The present study provides a comprehensive picture of the biological cycle of the M. merlangus stock in the NAS, focusing on the population structure, growth parameters, and reproductive traits. For this area, to our knowledge, the growth parameters and fecundity values have never been reported before. Moreover, this is the first time that populations sampled with a time-shift of 30 years from the same fishing ground are compared in terms of population structure and growth.
4.1. Historical Comparison
The temporal comparison revealed a reduction of the mean size over the considered period, with a higher proportion of large individuals in the 1990–1991 sample with respect to the 2020–2021 ones. This finding confirms the declining trend in size reported for several fish stocks at different latitudes mainly due to fishing exploitation [
54]. Fishing affects ecosystems by removing selectively larger specimens of commercial species, determining short-term (e.g., removal of spawners from the stock) and long-term effects (e.g., selection of fast-growing and early maturing specimens) [
18,
55,
56]. Nevertheless, since fishing exerts selective pressure by removing larger and, consequently, older individuals [
57,
58], it was expected for the same difference in the age structure to be observed. Conversely, our data revealed that in both populations, the maximum age was similar (30–32 months). An effect on mean size-at-age was observed instead, with higher sizes per age class in the 1990–1991 sample compared to the 2020–2021 ones. These results together suggest that the observed reduction in length classes is related to different growth performance, which may be explained by metabolic or trophic constraints.
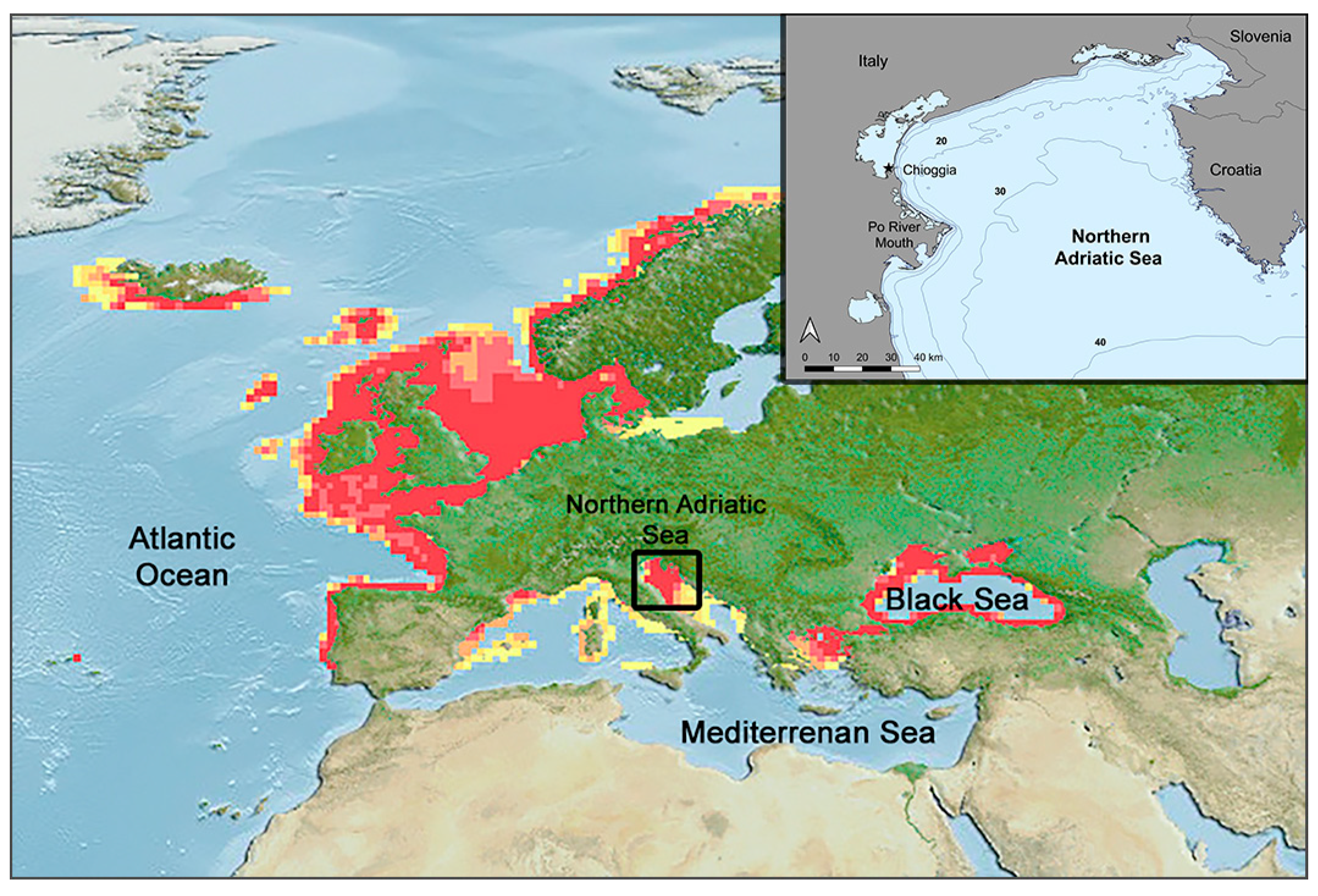
The physiological performance of marine fish, with them being ectotherms, is tightly controlled by water temperature, and their essential body functions (growth and reproduction) are optimal only within the thermal tolerance of the species [
59]. Oxygen consumption (i.e., metabolic rate) is higher in organisms living in warmer waters and it is proportional to body size [
60]. Thus, as the water temperature increases, the maximum body size decreases to balance the increase in the metabolic rate [
61,
62]. Generally, the sensitivity to temperature changes in temperate species is low but it may vary depending on the specific location within its distribution range. Several studies have shown that under ocean warming, sensible species have been shifting their distribution towards higher latitudes and deeper waters to maintain physiological homeostasis [
11,
19]. However, sometimes a geographic constraint may prevent poleward migration, as in the case of enclosed seas, such as the Mediterranean Sea. Within this area, species distributions are highly clustered depending on the local features of water masses, and it is possible to find a wide range of species along the latitudinal axis: from the cold waters to the sub-tropical ones [
63]. The coldest parts of the Mediterranean Sea are the Gulf of Lion and the NAS. Being located in the northern limit of the Mediterranean latitudinal range, these areas are defined as “cul-de-sacs”, from which cold-water species will have no possibility of escaping. Recent observational data showed that the AS, and in particular the NAS, is the sub-basin with the highest warming trend in sea surface temperature in the Mediterranean Sea over the last 20 years [
27,
64] (
Figure A2), which could determine a strong reduction in cold-adapted species during the ongoing century and, in the worst case scenario, even their local extinction [
29]. This phenomenon is probably occurring in the Adriatic population of whiting, whose distribution is restricted to the central and northern parts of the sub-basin, showing high abundance only in the northern part [
39]. Here, land boundaries could allow only a southward shift, which is prevented by the positive gradient of sea temperature and depth along the Adriatic latitudinal axis, creating a physical barrier. In fact, besides thermal tolerance, another peculiar feature of whiting is the preference for shallow waters, with the species being common at depths lower than 100 meters [
