storage are shown in Table 2.

Table 2.
Preharvest elicitors spray alleviates chilling injury in fruit.
3.2. Improving ROS Scavenging Ability
Oxygen burst caused by CI leads to excessive H2O2 accumulation that accelerates peroxidation of the cell membrane, leading to the production of malondialdehyde (MDA), which also destroys membrane integrity [5]. Preharvest elicitors sprayed as SA can increase the activity of antioxidant enzymes as well as the concentration of antioxidant compounds, which contribute to reducing H2O2 damage in fruit and vegetables [14]. Preharvest MeJA and SA spray improved the activities of catalase (CAT), ascorbate peroxidase (APX), and peroxidase (POD) as well as the biosynthesis of phenols and flavonoids in ‘Fino’ and ‘Verna’ lemon fruit during cold storage [14]. Moreover, during cold storage, higher activities of superoxide dismutase (SOD), CAT, APX, and POD and accumulations of total phenols, total anthocyanins, total carotenoids, and total flavonoids were found in ‘Black Splendor’ and ‘Royal Rosa’ plum fruit that was preharvest sprayed with SA, acetylsalicylic acid (ASA), or MeSA [19,36]. Preharvest MeJA spray increased total phenols, total anthocyanins, total flavonoids, and vitamin C contents, thereby improving ROS scavenging and antioxidant activity in fruit [17]. Moreover, preharvest spray with SA or chitosan oligochitosan in the ‘Xiaobai’ apricot orchard decreased MDA content and increased contents of total phenols, total flavonoids, and vitamin C. Additionally, preharvest chitosan oligochitosan and SA spray enhanced free radical-scavenging capacity, including DPPH free radical scavenging capacity, ABTS free radical scavenging capacity, cupric ion reducing antioxidant activity, and ferric reducing antioxidant activity, elevating scavenging ability of free radicals in fruit [7]. Although the effects of preharvest elicitors spray on activity of antioxidant enzymes and biosynthesis of antioxidants in harvested fruit have been studied at the biochemical level, the relevant molecular mechanisms remain to be further revealed.
3.3. Decreasing Membrane Lipid Metabolism
Destruction of the cell membrane is considered to bean important characteristic of CI. The content of unsaturated fatty acids (USFAs) in the cell membrane is closely related to CI in fruit [52]. A high content of USFAs contributes to the fluidity of cell membrane and membrane integrity and increases the cold tolerance of fruit [65,77]. Preharvest MeJA spray maintained USFAs content in ‘Mollar de Elche’ pomegranate fruit during cold storage and reduced the ratio of unsaturated/saturated fatty acids, contributing to maintaining the integrity of cell membrane and alleviating CI in fruit [78]. Low temperature leads to degradation and peroxidation of membrane lipids that cause the loss of membrane integrity and functionality, resulting in the appearance of CI symptoms in fruit [79,80]. Phospholipase D (PLD) is a key enzyme in membrane phospholipid metabolism, which can directly hydrolyze ester bonds of phosphoglyceride and catalyze the decomposition of phospholipid into phospholipid acid (PA) and diacylglycerol [81]. The occurrence of CI increases PLD activity in fruit that induces PA accumulation, while excessive production of PAleads to H2O2 accumulation, which destroys the integrity of the cell membrane [82]. Preharvest hexanal spray inhibited membrane phospholipid metabolism in ‘Fantasia’ nectarine fruit by down-regulating PLD expression, thereby maintaining the integrity of the cell membrane [66]. Although the effects of preharvest elicitors spray on membrane lipid metabolism in harvested fruit have been studied at the biochemical level, the relevant molecular mechanisms remain to be further proclaimed.
3.4. Reducing Pulp Browning
Epidermal or internal browning is a typical symptom of fruit CI. Preharvest SA and chitosan oligochitosan spray decreased polyphenol oxidase (PPO) activity in ‘Xiaobai’ apricot fruit, alleviating pulp browning in fruit during cold storage [7]. Preharvest sodium nitroprusside (SNP) spray alleviated CI-induced browning of pulp tissue of ‘G.H. Hill’ peach fruit by decreasing PPO activity [57]. Cell membrane degradation caused by CI increases the possibility of interaction between polyphenols and PPO, which oxidizes polyphenols to quinones, and further polymerizes and forms dark brown products [70]. However, the specific regulation of enzymatic browning in fruit by preharvest elicitors spray remains to be further explained.
4. Preharvest Elicitors Spray Delays Ripening and Maintains Postharvest Quality of Fruit
Fruit ripening caused by ethylene is an important cause of quality deterioration in harvested fruit. Many reports have elucidated that preharvest elicitors spray can effectively retard fruit ripening by inhibiting ethylene production, thereby maintaining postharvest quality and prolonging shelf life of fruit [83].
4.1. Retarding Ripening
Ethylene release and increased respiration are typical physical features of climacteric fruit, which play a critical role in fruit maturation and ripening [84]. The peak of ethylene in ‘Black Splendor’ plum fruit retarded after spraying with 0.5 mmol L−1 SA, 1 mmol L−1 ASA, or 0.5 mmol L−1 MeSA 3 times at 61, 76, and 94 days after full blossom, respectively [36]. Moreover, the respiration rate in ‘Dashehari’ mango fruit retarded by 7 days during cool storage after spraying with 1200, 1600, or 2000 µM hexanal before harvest [85]. Preharvest elicitors spray can retard the peak of ethylene and respiration in climacteric fruit as well as decrease ethylene release and respiratory rate. In ‘Zill’ mango fruit, sprayed with 150 µM SA before harvest delayed the peak of ethylene by 2 days and decreased the ethylene accumulation by 82% compared with the control during cool storage [83]. These elicitors spray also can inhibit ethylene release and respiratory rate in non-climacteric fruit. ‘Fino’ lemon fruit that was sprayed with 0.1 mM MeJA or 0.5 mM SA 4 times before harvest had a decrease in ethylene production and respiration rate [14]. Similarly, compared with the control, the respiratory rate in ‘Mollar de Elche’ pomegranate fruit decreased by 33.33% on the 60th of storage after 5 mmol L−1 MeJA sprayed 4 times at 94, 64, 34 and 4 days before harvest [17]. Preharvest elicitors spray on inhibiting ethylene production and respiration rate in fruit after harvest are shown in Table 3. Although the effect of preharvest elicitors spray on respiratory rate in fruit has been studied, how the spray affects respiratory metabolism needs to be further revealed.
Ethylene plays a dual role in fruit as it participates both in the fruit in the ripening and defense response of climacteric fruits [86]. 1-Aminocyclopropane-1-carboxylic acid synthase (ACS) and 1-aminocyclopropane-1-carboxylic acid oxidase (ACO) are two key enzymes involved in ethylene biosynthesis. Preharvest elicitors spray reduces ethylene release in climacteric and non-climacteric fruit during storage. The gene expression of MiACS and MiACO in ‘Zill’ mango fruit preharvest sprayed with SA was reduced by 90% and 96% on the 9th and 7th day of storage, respectively [83]. Preharvest chitosan spray decreased the gene expression of AdACS2 and AdACO2 by 30% and 50% in ‘Garmrok’ kiwifruit compared with the control on the 60th day of storage [86]. Similarly, preharvest chitosan oligosaccharides spray reduced FaACS and FaACO gene expression in ‘Qingxiang’ strawberry fruit by 92% and 66% compared with the control at harvest [43]. Ethylene receptors negatively regulate ethylene signal transduction, while ethylene response factors (ERFs) are positively regulated by ethylene [87]. Transcriptome analysis showed that preharvest BTH and chitosan spray decreased ERF expression in ‘Alba’ strawberries after harvest, thereby inhibiting ethylene signal transduction [35]. Moreover, at harvest, ETR2 was down-regulated in ‘Stark Red Gold’ peach fruit that was preharvest sprayed with MeJA [88]. Although the effects of preharvest elicitors spray on biosynthesis and signal transduction of ethylene in harvested fruit have been studied, the relevant regulation mechanisms remain to be further announced.


Table 3.
Preharvest elicitors spray maintains postharvest quality in fruit.
Table 3.
