Magnetic Particles-Based Analytical Platforms for Food Safety Monitoring
Abstract
:1. Introduction
2. Synthesis of MNPs
3. Surface Modification of MNPs
4. Applications of MNPs in Bioanalysis
4.1. Enzyme Immobilization Support
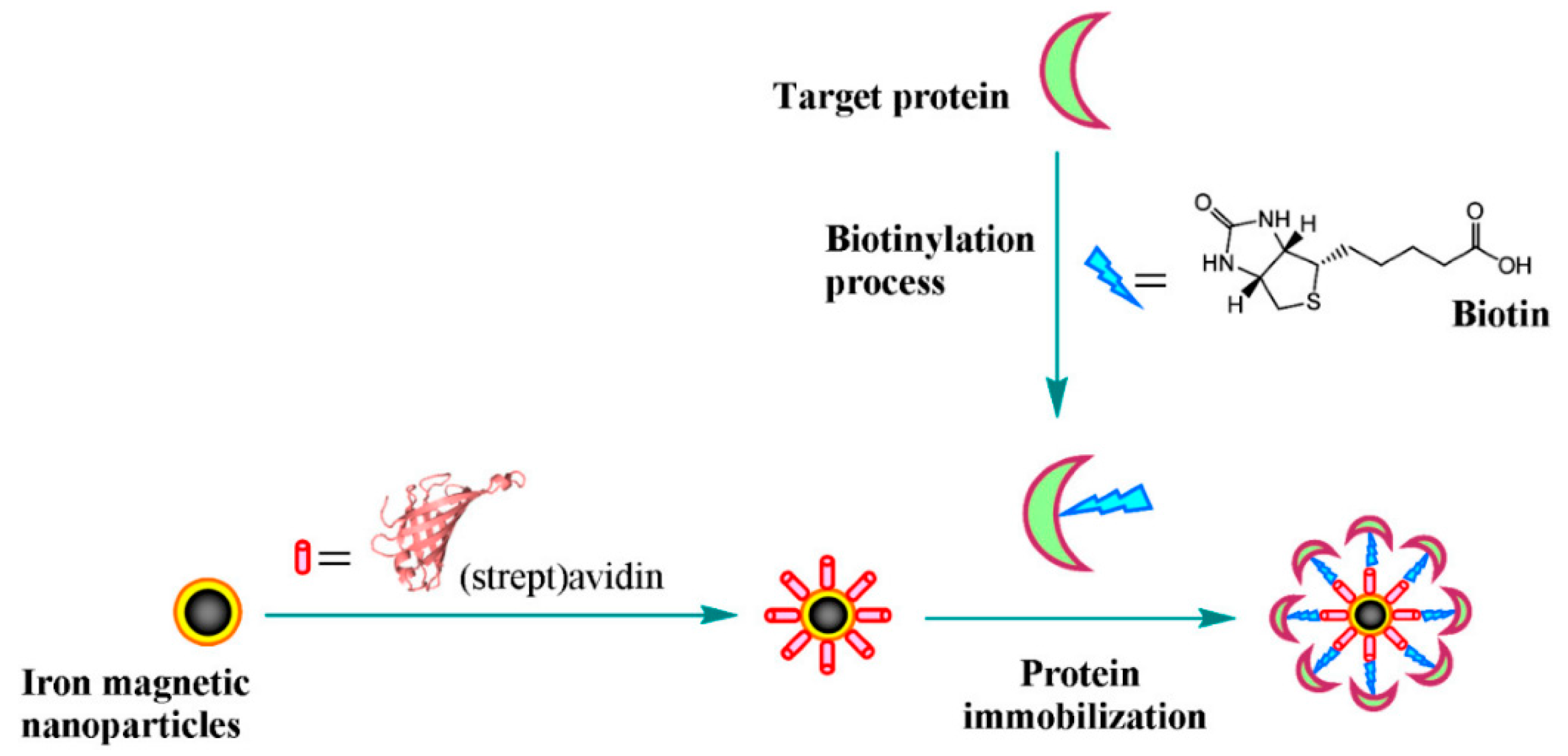
4.2. MNPs as Pre-Concentration and Capture Probe
5. Food Analysis Using MNPs
5.1. Analysis of Inorganic Species in Food Using MNPs
5.2. Analysis of Organic Species in Food Using MNPs
6. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Beveridge, J.S.; Stephens, J.R.; Williams, M.E. The Use of Magnetic Nanoparticles in Analytical Chemistry. Annu. Rev. Anal. Chem. 2011, 4, 251–273. [Google Scholar] [CrossRef] [PubMed]
- Muldoon, L.L.; Sàndor, M.; Pinkston, K.E.; Neuwelt, E.A. Imaging, distribution, and toxicity of superparamagnetic iron oxide magnetic resonance nanoparticles in the rat brain and intracerebral tumor. Neurosurgery 2005, 57, 785–796. [Google Scholar] [CrossRef] [PubMed]
- Puntes, V.F.; Krishnan, K.M.; Alivisatos, A.P. Colloidal nanocrystal shape and size control: The case of cobalt. Science 2001, 291, 2115–2117. [Google Scholar] [CrossRef] [PubMed]
- Akbarzadeh, A.; Samiei, M.; Davaran, S. Magnetic nanoparticles: Preparation, physical properties, and applications in biomedicine. Nanoscale Res. Lett. 2012, 7, 144. [Google Scholar] [CrossRef] [PubMed]
- Denizot, B.; Tanguy, G.; Hindre, F.; Rump, E.; Lejeune, J.; Jallet, P. The preparation of magnetite nanoparicles for biomedical. J. Colloid Interface Sci. 1999, 209, 10.1006. [Google Scholar]
- Wang, W.-W.; Zhu, Y.-J.; Ruan, M.-L. Microwave-assisted synthesis and magnetic property of magnetite and hematite nanoparticles. J. Nanopart. Res. 2007, 9, 419–426. [Google Scholar] [CrossRef]
- Wierucka, M.; Biziuk, M. Application of magnetic nanoparticles for magnetic solid-phase extraction in preparing biological, environmental and food samples. Trac-Trends Anal. Chem. 2014, 59, 50–58. [Google Scholar] [CrossRef]
- Latham, A.H.; Williams, M.E. Controlling transport and chemical functionality of magnetic nanoparticles. Acc. Chem. Res. 2008, 41, 411–420. [Google Scholar] [CrossRef]
- Burda, C.; Chen, X.B.; Narayanan, R.; El-Sayed, M.A. Chemistry and properties of nanocrystals of different shapes. Chem. Rev. 2005, 105, 1025–1102. [Google Scholar] [CrossRef]
- Qiao, R.R.; Yang, C.H.; Gao, M.Y. Superparamagnetic iron oxide nanoparticles: From preparations to in vivo MRI applications. J. Mater. Chem. 2009, 19, 6274–6293. [Google Scholar] [CrossRef]
- Gupta, A.K.; Gupta, M. Synthesis and surface engineering of iron oxide nanoparticles for biomedical applications. Biomaterials 2005, 26, 3995–4021. [Google Scholar] [CrossRef] [PubMed]
- Laurent, S.; Forge, D.; Port, M.; Roch, A.; Robic, C.; Elst, L.V.; Muller, R.N. Magnetic iron oxide nanoparticles: Synthesis, stabilization, vectorization, physicochemical characterizations, and biological applications. Chem. Rev. 2008, 108, 2064–2110. [Google Scholar] [CrossRef] [PubMed]
- Gao, J.H.; Gu, H.W.; Xu, B. Multifunctional Magnetic Nanoparticles: Design, Synthesis, and Biomedical Applications. Acc. Chem. Res. 2009, 42, 1097–1107. [Google Scholar] [CrossRef] [PubMed]
- Baumgartner, J.; Antonietta Carillo, M.; Eckes, K.M.; Werner, P.; Faivre, D. Biomimetic magnetite formation: From biocombinatorial approaches to mineralization effects. Langmuir 2014, 30, 2129–2136. [Google Scholar] [CrossRef] [PubMed]
- Biehl, P.; von der Lühe, M.; Dutz, S.; Schacher, F. Synthesis, characterization, and applications of magnetic nanoparticles featuring polyzwitterionic coatings. Polymers 2018, 10, 91. [Google Scholar] [CrossRef]
- Timko, M.; Molcan, M.; Hashim, A.; Skumiel, A.; Muller, M.; Gojzewski, H.; Jozefczak, A.; Kovac, J.; Rajnak, M.; Makowski, M. Hyperthermic effect in suspension of magnetosomes prepared by various methods. IEEE Trans. Magn. 2012, 49, 250–254. [Google Scholar] [CrossRef]
- Claudio, L.; Mitchell, B. Nanoparticles from Mechanical Attrition. Synthesis, Functionalization and Surface Treatment of Nanoparticles, Chapter 1; American Scientific Publishers: Valencia, CA, USA, 2002. [Google Scholar]
