Preparation of Magnetic Iron Oxide Incorporated Mesoporous Silica Hybrid Composites for pH and Temperature-Sensitive Drug Delivery
Abstract
:1. Introduction
2. Materials and Methods
2.1. Reagents
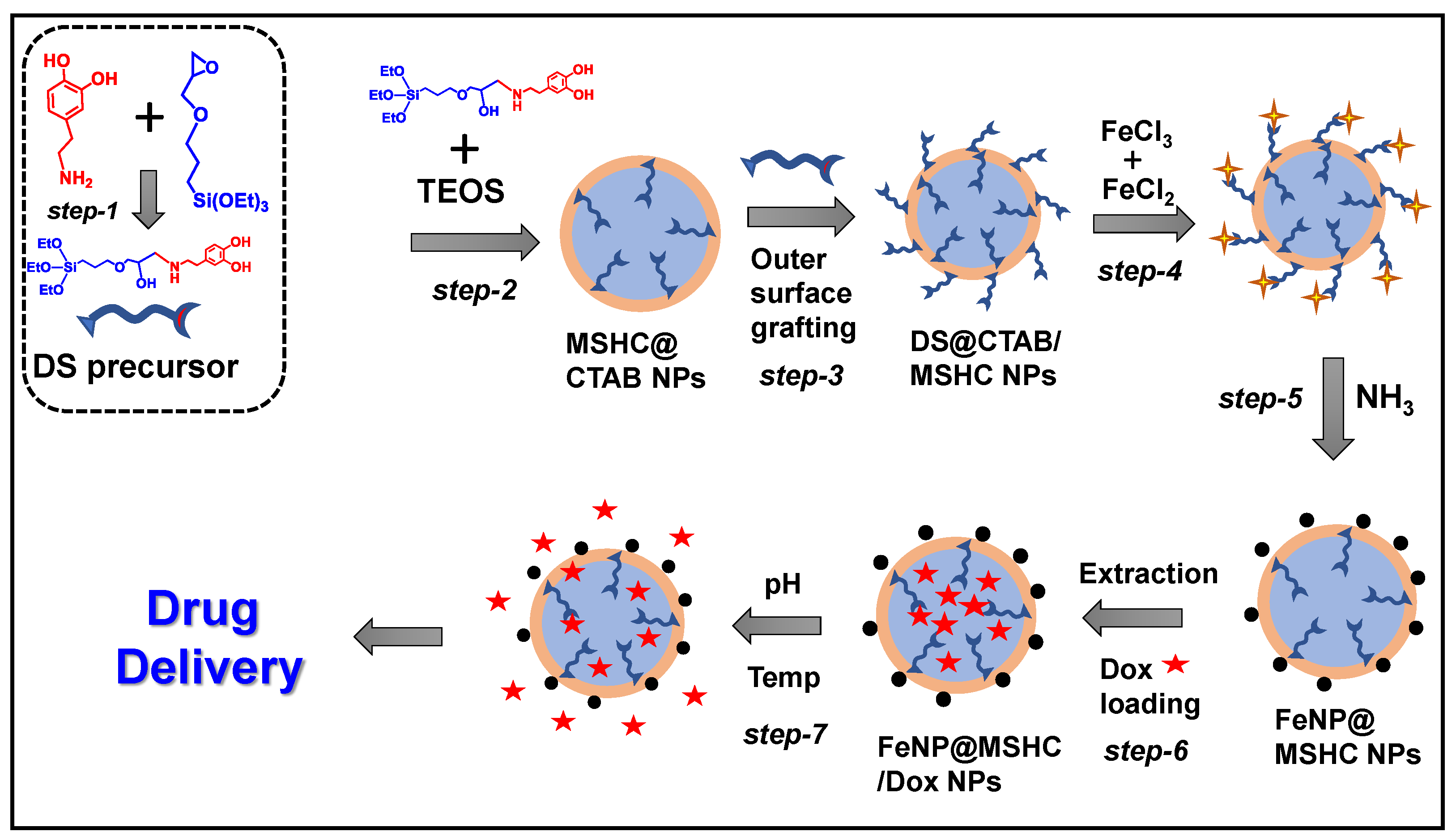
2.2. Synthesis of Dopamine-Silane (DS) Precursor
2.3. Synthesis of Dopamine-Silane Integrated with Mesoporous Silica Nanoparticles
2.4. Synthesis of Iron Oxide NPs Incorporated Mesoporous Silica Composite Nanoparticles
(FeNP@MSHC NPs)
2.5. Characterization
2.6. Drug Loading into the FeNP@ MSHC NPs
2.7. Drug Release Study from the FeNP@MSHC/Dox NPs
2.8. MTT Assay Analysis of the FeNP@MSHC/Dox NPs
2.9. Cell-Uptake Study
3. Results and Discussion
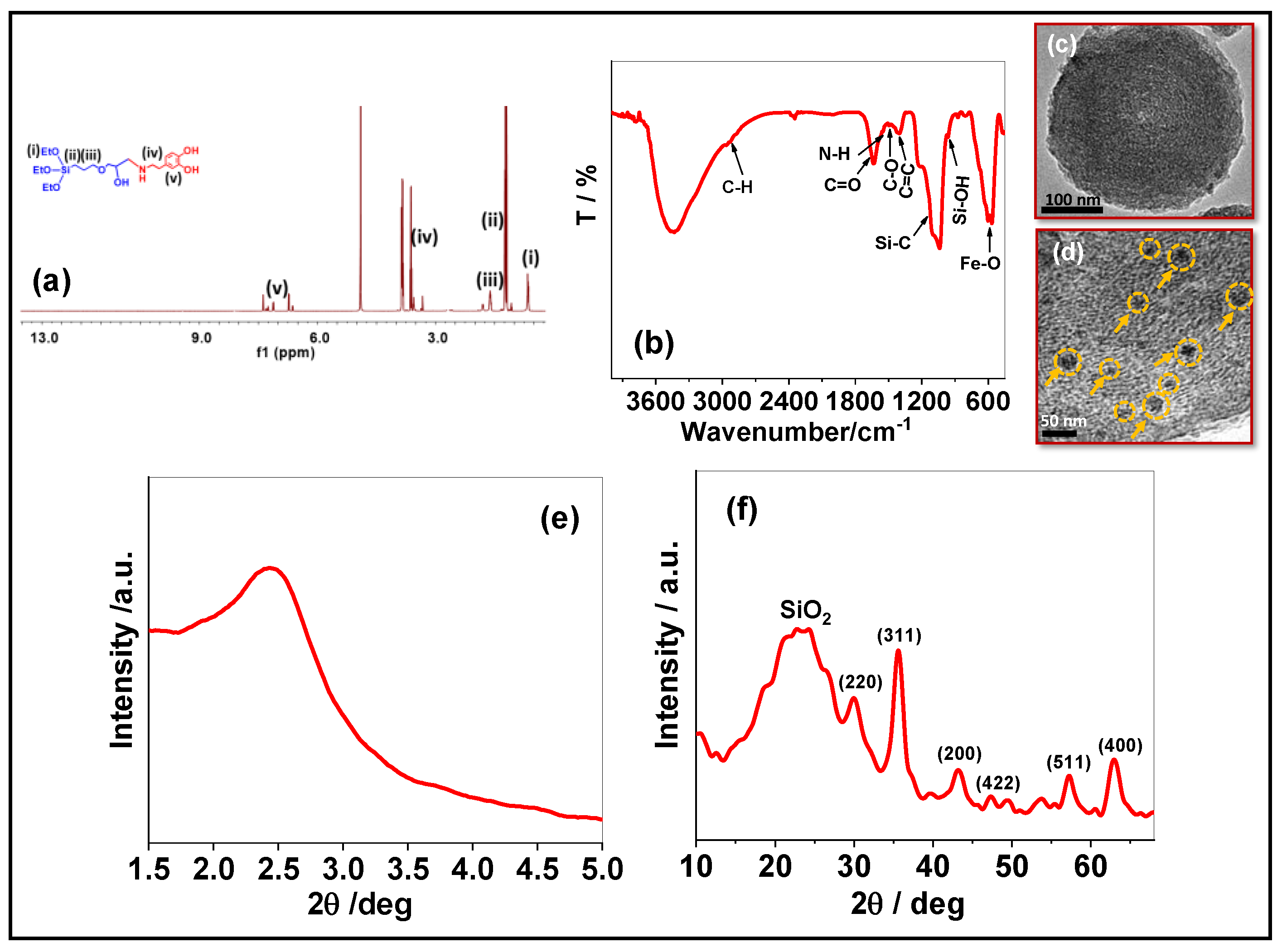
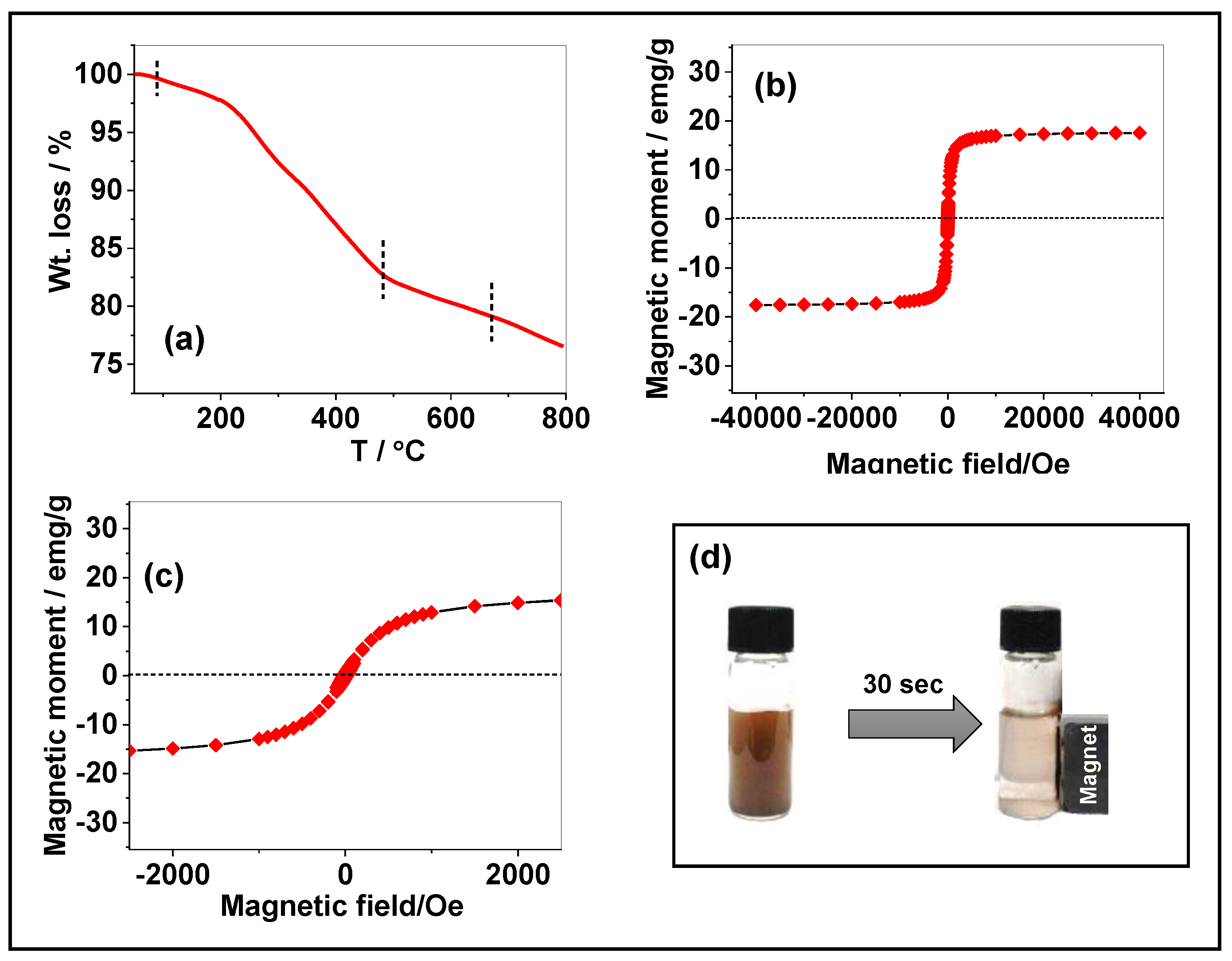
3.1. Characterization of the FeNP@MSHC NPs
3.2. Drug Loading into the FeNP@MSHC NPs
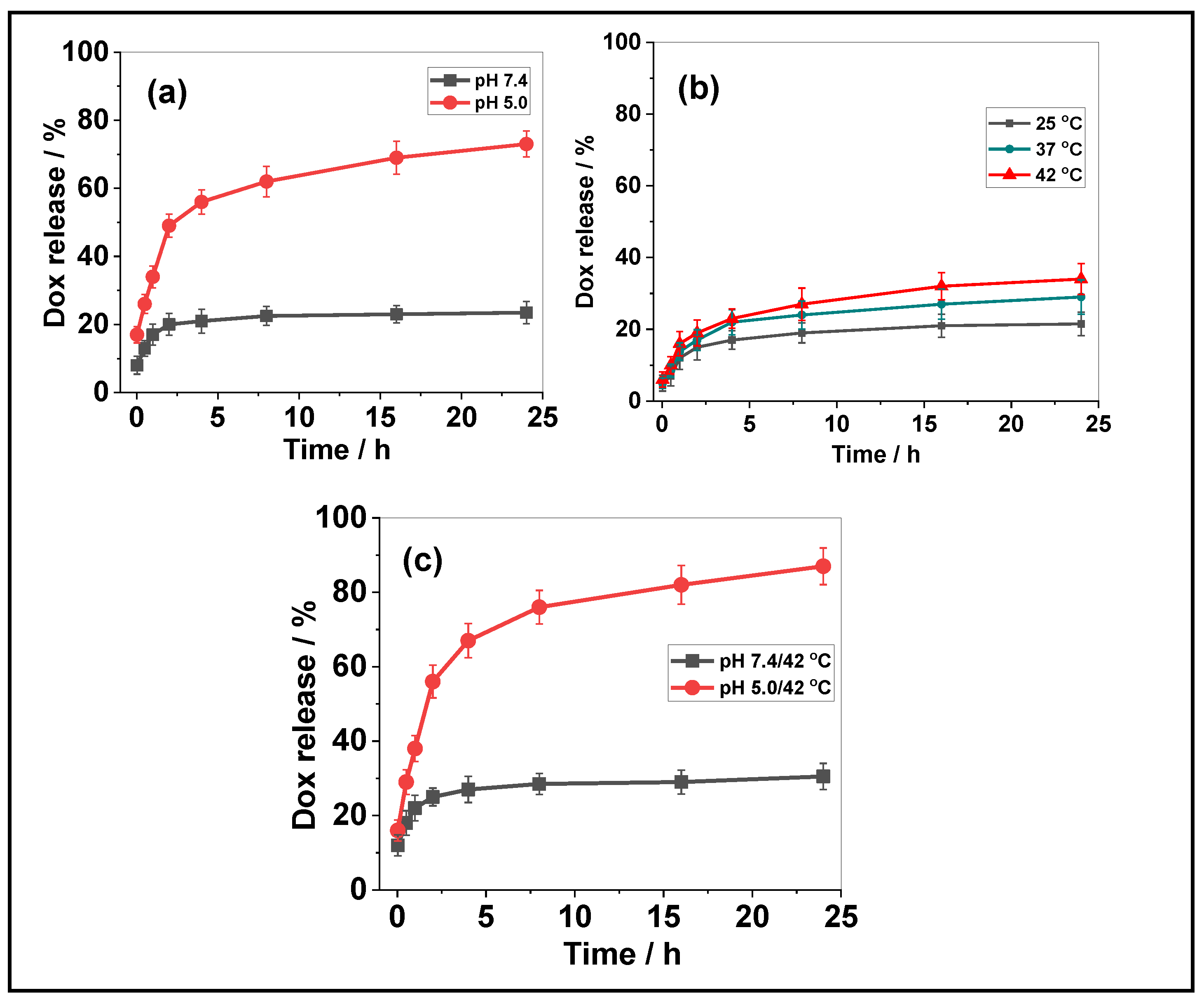
3.3. Drug Release from the FeNP@MSHC/Dox NPs
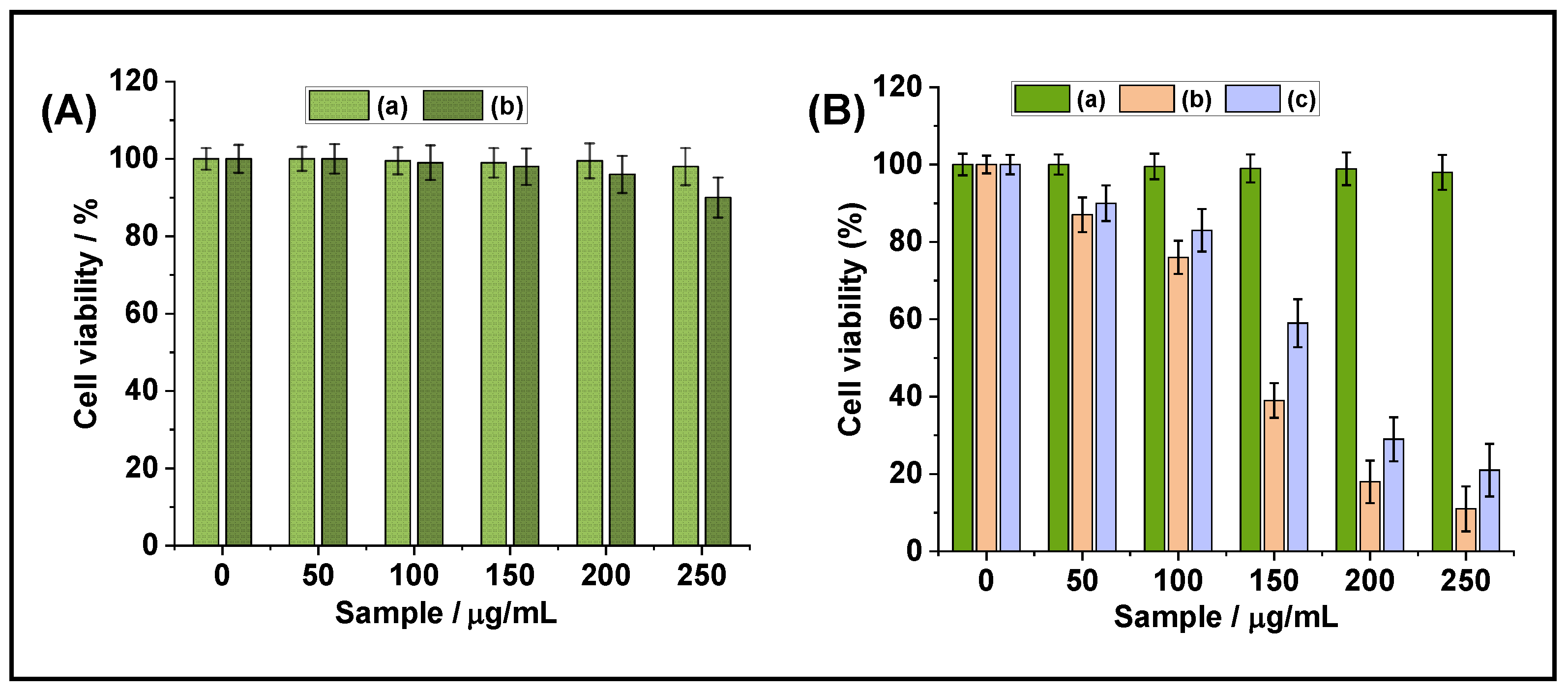
3.4. In Vitro Cytotoxicity Study
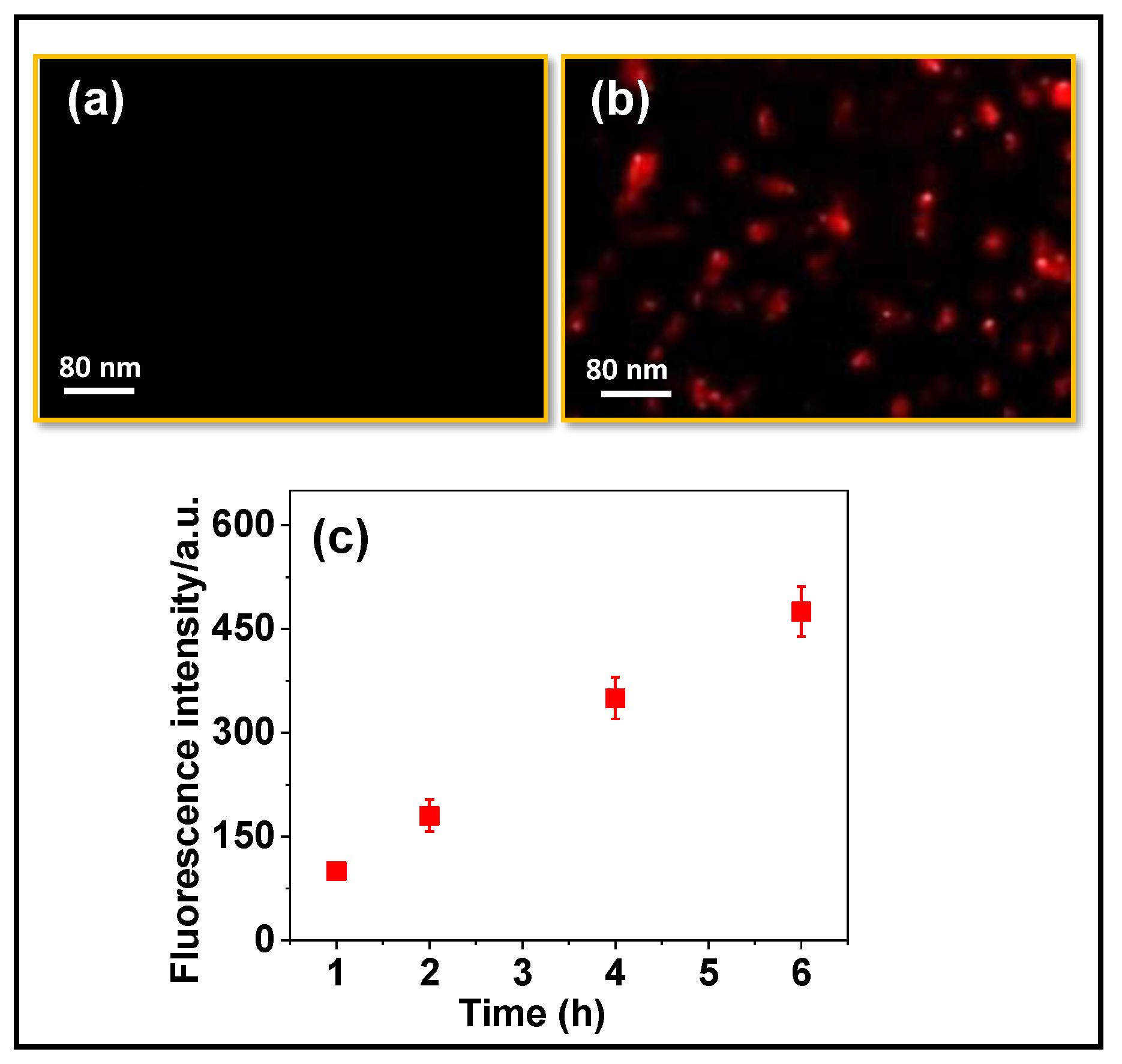
3.5. In Vitro Cell-Uptake Experiments
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Baig, N.; Kammakakam, I.; Falath, W. Nanomaterials: A review of synthesis methods, properties, recent progress, and challenges. Mater. Adv. 2021, 2, 1821–1871. [Google Scholar]
- Harish, V.; Tewari, D.; Gaur, M.; Yadav, A.B.; Swaroop, S.; Bechelany, M.; Barhoum, A. Review on Nanoparticles and Nanostructured Materials: Bioimaging, Biosensing, Drug Delivery, Tissue Engineering, Antimicrobial, and Agro-Food Applications. Nanomaterials 2022, 12, 457. [Google Scholar] [PubMed]