Preharvest elicitors spray maintains postharvest quality in fruit.
| Elicitor | Manufacturer | Fruit | Cultivar | Spraying Concentration | Spraying Times | Spraying Stage | Storage Temperature (°C)/Days | Postharvest Quality | References | |||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| TSS | TA | TSS/TA | Firmness | |||||||||
| SA | — | Navel orange | Lane Late | 2, 4, 6 or 8 mM | 1 | 10 days before harvest | 5/93 | √ a | √ | — c | √ | [67] |
| Sigma-Aldrich | Lemon | Fino | 0.5 mmol L−1 | 4 | 21 days intervals untile 3 days before harvest | 8/35 | √ | √ | — | √ | [14] | |
| Sigma-Aldrich | Mango | Zill | 150 µM | 1 | 100 days after anthesis | 25/11 | × b | √ | — | √ | [83] | |
| Sigma-Aldrich | Peach | Cresthaven | 1 or 2 mM | 2 | 23 and 15 days before harvest | 20/2 | × | √ | — | √ | [89] | |
| — | Flordaking | 1, 2 or 3 mM | 3 | cell division, pit hardening or lag phase and cell enlargement stage | 1/42 | × | √ | × | √ | [69] | ||
| — | Apricot | Xiaobai | 1 mmol L−1 | 2 | 7 and 2 days before harvest | 2/70 | √ | √ | — | √ | [7] | |
| Cornell Lab | Canino | 2 or 4 mM | 2 | 30 and 15 days before harvest | 0/28 | × | √ | × | √ | [37] | ||
| — | Grape | Thompson Seedless | 100 mg L−1 | 2 | pea and veraison stage | 20/7 | — | — | — | √ | [90] | |
| Sigma-Aldrich | Flame Seedless | 1.0, 1.5 or 2.0 mM | 2 | pea and veraision stage | 3–4/75 | √ | √ | √ | √ | [91] | ||
| Sigma-Aldrich | Magenta and Crimson | 1 mM | 3 | 40% berries, veraison stage and 3 days before harvest | 2/45 | √ | × | — | — | [50] | ||
| — | Superior Seedless | 1, 2, or 4 mM | 3 | fruit set, berry variation and 14 days before harvest | 28/44 | √ | √ | √ | √ | [92] | ||
| Sigma-Aldrich | Plum | Black Splendor | 0.5 mmol L−1 | 3 | 61,76 and 94 days after full blossom | 2/50 | √ | × | √ | √ | [19] | |
| — | Strawberry | Festival | 2 or 4 mmol | 3 | full flowering, green fruits and pink stage | 4/12 | √ | — | — | √ | [93] | |
| — | Sweet cherry | Sweet Heart, Sweet Late and Lapins | 0.5 mmol L−1 | 3 | 98, 112 and 126 days after full blossom (SE/SL)/66, 75 and 81 days after full blossom (L) | 2/28 | √ | √ | — | √ | [94] | |
| Sigma-Aldrich | Jujube | Dongzao | 2 mM | 4 | 30, 60, 90, and 110 days after full blossom | 0/60 | — | — | — | — | [95] | |
| — | Wax apple | Taaptipjaan | 0.5 or 1.0 mM | 1 | 24 h before harvest | 13/9 | — | — | — | √ | [96] | |
| — | Palm | Khesab | 3% | 3 | 5 and 15 weeks from pollination, and two weeks before harvest | 2/60 | × | — | — | — | [38] | |
| ASA | Sigma-Aldrich | Grape | Magenta and Crimson | 1 mM | 3 | 40% berries, veraison stage and 3 days before harvest | 2/45 | √ | × | — | — | [17] |
| — | Strawberry | Festival | 0.25 or 0.50 mmoL | 3 | full flowering, green fruits and pink stage | 4/12 | √ | — | — | √ | [93] | |
| — | Sweet cherry | Sweet Heart, Sweet Late and Lapins | 1.0 mmol L−1 | 3 | 98, 112 and 126 days after full blossom | 2/28 | √ | √ | — | √ | [94] | |
| Sigma-Aldrich | Plum | Black Splendor | 1 mmol L−1 | 3 | 61,76 and 94 days after full blossom | 2/50 | √ | × | √ | √ | [19,36] | |
| MeSA | — | Apricot | Kate | 0.05, 0.1 or 0.2 mmol L−1 | 2 | 72 d and 74 days after full blossom | 2/32 | √ | √ | — | √ | [72] |
| Sigma-Aldrich | Grape | Magenta and Crimson | 1 mM | 3 | 40% berries, veraison stage and 3 days before harvest | 2/45 | √ | × | — | — | [17] | |
| Sigma-Aldrich | Plum | Black Splendor | 0.5 mmol L−1 | 3 | 61,76 and 94days after full blossom | 2/50 | √ | × | √ | √ | [19] | |
| MeJA | Sigma-Aldrich | Lemon | Fino | 0.1 mmol L−1 | 4 | 21 days intervals untile 3 days before harvest | 8/35 | √ | √ | — | √ | [14] |
| Sigma-Aldrich | Mango | Mahachanok | 20, 40, 80 or 120 μL mL−1 | 1 | 90 days after anthesis | 15/24 | — | — | √ | √ | [97] | |
| — | Plum | Black Splendor and Royal Rosa | 0.5 or 1.0 mM | 3 | - | 20/9; 2/50 | × | √ | — | √ | [98] | |
| — | Fortune | 1120 or 2240 mg L−1 | 1 | 115 days after full blossom | 0/28 | × | — | — | √ | [99] | ||
| — | Strawberry | Chilean | 0.25 mM | 3 | 80% flowering, turning fruit and full ripe fruit stage | 22/3 | — | — | √ | √ | [100] | |
| Sigma-Aldrich | Camarosa | 250 mmol L−1 | 1; 2; 3 | 100% red stage; large green andafter 7 days at 100% red receptacle stages; flowering, after 24 days at the large green, and after 7 days at 100% red receptacle stages | 25/3 | — | — | √ | √ | [22] | ||
| Sigma-Aldrich | Pomegranate | Malas | 1 or 2 mM | 1 | 15 days before harvest | 4/80 | √ | √ | — | √ | [73] | |
| Sigma-Aldrich | Mollar de Elche | 1, 5 or 10 mmol L−1 | 4 | 94, 64, 34 and 4 days before harvest | 10/60 | — | — | — | √ | [78] | ||
| Prohydrojasmon | — | Mango | Kent, Shelly and Maya | 0.1%, 0.2% or 0.4% | 2 | 4 and 2 weeks before harvest | 12/21 | √ | × | — | × | [26] |
| Chitosan | Oxford Laboratory Reagents | Apricot | Canino | 1.5% or 2.5% | 2 | 30 and 15 days before harvest | 0/28 | × | √ | × | √ | [37] |
| — | Grape | Jingxiu | 1 g L−1 | 1 | 10 days before harvest | 20/16;0/42 | × | √ | × | — | [40] | |
| — | Yaghouti | 2% or 3% | 3 | fruit set, 25 and 50 days after fruit set | -/40 | √ | √ | √ | √ | [41] | ||
| — | Strawberry | Alba and Romina | 0.5% or 1% | 5 | flowering and followed every 5 days | 0.5/7 | — | — | — | — | [101] | |
| — | Chilean | 1.5% | 3 | 80% flowering, turning fruit stage and full ripe fruit stage | 22/3 | — | — | √ | √ | [100] | ||
| Nova-Chem | Seascape | 2, 4, or 6 g L−1 | 1 | just turning red | 3/28;13/35 | × | — | √ | [102] | |||
| Huarun Bioengineering | Kiwifruit | Guichang | 28.6% | 3 | budding phase, fruit setting phase and expanding final phase | 25/25 | × | × | — | √ | [42] | |
| Sigma-Aldrich | Garmrok | 100 or 500 mg·L−1 | 4 | 146, 154, 161 and 170 daysafter full blossom | 0/90 | × | × | — | √ | [103] | ||
| — | Palm | Khesab | 1% | 3 | 5 and 15 weeks from pollination and two weeks before harvest | 2/60 | × | — | — | — | [38] | |
| Chitosan oligochitosan | — | Apricot | Xiaobai | 0.05% | 2 | 7 and 2 days before harvest | 2/70 | × | — | — | — | [7] |
| Haidebei Marine Bioengineering | Jujube | Dongzao | 0.7 g L−1 2 kDa, 5 kDa or 10 kDa; 0.3, 0.7 or 1.0 g L−1 10 kDa | 4 | 30, 60, 90 and 110 days after full blossom | 0/60 | — | — | — | √ | [104] | |
| β-aminobutyric acid | — | Apple | Honeycrisp | 40 mM | 2 | 4, 2 and 1 weeks before harvest | 0.5 or 3/4 months; 0.5/5 months | — | √ | — | √ | [75] |
| Sigma-Aldrich | Blueberry | Bluecrop | 20 mM | 1 | 7 days before harvest | 2/20 | × | √ | — | √ | [45] | |
| Oxalic acid | Sigma-Aldrich | Lemon | Fino | 0.1, 0.5 or 1.0 mM | 5 | from physiological fruit dropto 3 days before harvest | 10/35 | √ | √ | — | √ | [52] |
| — | Apricot | Red Flesh | 0.5, 1 or 2 mM | 1 | fruit set stage | 25/5 | × | √ | × | √ | [51] | |
| — | Peach | Anjirymaleki | 1, 3 or 5 mmol L−1 | 1 | 15 days before harvest | 1/28 | — | — | — | √ | [46] | |
| — | Kiwifruit | Bruno | 5 mM | 3 | 130 days after full blossom and 2 times at 7 days intervals | 20/15 | × | × | — | √ | [47] | |
| — | Bruno | 5 mmol L−1 | 3 | 130, 137 and 144 days after the flowering | 20/13 | × | × | — | — | [49] | ||
| Sigma-Aldrich | Plum | Black Splendor | 1 mM | 3 | 63, 77 and 98 days after full blossom | 2/35 | × | √ | × | √ | [20] | |
| Hexanal | — | Apple | Honeycrisp | 0.02% | 2 | 30 and 15 days before harvest | 2.5/120 | √ | — | — | √ | [105] |
| — | Honeycrisp | 0.02% | 2 | 30 and 15 days before harvest | 2.5/120 | √ | — | — | √ | [106] | ||
| — | Mango | Dashehari | 800, 1200, 1600 or 2000 µM | 2 | 15 and 30 days before harvest | 12/35 | √ | √ | — | — | [85] | |
| Sigma-Aldrich | Neelum, Bangalora, Banganapalli and Alphonso | 0.02% | 1; 1; 2 | 15 days before harvest; 30 days before harvest; 15 and 30 days before harvest | 25/-;13/- | × | — | — | √ | [21] | ||
| — | Alphonso and Banganapalli | 2% | 2 | 30 and 15 days before harvest | 14/21; 28/21 | √ | √ | √ | — | [107] | ||
| — | Nectarines | Fantasia | 2% | 2 | 15 and 10 days before harvest | 2/45 | — | — | — | √ | [66] | |
| — | Strawberry | Jewel and Wendy | 0.01% or 0.02% | 2 | 7 and 3 days before harvest | 4/9 | — | × | — | √ | [108] | |
| — | Guava | Allahabad Safeda | 0.8, 1.2 or 1.6 mM | 2 | 4 and 2 weeks before harvest | 6–8/35 | √ | √ | — | √ | [23] | |
| Putrescine and spermidine | Olhoghi and Rishbaba | Grape | Olhoghi and Rishbaba | 1 or 2 mM/1 or 2 mM | 2 | 40 and 20 days before harvest | 1.5/55 | × | √ | — | — | [58] |
| Melatonin | — | Apricot | Colorado and Mikado | 0.1 mM | 3 | pit hardening, final fruit growth and 4 days before harvest | 1 or 8/28 | √ | × | — | √ | [60] |
a √ indicates an increase. b × indicates a decrease. c — indicates no assessment.