- LaMer, V.K.; Dinegar, R.H. Theory, production and mechanism of formation of monodispersed hydrosols. J. Am. Chem. Soc. 1950, 72, 4847–4854. [Google Scholar] [CrossRef]
- Khan, K.; Rehman, S.; Rahman, H.U.; Khan, Q. Synthesis and Application of Magnetic Nanoparticles; One Central Press (OCP): Atlantic Business Centre: Altrincham, UK, 2014; pp. 135–169. [Google Scholar]
- Kudr, J.; Haddad, Y.; Richtera, L.; Heger, Z.; Cernak, M.; Adam, V.; Zitka, O. Magnetic nanoparticles: From design and synthesis to real world applications. Nanomaterials 2017, 7, 243. [Google Scholar] [CrossRef]
- Salazar-Alvarez, G.; Qin, J.; Sepelak, V.; Bergmann, I.; Vasilakaki, M.; Trohidou, K.; Ardisson, J.; Macedo, W.; Mikhaylova, M.; Muhammed, M. Cubic versus spherical magnetic nanoparticles: The role of surface anisotropy. J. Am. Chem. Soc. 2008, 130, 13234–13239. [Google Scholar] [CrossRef]
- Jana, N.R.; Chen, Y.; Peng, X. Size-and shape-controlled magnetic (Cr, Mn, Fe, Co, Ni) oxide nanocrystals via a simple and general approach. Chem. Mater. 2004, 16, 3931–3935. [Google Scholar] [CrossRef]
- Rockenberger, J.; Scher, E.C.; Alivisatos, A.P. A new nonhydrolytic single-precursor approach to surfactant-capped nanocrystals of transition metal oxides. J. Am. Chem. Soc. 1999, 121, 11595–11596. [Google Scholar] [CrossRef]
- Zhang, H.; Ding, J.; Chow, G.; Ran, M.; Yi, J. Engineering magnetic properties of Ni nanoparticles by non-magnetic cores. Chem. Mater. 2009, 21, 5222–5228. [Google Scholar] [CrossRef]
- Sun, S.; Zeng, H.; Robinson, D.B.; Raoux, S.; Rice, P.M.; Wang, S.X.; Li, G. Monodisperse MFe2O4 (M = Fe, Co, Mn) nanoparticles. J. Am. Chem. Soc. 2004, 126, 273–279. [Google Scholar] [CrossRef] [PubMed]
- Adschiri, T.; Hakuta, Y.; Sue, K.; Arai, K. Hydrothermal synthesis of metal oxide nanoparticles at supercritical conditions. J. Nanopart. Res. 2001, 3, 227–235. [Google Scholar] [CrossRef]
- Yu, J.; Yu, X. Hydrothermal synthesis and photocatalytic activity of zinc oxide hollow spheres. Environ. Sci. Technol. 2008, 42, 4902–4907. [Google Scholar] [CrossRef]
- Yang, T.; Li, Y.; Zhu, M.; Li, Y.; Huang, J.; Jin, H.; Hu, Y. Room-temperature ferromagnetic Mn-doped ZnO nanocrystal synthesized by hydrothermal method under high magnetic field. Mater. Sci. Eng. B 2010, 170, 129–132. [Google Scholar] [CrossRef]
- Sander, D.; Oka, H.; Corbetta, M.; Stepanyuk, V.; Kirschner, J. New insights into nano-magnetism by spin-polarized scanning tunneling microscopy. J. Electron Spectrosc. Relat. Phenom. 2013, 189, 206–215. [Google Scholar] [CrossRef]
- Wu, W.; He, Q.; Chen, H.; Tang, J.; Nie, L. Sonochemical synthesis, structure and magnetic properties of air-stable Fe3O4/Au nanoparticles. Nanotechnology 2007, 18, 145609. [Google Scholar] [CrossRef]
- Ban, Z.; Barnakov, Y.A.; Li, F.; Golub, V.O.; O’Connor, C.J. The synthesis of core–shell iron@ gold nanoparticles and their characterization. J. Mater. Chem. 2005, 15, 4660–4662. [Google Scholar] [CrossRef]
- Hernández-Hernández, A.A.; Álvarez-Romero, G.A.; Contreras-López, E.; Aguilar-Arteaga, K.; Castañeda-Ovando, A. Food analysis by microextraction methods based on the use of magnetic nanoparticles as supports: Recent advances. Food Anal. Methods 2017, 10, 2974–2993. [Google Scholar] [CrossRef]
- Lu, H.; Yi, G.; Zhao, S.; Chen, D.; Guo, L.-H.; Cheng, J. Synthesis and characterization of multi-functional nanoparticles possessing magnetic, up-conversion fluorescence and bio-affinity properties. J. Mater. Chem. 2004, 14, 1336–1341. [Google Scholar] [CrossRef]
- Yang, H.H.; Zhang, S.Q.; Chen, X.L.; Zhuang, Z.X.; Xu, J.G.; Wang, X.R. Magnetite-containing spherical silica nanoparticles for biocatalysis and bioseparations. Anal. Chem. 2005, 77, 354. [Google Scholar] [CrossRef] [Green Version]
- Nakagawa, T.; Hashimoto, R.; Maruyama, K.; Tanaka, T.; Takeyama, H.; Matsunaga, T. Capture and release of DNA using aminosilane-modified bacterial magnetic particles for automated detection system of single nucleotide polymorphisms. Biotechnol. Bioeng. 2006, 94, 862–868. [Google Scholar] [CrossRef] [PubMed]
