Iron Sequestration by Galloyl–Silane Nano Coatings Inhibits Biofilm Formation of Sulfitobacter sp.
Abstract
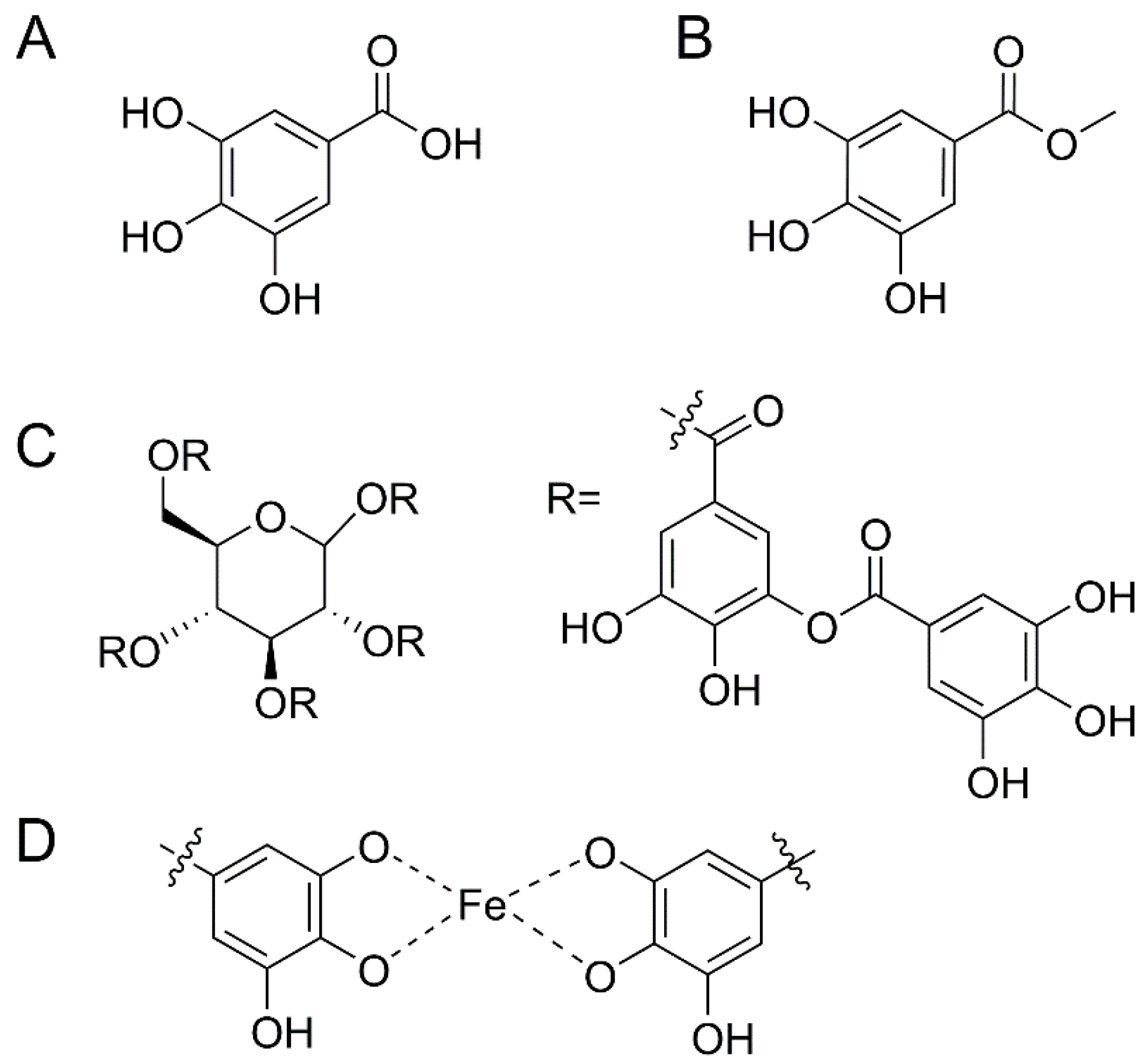
:1. Introduction
2. Materials and Methods
2.1. Bacterial Strain and Culture Conditions
2.2. Reagent and Media Preparations
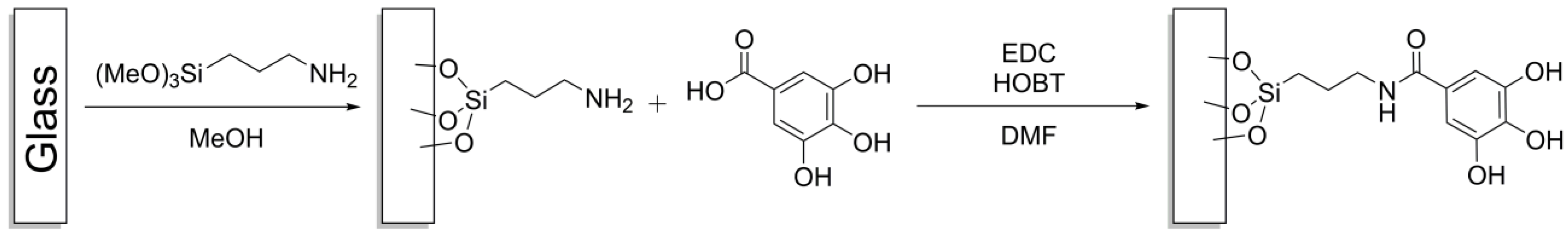
2.3. Glass Functionalization
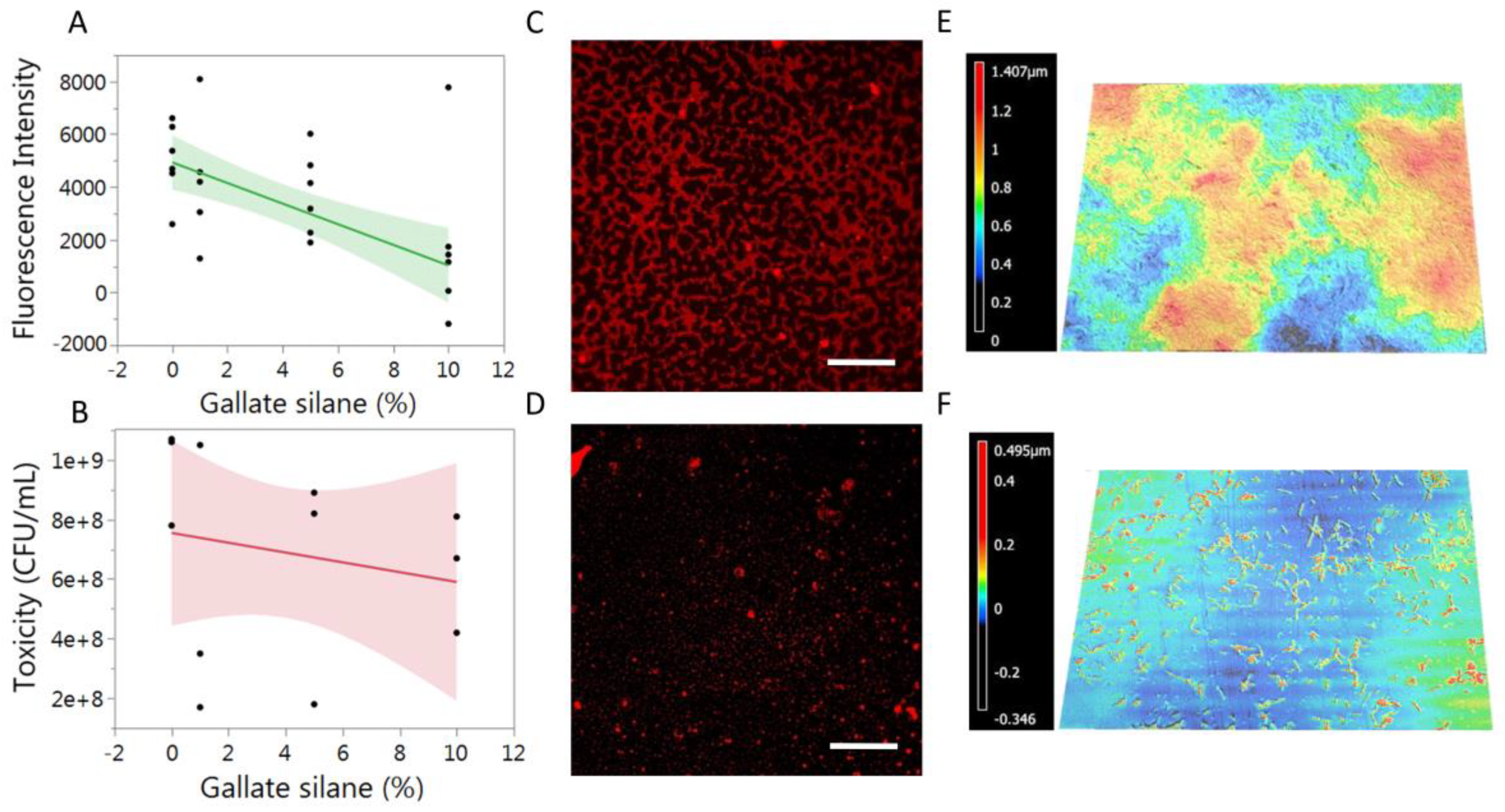
2.4. Biofilm Formation
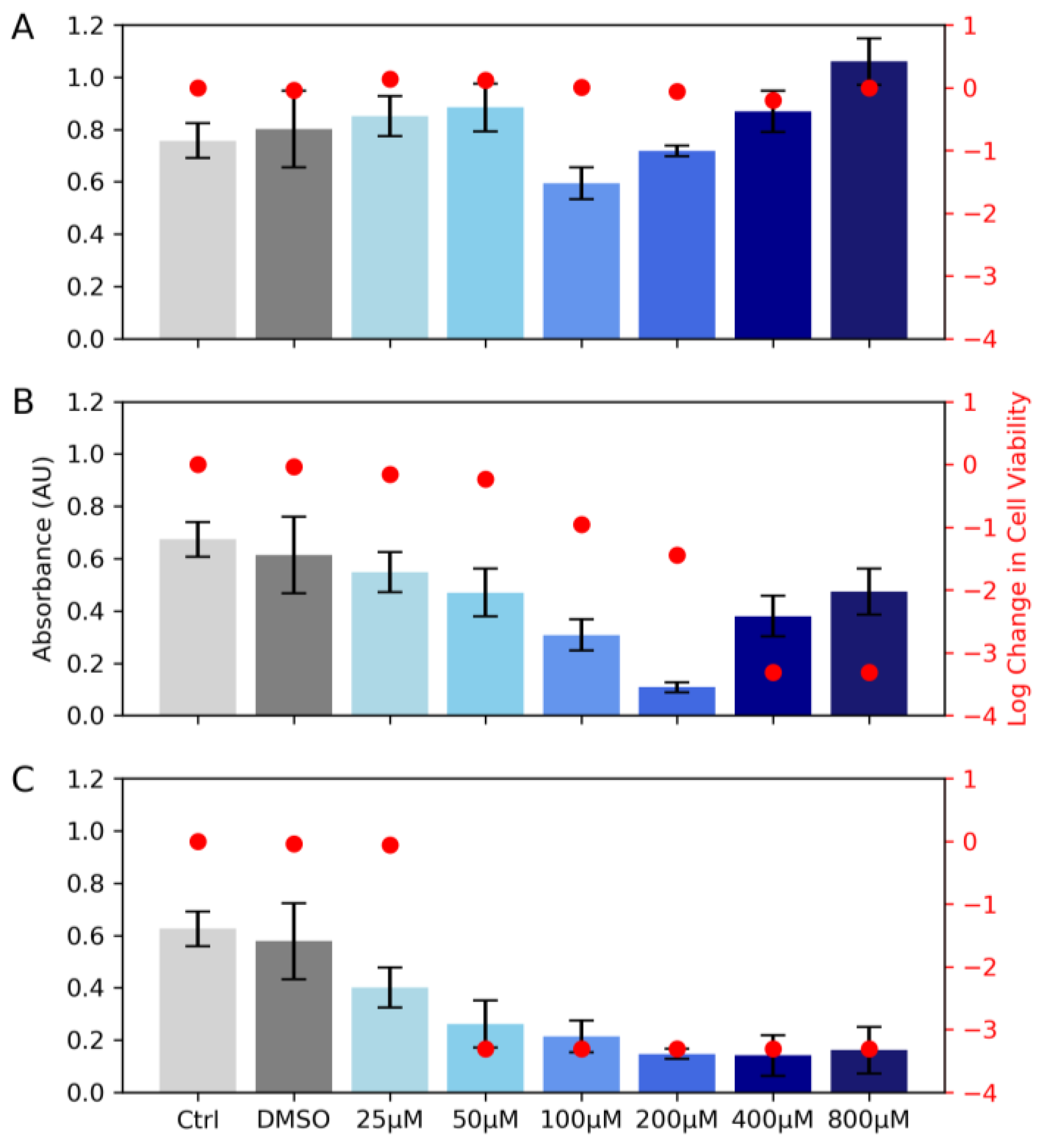
2.5. Cell Suspension Viability
2.6. Biofilm Imaging
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Blackwood, D.J. An Electrochemist Perspective of Microbiologically Influenced Corrosion. Corros. Mater. Degrad. 2018, 1, 59–76. [Google Scholar] [CrossRef]
- Rajala, P.; Carpén, L.; Vepsäläinen, M.; Raulio, M.; Sohlberg, E.; Bomberg, M. Microbially induced corrosion of carbon steel in deep groundwater environment. Front. Microbiol. 2015, 6, 647. [Google Scholar] [CrossRef] [PubMed]
- Sandy, M.; Butler, A. Microbial Iron Acquisition: Marine and Terrestrial Siderophores. Chem. Rev. 2009, 109, 4580–4595. [Google Scholar] [CrossRef] [PubMed]
- Hutchins, D.A.; Bruland, K.W. Iron-limited diatom growth and Si:N uptake ratios in a coastal upwelling regime. Nature 1998, 393, 561–564. [Google Scholar] [CrossRef]
- Qiao, J.; Purro, M.; Liu, Z.; Xiong, M.P. Terpyridine–Micelles for Inhibiting Bacterial Biofilm Development. ACS Infect. Dis. 2018, 4, 1346–1354. [Google Scholar] [CrossRef]