65]. It is possible, therefore, to explain the observed difference in growth efficiency between populations in terms of a metabolic constraint due to the thermal tolerance of this species, limiting the increase in body size [
59]. Moreover, the observed trend confirms what is expected from the gill–oxygen limitation theory [
12], according to which fish body growth is limited by the gills’ capability to provide oxygen through their surface. As oxygen solubility in water decreases with temperature [
66], body size is expected to decrease, through phenotypic plasticity, to maintain the scope for aerobic activity. This hypothesis is further corroborated by the reduction in the condition factor, which is associated not only with low oxygen levels but also food limitations [
67]. Furthermore, considering the potential effects of oxygen limitation on size at maturity, GSI, and egg production as well [
68], it can thus be suggested that reproductive functions have undergone important changes over the last few decades. Unfortunately, no data about the reproductive traits were available from the 1990–1991 samples to test this phenomenon.
One interesting finding is that the timing of otolith seasonal depositions revealed a different pattern between the investigated periods. The edge-zone analysis pointed out a difference in the opaque deposition timing over the warm season, showing an opaque deposition rate > 90% from April to September in the 1990–1991 samples and from May to July in the 2020–2021 samples (no samples in August), therefore with a reduction in the length of the period. Several factors are known to influence the periodicity of opaque and translucent zones in otoliths, such as geographical distributions, life-history events (settlement and reproduction), food availability, and water temperature [
69]. Nevertheless, temperature is known to play a primary role in determining deposition patterns [
41,
70,
71], and consequently, the increased temperature may have affected the biomineralization process negatively.
Another possible explanation for the decreased growth performance may be related to another environmental driver, primary production, which has been recognized as one of the main historical drivers in the Mediterranean ecosystem [
72]. For example, this is noticeable in the AS, where forage fish population dynamics can be influenced by river discharge, and consequently, by primary production [
73]. As a result, the declining trend in freshwater inputs observed in the Mediterranean Sea [
74] could have played a significant role in the observed pattern, reducing the prey quantity or quality for mesopredatory species such as whiting.
We observed an unexpected outcome when comparing the LFDs by sex related to the marked sexual dimorphism in size featuring in the 2020–2021 sample. While in the 1990–1991 sample, the modal distributions of the two sexes mostly overlap (except for the largest length classes, dominated by females), a 5–6 cm gap was noticed between the sexes in TL modal classes in 2020–2021. As already reported for other areas, whiting females attain a larger size than males, showing higher L
∞ and lower k [
75,
76,
77]. The increased difference in size may suggest a sex-dependent response related to the different energetic investments in gamete production and the effect of body size for each sex [
78,
79]. Although to our knowledge, a similar finding has never been pointed out in previous studies, we hypothesize that the integrated effect of fishing and warming towards smaller sizes exerts a stronger influence on males because of their lower reproductive cost with respect to females. On the other hand, females, despite being subjected to the same conditions, are less subjected to size decrease because their reproductive output (i.e., quantity and possibly quality of offspring) is positively correlated with size [
79]. Another possible explanation could be related to modifications in energy uptake patterns between males and females, controlled by sex-specific behaviour, as already reported for whiting by [
76]. Females ingest a significantly higher amount of food items and show lower percentages of empty stomachs compared to males. Fishing and warming could have acted in enhancing the degree of differentiation between these behavioral patterns, determining an increased dimorphism in growth rates over three decades.