- Bayda, S.; Adeel, M.; Tuccinardi, T.; Cordani, M.; Rizzolia, F. The History of Nanoscience and Nanotechnology: From Chemical–Physical Applications to Nanomedicine. Molecules 2020, 25, 112. [Google Scholar]
- Khan, I.; Saeed, K.; Khan, I. Nanoparticles: Properties, applications and toxicities. Arab. J. Chem. 2019, 12, 908–931. [Google Scholar]
- Navya, P.N.; Daima, H.K. Rational engineering of physicochemical properties of nanomaterials for biomedical applications with nanotoxicological perspectives. Nano Converg. 2016, 3, 1. [Google Scholar] [CrossRef] [Green Version]
- Chandrakala, V.; Aruna, V.; Angajala, G. Review on metal nanoparticles as nanocarriers: Current challenges and perspectives in drug delivery systems. Emergent Mater. 2022, 5, 1593–1615. [Google Scholar] [CrossRef]
- Chavali, M.S.; Nikolova, M.P. Metal oxide nanoparticles and their applications in nanotechnology. SN Appl. Sci. 2019, 1, 607. [Google Scholar] [CrossRef] [Green Version]
- Shen, Y.; Zhang, L.; Wang, K.; Li, X.; Li, J.; Zhang, S.; Zhao, H.; Jiang, X.; Guan, W.; Yang, L. Bio-mediated synthesis—A sustainable strategy for nanomaterials preparation: A comprehensive bibliometric review. Nanoselect 2021, 2, 2275–2290. [Google Scholar]
- Chircov, C.; Spoiala, A.; Puan, C.; Craciun, L.; Ficai, D.; Ficai, A.; Andronescu, E.; Turculet, S.C. Mesoporous Silica Platforms with Potential Applications in Release and Adsorption of Active Agents. Molecules 2020, 25, 3814. [Google Scholar]