- Yoza, B.; Arakaki, A.; Maruyama, K.; Takeyama, H.; Matsunaga, T. Fully automated DNA extraction from blood using magnetic particles modified with a hyperbranched polyamidoamine dendrimer. J. Biosci. Bioeng. 2003, 95, 21–26. [Google Scholar] [CrossRef]
- Nakagawa, T.; Tanaka, T.; Niwa, D.; Osaka, T.; Takeyama, H.; Matsunaga, T. Fabrication of amino silane-coated microchip for DNA extraction from whole blood. J. Biotechnol. 2005, 116, 105–111. [Google Scholar] [CrossRef] [PubMed]
- Anker, J.N.; Kopelman, R. Magnetically modulated optical nanoprobes. Appl. Phys. Lett. 2003, 82, 1102–1104. [Google Scholar] [CrossRef] [Green Version]
- Baker, I. Magnetic nanoparticle synthesis. In Nanobiomaterials; Elsevier: Amsterdam, The Netherlands, 2018; pp. 197–229. [Google Scholar]
- Berry, C.C.; Curtis, A.S. Functionalisation of magnetic nanoparticles for applications in biomedicine. J. Phys. D Appl. Phys. 2003, 36, R198. [Google Scholar]
- Mateo, C.; Palomo, J.M.; Fernandez-Lorente, G.; Guisan, J.M.; Fernandez-Lafuente, R. Improvement of enzyme activity, stability and selectivity via immobilization techniques. Enzym. Microb. Technol. 2007, 40, 1451–1463. [Google Scholar] [CrossRef]
- Bolivar, J.M.; Wilson, L.; Ferrarotti, S.A.; Fernandez-Lafuente, R.; Guisan, J.M.; Mateo, C. Stabilization of a formate dehydrogenase by covalent immobilization on highly activated glyoxyl-agarose supports. Biomacromolecules 2006, 7, 669–673. [Google Scholar] [CrossRef]
- Li, Y.; Xu, X.; Deng, C.; Yang, P.; Zhang, X. Immobilization of trypsin on superparamagnetic nanoparticles for rapid and effective proteolysis. J. Proteome Res. 2007, 6, 3849–3855. [Google Scholar] [CrossRef]
- Oh, C.; Lee, J.-H.; Lee, Y.-G.; Lee, Y.-H.; Kim, J.-W.; Kang, H.-H.; Oh, S.-G. New approach to the immobilization of glucose oxidase on non-porous silica microspheres functionalized by (3-aminopropyl) trimethoxysilane (APTMS). Colloids Surf. B Biointerfaces 2006, 53, 225–232. [Google Scholar] [CrossRef] [PubMed]
- Palomo, J.M.; Muñoz, G.; Fernández-Lorente, G.; Mateo, C.; Fuentes, M.; Guisan, J.M.; Fernández-Lafuente, R. Modulation of Mucor miehei lipase properties via directed immobilization on different hetero-functional epoxy resins: Hydrolytic resolution of (R, S)-2-butyroyl-2-phenylacetic acid. J. Mol. Catal. B Enzym. 2003, 21, 201–210. [Google Scholar] [CrossRef]
- Kim, J.; Grate, J.W.; Wang, P. Nanostructures for enzyme stabilization. Chem. Eng. Sci. 2006, 61, 1017–1026. [Google Scholar] [CrossRef]
- Liu, X.; Lei, L.; Li, Y.; Zhu, H.; Cui, Y.; Hu, H. Preparation of carriers based on magnetic nanoparticles grafted polymer and immobilization for lipase. Biochem. Eng. J. 2011, 56, 142–149. [Google Scholar] [CrossRef]
- Xie, T.; Wang, A.; Huang, L.; Li, H.; Chen, Z.; Wang, Q.; Yin, X. Recent advance in the support and technology used in enzyme immobilization. Afr. J. Biotechnol. 2009, 8, 4724–4733. [Google Scholar]
- Zhu, H.; Pan, J.; Hu, B.; Yu, H.-L.; Xu, J.-H. Immobilization of glycolate oxidase from Medicago falcata on magnetic nanoparticles for application in biosynthesis of glyoxylic acid. J. Mol. Catal. B Enzym. 2009, 61, 174–179. [Google Scholar] [CrossRef]
- Xu, J.; Sun, J.; Wang, Y.; Sheng, J.; Wang, F.; Sun, M. Application of iron magnetic nanoparticles in protein immobilization. Molecules 2014, 19, 11465–11486. [Google Scholar] [CrossRef]
- López-Gallego, F.; Betancor, L.; Mateo, C.; Hidalgo, A.; Alonso-Morales, N.; Dellamora-Ortiz, G.; Guisán, J.M.; Fernández-Lafuente, R. Enzyme stabilization by glutaraldehyde crosslinking of adsorbed proteins on aminated supports. J. Biotechnol. 2005, 119, 70–75. [Google Scholar] [CrossRef]
- Mateo, C.; Palomo, J.M.; Fuentes, M.; Betancor, L.; Grazu, V.; López-Gallego, F.; Pessela, B.C.; Hidalgo, A.; Fernández-Lorente, G.; Fernández-Lafuente, R. Glyoxyl agarose: A fully inert and hydrophilic support for immobilization and high stabilization of proteins. Enzym. Microb. Technol. 2006, 39, 274–280. [Google Scholar] [CrossRef]