4.2. Maintaining Sugarand Organic Acid
The ratio of sugars and organic acids is an important characteristic of fruit quality. Generally, total soluble solid (TSS) and titratable acid (TA) content are used to indicate sugars and organic acids in fruit. Preharvest elicitors spray can delay the increase of TSS content and maintain TA content [69].
In climacteric fruit, TSS content increases with ripening and then decreases with senescence, while TA only decreases after harvest [109,110]. On the 6th week of storage, compared with the control, TSS and TSS/TA content were 12.3% and 31.56% lower, while TA content was 23.19% higher in ‘Flordaking’ peach fruit sprayed with 3 mM SA before harvest [69]. Moreover, 0.5 mmol L−1 SA, 1 mmol L−1 ASA, or 0.5 mmol L−1 MeSA sprayed ‘Black Splendor’ plum fruit 3 times at 61, 76, and 94 days after anthesis, which increased TA content in fruit by 44.09%, 39.48% and 35.48% on the 50th of storage compared with the control, respectively [19]. In non-climacteric fruit, the concentration of TSS and TA gradually declined after harvest. Higher TSS and TA content were found in ‘Magenta’ and ‘Crimson’ seedless grapefruit sprayed with 1 mM ASA before harvest [50]. TSS content in ‘Fino’ lemon was 11% higher than that of the control on the 35th of storage after spraying with 0.1 mM MeJA 4 times before harvest [14]. Preharvest elicitors spray o maintaining TSS and TA in harvested fruit are shown in Table 3.
In fruits, sugars are stored in the form of starch, but there are few reports on the effect of preharvest elicitors spray on the degradation or accumulation of starch. However, preharvest elicitors spray delays ethylene biosynthesis and fruit ripening. Meanwhile, the TSS accumulation in fruit is delayed, which is associated with the inhibition of sucrose conversion. Higher sucrose content was found in ‘Kate’ apricot preharvest sprayed with MeSA during 32 days of storage [72]. In addition, a higher accumulation of sucrose, glucose, and fructose was found in ‘Fino’ lemon fruit preharvest sprayed with SA during 35 days of storage [14]. During 60 days of storage, preharvest MeJA spray kept fructose and glucose content in ‘Mollar de Elche’ pomegranate [17]. With fruit ripening and, organic acids are consumed as substrates of the tricarboxylic acid cycle (TCA) cycle in fruit. Preharvest elicitors spray delays respiration and fruit ripening [69]. Indeed, during 32 days of storage, the concentration of citric acid, malic acid, and total organic acids was higher in ‘Kate’ apricot fruit preharvest sprayed with MeSA [72]. Similarly, preharvest MeJA spray maintained the concentration of malic acid, succinic acid, oxalic acid, and ascorbic acid in ‘Mollar de Elche’ pomegranate during 60 days of storage [17].
4.3. Maintaing Fruit Firmness
Softening is an important cause of quality deterioration and shortens shelf life in climacteric and non-climacteric fruit. Preharvest elicitors spray can maintain firmness in fruit during storage.1, 2 or 3 mM SA sprayed ‘Flordaking’ peach fruit 3 times before harvest maintained the firmness during storage. On the 6th week of storage, the firmness in peach fruit sprayed with 3 mM SA was 1.68 fold higher than that of the control [69]. Moreover, 0.05% chitosan oligochitosan or 1 mmol L−1 SA sprayed twice on ‘Xiaobai’ apricot fruit at 7 and 2 days before harvest increased the firmness in fruit by 24% and 35% compared to the control on the 14th day of storage, respectively [7]. Similarly, the firmness in the ‘Mahachanok’ mango sprayed with 20, 40, 80, or 120 μL mL−1 MeJA at 90 days after anthesis was higher than that of the control during storage. Among them, 80 μL mL−1 MeJA spray showed the best effect, which increased the firmness by 20.75% compared with the control on the 24th of storage [97]. Compared with the control, the firmness showed 2 folds higher in ‘Flame Seedless’ table grapes sprayed with 2.0 mM SA twice at the pea stage and veraison stage on the 75th day of storage [91]. Preharvest elicitors spray on maintaining firmness in harvested fruit is shown in Table 3. This delay in fruit softening that was observed in the preharvest spray of elicitors could be connected to the inhibition in the ethylene released and the respiration rate that delayed the fruit ripening.
Fruit respiration is associated with the degree of water transpiration and weight loss [111]. Preharvest elicitors spray inhibited the water transpiration in fruit after harvest [92]. This is correlated to the inhibition of respiration that was observed in fruit preharvest treated with elicitors. On the other hand, preharvest application of elicitors strengthened the structure of the epidermal tissue of fruit and enhanced the thickness of the cuticle and density of wax, thereby further inhibiting water transpiration in harvested fruit [39,112]. Additionally, some elicitors spray, such as chitosan, can form a protective film on the surface of the fruit, which could further hinder water transpiration of the fruit [7].
Fruit softening also relates to the activity of cell wall degradation enzymes (CWDEs) [111]. Pectin, hemicellulose, and cellulose are the main components of the cell wall in fruit, and they can be depolymerized and degraded under the action of CWDEs, which causes the collapse of the cell wall, leading to fruit softening. The CWDEs mainly include polygalacturonases (PG), pectin methyl esterases (PME), and cellulases [44]. Cellulose content increased by 20.08% in ‘Qingxiang’ strawberry fruit preharvest sprayed with chitosan oligosaccharides compared with the control [43]. On the 7th day of storage, the activities of PME, PG, and cellulose were 26%, 17.43%, and 79.16% lower in ‘Agate’ muskmelon fruit sprayed with ASA before harvest than that in the control, respectively [113]. On the 9th day of storage, preharvest hexanal spray reduced the activities of PME and PG in ‘Neelum’ mango fruit, which was 50% and 42% lower than that of control, respectively [21]. Moreover, on the 14th day of storage, the PME activity decreased and protopectin content increased in ‘Allahabad Safeda’ guava fruit preharvest sprayed with hexanal, which was 33.3% lower and 1.3 folds higher than that of the control, respectively [23]. Compared with the control, the PME activity was 25% lower in ‘Flame Seedless’ grapefruit preharvest sprayed with SA [91]. In addition, preharvest SA and chitosan spray decreased PG activity in ‘Tupi’ blackberry fruit during storage. At the 6 days of storage, the PG activity in SA and chitosan-sprayed fruit was 5.5% and 14.4% lower than that of the control, respectively [9]. On the 21stday of storage, higher protopectin content was found in ‘Osbeck’ navel orange preharvest sprayed with oligochitosan, which was 19.6% higher than that of the control. Moreover, the sprayed fruit showed lower PG and PME activity, which was 58.3% and 30.8% lower than that of the control, respectively [44]. Furthermore, preharvest hexanal spray down-regulated PME expression in ‘Jewel’ and ‘Wendy’ strawberry fruit during storage [108]. Thus, in all, preharvest treatments with elicitors inhibit the expression of CWDE in harvested fruit during storage, while fruit firmness also involves in water transpiration and epidermal structure. Further studies are required to elucidate the relevant molecular mechanisms modulated by preharvest elicitors spray.
