Improving the Visualization of the Adrenal Veins Using Virtual Monoenergetic Images from Dual-Energy Computed Tomography before Adrenal Venous Sampling
Abstract
:1. Introduction
2. Materials and Methods
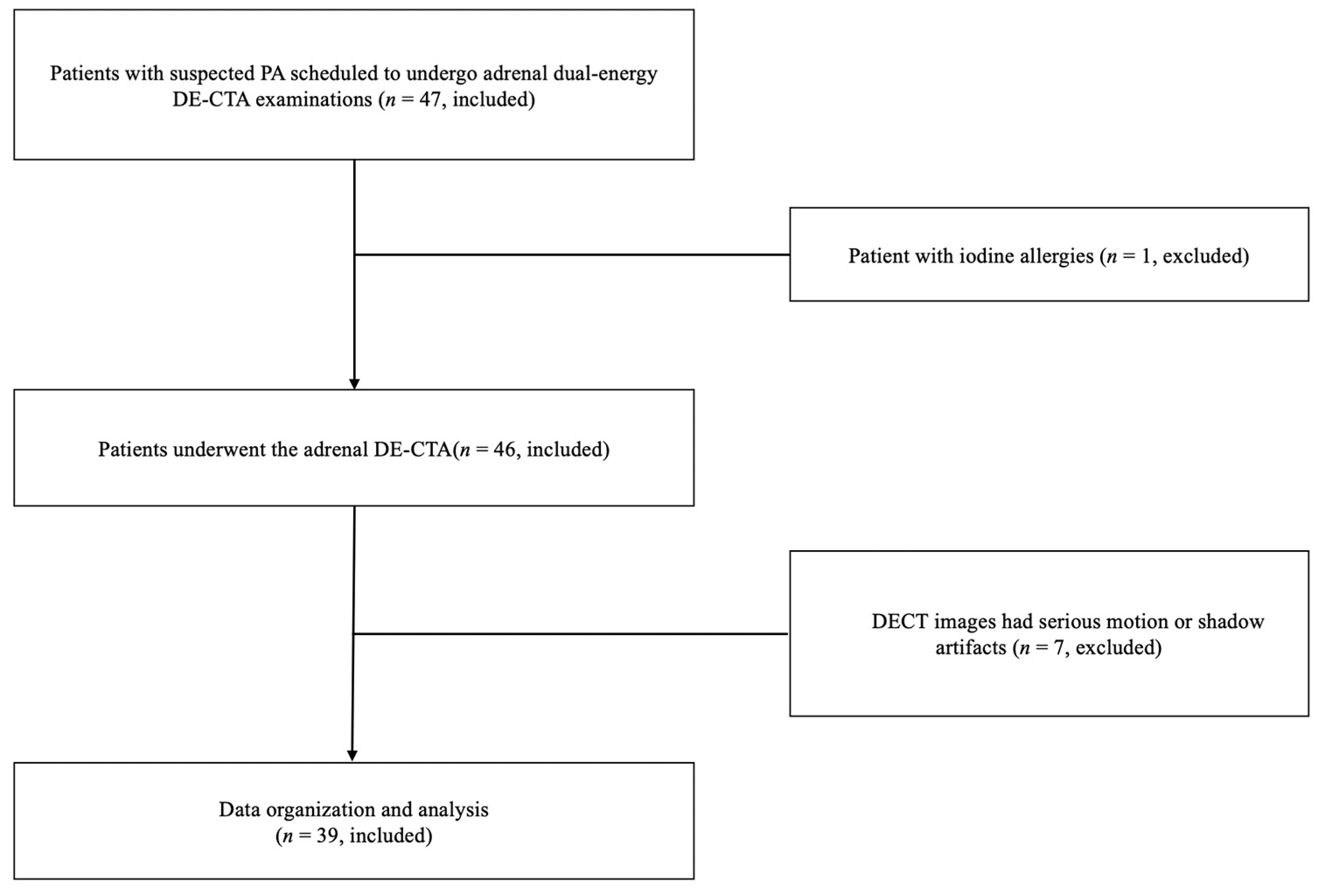
2.1. Patients
2.2. CT Examinations
2.3. Image Reconstruction
2.4. DE-CTA Objective Image Analysis
2.5. DECT Subjective Image Analysis
2.6. Statistical Analysis
3. Results
3.1. Participant Characteristics
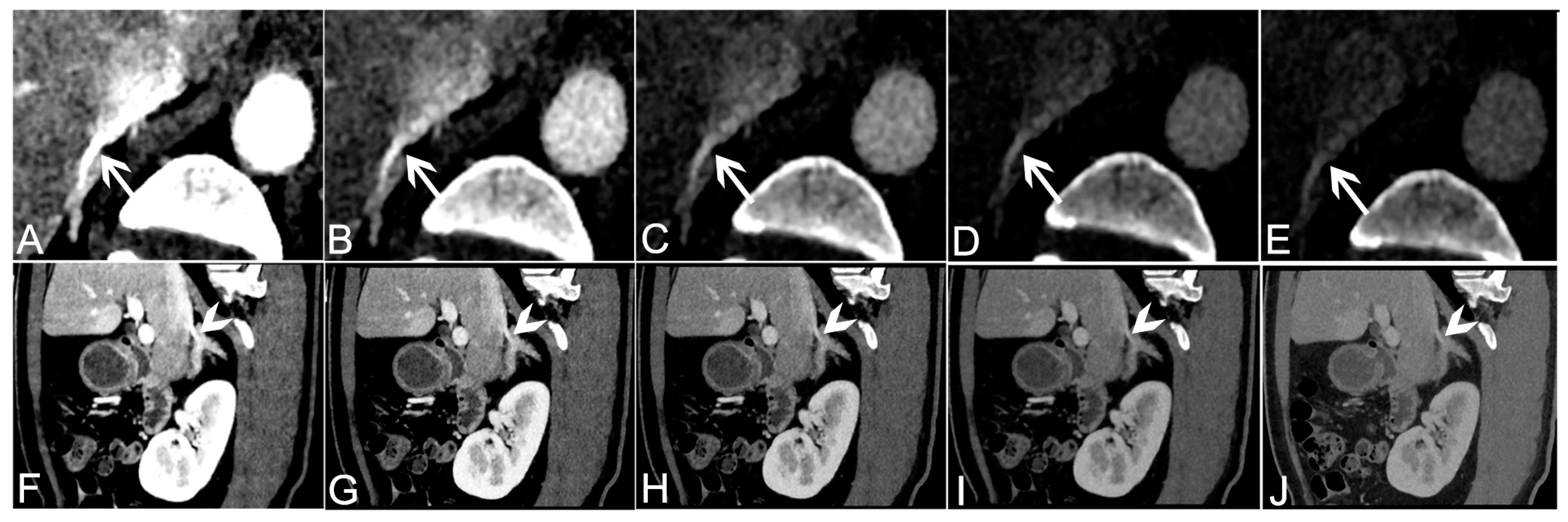
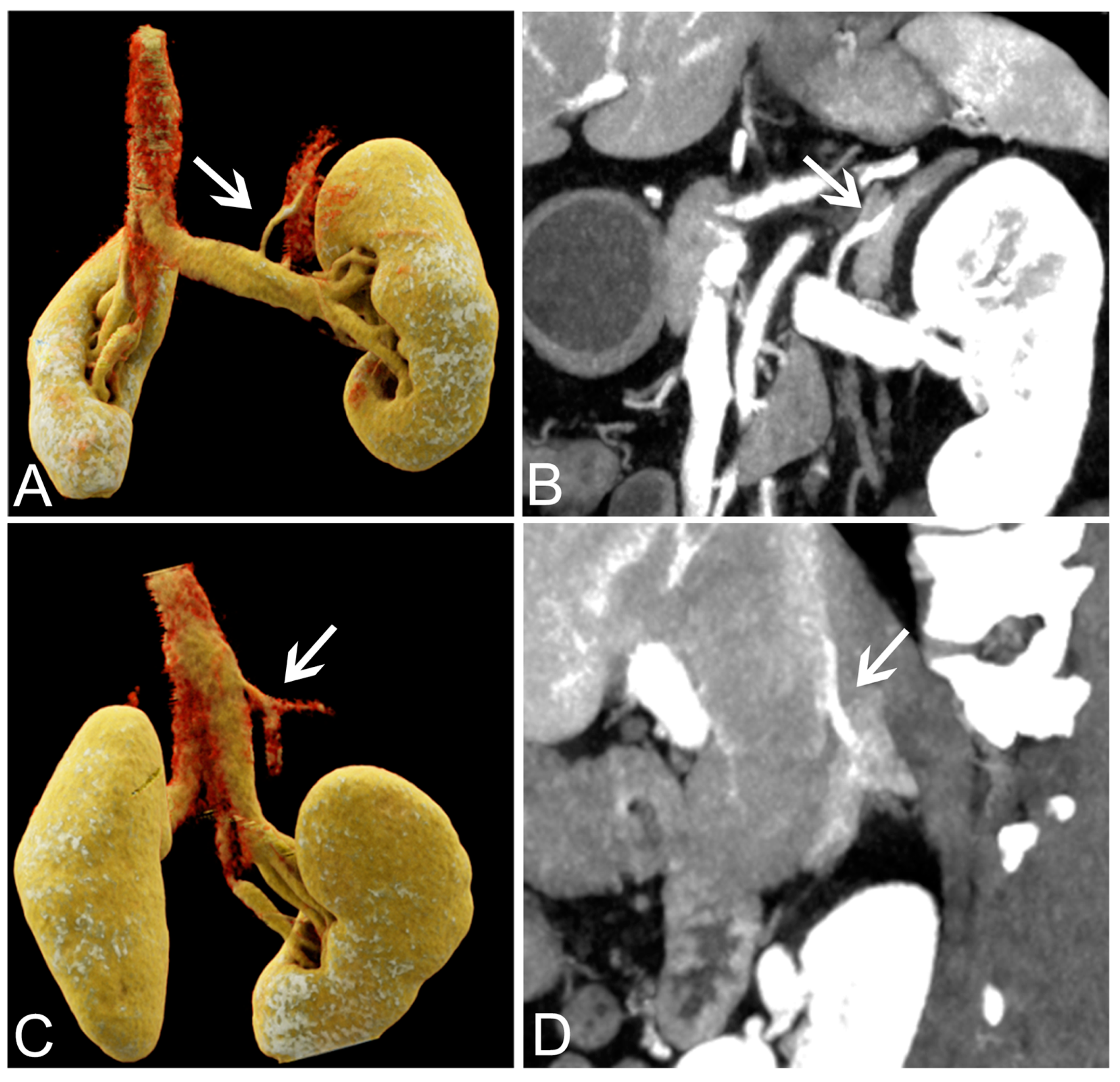
3.2. Objective Analysis of VMI+ Series from Venous Phase Data
3.3. Subjective Analysis of VMI+ Series from Venous Phase Data
3.4. Success Rate of AVS
4. Discussions
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Rimoldi, S.F.; Scherrer, U.; Messerli, F.H. Secondary arterial hypertension: When, who, and how to screen? Eur. Heart J. 2014, 35, 1245–1254. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rossi, G.P. Primary Aldosteronism: JACC State-of-the-Art Review. J. Am. Coll. Cardiol. 2019, 74, 2799–2811. [Google Scholar] [CrossRef] [PubMed]
- Williams, T.A.; Reincke, M. Management of endocrine disease: Diagnosis and management of primary aldosteronism: The Endocrine Society guideline 2016 revisited. Eur. J. Endocrinol. 2018, 179, R19–R29. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Melby, J.C.; Spark, R.F.; Dale, S.L.; Egdahl, R.H.; Kahn, P.C. Diagnosis and localization of aldosterone-producing adenomas by adrenal-vein cateterization. N. Engl. J. Med. 1967, 277, 1050–1056. [Google Scholar] [CrossRef] [PubMed]
- Rossi, G.P.; Maiolino, G.; Seccia, T.M. Adrenal Venous Sampling: Where Do We Stand? Endocrinol. Metab. Clin. N. Am. 2019, 48, 843–858. [Google Scholar] [CrossRef] [PubMed]
- Cesmebasi, A.; Du Plessis, M.; Iannatuono, M.; Shah, S.; Tubbs, R.S.; Loukas, M. A review of the anatomy and clinical significance of adrenal veins. Clin. Anat. 2014, 27, 1253–1263. [Google Scholar] [CrossRef]
- Stewart, P.M.; Allolio, B. Adrenal vein sampling for Primary Aldosteronism: Time for a reality check. Clin. Endocrinol. (Oxf.) 2010, 72, 146–148. [Google Scholar] [CrossRef]