- Chang, M.-Y.; Juang, R.-S. Use of chitosan–clay composite as immobilization support for improved activity and stability of β-glucosidase. Biochem. Eng. J. 2007, 35, 93–98. [Google Scholar] [CrossRef]
- Pan, C.; Hu, B.; Li, W.; Sun, Y.; Ye, H.; Zeng, X. Novel and efficient method for immobilization and stabilization of β-d-galactosidase by covalent attachment onto magnetic Fe3O4–chitosan nanoparticles. J. Mol. Catal. B Enzym. 2009, 61, 208–215. [Google Scholar] [CrossRef]
- Huang, S.H.; Liao, M.H.; Chen, D.H. Direct binding and characterization of lipase onto magnetic nanoparticles. Biotechnol. Prog. 2003, 19, 1095–1100. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Bai, S.; Sun, Y. Preparation of magnetic nanoparticles and its application to enzyme immobilization. Chin. J. Process Eng. 2004, 4, 362–366. [Google Scholar]
- Lu, A.H.; Salabas, E.e.L.; Schüth, F. Magnetic nanoparticles: Synthesis, protection, functionalization, and application. Angew. Chem. Int. Ed. 2007, 46, 1222–1244. [Google Scholar] [CrossRef] [PubMed]
- Hemmati, M.; Rajabi, M.; Asghari, A. Magnetic nanoparticle based solid-phase extraction of heavy metal ions: A review on recent advances. Microchim. Acta 2018, 185, 160. [Google Scholar] [CrossRef] [PubMed]
- Shegefti, S.; Mehdinia, A.; Shemirani, F. Preconcentration of cobalt (II) using polythionine-coated Fe3O4 nanocomposite prior its determination by AAS. Microchim. Acta 2016, 183, 1963–1970. [Google Scholar] [CrossRef]
- Bagheri, H.; Afkhami, A.; Saber-Tehrani, M.; Khoshsafar, H. Preparation and characterization of magnetic nanocomposite of Schiff base/silica/magnetite as a preconcentration phase for the trace determination of heavy metal ions in water, food and biological samples using atomic absorption spectrometry. Talanta 2012, 97, 87–95. [Google Scholar] [CrossRef]
- Chou, P.-H.; Chen, S.-H.; Liao, H.-K.; Lin, P.-C.; Her, G.-R.; Lai, A.C.-Y.; Chen, J.-H.; Lin, C.-C.; Chen, Y.-J. Nanoprobe-based affinity mass spectrometry for selected protein profiling in human plasma. Anal. Chem. 2005, 77, 5990–5997. [Google Scholar] [CrossRef]
- Li, Y.-C.; Lin, Y.-S.; Tsai, P.-J.; Chen, C.-T.; Chen, W.-Y.; Chen, Y.-C. Nitrilotriacetic acid-coated magnetic nanoparticles as affinity probes for enrichment of histidine-tagged proteins and phosphorylated peptides. Anal. Chem. 2007, 79, 7519–7525. [Google Scholar] [CrossRef]
- Huang, C.; Hu, B. Silica-coated magnetic nanoparticles modified with γ-mercaptopropyltrimethoxysilane for fast and selective solid phase extraction of trace amounts of Cd, Cu, Hg, and Pb in environmental and biological samples prior to their determination by inductively coupled plasma mass spectrometry. Spectrochim. Acta Part B At. Spectrosc. 2008, 63, 437–444. [Google Scholar]
- Shishehbore, M.R.; Afkhami, A.; Bagheri, H. Salicylic acid functionalized silica-coated magnetite nanoparticles for solid phase extraction and preconcentration of some heavy metal ions from various real samples. Chem. Cent. J. 2011, 5, 41. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Huang, C.; Hu, B. Speciation of inorganic tellurium from seawater by ICP-MS following magnetic SPE separation and preconcentration. J. Sep. Sci. 2008, 31, 760–767. [Google Scholar] [CrossRef] [PubMed]
- Huo, S.-H.; Yan, X.-P. Facile magnetization of metal–organic framework MIL-101 for magnetic solid-phase extraction of polycyclic aromatic hydrocarbons in environmental water samples. Analyst 2012, 137, 3445–3451. [Google Scholar] [CrossRef] [PubMed]
- Bianchi, F.; Chiesi, V.; Casoli, F.; Luches, P.; Nasi, L.; Careri, M.; Mangia, A. Magnetic solid-phase extraction based on diphenyl functionalization of Fe3O4 magnetic nanoparticles for the determination of polycyclic aromatic hydrocarbons in urine samples. J. Chromatogr. A 2012, 1231, 8–15. [Google Scholar] [CrossRef] [PubMed]
- Huang, C.; Xie, W.; Li, X.; Zhang, J. Speciation of inorganic arsenic in environmental waters using magnetic solid phase extraction and preconcentration followed by ICP-MS. Microchim. Acta 2011, 173, 165–172. [Google Scholar] [CrossRef]