5. Concluding Remarks and Perspectives
In addition to inducing SAR in fruit, preharvest elicitors spray can improve phenylpropanoids metabolism and carotenoids biosynthesis, whichin crease the accumulation of flavonoids, carotenoids, and ascorbic acid, thereby enhancing antioxidant activity in harvested fruit. Antioxidant activity is an important property of fruits, which is correlated to scavenging ability of ROS. The increase in antioxidant activity maintains the integrity of the cell membrane and reduces lipid peroxidation, which is probably related to the reduced ROS in fruits, thereby improving cold tolerance of fruit. Moreover, the spray also inhibits membrane phospholipid metabolism, and maintains the function and fluidity of cell membrane in fruits, which alleviate CI in harvested fruit during cold storage reduces CI, maintaining postharvest quality of fruits. In addition, preharvest elicitors spray retards fruit ripening by inhibiting ethylene production and respiration rate, which maintains the levels of sugars and organic acids as well as firmness in harvested fruit during storage (Figure 1).
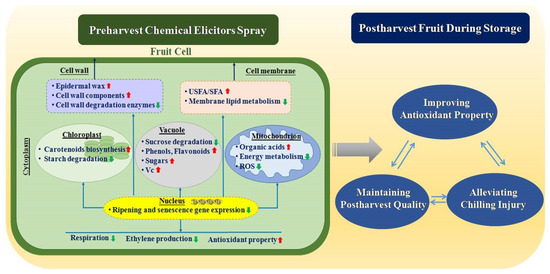
Figure 1.
A possible mode of action of preharvest elicitors spray improves antioxidant property, alleviates chilling injury, and maintains quality in harvested fruit. ROS indicates reactive oxygen species; USFS/SFA indicates unsaturated/saturated fatty acids; Vc indicates vitamin C.
A possible mode of action of how preharvest elicitors spray alleviates CI, maintains quality, and improves antioxidant capacity in harvested fruit is shown in Figure 1. Preharvest elicitors spray is a simple and effective strategy in controlling CI and maintaining quality in harvested fruit, which reduces the postharvest process and costs. However, preharvest elicitors spray still has challenges in practical agricultural applications. Firstly, most of the elicitors are registered for disease control, but only a few of them have been registered for improving antioxidant activity, controlling CI, maintaining quality, and in harvested fruit. Therefore, it is necessary to improve the register of the relevance elicitors. Secondly, there are tremendous differences in the effect of different elicitors on different species or cultivars of fruits. Therefore, it is important to screen the appropriate elicitor, concentration and application time according to the different fruits. Thirdly, fertilization, irrigation, pruning and other practices modulate the elicitors’ action. Moreover, the spraying method, such as the density and intensity of the spray, and the equipment used also affect the effect of the application of preharvest elicitor. Hence, the time, concentration, and frequency of elicitors spraying need to be screened based on different cultivation techniques. Fourthly, climate factors, such as temperature, light and rainfall, affect preharvest application. Therefore, it is important to investigate the influence of these climatic factors and their changes on the effect of applying these measures. Fifthly, postharvest environmental factors, including temperature, relative humidity, and gas composition, also affect the application effects of the elicitor, the optimum combination of pre-and postharvest environmental factors needs to be screened. Finally, although some researchers have elucidated the effects of preharvest elicitors spray on postharvest properties of fruits and revealed the preliminary mechanisms of action at physiological and cytological levels, the in-depth mechanisms of action are still unclear. Hence, approaches of multi-omic, epigenetic and molecular biology need to be used to further reveal the mechanism of preharvest elicitors spray on controlling CI and maintaining the quality of fruit during storage.
Due to the rapid cell division and significant cell expansion in fruit during the growth and development stage, the fruit is more sensitive to the stimuli of external elicitors. Additionally, preharvest fruit has a better active metabolism than the harvested fruit which is usually stored at cold temperatures. Therefore, in a way, the effects of preharvest elicitors spray on delaying ripening and senescence and improving resistance are better than that of postharvest treatment. Moreover, the elicitors are metabolized in the fruit and induce the biosynthesis of various natural metabolites that are safer than chemical fungicides, have a broader spectrum of activity, and will not be easily braked by a resistant isolate. Finally, more attention needs to be paid to strengthening the relationship between pre- and postharvest, expanding the breadth and depth of pre- and postharvest research.
Author Contributions
Writing—original draft preparation, D.G.; writing—review and editing, Y.B.; investigation, Y.L.; visualization, Y.W.; supervision, D.P. and N.A.; funding acquisition, Y.B. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by the Israel–China Project of the National Natural Science Foundation of China (31861143046). And the APC was funded by 31861143046.
Acknowledgments
This work was supported by the National Natural Science Foundation of China-Israel International Cooperation Project (31861143046).
Conflicts of Interest
The authors declare no conflict of interest.
References
- FAO. Fruit and vegetables-your dietary essentials. In The International Year of Fruits and Vegetables, 2021, Background Paper; FAO: Rome, Italy, 2020. [Google Scholar] [CrossRef]
- Ziv, C.; Fallik, E. Postharvest storage techniques and quality evaluation of fruits and vegetables for reducing food loss. Agronomy 2021, 11, 1133. [Google Scholar] [CrossRef]
- Thompson, A.K. Chaper 17-Postharvest chemical and physical deterioration of fruit and vegetables. In Chemical Deterioration and Physical Instability of Food and Beverages; Skibsted, L.H., Risbo, J., Andersen, M.L., Eds.; Woodhead Publishing: London, UK, 2010; pp. 483–518. [Google Scholar]
- Aghdam, M.S.; Asghari, M.; Babalar, M.; Sarcheshmeh, M.A.A. Chapter 8-Impact of salicylic acid on postharvest physiology of fruits and vegetables. In Eco-Friendly Technology for Postharvest Produce Quality; Siddiqui, M.W., Ed.; Academic Press: Cambridge, MA, USA, 2016; pp. 243–268. [Google Scholar]
- Sevillano, L.; Sanchez-Ballesta, M.T.; Romojaro, F.; Flores, F.B. Physiological, hormonal and molecular mechanisms regulating chilling injury in horticultural species. Postharvest technologies applied to reduce its impact. J. Sci. Food Agric. 2009, 89, 555–573. [Google Scholar] [CrossRef]
- Romanazzi, G.; Sanzani, S.M.; Bi, Y.; Tian, S.; Guti´errez Martínez, P.; Alkan, N. Induced resistance to control postharvest decay of fruit and vegetables. Postharvest Biol. Technol. 2016, 122, 82–94. [Google Scholar] [CrossRef]
- Cui, K.B.; Shu, C.; Zhao, H.D.; Fan, X.G.; Cao, J.K.; Jiang, W.B. Preharvest chitosan oligochitosan and salicylic acid treatments enhance phenol metabolism and maintain the postharvest quality of apricots (Prunus armeniaca L.). Sci. Hortic. 2020, 267, 109334. [Google Scholar] [CrossRef]