- Ota, H.; Seiji, K.; Kawabata, M.; Satani, N.; Omata, K.; Ono, Y.; Iwakura, Y.; Morimoto, R.; Matsuura, T.; Kudo, M.; et al. Dynamic multidetector CT and non-contrast-enhanced MR for right adrenal vein imaging: Comparison with catheter venography in adrenal venous sampling. Eur. Radiol. 2016, 26, 622–630. [Google Scholar] [CrossRef]
- Noda, Y.; Goshima, S.; Nagata, S.; Kawada, H.; Kawai, N.; Koyasu, H.; Matsuo, M. Visualization of right adrenal vein: Comparison with three phase dynamic contrast-enhanced CT. Eur. J. Radiol. 2017, 96, 104–108. [Google Scholar] [CrossRef]
- Morita, S.; Nishina, Y.; Yamazaki, H.; Sonoyama, Y.; Ichihara, A.; Sakai, S. Dual adrenal venous phase contrast-enhanced MDCT for visualization of right adrenal veins in patients with primary aldosteronism. Eur. Radiol. 2016, 26, 2073–2077. [Google Scholar] [CrossRef]
- Albrecht, M.H.; Vogl, T.J.; Martin, S.S.; Nance, J.W.; Duguay, T.M.; Wichmann, J.L.; De Cecco, C.N.; Varga-Szemes, A.; van Assen, M.; Tesche, C.; et al. Review of clinical applications for virtual monoenergetic dual-energy CT. Radiology 2019, 293, 260–271. [Google Scholar] [CrossRef] [PubMed]
- Parakh, A.; Lennartz, S.; An, C.; Rajiah, P.; Yeh, B.M.; Simeone, F.J.; Sahani, D.V.; Kambadakone, A.R. Dual-energy CT images: Pearls and pitfalls. Radiographics 2021, 41, 98–119. [Google Scholar] [CrossRef] [PubMed]
- Parakh, A.; An, C.; Lennartz, S.; Rajiah, P.; Yeh, B.M.; Simeone, F.J.; Sahani, D.V.; Kambadakone, A.R. Recognizing and minimizing artifacts at dual-energy CT. Radiographics 2021, 41, 509–523. [Google Scholar] [CrossRef] [PubMed]
- Zhuo, S.Q.; Ma, H.L.; Liu, Z.L.; Ruan, G.Y.; Xie, C.B.; Huang, W.J.; Yang, G.J.; Li, Z.P.; Zeng, S.H. Evaluation of a second-generation monoenergetic reconstruction algorithm for lesion contrast and venous invasion in pancreatic ductal adenocarcinomas. Eur. Rev. Med. Pharmacol. Sci. 2019, 23, 9341–9350. [Google Scholar]