- Zhang, N.; Peng, H.; Wang, S.; Hu, B. Fast and selective magnetic solid phase extraction of trace Cd, Mn and Pb in environmental and biological samples and their determination by ICP-MS. Microchim. Acta 2011, 175, 121. [Google Scholar] [CrossRef]
- Suleiman, J.S.; Hu, B.; Peng, H.; Huang, C. Separation/preconcentration of trace amounts of Cr, Cu and Pb in environmental samples by magnetic solid-phase extraction with Bismuthiol-II-immobilized magnetic nanoparticles and their determination by ICP-OES. Talanta 2009, 77, 1579–1583. [Google Scholar] [CrossRef]
- Zhang, N.; Peng, H.; Hu, B. Light-induced pH change and its application to solid phase extraction of trace heavy metals by high-magnetization Fe3O4@ SiO2@ TiO2 nanoparticles followed by inductively coupled plasma mass spectrometry detection. Talanta 2012, 94, 278–283. [Google Scholar] [CrossRef]
- Wang, Y.; Tian, T.; Wang, L.; Hu, X. Solid-phase preconcentration of cadmium (II) using amino-functionalized magnetic-core silica-shell nanoparticles, and its determination by hydride generation atomic fluorescence spectrometry. Microchim. Acta 2013, 180, 235–242. [Google Scholar] [CrossRef]
- Ozmen, E.Y.; Sezgin, M.; Yilmaz, A.; Yilmaz, M. Synthesis of β-cyclodextrin and starch based polymers for sorption of azo dyes from aqueous solutions. Bioresour. Technol. 2008, 99, 526–531. [Google Scholar] [CrossRef]
- Ji, Y.; Liu, X.; Guan, M.; Zhao, C.; Huang, H.; Zhang, H.; Wang, C. Preparation of functionalized magnetic nanoparticulate sorbents for rapid extraction of biphenolic pollutants from environmental samples. J. Sep. Sci. 2009, 32, 2139–2145. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Sun, Y.; Wang, Y.; Jiang, C.; Yu, X.; Gao, Y.; Zhang, H.; Song, D. Determination of Sudan dyes in environmental water by magnetic mesoporous microsphere-based solid phase extraction ultra fast liquid chromatography. Anal. Methods 2013, 5, 1399–1406. [Google Scholar] [CrossRef]
- Zhai, Y.; He, Q.; Han, Q. Solid-phase extraction of trace metal ions with magnetic nanoparticles modified with 2, 6-diaminopyridine. Microchim. Acta 2012, 178, 405–412. [Google Scholar] [CrossRef]
- Zhai, Y.; He, Q.; Yang, X.; Han, Q. Solid phase extraction and preconcentration of trace mercury (II) from aqueous solution using magnetic nanoparticles doped with 1, 5-diphenylcarbazide. Microchim. Acta 2010, 169, 353–360. [Google Scholar] [CrossRef]
- Tian, H.; Li, J.; Shen, Q.; Wang, H.; Hao, Z.; Zou, L.; Hu, Q. Using shell-tunable mesoporous Fe3O4@ HMS and magnetic separation to remove DDT from aqueous media. J. Hazard. Mater. 2009, 171, 459–464. [Google Scholar] [CrossRef] [PubMed]
- Fang, X.; Zhang, W.-W. Affinity separation and enrichment methods in proteomic analysis. J. Proteom. 2008, 71, 284–303. [Google Scholar] [CrossRef] [PubMed]
- Sahu, S.K.; Chakrabarty, A.; Bhattacharya, D.; Ghosh, S.K.; Pramanik, P. Single step surface modification of highly stable magnetic nanoparticles for purification of His-tag proteins. J. Nanopart. Res. 2011, 13, 2475–2484. [Google Scholar] [CrossRef]
- Zhang, G.; Cao, Q.; Li, N.; Li, K.; Liu, F. Tris (hydroxymethyl) aminomethane-modified magnetic microspheres for rapid affinity purification of lysozyme. Talanta 2011, 83, 1515–1520. [Google Scholar] [CrossRef]
- Vijayalakshmi, M. Pseudo-biospecific affinity ligand chromatography. In Molecular Interactions in Bioseparations; Springer: Berlin, Germany, 1993; pp. 257–275. [Google Scholar]
- Hilbrig, F.; Freitag, R. Protein purification by affinity precipitation. J. Chromatogr. B 2003, 790, 79–90. [Google Scholar] [CrossRef]
- Batalha, I.L.; Hussain, A.; Roque, A. Gum Arabic coated magnetic nanoparticles with affinity ligands specific for antibodies. J. Mol. Recognit. 2010, 23, 462–471. [Google Scholar] [CrossRef]
- Huang, S.-H.; Liao, M.-H.; Chen, D.-H. Fast and efficient recovery of lipase by polyacrylic acid-coated magnetic nano-adsorbent with high activity retention. Sep. Purif. Technol. 2006, 51, 113–117. [Google Scholar] [CrossRef]