- Wang, D.; Chen, X.; Feng, J.; Sun, M. Recent advances of ordered mesoporous silica materials for solid-phase extraction. J. Chhromatogr. A 2022, 1675, 463157. [Google Scholar]
- Feng, Y.; Liao, Z.; Li, M.; Zhang, H.; Li, T.; Qin, X.; Li, S.; Wu, C.; You, F.; Liao, X.; et al. Mesoporous Silica Nanoparticles-Based Nanoplatforms: Basic Construction, Current State, and Emerging Applications in Anticancer Therapeutics. Adv. Healthcare Mater. 2022, 18, 2201884. [Google Scholar] [CrossRef]
- Prabha, S.; Durgalakshmi, D.; Rajendran, S.; Lichtfouse, E. Plant-derived silica nanoparticles and composites for biosensors, bioimaging, drug delivery and supercapacitors: A review. Environ. Chem. Lett. 2021, 19, 1667–1691. [Google Scholar] [CrossRef] [PubMed]
- Guimaraes, R.; Rodrigues, C.; Moreira, A.F.; Correia, I. Overview of stimuli-responsive mesoporous organosilica nanocarriers for drug delivery. Pharmacol. Res. 2020, 155, 104742. [Google Scholar] [PubMed]
- Park, S.S.; Moorthy, M.S.; Ha, C.S. Periodic mesoporous organosilicas for advanced applications. NPG Asia Mater. 2014, 6, e96. [Google Scholar] [CrossRef]
- Moorthy, M.S.; Park, S.S.; Fuping, D.; Hong, S.-H.; Selvaraj, M.; Ha, C.S. Step-up synthesis of amidoxime-functionalised periodic mesoporous organosilicas with an amphoteric ligand in the framework for drug delivery. J. Mater. Chem. 2012, 22, 9100–9108. [Google Scholar]
- Moorthy, M.S.; Hoang, G.; Subramanian, B.; Bui, N.Q.; Panchanathan, M.; Mondal, S.; Tuong, V.P.T.; Kim, H.; Oh, J. Prussian blue decorated mesoporous silica hybrid nanocarriers for photoacoustic imaging-guided synergistic chemo-photothermal combination therapy. J. Mater. Chem. B 2018, 6, 5220–5233. [Google Scholar]