- Grant, K.L.; Flohr, T.G.; Krauss, B.; Sedlmair, M.; Thomas, C.; Schmidt, B. Assessment of an advanced image-based technique to calculate virtual monoenergetic computed tomographic images from a dual-energy examination to improve contrast-to-noise ratio in examinations using iodinated contrast media. Invest. Radiol. 2014, 49, 586–592. [Google Scholar] [CrossRef]
- Albrecht, M.H.; Scholtz, J.E.; Hüsers, K.; Beeres, M.; Bucher, A.M.; Kaup, M.; Martin, S.S.; Fischer, S.; Bodelle, B.; Bauer, R.W.; et al. Advanced image-based virtual monoenergetic dual-energy CT angiography of the abdomen: Optimization of kiloelectron volt settings to improve image contrast. Eur. Radiol. 2016, 26, 1863–1870. [Google Scholar] [CrossRef]
- Nomura, T.; Niwa, T.; Ozawa, S.; Imai, Y.; Hashimoto, J. Visibility of bronchial arteries using virtual and advanced virtual monoenergetic imaging. Acta Radiol. 2020, 61, 1618–1627. [Google Scholar] [CrossRef]
- Arendt, C.T.; Czwikla, R.; Lenga, L.; Wichmann, J.L.; Albrecht, M.H.; Booz, C.; Martin, S.S.; Leithner, D.; Tischendorf, P.; Blandino, A.; et al. Improved coronary artery contrast enhancement using noise-optimised virtual monoenergetic imaging from dual-source dual-energy computed tomography. Eur. J. Radiol. 2020, 122, 108666. [Google Scholar] [CrossRef] [Green Version]
- Liang, H.W.; Zhou, Y.; Zhang, Z.W.; Yan, G.W.; Du, S.L.; Zhang, X.H.; Li, X.Y.; Lv, F.J.; Zheng, Q.; Li, Y.M. Dual-energy CT with virtual monoenergetic images to improve the visualization of pancreatic supplying arteries: The normal anatomy and variations. Insights Imaging 2022, 13, 21. [Google Scholar] [CrossRef]
- Schabel, C.; Bongers, M.; Sedlmair, M.; Korn, A.; Grosse, U.; Mangold, S.; Claussen, C.D.; Thomas, C. Assessment of the hepatic veins in poor contrast conditions using dual energy CT: Evaluation of a novel monoenergetic extrapolation software algorithm. Rofo 2014, 186, 591–597. [Google Scholar] [CrossRef] [Green Version]
- Nakayama, K.; Shimohira, M.; Nakagawa, M.; Ozawa, Y.; Sawada, Y.; Ohta, K.; Ohashi, K.; Shibamoto, Y. Advanced monoenergetic reconstruction technique in dual-energy computed tomography for evaluation of vascular anatomy before adrenal vein sampling. Acta Radiol. 2020, 61, 282–288. [Google Scholar] [CrossRef] [PubMed]