- Meyer, A.; Hansen, D.B.; Gomes, C.S.; Hobley, T.J.; Thomas, O.R.; Franzreb, M. Demonstration of a strategy for product purification by high-gradient magnetic fishing: Recovery of superoxide dismutase from unconditioned whey. Biotechnol. Prog. 2005, 21, 244–254. [Google Scholar] [CrossRef] [PubMed]
- Ma, Z.-Y.; Liu, X.-Q.; Guan, Y.-P.; Liu, H.-Z. Synthesis of magnetic silica nanospheres with metal ligands and application in affinity separation of proteins. Colloids Surf. A Physicochem. Eng. Asp. 2006, 275, 87–91. [Google Scholar] [CrossRef]
- Chen, L.; Guo, C.; Guan, Y.; Liu, H. Isolation of lactoferrin from acid whey by magnetic affinity separation. Sep. Purif. Technol. 2007, 56, 168–174. [Google Scholar] [CrossRef]
- Chen, D.-H.; Huang, S.-H. Fast separation of bromelain by polyacrylic acid-bound iron oxide magnetic nanoparticles. Process Biochem. 2004, 39, 2207–2211. [Google Scholar] [CrossRef]
- Başar, N.; Uzun, L.; Güner, A.; Denizli, A. Lysozyme purification with dye-affinity beads under magnetic field. Int. J. Biol. Macromol. 2007, 41, 234–242. [Google Scholar] [CrossRef] [PubMed]
- Sun, J.; Su, Y.; Rao, S.; Yang, Y. Separation of lysozyme using superparamagnetic carboxymethyl chitosan nanoparticles. J. Chromatogr. B 2011, 879, 2194–2200. [Google Scholar] [CrossRef]
- Liao, M.-H.; Chen, D.-H. Fast and efficient adsorption/desorption of protein by a novel magnetic nano-adsorbent. Biotechnol. Lett. 2002, 24, 1913–1917. [Google Scholar] [CrossRef]
- Speroni, F.; Elviri, L.; Careri, M.; Mangia, A. Magnetic particles functionalized with PAMAM-dendrimers and antibodies: A new system for an ELISA method able to detect Ara h3/4 peanut allergen in foods. Anal. Bioanal. Chem. 2010, 397, 3035–3042. [Google Scholar] [CrossRef]
- Yang, H.; Qu, L.; Wimbrow, A.N.; Jiang, X.; Sun, Y. Rapid detection of Listeria monocytogenes by nanoparticle-based immunomagnetic separation and real-time PCR. Int. J. Food Microbiol. 2007, 118, 132–138. [Google Scholar] [CrossRef]
- Yang, K.; Jenkins, D.M.; Su, W.W. Rapid concentration of bacteria using submicron magnetic anion exchangers for improving PCR-based multiplex pathogen detection. J. Microbiol. Methods 2011, 86, 69–77. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Chen, L.; Xu, Y.; Wang, H.; Zeng, Q.; Zhao, Q.; Ren, N.; Ding, L. Determination of β-lactam antibiotics in milk based on magnetic molecularly imprinted polymer extraction coupled with liquid chromatography–tandem mass spectrometry. J. Chromatogr. B 2010, 878, 3421–3426. [Google Scholar] [CrossRef] [PubMed]
- Varshney, M.; Li, Y. Interdigitated array microelectrode based impedance biosensor coupled with magnetic nanoparticle–antibody conjugates for detection of Escherichia coli O157: H7 in food samples. Biosens. Bioelectron. 2007, 22, 2408–2414. [Google Scholar] [CrossRef] [PubMed]
- Varshney, M.; Li, Y. Interdigitated array microelectrodes based impedance biosensors for detection of bacterial cells. Biosens. Bioelectron. 2009, 24, 2951–2960. [Google Scholar] [CrossRef]
- Loh, K.-S.; Lee, Y.; Musa, A.; Salmah, A.; Zamri, I. Use of Fe3O4 nanoparticles for enhancement of biosensor response to the herbicide 2, 4-dichlorophenoxyacetic acid. Sensors 2008, 8, 5775–5791. [Google Scholar] [CrossRef]
- Kaushik, A.; Khan, R.; Solanki, P.R.; Pandey, P.; Alam, J.; Ahmad, S.; Malhotra, B. Iron oxide nanoparticles–chitosan composite based glucose biosensor. Biosens. Bioelectron. 2008, 24, 676–683. [Google Scholar] [CrossRef]
- Mirabi, A.; Dalirandeh, Z.; Rad, A.S. Preparation of modified magnetic nanoparticles as a sorbent for the preconcentration and determination of cadmium ions in food and environmental water samples prior to flame atomic absorption spectrometry. J. Magn. Magn. Mater. 2015, 381, 138–144. [Google Scholar] [CrossRef]
- Pirouz, M.J.; Beyki, M.H.; Shemirani, F. Anhydride functionalised calcium ferrite nanoparticles: A new selective magnetic material for enrichment of lead ions from water and food samples. Food Chem. 2015, 170, 131–137. [Google Scholar] [CrossRef]