Consideration must be made about the timing of the sample collection. Even if the same methodology was used, these results need to be interpreted with caution because of the longtime shift between sampling collection and its related issues. First, the samples came from two discrete sampling activities, thus providing detailed pictures of two restricted periods that were not appropriate to highlight a trend over three decades. The adaptive phenotypic plasticity may influence population dynamics even on a short-term scale, and it is favored by heterogeneous environments such as the AS [
13]. Consequently, populations that are subjected to environmental variability may have natural oscillations in biological parameters in relation to external conditions. Finally, the skewed distribution observed in 1990–1991 is probably due to the higher proportion of specimens coming from the summer months, a period during which YOY are more abundant than adults [
80]. In addition, it may be also related to the high fishing pressure exerted in the late 20th century, being the typical length structure featuring heavily exploited stocks [
81].
4.2. Population Biological Traits
The size range found was smaller than that previously observed in the AS [
31] and similar to the Black Sea [
82,
83]. As already reported for other gadoids [
84], the Mediterranean maximum size was much smaller than the Atlantic one [
76,
85,
86]. The presence of the opaque margin during the warmer months in the otoliths analyzed confirms the deposition pattern of the species, indicating that an opaque deposition is associated with the fast growth season [
84,
87,
88].
The age classes and maximum age estimated in the present study were similar to those previously observed in the AS [
30,
80] and lower than those reported in the Atlantic Ocean and Black Sea [
76,
82,
85,
89]. Surprisingly, despite similar length ranges, our age estimates are lower than the Black Sea population, indicating faster growth and a shorter lifespan in the AS population. Moreover, this result is noticeable by comparing the growth rate and the growth performance between different areas, values of which in the AS population are the highest reported in the literature [
46,
77] (
Figure 9). In particular, our estimates of
k were higher than those previously reported probably because of the inclusion of the YOY data in the VBGF, whose age estimation (in months) was validated through the analysis of the YOY LFDs collected between April and June (
Figure A1). Generally, it is difficult to obtain length-at-age data of juvenile stages (when the growth rate reaches its maximum), and this often leads to an underestimation of k values [
84]. Another explanation for the difference in the age–length results could be attributed to the different methodologies used to perform the age readings. After several attempts, we obtained consistent results with the “burnt and broken” otoliths, but there is no general agreement about the best method to use, even if the latest ICES guidelines [
41] suggest using broken or sectioned preparations in this species. Additionally, age reading studies often lack a validation step (e.g., edge/length–frequency analysis, marking and recapturing, and tagging), limiting the comparability the results.
Sexual maturity is reached within the first year of life in males and females at 16.1 and 16.8 cm TL, respectively. These estimates are lower than previous observations carried out in the AS [
30,
80] and, as expected, in the Atlantic Ocean [
85]; on the other hand, our estimates were higher than those reported in the Black Sea [
90]. The difference observed between our updated data and the previous studies in the AS agrees with our expectations, considering the reduction in L
∞ and the positive relationship between L
∞ and L
50 [
91]. Macroscopic observations on gonads and GSI trends indicated clearly that spawning takes place in winter, showing a narrower spawning season compared to the Atlantic Ocean and Black Sea, where it occurs from January to September and throughout the year, respectively [
65,
90]. During the spawning peak (December), the mean GSI value of the females (11%) is one order of magnitude higher than the males (1%), confirming the highly different reproductive efforts between the sexes [
30,
90].
The present study has provided fecundity estimates for the first time for whiting in the AS, which are within the range provided by previous studies carried out in the north east Atlantic Ocean and the Black Sea [
65,
92]. Considering that whiting is a batch spawner [
52] and that the average F was 18.1 times higher than the average F
b, a female could potentially lay 18 batches during the spawning season. Regarding the OFDs, the presence of at least two separated modal groups was clear only in females in advanced ovarian developmental stages (with hydrated oocytes) and we often observed nonhomogeneous oocyte size distributions, without any recognizable modal group. Although whiting is considered to have a group-synchronous ovarian organization, our data suggest the possibility of an asynchronous organization [
52], but the low number (15) of females used may not be adequate to infer the spawning pattern of this species.
The differences in life history traits observed between the Mediterranean Sea and Black Sea populations can be related to genetic divergence between them. Previous studies, based on morphological and meristic features, revealed differentiation between the AS and the Black Sea, raising a debate about the existence of two subspecies [
93,
94]. Recent molecular analyses have attempted to answer this question, stating that, despite some genetic differentiation between different sampling sites in the Black and Aegean Seas, there is no evidence supporting the existence of the presumed subspecies [
95].