- Kim, T.M.; Choi, Y.H.; Cheon, J.E.; Kim, W.S.; Kim, I.O.; Park, J.E.; Shin, S.M.; Pak, S.Y.; Krauss, B. Optimal Kiloelectron Volt for Noise-Optimized Virtual Monoenergetic Images of Dual-Energy Pediatric Abdominopelvic Computed Tomography: Preliminary Results. Korean J. Radiol. 2019, 20, 283–294. [Google Scholar] [CrossRef] [PubMed]
- Lv, P.; Liu, J.; Wu, R.; Hou, P.; Hu, L.; Gao, J. Use of non-linear image blending with dual-energy CT improves vascular visualization in abdominal angiography. Clin. Radiol. 2014, 69, e93–e99. [Google Scholar] [CrossRef] [PubMed]
- Sudarski, S.; Apfaltrer, P.; Nance, J.W., Jr.; Schneider, D.; Meyer, M.; Schoenberg, S.O.; Fink, C.; Henzler, T. Optimization of keV-settings in abdominal and lower extremity dual-source dual-energy CT angiography determined with virtual monoenergetic imaging. Eur. J. Radiol. 2013, 82, e574–e581. [Google Scholar] [CrossRef]
- Ren, H.; Zhen, Y.; Gong, Z.; Wang, C.; Chang, Z.; Zheng, J. Assessment of Virtual Monoenergetic Images in Run-off Computed Tomography Angiography: A Comparison Study to Conventional Images From Spectral Detector Computed Tomography. J. Comput. Assist. Tomogr. 2021, 45, 232–237. [Google Scholar] [CrossRef]
- Albrecht, M.H.; Trommer, J.; Wichmann, J.L.; Scholtz, J.E.; Martin, S.S.; Lehnert, T.; Vogl, T.J.; Bodelle, B. Comprehensive comparison of virtual monoenergetic and linearly blended reconstruction techniques in third-generation dual-source dual-energy computed tomography angiography of the thorax and abdomen. Invest. Radiol. 2016, 51, 582–590. [Google Scholar] [CrossRef]
- Albrecht, M.H.; Scholtz, J.E.; Kraft, J.; Bauer, R.W.; Kaup, M.; Dewes, P.; Bucher, A.M.; Burck, I.; Wagenblast, J.; Lehnert, T.; et al. Assessment of an advanced monoenergetic reconstruction technique in dual-energy computed tomography of head and neck cancer. Eur. Radiol. 2015, 25, 2493–2501. [Google Scholar] [CrossRef]