- Yang, M.; Wu, X.; Jia, Y.; Xi, X.; Yang, X.; Lu, R.; Zhang, S.; Gao, H.; Zhou, W. Use of magnetic effervescent tablet-assisted ionic liquid dispersive liquid–liquid microextraction to extract fungicides from environmental waters with the aid of experimental design methodology. Anal. Chim. Acta 2016, 906, 118–127. [Google Scholar] [CrossRef]
- Chen, J.; Zhu, X. Ionic liquid coated magnetic core/shell Fe3O4@ SiO2 nanoparticles for the separation/analysis of linuron in food samples. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2015, 137, 456–462. [Google Scholar] [CrossRef]
- Mehdinia, A.; Shegefti, S.; Shemirani, F. A novel nanomagnetic task specific ionic liquid as a selective sorbent for the trace determination of cadmium in water and fruit samples. Talanta 2015, 144, 1266–1272. [Google Scholar] [CrossRef] [PubMed]
- Soylak, M.; Yilmaz, E. Determination of cadmium in fruit and vegetables by ionic liquid magnetic microextraction and flame atomic absorption spectrometry. Anal. Lett. 2015, 48, 464–476. [Google Scholar] [CrossRef]
- Farahani, M.D.; Shemirani, F.; Ramandi, N.F.; Gharehbaghi, M. Ionic liquid as a ferrofluid carrier for dispersive solid phase extraction of copper from food samples. Food Anal. Methods 2015, 8, 1979–1989. [Google Scholar] [CrossRef]
- Najafi, E.; Aboufazeli, F.; Zhad, H.R.L.Z.; Sadeghi, O.; Amani, V. A novel magnetic ion imprinted nano-polymer for selective separation and determination of low levels of mercury (II) ions in fish samples. Food Chem. 2013, 141, 4040–4045. [Google Scholar] [CrossRef] [PubMed]
- Mashhadizadeh, M.H.; Amoli-Diva, M.; Shapouri, M.R.; Afruzi, H. Solid phase extraction of trace amounts of silver, cadmium, copper, mercury, and lead in various food samples based on ethylene glycol bis-mercaptoacetate modified 3-(trimethoxysilyl)-1-propanethiol coated Fe3O4 nanoparticles. Food Chem. 2014, 151, 300–305. [Google Scholar] [CrossRef]
- Hao, Y.; Gao, R.; Liu, D.; Tang, Y.; Guo, Z. Selective extraction of gallic acid in pomegranate rind using surface imprinting polymers over magnetic carbon nanotubes. Anal. Bioanal. Chem. 2015, 407, 7681–7690. [Google Scholar] [CrossRef] [PubMed]
- Hao, Y.; Gao, R.; Liu, D.; He, G.; Tang, Y.; Guo, Z. Selective extraction and determination of chlorogenic acid in fruit juices using hydrophilic magnetic imprinted nanoparticles. Food Chem. 2016, 200, 215–222. [Google Scholar] [CrossRef]
- Zhang, L.; Wu, H.; Liu, Z.; Gao, N.; Du, L.; Fu, Y. Ionic liquid-magnetic nanoparticle microextraction of safranin T in food samples. Food Anal. Methods 2015, 8, 541–548. [Google Scholar] [CrossRef]
- Piao, C.; Chen, L. Separation of Sudan dyes from chilli powder by magnetic molecularly imprinted polymer. J. Chromatogr. A 2012, 1268, 185–190. [Google Scholar] [CrossRef]
- Luo, Y.-B.; Yu, Q.-W.; Yuan, B.-F.; Feng, Y.-Q. Fast microextraction of phthalate acid esters from beverage, environmental water and perfume samples by magnetic multi-walled carbon nanotubes. Talanta 2012, 90, 123–131. [Google Scholar] [CrossRef]




| Magnetic Nanomaterial | Analytes | Detection Technique | Amount of Sorbent (mg) | Sample Volume (mL) | LOD (ng L−1) | Enrichment Factor | Adsorption Capacity (mg g−1) | Precision (RSD, %) | Recovery (%) | Ref. |
|---|---|---|---|---|---|---|---|---|---|---|
| Fe3O4-SiO2-γ-MPTMS | Cd Cu Hg Pb | ICP-MS | 50 | 250 | 0.024 0.092 0.107 0.056 | 500 | 45.2 56.8 83.8 70.4 | 6.7 9.6 8.3 3.7 | 94 | [63] |
| Fe3O4-SiO2-SA | Cr(III) Cu(II) Ni(II) Cd(II) | FAAS | 110 | 4 | 150 220 270 110 | 200 | 39.9 39.8 27.8 17.3 | 4.0 3.1 2.2 3.3 | - | [64] |
| Fe3O4-SiO2-γ-MPTMS | Te(IV) | ICP-MS | 50 | 160 | 0.079 | 320 | 10.1 | 7.0 | 88–109 | [65] |
| Fe3O4-SiO2-L | Pb(II) Cd(II) Cu(II) | FAAS | 130 | 350 | 140 190 120 | - | - | 1.4 1.6 1.8 | 97.8–102.9 | [60] |
| Fe3O4-SiO2-MIL-101 | PAHs | HPLC-PDA | 0.6+1.0 | 20 | 2.8–27.2 | 101-180 | - | 3.1-8.7 | 81.3–105 | [66] |
| Fe3O4-SiO2-diphenyl | PAHs | GC-MS | 25 | 0.8 | - | - | - | - | 88–97 | [67] |
| Fe3O4-SiO2–AAPTS | As(V) | ICP-MS | 50 | 150 | 0.21 | 3001 | 13.1 | 6.8 | 104.1 | [68] |