- Huang, Y.; Zhou, E.; Jiang, C.; Jia, R.; Liu, S.; Xu, D.; Gu, T.; Wang, F. Endogenous phenazine-1-carboxamide encoding gene PhzH regulated the extracellular electron transfer in biocorrosion of stainless steel by marine Pseudomonas aeruginosa. Electrochem. Commun. 2018, 94, 9–13. [Google Scholar] [CrossRef]
- Widdel, F.; Schnell, S.; Heising, S.; Ehrenreich, A.; Assmus, B.; Schink, B. Ferrous iron oxidation by anoxygenic phototrophic bacteria. Nature 1993, 362, 834–836. [Google Scholar] [CrossRef]
- Bose, A.; Gardel, E.J.; Vidoudez, C.; Parra, E.A.; Girguis, P.R. Electron uptake by iron-oxidizing phototrophic bacteria. Nat. Commun. 2014, 5, 3391. [Google Scholar] [CrossRef]
- Pasmore, M.; Costerton, J.W. Biofilms, bacterial signaling, and their ties to marine biology. J. Ind. Microbiol. Biotechnol. 2003, 30, 407–413. [Google Scholar] [CrossRef]
- McRose, D.L.; Baars, O.; Seyedsayamdost, M.R.; Morel, F.M.M. Quorum sensing and iron regulate a two-for-one siderophore gene cluster in Vibrio harveyi. Proc. Natl. Acad. Sci. USA 2018, 115, 7581–7586. [Google Scholar] [CrossRef] [Green Version]
- Guan, L.L.; Kamino, K. Bacterial response to siderophore and quorum-sensing chemical signals in the seawater microbial community. BMC Microbiol. 2001, 1, 27. [Google Scholar] [CrossRef]
- Chen, J.; Guo, Y.; Lu, Y.; Wang, B.; Sun, J.; Zhang, H.; Wang, H. Chemistry and Biology of Siderophores from Marine Microbes. Mar. Drugs 2019, 17, 562. [Google Scholar] [CrossRef]