- Ahmad, S.; Singh, Z.; Khan, A.S.; Iqbal, Z. Pre-harvest application of salicylic acid maintain the rind textural properties and reduce fruit rot and chilling injury of sweet orange during cold storage. Pak. J Agric. Sci. 2013, 50, 559–569. [Google Scholar]
- Martínez-Camacho, J.E.; Guevara-González, R.G.; Rico-García, E.; Tovar-Pérez, E.G.; Torres-Pacheco, I. Delayed senescence and marketability index preservation of blackberry fruit by preharvest application of chitosan and salicylic acid. Front. Plant Sci. 2022, 13, 796393. [Google Scholar] [CrossRef]
- Segado, P.; Domínguez, E.; Heredia, A. Ultrastructure of the epidermal cell wall and cuticle of tomato fruit (Solanum lycopersicum L.) during development. Plant Physiol. 2016, 170, 935–946. [Google Scholar] [CrossRef]
- Seymour, G.B.; Østergaard, L.; Chapman, N.H.; Knapp, S.; Martin, C. Fruit development and ripening. Annu. Rev. Plant Biol. 2013, 64, 219. [Google Scholar] [CrossRef]
- Bi, X.Y.; Liao, L.; Deng, L.J.; Jin, Z.H.; Huang, Z.H.; Sun, G.C.; Xiong, B.; Wang, Z.H. Combined transcriptome and metabolome analyses reveal candidate genes involved in Tangor (Citrus reticulata × Citrus sinensis) fruit development and quality formation. Int. J. Mol. Sci. 2022, 23, 5457. [Google Scholar] [CrossRef]
- Harasym, J.; Oledzki, R. Effect of fruit and vegetable antioxidants on total antioxidant capacity of blood plasma. Nutrition 2014, 30, 511–517. [Google Scholar] [CrossRef]
- Serna-Escolano, V.; Martínez-Romero, D.; Giménez, M.J.; Serrano, M.; García-Martínez, S.; Valero, D.; Valverde, J.M.; Zapata, P.J. Enhancing antioxidant systems by preharvest treatments with methyl jasmonate and salicylic acid leads to maintain lemon quality during cold storage. Food Chem. 2021, 338, 128044. [Google Scholar] [CrossRef] [PubMed]
- Fanyuk, M.; Kumar Patel, M.; Ovadia, R.; Maurer, D.; Feygenberg, O.; Oren-Shamir, M.; Alkan, N. Preharvest application of phenylalanine induces red color in mango and apple fruit’s skin. Antioxidants 2022, 11, 491. [Google Scholar] [CrossRef] [PubMed]
- Sudheeran, P.K.; Love, C.; Feygenberg, O.; Maurer, D.; Ovadia, R.; Oren-Shamir, M.; Alkan, N. Induction of red skin and improvement of fruit quality in ‘Kent’, ‘Shelly’ and ‘Maya’ mangoes by preharvest spraying of prohydrojasmon at the orchard. Postharvest Biol. Technol. 2019, 149, 18–26. [Google Scholar] [CrossRef]
- García-Pastor, M.E.; Serrano, M.; Guillén, F.; Giménez, M.J.; Martínez-Romero, D.; Valero, D.; Zapata, P.J. Preharvest application of methyl jasmonate increases crop yield, fruit quality and bioactive compounds in pomegranate ‘Mollar de Elche’ at harvest and during postharvest storage. J. Sci. Food Agric. 2020, 100, 145–153. [Google Scholar] [CrossRef] [PubMed]
- Alrashdi, A.M.A.; Al-Qurashi, A.D.; Awad, M.A.; Mohamed, S.A.; Alrashdi, A.A. Quality, antioxidant compounds, antioxidant capacity and enzymes activity of ‘El-Bayadi’ table grapes at harvest as affected by preharvest salicylic acid and gibberellic acid spray. Sci. Hortic. 2017, 220, 243–249. [Google Scholar] [CrossRef]
- Martínez-Esplá, A.; Serrano, M.; Valero, D.; Martínez-Romero, D.; Castillo, S.; Zapata, P.J. Enhancement of antioxidant systems and storability of two plum cultivars by preharvest treatments with salicylates. Int. J. Mol. Sci. 2017, 18, 1911. [Google Scholar] [CrossRef]
- Serrano, M.; Marínez-Esplá, A.; Giménez, M.J.; Valero, D.; Zapata, P.J.; Guillén, F.; Castillo, S. Preharvest application of oxalic acid improves antioxidant systems in plums. Acta Hortic. 2016, 19, 1194. [Google Scholar] [CrossRef]
- Preethi, P.; Soorianathasundaram, K.; Sadasakthi, A.; Subramanian, K.S.; Vijay Rakesh Reddy, S.; Paliyath, G.; Subramanian, J. Preharvest application of hexanal as a surface treatment improved the storage life and quality of mango fruits. Coatings 2021, 11, 1267. [Google Scholar] [CrossRef]
- Zuñiga, P.E.; Castañeda, Y.; Arrey-Salas, O.; Fuentes, L.; Aburto, F.; Figueroa, C.R. Methyl jasmonate applications from flowering to ripe fruit stages of strawberry (Fragaria × ananassa ‘Camarosa’) reinforce the fruit antioxidant response at post-harvest. Front. Plant Sci. 2020, 11, 538. [Google Scholar] [CrossRef]
- Gill, K.S.; Dhaliwal, H.S.; Mahajan, B.V.C.; Paliyath, G.; Boora, R.S. Enhancing postharvest shelf life and quality of guava (Psidium guajava L.) cv. Allahabad Safeda by pre-harvest application of hexanal containing aqueous formulation. Postharvest Biol. Technol. 2016, 112, 224–232. [Google Scholar] [CrossRef]
- Ruiz-García, Y.; Gómez-Plaza, E. Elicitors: A tool for improving fruit phenolic content. Agriculture 2013, 3, 33–52. [Google Scholar] [CrossRef]
- Shen, N.; Wang, T.F.; Gan, Q.; Liu, S.; Wang, L.; Jin, B. Plant flavonoids: Classification, distribution, biosynthesis, and antioxidant activity. Food Chem. 2022, 383, 132531. [Google Scholar] [CrossRef] [PubMed]
- Xie, P.D.; Yang, Y.Y.; Gong, D.; Li, Z.C.; Wang, B.; Xue, S.L.; Oyom, W.; Prusky, D.; Bi, Y. Preharvest L-phenylalanine sprays accelerated wound healing of harvested muskmelons by eliciting phenylpropanoid metabolism and enzymatic browning. Postharvest Biol. Technol. 2022, 193, 112053. [Google Scholar] [CrossRef]
- Li, S.E.; Cheng, Y.; Yan, R.; Liu, Y.; Huan, C.; Zheng, X.L. Preharvest spray with melatonin improves postharvest disease resistance in cherry tomato fruit. Postharvest Biol. Technol. 2022, 193, 112055. [Google Scholar] [CrossRef]
- Wang, X.; Cao, X.; Shang, Y.; Bu, H.; Wang, T.; Lyu, D.; Du, G. Preharvest application of prohydrojasmon affects color development, phenolic metabolism, and pigment-related gene expression in red pear (Pyrus ussuriensis). J. Sci. Food Agric. 2020, 100, 4766–4775. [Google Scholar] [CrossRef]
- Flores, G.; del Castillo, M.L.R. Influence of preharvest and postharvest methyl jasmonate treatments on flavonoid content and metabolomic enzymes in red raspberry. Postharvest Biol. Technol. 2014, 97, 77–82. [Google Scholar] [CrossRef]
- Wang, J.; Bi, Y.; Zhang, Z.; Zhang, H.; Ge, Y.H. Reduction of latent infection and enhancement of disease resistance in muskmelon by preharvest application of harpin. J. Agric. Food Chem. 2011, 59, 12527. [Google Scholar] [CrossRef]
- Li, Z.C.; Xu, X.Q.; Xue, S.L.; Gong, D.; Wang, B.; Zheng, X.Y.; Xie, P.D.; Bi, Y.; Prusky, D. Preharvest multiple sprays with chitosan promotes the synthesis and deposition of lignin at wounds of harvested muskmelons. Int. J. Biol. Macromol. 2022, 206, 167–174. [Google Scholar] [CrossRef]
- Li, Z.C.; Xue, S.L.; Xu, X.Q.; Wang, B.; Zheng, X.Y.; Li, B.J.; Xie, P.D.; Bi, Y.; Prusky, D. Preharvest multiple sprays with chitosan accelerate the deposition of suberin poly phenolic at wound sites of harvested muskmelons. Postharvest Biol. Technol. 2021, 179, 111565. [Google Scholar] [CrossRef]
- Fenech, M.; Amaya, I.; Valpuesta, V.; Botella, M.A. Vitamin C content in fruits: Biosynthesis and regulation. Front. Plant Sci. 2019, 9, 2006. [Google Scholar] [CrossRef]
- Kato, M.; Ikoma, Y.; Matsumoto, H.; Sugiura, M.; Hyodo, H.; Yano, M. Accumulation of carotenoids and expression of carotenoid biosynthetic genes during maturation in citrus fruit. Plant Physiol. 2004, 134, 824–837. [Google Scholar] [CrossRef] [PubMed]