- Yu, L.; Chen, Y.; Lin, H.; Du, W.; Chen, H.; Shi, J. Ultrasmall mesoporous organosilica nanoparticles: Morphology modulations and redox-responsive biodegradability for tumor-specific drug delivery. Biomaterials 2018, 161, 292–305. [Google Scholar]
- Ashouri, A.; Samadi, S.; Nasiri, B.; Bahrami, Z. Iron-based nanomaterials used as magnetic mesoporous nanocomposites to catalyze the preparation of N-sulfonylimines. Comptes Rendus Chim. 2019, 22, 549–559. [Google Scholar]
- Tran, H.-V.; Ngo, N.M.; Medhi, R.; Srinoi, P.; Liu, T.; Rittikulsittichai, S.; Lee, T.R. Multifunctional Iron Oxide Magnetic Nanoparticles for Biomedical Applications: A Review. Materials 2022, 15, 503. [Google Scholar] [CrossRef]
- Nemec, S.; Kralj, S. A Versatile Interfacial Coassembly Method for Fabrication of Tunable Silica Shells with Radially Aligned Dual Mesopores on Diverse Magnetic Core Nanoparticles. ACS Appl. Mater. Interfaces 2021, 13, 1883–1894. [Google Scholar]
- Chang, Z.; Wang, Z.; Shao, D.; Yue, J.; Xing, H.; Li, L.; Ge, M.; Li, M.; Yan, H.; Hu, H.; et al. Shape Engineering Boosts Magnetic Mesoporous Silica Nanoparticle-Based Isolation and Detection of Circulating Tumor Cells. ACS Appl. Mater. Interfaces 2018, 10, 10656–10663. [Google Scholar] [PubMed]
- Moorthy, M.S.; Subramanian, B.; Panchanathan, M.; Mondal, S.; Kim, H.; Lee, K.D.; Oh, J. Fucoidan-coated core–shell magnetic mesoporous silica nanoparticles for chemotherapy and magnetic hyperthermia-based thermal therapy applications. New J. Chem. 2017, 41, 15334–15346. [Google Scholar]