- Frellesen, C.; Fessler, F.; Hardie, A.D.; Wichmann, J.L.; De Cecco, C.N.; Schoepf, U.J.; Kerl, J.M.; Schulz, B.; Hammerstingl, R.; Vogl, T.J.; et al. Dual-energy CT of the pancreas: Improved carcinoma-to-pancreas contrast with a noise-optimized monoenergetic reconstruction algorithm. Eur. J. Radiol. 2015, 84, 2052–2058. [Google Scholar] [CrossRef]
| Patient Characteristics | Value (%) |
|---|---|
| Age (years old) | 45.08 ± 11.98 |
| Sex | |
| male | n = 19 (48.71) |
| female | n = 20 (51.29) |
| BMI (kg/m2) | 24.90 ± 3.41 |
| 40 keV | 50 keV | 60 keV | 70 keV | 80 keV | pa | pb | ||||
|---|---|---|---|---|---|---|---|---|---|---|
| p 40 vs. 50 | p 40 vs. 60 | p 40 vs. 70 | p 40 vs. 80 | |||||||
| Right adrenal vein | ||||||||||
| CT value | 489.72 ± 119.72 | 377.23 ± 79.42 | 244.49 ± 55.15 | 187.30 ± 40.44 | 150.63 ± 31.29 | <0.001 * | 0.005 * | <0.001 * | <0.001 * | <0.001 * |
| noise | 31.60 ± 8.81 | 21.95 ± 5.86 | 16.11 ± 4.14 | 12.54 ± 3.17 | 11.62 ± 3.58 | <0.001 * | 0.005 * | <0.001 * | <0.001 * | <0.001 * |
| CNR | 14.82 ± 5.39 | 13.05 ± 4.78 | 11.16 ± 4.09 | 9.36 ± 3.54 | 8.57 ± 1.06 | <0.001 * | 0.005 * | <0.001 * | <0.001 * | <0.001 * |
| SNR | 16.18 ± 4.88 | 16.02 ± 4.73 | 15.83 ± 4.64 | 15.62 ± 4.62 | 14.39 ± 6.32 | <0.001 * | 1 | 0.88 | 0.28 | <0.001 * |
| Left adrenal vein | ||||||||||
| CT value | 528.05 ± 98.26 | 362.44 ± 65.69 | 261.72 ± 46.34 | 199.60 ± 34.91 | 150.63 ± 31.29 | <0.001 * | 0.005 * | <0.001 * | <0.001 * | <0.001 * |
| noise | 33.95 ± 9.04 | 23.78 ± 6.06 | 17.65 ± 4.28 | 13.91 ± 3.22 | 11.55 ± 2.60 | <0.001 * | 0.005 * | <0.001 * | <0.001 * | <0.001 * |
| CNR | 17.42 ± 5.08 | 15.27 ± 4.62 | 12.98 ± 4.06 | 10.77 ± 3.49 | 8.83 ± 3.02 | <0.001 * | 0.005 * | <0.001 * | <0.001 * | <0.001 * |
| SNR | 16.48 ± 4.89 | 16.08 ± 4.71 | 15.59 ± 4.53 | 15.03 ± 4.40 | 14.51 ± 4.33 | 0.253 | 0.053 | <0.001 * | <0.001 * | <0.001 * |
| pc | ||||||||||
| p 50 vs. 40 | p 50 vs. 60 | p 50 vs.70 | p 50 vs.80 | |||||||
| Right adrenal gland | ||||||||||
| CT value | 331.10 ± 88.59 | 227.49 ± 59.53 | 164.48 ± 42.02 | 125.62 ± 31.39 | 100.71 ± 24.74 | <0.001 * | 0.005 * | 0.005 * | <0.001 * | <0.001 * |
| noise | 28.45 ± 5.48 | 20.46 ± 3.83 | 15.69 ± 2.88 | 12.82 ± 2.35 | 11.03 ± 2.06 | <0.001 * | 0.005 * | 0.005 * | <0.001 * | <0.001 * |