- Landi, L.; De Miccolis Angelini, R.M.; Pollastro, S.; Feliziani, E.; Faretra, F.; Romanazzi, G. Global transcriptome analysis and identification of differentially expressed genes in strawberry after preharvest application of benzothiadiazole and chitosan. Front. Plant Sci. 2017, 8, 235. [Google Scholar] [CrossRef] [PubMed]
- Martínez-Esplá, A.; Zapata, P.J.; Valero, D.; Martínez-Romero, D.; Díaz-Mula, H.M.; Serrano, M. Preharvest treatments with salicylates enhance nutrient and antioxidant compounds in plum at harvest and after storage. J. Sci. Food Agric. 2018, 98, 2742–2750. [Google Scholar] [CrossRef] [PubMed]
- Elmenofy, H.M.; Okba, S.K.; Salama, A.M.; Alam-Eldein, S.M. Yield, fruit quality, and storability of ‘Canino’ apricot in response to aminoethoxyvinylglycine, salicylic acid, and chitosan. Plants 2021, 10, 1838. [Google Scholar] [CrossRef]
- Ahmed, Z.F.R.; Alblooshi, S.S.N.A.; Kaur, N.; Maqsood, S.; Schmeda-Hirschmann, G. Synergistic effect of preharvest spray application of natural elicitors on storage life and bioactive compounds of date palm (Phoenix dactylifera L., cv. Khesab). Horticulturae 2021, 7, 145. [Google Scholar] [CrossRef]
- Zhang, Z.K.; Bi, Y.; Ge, Y.H.; Wang, J.J.; Deng, J.J.; Xie, D.F.; Wang, Y. Multiple pre-harvest treatments with acibenzolar-S-methyl reduce latent infection and induce resistance in muskmelon fruit. Sci. Hortic. 2011, 130, 126–132. [Google Scholar] [CrossRef]
- Meng, X.H.; Li, B.Q.; Liu, J.; Tian, S.P. Physiological responses and quality attributes of table grape fruit to chitosan preharvest spray and postharvest coating during storage. Food Chem. 2008, 106, 501–508. [Google Scholar] [CrossRef]
- Nia, A.E.; Taghipour, S.; Siahmansour, S. Pre-harvest application of chitosan and postharvest Aloe vera gel coating enhances quality of table grape (Vitis vinifera L. cv. ‘Yaghouti’) during postharvest period. Food Chem. 2021, 347, 129012. [Google Scholar] [CrossRef]
- Zhang, C.; Long, Y.H.; Wang, P.Q.; Li, J.H.; An, H.M.; Wu, X.M.; Li, M. The effect of preharvest 28.6% chitosan composite film sprays for controlling the soft rot on kiwifruit and its defence responses. Hortic. Sci. 2019, 46, 180–194. [Google Scholar] [CrossRef]
- He, Y.; Bose, S.K.; Wang, W.; Jia, X.; Lu, H.; Yin, H. Pre-harvest treatment of chitosan oligosaccharides improved strawberry fruit quality. Int. J. Mol. Sci. 2018, 19, 2194. [Google Scholar] [CrossRef]
- Deng, L.L.; Zhou, Y.H.; Zeng, K.F. Pre-harvest spray of oligochitosan induced the resistance of harvested navel oranges to anthracnose during ambient temperature storage. Crop. Prot. 2015, 70, 70–76. [Google Scholar] [CrossRef]
- Chea, S.; Yu, D.J.; Park, J.; Oh, H.D.; Chung, S.W.; Lee, H.J. Preharvest β-aminobutyric acid treatment alleviates postharvest deterioration of ‘Bluecrop’ highbush blueberry fruit during refrigerated storage. Sci. Hortic. 2019, 246, 95–103. [Google Scholar] [CrossRef]
- Razavi, F.; Hajilou, J. Enhancement of postharvest nutritional quality and antioxidant capacity of peach fruits by preharvest oxalic acid treatment. Sci. Hortic. 2016, 200, 95–101. [Google Scholar] [CrossRef]
- Zhu, Y.Y.; Yu, J.; Brecht, J.K.; Jiang, T.J.; Zheng, X.L. Pre-harvest application of oxalic acid increases quality and resistance to Penicillium expansum in kiwifruit during postharvest storage. Food Chem. 2016, 190, 537–543. [Google Scholar] [CrossRef] [PubMed]
- Martinez-Espla, A.; Zapata, P.J.; Valero, D.; Garcia-Viguera, C.; Castillo, S.; Serrano, M. Preharvest application of oxalic acid increased fruit size, bioactive compounds, and antioxidant capacity in sweet cherry cultivars (Prunus avium L.). J. Agric. Food Chem. 2014, 62, 3432–3437. [Google Scholar] [CrossRef] [PubMed]
- Ali, M.; Liu, M.M.; Wang, Z.E.; Li, S.E.; Jiang, T.J.; Zheng, X.L. Pre-harvest spraying of oxalic acid improves postharvest quality associated with increase in ascorbic acid and regulation of ethanol fermentation in kiwifruit cv. Bruno during storage. J. Integr. Agric. 2019, 18, 2514–2520. [Google Scholar] [CrossRef]
- García-Pastor, M.E.; Zapata, P.J.; Castillo, S.; Martínez-Romero, D.; Valero, D.; Serrano, M.; Guillén, F. Preharvest salicylate treatments enhance antioxidant compounds, color and crop yield in low pigmented-table grape cultivars and preserve quality traits during storage. Antioxidants 2020, 9, 832. [Google Scholar] [CrossRef]
- Ahmed, M.; Ullah, S.; Razzaq, K.; Rajwana, I.A.; Akhtar, G.; Naz, A.; Amin, M.; Khalid, M.S.; Khalid, S. Pre-harvest oxalic acid application improves fruit size at harvest, physico-chemical and sensory attributes of ‘Red Flesh’ apricot during fruit ripening. J. Hortic. Sci. Technol. 2021, 4, 48–55. [Google Scholar] [CrossRef]
- Serna-Escolano, V.; Giménez, M.J.; Castillo, S.; Valverde, J.M.; Martínez-Romero, D.; Guillén, F.; Serrano, M.; Valero, D.; Zapata, P.J. Preharvest Treatment with Oxalic Acid Improves Postharvest Storage of Lemon Fruit by Stimulation of the Antioxidant System and Phenolic Content. Antioxidants 2021, 10, 963. [Google Scholar] [CrossRef]
- Wang, J.; Bi, Y.; Wang, Y.; Deng, J.; Zhang, H.; Zhang, Z. Multiple preharvest treatments with harpin reduce postharvest disease and maintain quality in muskmelon fruit (cv. Huanghemi). Phytoparasitica 2014, 42, 155–163. [Google Scholar] [CrossRef]
- Misran, A.; Padmanabhan, P.; Sullivan, J.A.; Khanizadeh, S.; Paliyath, G. Composition of phenolics and volatiles in strawberry cultivars and influence of preharvest hexanal treatment on their profiles. Can. J. Plant Sci. 2015, 95, 115–126. [Google Scholar] [CrossRef]
- Wang, B.; He, X.F.; Bi, Y. Preharvest sprays with sodium nitroprusside induce resistance in harvested muskmelon against the pink rot disease. J. Food Process Preserv. 2021, 45, e15339. [Google Scholar] [CrossRef]
- Wang, B.; Jiang, H.; Bi, Y.; He, X.F.; Wang, Y.; Li, Y.C.; Zheng, X.Y.; Prusky, D. Preharvest multiple sprays with sodium nitroprusside promote wound healing of harvested muskmelons by activation of phenylpropanoid metabolism. Postharvest Biol. Technol. 2019, 158, 110988. [Google Scholar] [CrossRef]
- Saba, M.K.; Moradi, S. Sodium nitroprusside (SNP) spray to maintain fruit quality and alleviate postharvest chilling injury of peach fruit. Sci. Hortic. 2017, 216, 193–199. [Google Scholar] [CrossRef]
- Mirdehghan, S.H.; Rahimi, S. Pre-harvest application of polyamines enhances antioxidants and table grape (Vitis vinifera L.) quality during postharvest period. Food Chem. 2016, 196, 1040–1047. [Google Scholar] [CrossRef] [PubMed]
- Babalar, M.; Pirzad, F.; Sarcheshmeh, M.A.A.; Talaei, A.; Lessani, H. Arginine treatment attenuates chilling injury of pomegranate fruit during cold storage by enhancing antioxidant system activity. Postharvest Biol. Technol. 2018, 137, 31–37. [Google Scholar] [CrossRef]
- Medina-Santamarina, J.; Zapata, P.J.; Valverde, J.M.; Valero, D.; Serrano, M.; Guillén, F. Melatonin treatment of apricot trees leads to maintenance of fruit quality attributes during storage at chilling and non-chilling temperatures. Agronomy 2021, 11, 917. [Google Scholar] [CrossRef]