- Moorthy, M.S.; Kim, H.-B.; Bae, J.-H.; Kim, S.-H.; Ha, C.S. Design of core–shell magnetic mesoporous silica hybrids for pH and UV light stimuli-responsive cargo release. RSC Adv. 2016, 6, 29106–29115. [Google Scholar]
- Zhu, N.; Ji, H.; Yu, P.; Niu, J.; Farooq, M.U.; Akram, M.W.; Udego, I.O.; Li, H.; Niu, X. Surface Modification of Magnetic Iron Oxide Nanoparticles. Nanomaterials 2018, 8, 810. [Google Scholar] [CrossRef] [Green Version]
- El-Boubbou, K.; Ali, R.; Al-Zahrani, H.; Trivilegio, T.; Alanazi, A.H.; Khan, A.L.; Boudjelal, M.; Alkushi, A. Preparation of iron oxide mesoporous magnetic microparticles as novel multidrug carriers for synergistic anticancer therapy and deep tumor penetration. Sci. Rep. 2019, 9, 9481. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bruckmann, F.S.; Nunes, F.B.; Salles, T.R.; Franco, C.; Cadona, F.C.; Rhoden, C.R.B. Biological Applications of Silica-Based Nanoparticles. Magnetochemistry 2022, 8, 131. [Google Scholar] [CrossRef]
- Yu, X.; Wang, L.; Li, K.; Mi, Y.; Li, Z.; Wu, D.; Sun, F.; He, S.; Zeng, H. Tuning dipolar effects on magnetic hyperthermia of Zn0.3Fe2.7O4/SiO2 nanoparticles by silica shell. J. Magn. Magn. Mater. 2021, 521, 167483. [Google Scholar]
- Zhang, J.; Lin, Y.; Lin, Z.; Wei, Q.; Quan, J.; Ruan, R.; Jiang, X.; Hou, L.; Song, J.; Ding, J.; et al. Stimuli-Responsive Nanoparticles for Controlled Drug Delivery in Synergistic Cancer Immunotherapy. Adv. Sci. 2021, 9, 2103444. [Google Scholar]