- Kaufmann, G.F.; Sartorio, R.; Lee, S.-H.; Rogers, C.J.; Meijler, M.M.; Moss, J.A.; Clapham, B.; Brogan, A.P.; Dickerson, T.J.; Janda, K.D. Revisiting quorum sensing: Discovery of additional chemical and biological functions for 3-oxo-N-acylhomoserine lactones. Proc. Natl. Acad. Sci. USA 2005, 102, 309–314. [Google Scholar] [CrossRef]
- Daniel, R.; Simon, M.; Wemheuer, B. Editorial: Molecular Ecology and Genetic Diversity of the Roseobacter Clade. Front. Microbiol. 2018, 9, 1185. [Google Scholar] [CrossRef]
- Luo, H.; Moran, M.A. Evolutionary ecology of the marine Roseobacter clade. Microbiol Mol Biol Rev 2014, 78, 573–587. [Google Scholar] [CrossRef]
- Buchan, A.; Gonzalez, J.M.; Moran, M.A. Overview of the marine roseobacter lineage. Appl. Environ. Microbiol. 2005, 71, 5665–5677. [Google Scholar] [CrossRef]
- Cude, W.N.; Buchan, A. Acyl-homoserine lactone-based quorum sensing in the Roseobacter clade: Complex cell-to-cell communication controls multiple physiologies. Front. Microbiol. 2013, 4, 336. [Google Scholar] [CrossRef]
- González, J.M.; Kiene, R.P.; Moran, M.A. Transformation of Sulfur Compounds by an Abundant Lineage of Marine Bacteria in the α-Subclass of the Class Proteobacteria. Appl. Environ. Microbiol. 1999, 65, 3810–3819. [Google Scholar] [CrossRef]