- Michailidis, M.; Tanou, G.; Sarrou, E.; Karagiannis, E.; Ganopoulos, I.; Martens, S.; Molassiotis, A. Pre-and post-harvest melatonin application boosted phenolic compounds accumulation and altered respiratory characters in sweet cherry fruit. Front. Nutr. 2021, 8, 306. [Google Scholar] [CrossRef]
- Sun, H.L.; Wang, X.Y.; Shang, Y.; Wang, X.Q.; Du, G.D.; Lv, D.G. Preharvest application of melatonin induces anthocyanin accumulation and related gene upregulation in red pear (Pyrus ussuriensis). J. Integr. Agric. 2021, 20, 2126–2137. [Google Scholar] [CrossRef]
- Andrys, D.; Kulpa, D.; Grzeszczuk, M.; Bihun, M.; Dobrowolska, A. Antioxidant and antimicrobial activities of Lavandulaangustifolia Mill. field-grown and propagated in vitro. Folia Hortic. 2017, 29, 161–180. [Google Scholar] [CrossRef]
- Gupta, M.; Karmakar, N.; Sasmal, S.; Chowdhury, S.; Biswas, S. Free radical scavenging activity of aqueous and alcoholic ex-tracts of Glycyrrhizaglabra Linn. measured by ferric reducing antioxidant power (FRAP), ABTS bleaching assay (αTEAC), DPPH assay and peroxyl radical antioxidant assay. Int. J. Pharmacol. Toxicol. 2016, 4, 235–240. [Google Scholar] [CrossRef]
- Zhang, W.L.; Jiang, H.T.; Cao, J.K.; Jiang, W.B. Advances in biochemical mechanisms and control technologies to treat chilling injury in postharvest fruits and vegetables. Trends Food Sci. Technol. 2021, 113, 355–365. [Google Scholar] [CrossRef]
- Kumar, S.K.; Kayal, W.E.; Sullivan, J.A.; Paliyath, G.; Jayasankar, S. Pre-harvest application of hexanal formulation enhances shelf life and quality of ‘Fantasia’ nectarines by regulating membrane and cell wall catabolism-associated genes. Sci. Hortic. 2018, 229, 117–124. [Google Scholar] [CrossRef]
- Ahmad, S.; Singh, Z.; Iqbal, Z. Effect of preharvest sprays of salicylic acid on the shelf life and quality of ‘Lane Late’ sweet orange (Citrus sinensis L.) cold storage. Acta Hortic. 2013, 1012, 103–112. [Google Scholar] [CrossRef]
- Ahmed, W.; Ahmed, S.; Ali, L. Effect of pre-harvest spray of salicylic (SA) and methyl jasmonate (MeJA) on the phyrtochemicals and physiological changes during the storage of grapefruit cv. Ray ruby. Int. J. Biosci. 2015, 6, 269–282. [Google Scholar]
- Ali, I.; Wang, X.; Tareen, M.J.; Wattoo, F.M.; Qayyum, A.; Hassan, M.U.; Shafique, M.; Liaquat, M.; Asghar, S.; Hussain, T.; et al. Foliar application of salicylic acid at different phenological stages of peach fruit CV. ‘Flordaking’ improves harvest quality and reduces chilling injury during low temperature storage. Plants 2021, 10, 1981. [Google Scholar] [CrossRef]
- Lu, X.H.; Sun, D.Q.; Li, Y.H.; Shi, W.Q.; Sun, G.M. Pre- and post-harvest salicylic acid treatments alleviate internal browning and maintain quality of winter pineapple fruit. Sci. Hortic. 2011, 130, 97–101. [Google Scholar] [CrossRef]
- Al-Obeed, R.S. Jujube post-harvest fruit quality and storability in response to agro-chemicals preharvest application. Afr. J. Agric. Res. 2012, 7, 5099–5107. [Google Scholar]
- Fan, X.G.; Du, Z.L.; Cui, X.Z.; Ji, W.J.; Ma, J.T.; Li, X.L.; Wang, X.M.; Zhao, H.D.; Liu, B.D.; Guo, F.J.; et al. Preharvest methyl salicylate treatment enhance the chilling tolerance and improve the postharvest quality of apricot during low temperature storage. Postharvest Biol. Technol. 2021, 177, 111535. [Google Scholar] [CrossRef]
- Saba, K.M.; Zarei, L. Preharvest methyl jasmonate’s impact on postharvest chilling sensitivity, antioxidant activity, and pomegranate fruit quality. J. Food Biochem. 2019, 43, e12763. [Google Scholar] [CrossRef]
- García-Pastor, M.E.; Giménez, M.J.; Zapata, P.J.; Guillén, F.; Valverde, J.M.; Serrano, M.; Valero, D. Preharvest application of methyl salicylate, acetyl salicylic acid and salicylic acid alleviated disease caused by Botrytis cinerea through stimulation of antioxidant system in table grapes. Int. J. Food Microbiol. 2020, 334, 108807. [Google Scholar] [CrossRef] [PubMed]
- Shoffe, Y.A.; Nock, J.F.; Zhang, Y.Y.; Watkins, C.B. Pre- and post-harvest γ-aminobutyric acid application in relation to fruit quality and physiological disorder development in ‘Honeycrisp’ apples. Sci. Hortic. 2021, 289, 110431. [Google Scholar] [CrossRef]
- Hosseini, M.S.; Fakhar, Z.; Babalar, M.; Askari, M.A. Effect of pre-harvest putrescine treatment on quality and postharvest life of pear cv. Spadona. Adv. Hortic. Sci. 2017, 31, 11–17. [Google Scholar]
- Zhang, L.; Cao, X.; Wang, Z.Q.; Zhang, Z.K.; Li, J.K.; Wang, Q.; Xu, X.B. Brassinolide alleviated chilling injury of banana fruit by regulating unsaturated fatty acids and phenolic compounds. Sci. Hortic. 2022, 297, 110922. [Google Scholar] [CrossRef]
- García-Pastor, M.E.; Serrano, M.; Guillén, F.; Zapata, P.J.; Valero, D. Preharvest or a combination of preharvest and postharvest treatments with methyl jasmonate reduced chilling injury, by maintaining higher unsaturated fatty acids, and increased aril colour and phenolics content in pomegranate. Postharvest Biol. Technol. 2020, 167, 111226. [Google Scholar] [CrossRef]
- Zhang, X.; Liu, T.; Zhu, S.; Wang, D.; Sun, S.; Xin, L. Short-term hypobaric treatment alleviates chilling injury by regulating membrane fatty acids metabolism in peach fruit. J. Food Biochem. 2022, 64, e14113. [Google Scholar] [CrossRef]
- Chen, G.F.; Hou, Y.Y.; Zheng, Y.H.; Jin, P. 2,4-Epibrassinolide enhance chilling tolerance of loquat fruit by regulating cell wall and membrane fatty acid metabolism. Sci. Hortic. 2022, 295, 110813. [Google Scholar] [CrossRef]
- Gong, D.; Bi, Y.; Zhang, X.M.; Han, Z.H.; Zong, Y.Y.; Li, Y.C.; Sionov, E.; Prusky, D. Benzothiadiazole treatment inhibits membrane lipid metabolism and straight-chain volatile compound release in Penicillium expansum-inoculated apple fruit. Postharvest Biol. Technol. 2021, 181, 111671. [Google Scholar] [CrossRef]
- Song, C.B.; Wang, K.; Xiao, X.; Liu, Q.L.; Yang, M.J.; Li, X.; Feng, Y.B.; Li, S.S.; Shi, L.Y.; Chen, W.; et al. Membrane lipid metabolism influences chilling injury during cold storage of peach fruit. Food Res. Int. 2022, 157, 111249. [Google Scholar] [CrossRef]
- Hong, K.; Gong, D.; Xu, H.; Wang, S.; Jia, Z.; Chen, J.; Zhang, L. Effects of salicylic acid and nitric oxide pretreatment on the expression of genes involved in the ethylene signaling pathway and the quality of postharvest mango fruit. N. Z. J. Crop. Hortic. Sci. 2014, 42, 205–216. [Google Scholar] [CrossRef]
- Asghari, M.; Aghdam, M.S. Impact of salicylic acid on post-harvest physiology of horticultural crops. Trends Food Sci. Technol. 2010, 21, 502–509. [Google Scholar] [CrossRef]
- Kaur, K.; Kaur, G.; Brar, J.S. Pre-harvest application of hexanal formulations for improving post-harvest life and quality of mango (Mangifera indica L.) cv. Dashehari. J. Food Sci. Technol. 2020, 57, 4257–4264. [Google Scholar] [CrossRef] [PubMed]
- Alkan, N.; Fortes, A.M. Insights into molecular and metabolic events associated with fruit response to post-harvest fungal pathogens. Front. Plant Sci. 2015, 6, 889. [Google Scholar] [CrossRef] [PubMed]
- Binder, B.M. Ethylene signaling in plants. J. Biol. Chem. 2020, 295, 7710–7725. [Google Scholar] [CrossRef]