- Hershberger, K.K.; Gauger, A.J.; Bronstein, L.M. Utilizing Stimuli Responsive Linkages to Engineer and Enhance Polymer Nanoparticle-Based Drug Delivery Platforms. ACS Appl. Bio. Mater. 2021, 4, 4720–4736. [Google Scholar]
- Au, J.L.-S.; Yeung, B.Z.; Wientjes, M.G.; Lu, Z.; Wientjes, M.G. Delivery of cancer therapeutics to extracellular and intracellular targets: Determinants, barriers, challenges and opportunities. Adv. Drug Deliv. Rev. 2016, 97, 280–301. [Google Scholar] [PubMed] [Green Version]
- Ponomareva, S.; Joisten, H.; Francois, T.; Naud, C.; Morel, R.; Hou, Y.; Myer, T.; Joumard, I.; Dieny, B.; Carriere, M. Magnetic particles for triggering insulin release in INS-1E cells subjected to a rotating magnetic field. Nanoscale 2022, 14, 13274–13283. [Google Scholar] [PubMed]
- Moorthy, M.S.; Park, J.-H.; Bae, J.-H.; Kim, S.-H.; Ha, C.S. Mesoporous organosilica hybrids with a tunable amphoteric framework for controlled drug delivery. J. Mater. Chem. B 2014, 2, 6487–6499. [Google Scholar] [PubMed]
- Cui, R.; Zhang, K.; Wu, X.; Zhang, H.; Wang, C. Determination of Fe3+ upon Special “Upconversion Luminescence” of Dopamine. ACS Omega 2019, 4, 9918–9924. [Google Scholar] [PubMed] [Green Version]
- Kumar, P.; Nagarajan, A.; Uchil, P.D. Analysis of Cell Viability by the MTT Assay. Cold Spring Harb. Protoc. 2018, 2018, pdb-prot095505. [Google Scholar] [CrossRef]
- Gao, J.; Lei, H.; Han, Z.; Shi, Q.; Chen, Y.; Jinag, Y. Dopamine functionalized tannic-acid-templated mesoporous silica nanoparticles as a new sorbent for the efficient removal of Cu2+ from aqueous solution. Sci. Rep. 2017, 7, 45215. [Google Scholar] [CrossRef] [Green Version]