| CNR | 8.95 ± 3.85 | 7.55 ± 3.42 | 6.08 ± 2.98 | 4.67 ± 2.61 | 1.94 ± 1.32 | <0.001 * | 0.005 * | 0.005 * | 0.001 * | <0.001 * |
| SNR | 11.87 ± 3.15 | 11.37 ± 3.09 | 10.74 ± 2.99 | 10.06 ± 2.84 | 9.39 ± 2.68 | <0.001 * | 0.012 | 0.012 | <0.001 * | <0.001 * |
| Left adrenal gland | ||||||||||
| CT value | 324.22 ± 101.86 | 220.39 ± 68.26 | 157.71 ± 48.32 | 119.06 ± 36.20 | 94.28 ± 28.60 | <0.001 * | 0.005 * | 0.005 * | <0.001 * | <0.001 * |
| noise | 31.51 ± 5.31 | 22.70 ± 3.88 | 17.40 ± 3.12 | 14.20 ± 2.75 | 12.18 ± 2.57 | <0.001 * | 0.005 * | 0.005 * | <0.001 * | <0.001 * |
| CNR | 9.25 ± 4.08 | 7.55 ± 3.42 | 5.84 ± 2.92 | 4.22 ± 2.51 | 2.83 ± 2.21 | <0.001 * | 0.005 * | 0.005 * | <0.001 * | <0.001 * |
| SNR | 10.51 ± 3.61 | 9.95 ± 3.44 | 9.33 ± 3.29 | 8.69 ± 3.13 | 8.07 ± 2.98 | <0.001 * | 0.081 | 0.081 | <0.001 * | <0.001 * |
| Right kidney | ||||||||||
| CT value | 240.82 ± 59.67 | 167.23 ± 39.53 | 122.47 ± 27.40 | 94.87 ± 20.06 | 77.18 ± 15.49 | <0.001 * | 0.005 * | 0.005 * | <0.001 * | <0.001 * |
| noise | 31.44 ± 4.66 | 21.91 ± 3.33 | 16.17 ± 2.61 | 12.61 ± 2.23 | 10.52 ± 2.02 | <0.001 * | 0.005 * | 0.005 * | <0.001 * | <0.001 * |
| CNR | 19.48 ± 7.51 | 12.83 ± 6.56 | 14.41 ± 5.84 | 11.9 ± 5.05 | 5.46 ± 2.36 | <0.001 * | 0.005 * | 0.005 * | <0.001 * | <0.001 * |
| SNR | 7.76 ± 2.02 | 7.75 ± 1.98 | 7.72 ± 1.98 | 7.66 ± 2.02 | 7.58 ± 2.10 | 0.023 | 0.72 | 0.56 | 0.13 | <0.01 * |
| Left kidney | ||||||||||
| CT value | 255.30 ± 60.60 | 176.41 ± 40.23 | 128.43 ± 27.98 | 98.84 ± 20.57 | 79.88 ± 15.98 | <0.001 * | 0.006 * | 0.006 * | <0.001 * | <0.001 * |
| noise | 29.75 ± 5.38 | 20.65 ± 3.72 | 15.17 ± 2.77 | 12.42 ± 1.39 | 10.46 ± 1.22 | <0.001 * | 0.006 * | 0.006 * | <0.001 * | <0.001 * |
| CNR | 20.23 ± 7.71 | 17.53 ± 6.73 | 15.13 ± 5.04 | 11.92 ± 4.83 | 9.52 ± 4.03 | <0.001 * | 0.006 * | 0.006 * | <0.001 * | <0.001 * |
| SNR | 8.89 ± 2.75 | 8.86 ± 2.67 | 8.79 ± 2.62 | 8.68 ± 2.69 | 8.54 ± 2.63 | <0.001 * | 0.51 | 0.38 | 0.03 * | 0.005 * |
| Liver | ||||||||||