| Fe3O4-SiO2-IDA | Cd(II) Mn(II) Pb(II) | ICP-MS | 40 | 100 | 0.16 0.26 0.26 | 200 | 45.1 30.5 73.1 | 4.8 4.6 7.4 | 95–106.6 | [69] |
| Fe3O4-SiO2-Bismuthiol-II | Cr Cu Pb | ICP-OES | 100 | 100 | 43 58 85 | 96 95 87 | 8.6 5.3 9.4 | 3.5 4.6 3.7 | 90–104 | [70] |
| Fe3O4-SiO2-TiO2 | Cd(II) Cr(III) Mn(II) Cu(II) | ICP-MS | 40 | 50 | 4.0 2.6 1.6 2.3 | 100 | 59.3 27.8 15.4 33.2 | 3.6 4.5 4.0 4.1 | 100–109 | [71] |
| Aminated-CoFe2O4-SiO2 | Cd(II) | HG-AFS | 20 | 50 | 3.15 | 50 | 5.0 | 4.9 | 98.0–100.4 | [72] |
| Fe3O4-SiO2-Zincon | Pb | GFAAS | 20 | 100 | 10 | 200 | 21.5 | 7.8–9.2 | 84–104 | [73] |
| Fe3O4-SiO2-β-CD | BPA DES | HPLC | 100 | 250 | 20.0 23.0 | 100-390 | - | <7 | 80–105 | [74] |
| Fe3O4-SiO2 | Sudan dyes | UFLC | 40 | 4 | 82–120 | 500 | - | 1.93–8.11 | 87.10–111.4 | [75] |
| Fe3O4-SiO2-DAPD | Cu(II) Zn(II) | FAAS | 10 | 100 | 140 220 | 125 | 45 32 | 2.3 3.6 | 97–104 | [76] |
| Fe3O4-SiO2-DPC | Hg(II) | AAS | 100 | 200 | 160 | 100 | - | 2.2 | 97.5 | [77] |
| Fe3O4-HMS | DDT | GC-MS | 10 | 35 | - | - | - | - | - | [78] |
| Protein | Magnetic Carrier | Ligand | Elution Method | Binding Capacity (mg/g) | Reference |
|---|---|---|---|---|---|
| Lipase | Fe3O4-PAA | –COOH | Phosphate buffer (pH 9) | 605 | [85] |
| Antibody | Fe3O4-gum Arabic-Artificial Protein | Artificial Protein A/artificial Protein L | Citrate buffer (pH 3) and Glycine NaOH buffer (pH 11) | 133 65 | [84] |
| Superoxide dismutase | Fe3O4-IDA-Cu+2 | IDA-Cu+2 | Potassium phosphate containing NH4Cl | n/a | [86] |
| Bovine haemoglobin | Fe3O4-SiO2-GPS–IDA-Zn+2 | IDA-Zn+2 | n/a | 207.2 | [87] |
| Lactoferrin | Fe3O4-PGMA-EA-heparin | Heparin | NaCl Solution | 164 | [88] |
| His-Tagged Protein | Fe3O4-PMIDA-Ni+2 | PMIDA-Ni+2 | Sodium phosphate, imidazole and NaCl | n/a | [80] |
| Bromelain | Fe3O4-PAA | –COOH | Phosphate buffer containing NaCl | 476 | [89] |
| Lysozyme | Magnetic PHEMA beads-Cibacron Blue-F3GA | Cibacron Blue F3GA | Tris/HCl buffer containing NaCl | 342 | [90] |
| Fe3O4-PEG-CMCTs | –COOH | PBS + NaCl | 256.6 | [91] | |
| Fe3O4-PAA | –COOH | PBS + NaSCN | 224 | [92] | |
| Fe3O4-SiO2-GPS-Tris | Tris | 108.6 | [81] |
| Analyte | Samples | Magnetic Functionalized Material | Detection Technique | Eluent | LOD (ng mL−1) | Recovery % | Reference |
|---|---|---|---|---|---|---|---|
| Hg | Fish | Fe3O4-IIP | ICP-OES | EDTA | 0.03 | 98.4–102.4 | [108] |
| Pb | Water, milk, canned tuna fish, parsley and canned tomato paste | CaFe2O4-SiO2-NH2-PA | FAAS | HNO3 | 0.78 | 91.3–100 | [102] |
| Ag | Rice, tuna fish and tea leaves | Fe3O4-SiO2-SH | ICP-OES CV-AAS | Thiourea | 0.07 | 96.2 | [109] |
| Cu | 0.09 | 99.8 | |||||
| Cd | 0.06 | 98.4 | |||||
| Pb | 0.08 | 95.4 | |||||
| Hg | 0.01 | 97.1 | |||||
| Cd | Green tea. Lettuce, ginseng, rice, spice and carrot | Fe3O4-SDS-carbazone | FAAS | HCL | 3.71 | NR | [101] |
| Cd | Milk powder | Fe3O4-SiO2-SH | ICP-MS | HCL | 2 × 10−5 | 97 | [63] |
| Cu | 9 × 10−5 | 96 | |||||
| Hg | 1.1× 10−4 | 104 | |||||
| Pb | 6 × 10−5 | 97 | |||||
| Pb | Tuna fish, rice and shrimp | Fe3O4-SiO2-3-(4-methoxybenzylideneamino)-2-Thioxothiazolodin-4-0ne | FAAS | MeOH-HNO3 | 0.14 | 97.8–102.9 | [60] |
| Cd | 0.19 | 98.7–101.4 | |||||
| Cu | 0.12 | 98.6–102.6 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Khan, R.; Rehman, A.; Hayat, A.; Andreescu, S. Magnetic Particles-Based Analytical Platforms for Food Safety Monitoring. Magnetochemistry 2019, 5, 63. https://doi.org/10.3390/magnetochemistry5040063
Khan R, Rehman A, Hayat A, Andreescu S. Magnetic Particles-Based Analytical Platforms for Food Safety Monitoring. Magnetochemistry. 2019; 5(4):63. https://doi.org/10.3390/magnetochemistry5040063
Chicago/Turabian StyleKhan, Reem, Abdur Rehman, Akhtar Hayat, and Silvana Andreescu. 2019. "Magnetic Particles-Based Analytical Platforms for Food Safety Monitoring" Magnetochemistry 5, no. 4: 63. https://doi.org/10.3390/magnetochemistry5040063
APA StyleKhan, R., Rehman, A., Hayat, A., & Andreescu, S. (2019). Magnetic Particles-Based Analytical Platforms for Food Safety Monitoring. Magnetochemistry, 5(4), 63. https://doi.org/10.3390/magnetochemistry5040063