- Tian, Z.; Yu, X.; Ruan, Z.; Zhu, M.; Zhu, Y.; Hanagata, N. Magnetic mesoporous silica nanoparticles coated with thermo-responsive copolymer for potential chemo- and magnetic hyperthermia therapy. Micropor. Mesopor. Mater. 2018, 256, 1–9. [Google Scholar]
- Moorthy, M.S.; Seo, D.-J.; Song, H.-J.; Park, S.S.; Ha, C.S. Magnetic mesoporous silica hybrid nanoparticles for highly selective boron adsorption. J. Mater. Chem. A. 2013, 40, 12485–12496. [Google Scholar]
- Baeza, A.; Guisasola, E.; Ruiz-Hernandez, E.; Vallet-Regi, M. Magnetically Triggered Multidrug Release by Hybrid Mesoporous Silica Nanoparticles. Chem. Mater. 2012, 24, 517–524. [Google Scholar]
- Sims, K.R., Jr.; He, B.; Koo, H.; Benoit, D.S.W. Electrostatic Interactions Enable Nanoparticle Delivery of the Flavonoid Myricetin. ACS Omega 2020, 5, 12649–12659. [Google Scholar]
- Farjadian, F.; Rezaeifard, S.; Naeimi, M.; Ghasemi, S.; Mohammadi-Samani, S.; Welland, M.E.; Tayebi, L. Temperature and pH-responsive nano-hydrogel drug delivery system based on lysine-modified poly (vinylcaprolactam). Int. J. Nanomed. 2019, 14, 6901–6915. [Google Scholar] [CrossRef] [Green Version]
- Saputra, O.A.; Lestari, W.A.; Kurniansyah, V.; Lestari, W.W.; Sugiura, T.; Mukti, R.R.; Martien, R.; Wibowo, F.R. Organically surface engineered mesoporous silica nanoparticles control the release of quercetin by pH stimuli. Sci. Rep. 2022, 12, 20661. [Google Scholar] [CrossRef] [PubMed]

| Sample | Surface Area (m2/g) | Pore Size (nm) | Pore Volume (cm3/g) | Dox Loading (%) |
|---|---|---|---|---|