| CT value | 232.86 ± 42.20 | 182.31 ± 39.80 | 141.17 ± 28.70 | 115.81 ± 22.09 | 99.55 ± 18.02 | <0.001 * | 0.005 * | 0.005 * | <0.001 * | <0.001 * |
| noise | 29.87 ± 3.34 | 20.93 ± 2.30 | 15.60 ± 1.71 | 12.42 ± 1.39 | 10.46 ± 1.22 | <0.001 * | 0.005 * | 0.005 * | <0.001 * | <0.001 * |
| CNR | 5.87 ± 2.43 | 5.23 ± 2.19 | 4.54 ± 1.97 | 3.89 ± 1.81 | 1.91 ± 1.12 | <0.001 * | 0.008 * | 0.01 * | <0.001 * | <0.001 * |
| SNR | 8.45 ± 2.06 | 8.81 ± 2.13 | 9.18 ± 2.22 | 9.48 ± 2.32 | 9.71 ± 2.44 | <0.001 * | 0.02 * | 0.03 * | <0.001 * | <0.001 * |
| Spleen | ||||||||||
| CT value | 381.84 ± 78.21 | 266.61 ± 51.41 | 196.52 ± 35.14 | 153.30 ± 25.15 | 125.59 ± 18.79 | <0.001 * | 0.005 * | 0.005 * | <0.001 * | <0.001 * |
| noise | 28.73 ± 4.46 | 19.95 ± 2.87 | 14.71 ± 2.01 | 11.56 ± 1.55 | 9.61 ± 1.33 | <0.001 * | 0.005 * | 0.005 * | <0.001 * | <0.001 * |
| CNR | 11.52 ± 3.4 | 10.02 ± 2.91 | 8.45 ± 2.44 | 6.96 ± 2.03 | 5.65 ± 1.71 | <0.001 * | 0.005 * | 0.005 * | <0.001 * | <0.001 * |
| SNR | 13.52 ± 3.02 | 13.59 ± 3.01 | 13.61 ± 3.01 | 13.54 ± 3.02 | 13.39 ± 3.07 | 0.62 | ||||
| Pancreas | ||||||||||
| CT value | 279.40 ± 61.93 | 198.23 ± 40.64 | 148.81 ± 27.85 | 118.33 ± 20.16 | 98.79 ± 15.43 | <0.001 * | 0.005 * | 0.005 * | <0.001 * | <0.001 * |
| noise | 29.12 ± 4.00 | 20.63 ± 2.64 | 15.57 ± 1.92 | 12.56 ± 1.55 | 10.17 ± 1.77 | <0.001 * | 0.005 * | 0.005 * | <0.001 * | <0.001 * |
| CNR | 7.52 ± 4.46 | 6.43 ± 2.3 | 5.3 ± 1.91 | 4.23 ± 1.59 | 3.30 ± 1.36 | <0.001 * | 0.005 * | 0.005 * | <0.001 * | <0.001 * |
| SNR | 9.71 ± 2.28 | 9.74 ± 2.26 | 7.50 ± 2.11 | 9.61 ± 2.37 | 10.13 ± 3.61 | <0.001 * | 1 | <0.001 * | 0.25 | 0.77 |
| 40 keV VMI+ | 50 keV VMI+ | 60 keV VMI+ | 70 keV VMI+ | 80 keV VMI+ | |
|---|---|---|---|---|---|
| Right Adrenal Vein | |||||
| 4 (excellent) | 26 | 3 | 3 | 0 | 0 |
| 3 (good) | 13 | 31 | 15 | 4 | 0 |
| 2 (fair) | 0 | 5 | 21 | 6 | 7 |
| 1 (poor) | 0 | 0 | 0 | 29 | 32 |
| detectability | 100% (39/39) | 87.18% (34/39) | 46.15% (18/39) | 10.26% (4/39) | 0% (0/39) |
| score | 4 (4,4) | 3 (3,3) | 2 (2,3) | 2 (2,2) | 1 (1,1) |
| p | <0.01 * | <0.001 * | <0.001 * | <0.001 * | |
| kappa | 0.80 | 0.86 | 0.86 | 0.73 | 0.80 |