- Slightom, R.N. Characterization of Motility and Surface Attachment in Thirteen Members of the Roseobacter Clade. Master’s Thesis, University of Tennessee, Knoxville, TN, USA, 2008. [Google Scholar]
- Buchan, A.; Collier, L.S.; Neidle, E.L.; Moran, M.A. Key Aromatic-Ring-Cleaving Enzyme, Protocatechuate 3,4-Dioxygenase, in the Ecologically Important Marine Roseobacter Lineage. Appl. Environ. Microbiol. 2000, 66, 4662–4672. [Google Scholar] [CrossRef]
- Tsukidate, H.; Otake, S.; Kato, Y.; Yoshimura, K.; Kitatsuji, M.; Yoshimura, E.; Suzuki, M. Iron Elution from Iron and Steel Slag Using Bacterial Complex Identified from the Seawater. Materials 2021, 14, 1477. [Google Scholar] [CrossRef]
- Fazary, A.E.; Taha, M.; Ju, Y.-H. Iron Complexation Studies of Gallic Acid. J. Chem. Eng. Data 2009, 54, 35–42. [Google Scholar] [CrossRef]
- Tamilmani, P.; Pandey, M.C. Iron binding efficiency of polyphenols: Comparison of effect of ascorbic acid and ethylenediaminetetraacetic acid on catechol and galloyl groups. Food Chem. 2016, 197, 1275–1279. [Google Scholar] [CrossRef] [PubMed]
- Singh, P.K.; Parsek, M.R.; Greenberg, E.P.; Welsh, M.J. A component of innate immunity prevents bacterial biofilm development. Nature 2002, 417, 552–555. [Google Scholar] [CrossRef] [PubMed]
- Bijlsma, J.; de Bruijn, W.J.C.; Hageman, J.A.; Goos, P.; Velikov, K.P.; Vincken, J.-P. Revealing the main factors and two-way interactions contributing to food discolouration caused by iron-catechol complexation. Sci. Rep. 2020, 10, 8288. [Google Scholar] [CrossRef]