| FeNP@MSHC NPs | 364 ± 2.5 | 3.2 ± 0.1 | 0.048 ± 1.5 | ~62 ± 1.5 |
| pH | Release Efficiency (%) |
|---|---|
| 7.4 | 23.5 |
| 5.0 | 52.4 |
| 7.4/25 °C | 21.5 |
| 7.4/42 °C | 34 |
| 7.4/42 °C | 34 |
| 5.0/42 °C | 87 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Santhamoorthy, M.; Thirupathi, K.; Krishnan, S.; Guganathan, L.; Dave, S.; Phan, T.T.V.; Kim, S.-C. Preparation of Magnetic Iron Oxide Incorporated Mesoporous Silica Hybrid Composites for pH and Temperature-Sensitive Drug Delivery. Magnetochemistry 2023, 9, 81. https://doi.org/10.3390/magnetochemistry9030081
Santhamoorthy M, Thirupathi K, Krishnan S, Guganathan L, Dave S, Phan TTV, Kim S-C. Preparation of Magnetic Iron Oxide Incorporated Mesoporous Silica Hybrid Composites for pH and Temperature-Sensitive Drug Delivery. Magnetochemistry. 2023; 9(3):81. https://doi.org/10.3390/magnetochemistry9030081
Chicago/Turabian StyleSanthamoorthy, Madhappan, Kokila Thirupathi, Selvakumar Krishnan, Loganathan Guganathan, Sushma Dave, Thi Tuong Vy Phan, and Seong-Cheol Kim. 2023. "Preparation of Magnetic Iron Oxide Incorporated Mesoporous Silica Hybrid Composites for pH and Temperature-Sensitive Drug Delivery" Magnetochemistry 9, no. 3: 81. https://doi.org/10.3390/magnetochemistry9030081
APA StyleSanthamoorthy, M., Thirupathi, K., Krishnan, S., Guganathan, L., Dave, S., Phan, T. T. V., & Kim, S.-C. (2023). Preparation of Magnetic Iron Oxide Incorporated Mesoporous Silica Hybrid Composites for pH and Temperature-Sensitive Drug Delivery. Magnetochemistry, 9(3), 81. https://doi.org/10.3390/magnetochemistry9030081