| Left adrenal vein | |||||
| 4 (excellent) | 24 | 7 | 0 | 0 | 0 |
| 3 (good) | 14 | 30 | 25 | 10 | 0 |
| 2 (fair) | 1 | 2 | 14 | 26 | 20 |
| 1 (poor) | 0 | 0 | 0 | 3 | 19 |
| detectability | 97.44% (38/39) | 94.87% (37/39) | 64.11% (25/39) | 25.64% (10/39) | 0% (0/39) |
| score | 4 (4,4) | 3 (3,3) | 3 (2,3) | 2 (2,3) | 2 (1,2) |
| p | 0.008 * | <0.001 * | <0.001 * | <0.001 * | |
| kappa | 0.79 | 0.87 | 0.89 | 0.78 | 0.89 |
| Image Quality | p | Kappa | |
|---|---|---|---|
| 40 keV VMI+ | 3 (3,4) | <0.001 * | 0.86 |
| 50 keV VMI+ | 5 (5,5) | 0.72 | |
| 60 keV VMI+ | 3 (3,3) | <0.001 * | 0.72 |
| 70 keV VMI+ | 2 (2,3) | <0.001 * | 0.77 |
| 80 keV VMI+ | 2 (1,2) | <0.001 * | 0.85 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, Y.; Chen, X.; Lu, G.; Su, Y.; Yang, L.; Shi, G.; Zhang, F.; Zhuo, J.; Duan, X.; Hu, H. Improving the Visualization of the Adrenal Veins Using Virtual Monoenergetic Images from Dual-Energy Computed Tomography before Adrenal Venous Sampling. Tomography 2023, 9, 485-496. https://doi.org/10.3390/tomography9020040
Wang Y, Chen X, Lu G, Su Y, Yang L, Shi G, Zhang F, Zhuo J, Duan X, Hu H. Improving the Visualization of the Adrenal Veins Using Virtual Monoenergetic Images from Dual-Energy Computed Tomography before Adrenal Venous Sampling. Tomography. 2023; 9(2):485-496. https://doi.org/10.3390/tomography9020040
Chicago/Turabian StyleWang, Yu, Xiaohong Chen, Guoxiong Lu, Yun Su, Lingjie Yang, Guangzi Shi, Fang Zhang, Jiayi Zhuo, Xiaohui Duan, and Huijun Hu. 2023. "Improving the Visualization of the Adrenal Veins Using Virtual Monoenergetic Images from Dual-Energy Computed Tomography before Adrenal Venous Sampling" Tomography 9, no. 2: 485-496. https://doi.org/10.3390/tomography9020040
APA StyleWang, Y., Chen, X., Lu, G., Su, Y., Yang, L., Shi, G., Zhang, F., Zhuo, J., Duan, X., & Hu, H. (2023). Improving the Visualization of the Adrenal Veins Using Virtual Monoenergetic Images from Dual-Energy Computed Tomography before Adrenal Venous Sampling. Tomography, 9(2), 485-496. https://doi.org/10.3390/tomography9020040