- Ruiz, K.B.; Trainotti, L.; Bonghi, C.; Ziosi, V.; Costa, G.; Torrigiani, P. Early methyl jasmonate application to peach delays fruit/seed development by altering the expression of multiple hormone-related genes. J. Plant Growth Regul. 2013, 32, 852–864. [Google Scholar] [CrossRef]
- Erogul, D.; Özsoydan, İ. Effect of pre-harvest salicylic acid treatments on the quality and shelf life of the ‘Cresthaven’ peach cultivar. Folia Hortic. 2020, 32, 221–227. [Google Scholar] [CrossRef]
- Marzouk, H.A.; Kassem, H.A. Improving yield, quality, and shelf life of Thompson seedless grapevine by preharvest foliar applications. Sci. Hortic. 2011, 130, 425–430. [Google Scholar] [CrossRef]
- Champa, W.A.H.; Gill, M.I.S.; Mahajan, B.V.C.; Arora, N.K. Preharvest salicylic acid treatments to improve quality and postharvest life of table grapes (Vitisvinífera L.) cv. Flame Seedless. J. Food Sci. Technol. 2015, 52, 3607–3616. [Google Scholar] [CrossRef]
- Lo’ay, A.A. Preharvest salicylic acid and delay ripening of ‘superior seedless’ grapes. Egypt J. Basic Appl. Sci. 2017, 4, 227–230. [Google Scholar] [CrossRef][Green Version]
- Darwish, O.S.; Ali, M.R.; Khojah, E.; Samra, B.N.; Ramadan, K.M.; El-Mogy, M.M. Pre-harvest application of salicylic acid, abscisic acid, and methyl jasmonate conserve bioactive compounds of strawberry fruits during refrigerated storage. Horticulturae 2021, 7, 568. [Google Scholar] [CrossRef]
- Giménez, M.J.; Serrano, M.; Valverde, J.M.; Martínez-Romero, D.; Castillo, S.; Valero, D.; Guillén, F. Preharvest salicylic acid and acetylsalicylic acid treatments preserve quality and enhance antioxidant systems during postharvest storage of sweet cherry cultivars. J. Sci. Food Agric. 2017, 97, 1220–1228. [Google Scholar] [CrossRef] [PubMed]
- Cao, J.K.; Yan, J.Q.; Zhao, Y.M.; Jiang, W.B. Effects of fourpre-harvest foliar sprays with β-aminobutyric acid or salicylic acid on the incidence of postharvest disease and induced defence responses in jujube (Zizyphus jujuba Mill.) fruit after storage. J. Hortic. Sci. Biotechnol. 2013, 88, 338–344. [Google Scholar] [CrossRef]
- Supapvanich, S.; Mitsang, P.; Youryon, P. Preharvest salicylic acid application maintains physicochemical quality of ‘Taaptimjaan’ wax apple fruit (Syzygium samarangenese) during short-term storage. Sci. Hortic. 2017, 215, 178–183. [Google Scholar] [CrossRef]
- Muengkaew, R.; Chaiprasart, P.; Warrington, I. Changing of physiochemical properties and color development of mango fruit sprayed methyl jasmonate. Sci. Hortic. 2016, 198, 70–77. [Google Scholar] [CrossRef]
- Zapata, P.J.; Martínez-Esplá, A.; Guillén, F.; Díaz-Mula, H.M.; Martínez-Romero, D.; Serrano, M.; Valero, D. Preharvest application of methyl jasmonate (MeJA) in two plum cultivars. 2. Improvement of fruit quality and antioxidant systems during postharvest storage. Postharvest Biol. Technol. 2014, 98, 115–122. [Google Scholar] [CrossRef]
- Karaman, S.; Ozturk, B.; Genc, N.; Celik, S.M. Effect of preharvest application of methyl jasmonate on fruit quality of plum (Prunus Salicina L indell cv.“Fortune”) at harvest and during cold storage. J. Food Process. Preserv. 2012, 37, 1049–1059. [Google Scholar] [CrossRef]
- Saavedra, G.M.; Figueroa, N.E.; Poblete, L.A.; Cherian, S.; Figueroa, C.R. Effects of preharvest applications of methyl jasmonate and chitosan on postharvest decay, quality and chemical attributes of Fragaria chiloensis fruit. Food Chem. 2016, 190, 448–453. [Google Scholar] [CrossRef]
- Feliziani, E.; Landi, L.; Romanazzi, G. Preharvest treatments with chitosan and other alternatives to conventional fungicides to control postharvest decay of strawberry. Carbohydr. Polym. 2015, 5, 111–117. [Google Scholar] [CrossRef]
- Bhaskara-Reddy, M.V.; Belkacemi, K.; Corcuff, R.; Castaigne, F.; Arul, J. Effect of pre-harvest chitosan sprays on post-harvest infection by Botrytis cinerea and quality of strawberry fruit. Postharvest Biol. Technol. 2000, 20, 39–51. [Google Scholar] [CrossRef]
- Kumarihami, H.M.P.C.; Kim, J.G.; Kim, Y.H.; Lee, M.; Lee, Y.S.; Kwack, Y.B.; Kim, J. Preharvest application of chitosan improves the postharvest life of ‘Garmrok’ kiwifruit through the modulation of genes related to ethylene biosynthesis, cell wall modification and lignin metabolism. Foods 2021, 10, 373. [Google Scholar] [CrossRef]
- Yan, J.Q.; Cao, J.K.; Jiang, W.B.; Zhao, Y.M. Effects of preharvest oligochitosan sprays on postharvest fungal diseases, storage quality, and defense responses in jujube (Zizyphus jujuba Mill. cv. Dongzao) fruit. Sci. Hortic. 2012, 142, 196–204. [Google Scholar] [CrossRef]
- DeBrouwer, E.J.; Sriskantharajah, K.; Kayal, W.E.; Sullivan, J.A.; Paliyath, G.; Subramanian, J. Pre-harvest hexanal spray reduces bitter pit and enhances post-harvest quality in ‘Honeycrisp’ apples (Malus domestica Borkh.). Sci. Hortic. 2020, 273, 109610. [Google Scholar] [CrossRef]
- Sriskantharajah, K.; El Kayal, W.; Torkamaneh, D.; Ayyanath, M.M.; Saxena, P.K.; Sullivan, A.J.; Paliyath, G.; Subramanian, J. Transcriptomics of improved fruit retention by hexanal in ‘Honeycrisp’ reveals hormonal crosstalk and reduced cell wall degradation in the fruit abscission zone. Int. J. Mol. Sci. 2021, 22, 8830. [Google Scholar] [CrossRef] [PubMed]
- Anusuya, P.; Nagaraj, R.; Janavi, G.J.; Subramanian, K.S.; Paliyath, G.; Subramanian, J. Pre-harvest sprays of hexanal formulation for extending retention and shelf-life of mango (Mangifera indica L.) fruits. Sci. Hortic. 2016, 211, 231–240. [Google Scholar] [CrossRef]
- El Kayal, W.; El-Sharkawy, I.; Dowling, C.; Paliyath, G.; Sullivan, J.A.; Subramanian, J. Effect of preharvest application of hexanal and growth regulators in enhancing shelf life and regulation of membrane-associated genes in strawberry. Can. J. Plant Sci. 2017, 97, 1109–1120. [Google Scholar] [CrossRef]
- Durán-Soria, S.; Pott, D.M.; Osorio, S.; Vallarino, J.G. Sugar signaling during fruit ripening. Front. Plant Sci. 2020, 11, 564917. [Google Scholar] [CrossRef]
- Batista-Silva, W.; Nascimento, V.L.; Medeiros, D.B.; Nunes-Nesi, A.; Ribeiro, D.M.; Zsögön, A.; Araújo, W.L. Modifications in organic acid profiles during fruit development and ripening: Correlation or causation? Front. Plant Sci. 2018, 9, 1689. [Google Scholar] [CrossRef]
- Liu, S.M.; Huang, H.; Huber, D.J.; Pan, Y.G.; Shi, X.Q.; Zhang, Z.K. Delay of ripening and softening in ‘Guifei’ mango fruit by postharvest application of melatonin. Postharvest Biol. Technol. 2020, 163, 111136. [Google Scholar] [CrossRef]
- Fekry, W.M.E.; Rashad, Y.M.; Alaraidh, I.A.; Mehany, T. Exogenous application of melatonin and methyl jasmonate as a pre-harvest treatment enhances growth of Barhi date palm trees, prolongs storability, and maintains quality of their fruits under storage conditions. Plants 2022, 11, 96. [Google Scholar] [CrossRef]
- Liu, Y.N.; Wang, Y.; Bi, Y.; Li, S.E.; Jiang, H.; Zhu, Y.; Wang, B. Effect of preharvest acetylsalicylic acid treatments on ripening and softening of harvested muskmelon fruit. Sci. Agric. Sin. 2017, 50, 1862–1872, (English abstract). [Google Scholar]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).