- Andjelković, M.; Van Camp, J.; De Meulenaer, B.; Depaemelaere, G.; Socaciu, C.; Verloo, M.; Verhe, R. Iron-chelation properties of phenolic acids bearing catechol and galloyl groups. Food Chem. 2006, 98, 23–31. [Google Scholar] [CrossRef]
- Zhang, W.; Wang, R.; Sun, Z.; Zhu, X.; Zhao, Q.; Zhang, T.; Cholewinski, A.; Yang, F.; Zhao, B.; Pinnaratip, R.; et al. Catechol-functionalized hydrogels: Biomimetic design, adhesion mechanism, and biomedical applications. Chem. Soc. Rev. 2020, 49, 433–464. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Messersmith, R.E.; Sage, F.C.; Johnson, J.K.; Langevin, S.A.; Forsyth, E.R.; Hart, M.T.; Hoffman, C.M. Iron Sequestration by Galloyl–Silane Nano Coatings Inhibits Biofilm Formation of Sulfitobacter sp. Biomimetics 2023, 8, 79. https://doi.org/10.3390/biomimetics8010079
Messersmith RE, Sage FC, Johnson JK, Langevin SA, Forsyth ER, Hart MT, Hoffman CM. Iron Sequestration by Galloyl–Silane Nano Coatings Inhibits Biofilm Formation of Sulfitobacter sp. Biomimetics. 2023; 8(1):79. https://doi.org/10.3390/biomimetics8010079
Chicago/Turabian StyleMessersmith, Reid E., F. Connor Sage, James K. Johnson, Spencer A. Langevin, Ellen R. Forsyth, Meaghan T. Hart, and Christopher M. Hoffman. 2023. "Iron Sequestration by Galloyl–Silane Nano Coatings Inhibits Biofilm Formation of Sulfitobacter sp." Biomimetics 8, no. 1: 79. https://doi.org/10.3390/biomimetics8010079




