1. Introduction
The aquaculture industry is experiencing rapid global growth and it is recognized as the fastest-growing sector in worldwide food production, accompanied by a surge in demand for fish products in the consumer market [
1]. Ensuring the sustainable health of aquatic organisms is an important endeavor for the human food supply chain. Disease outbreaks are widely acknowledged as a major impediment to the growth of the aquaculture industry, resulting in substantial global losses annually [
2]. Antibiotics are now widely used in the aquaculture industry to minimize losses of aquatic products caused by bacterial pathogens [
3]. Yet, antibiotic-resistant (AMR) bacteria are also born out of the feed industry’s practice of indiscriminate and constant abuse of production [
4], its genes and residues come with it [
5]. When AMR bacteria evolve and spread in natural water bodies, they not only cause disease and death in aquatic animals but also have a profoundly negative impact on economic development [
6]. Leading some countries to impose restrictions or bans on the widespread antimicrobial agent usage in aquaculture [
7]. Consequently, there is an imperative need for non-toxic, environmentally friendly, and highly efficient biological agents that can serve as alternatives to antibiotics within the aquaculture industry. Immunostimulants represent one such option that can effectively mitigate the outbreak of aquatic animal diseases [
8]. Understanding natural immune responses in crustaceans plays an important role in studies applying immunotherapy to enhance disease resistance among cultured animals.
Crustaceans are known to lack an acquired immune system, relying mainly on innate immunity [
9]. PRRs (pattern recognition receptors) enable the immune system to distinguish between “self” and not-self”, playing a crucial role in initiating the immune response [
10]. Various PRRs have been established in crustaceans, including
LBPs (lipopolysaccharide-binding proteins) [
11],
LGBPs (lipopolysaccharide and β-1,3-glucan binding proteins) [
12],
GNBPs (gram-negative binding proteins) [
13],
PGRPs (peptidoglycan recognition proteins binding proteins) [
14],
bGBPs (β-glucan binding proteins) [
15],
CTLs (C-type lectins) [
16],
TEPs (sulfate-containing proteins) [
17],
SCRs (scavenger receptors) [
18],
FBGLs (fibrinogen-like structural domain immunoselectors) [
19], and
TLRs (Toll-like receptors) [
20]. Upon recognition of corresponding PAMPs, PRR-PAMP interactions trigger a cascade of immune reactions. Phagocytosis mediated primarily by hyalinocytes eliminates foreign invasive substances [
21]; primarily encapsulation and cytotoxicity predominantly carried out by semigranular cells counteract parasites and fungi invading the host [
22]; activation of Toll and IMD (immune deficiency) signaling pathways regulates antimicrobial peptides production [
23,
24];
lectin secretion and proPO-AS (Prophenoloxidase activating system) activation occur as well [
25,
26]. Furthermore, we observed a correlation between immunization and growth performance in crustaceans [
27,
28,
29]. Therefore, it is worthwhile to explore the research directions that stimulate the interaction between PRRs and PAMPs using immunostimulants to enhance environmental resistance and growth performance in crustaceans.
Immunostimulants are effective in boosting non-specific immune responses and improving overall immune system function [
30]. Numerous studies have demonstrated that incorporating immunostimulants into feeds leads to improved immunity in crustaceans [
31,
32,
33]. One prominent immunostimulant in crustaceans is β-1,3-glucans, a carbohydrate group synthesized by the cell walls of plants, bacteria, fungi, and some freshwater algae. These glucans are well-sourced and inexpensive [
34]. They can be either branched or linear polymers (polysaccharides) composed of repeating glucose units linked by β-glycosidic bonds. Remarkably, previous studies have shown that β-1,3-glucan acts as a typical PAMP interacting with PRRs present in immune cells to trigger an innate immune response [
35,
36]. As a dietary additive, β-1,3-glucan is widely used in aquatic animals to bolster immunity, enhance disease resistance, and stimulate growth [
37]. For instance, feeding juvenile tiger shrimp (
Penaeus esculentus) with diets containing β-glucan and mannan oligosaccharide improves immune response and survival when exposed to WSSV (
white Spot Syndrome Virus) infection [
38], while feeding
M. nipponense diets enriched with β-1,3-glucan not only improves its innate immunity and growth performance but also modulates the diversity of gut microbiota for maintaining gut health [
39,
40]. In another study involving
Eriocheir sinensis fed β-1,3-glucan followed by
Vibrio parahaemolyticus infestation after knocking out
LGBP at the entry point of PRRs for β-1,3-glucan resulted in increased mortality rates [
41]. Similarly, higher expression levels of LGBP were detected in the hepatopancreas of
Litopenaeus vannamei fed a diet supplemented with β-glucan [
42]. These findings highlight the prominent research interest surrounding the use of β-1,3-glucans as trace additives in aquaculture. Therefore, β-1,3-glucan represents a promising micro-additive for immune-enhancing feeds with the potential to reduce and replace the dependence on antibiotics in the aquaculture industry.
In China and other Asian countries, the oriental river prawn,
M. nipponense, is widely distributed in freshwater basins. Specifically in China, the annual production of this prawn is 240,739 tons, making it one of the most commercially viable species [
43,
44,
45]. However, intensive prawn farming in
M. nipponense has led to deteriorating water quality and environmental stress, making prawns susceptible to bacterial diseases, mass death events caused by
Aeromonas veronii [
46], and “red gill disease” from
M. nipponense [
47]. Due to multiple adverse effects associated with antibiotic usage [
48], many countries have prohibited their use in aquatic organisms intended for human consumption like fish and shrimp due to concerns over negative impacts on human health, development of antibiotic-resistant strains, reduced efficacy from repeated applications, and environmental contamination issues [
49,
50,
51]. Consequently, finding viable alternatives that are effective yet somewhat economical and environmentally safe has become an urgent requirement for restraining antibiotic usage in
M. nipponense farming practices. This study sought to evaluate the impacts of β-1,3-glucan diets on growth capability, antioxidant power, immune response, digestive well-being, and immunity against
M. nipponense bacterial infections. Practical insight into alternative treatment options for ensuring sustainability within
M. nipponense aquaculture is provided by the results obtained.
2. Materials and Methods
2.1. The Origin of Prawns and β-1,3-Glucan
The prawns selected in this experiment were healthy, with similar growth age, and no obvious scar. The prawns were all supplied by Wuyue Agricultural Co., Ltd. (Huzhou, China). The β-1,3-glucan utilized in the study was obtained from Nanjing Taixin Biotechnology Co., Ltd. (Nanjing, China) with a purity of 95.21%.
2.2. Experimental Design and Dietary Composition
The basal diet was made as an isonitrogenous and isocaloric diet using fishmeal, soybean meal, and rapeseed meal as protein sources, and fish oil and soybean oil as lipid sources. The experimental diets were prepared by supplementing the basic diet with 0% (control), 0.05%, 0.1%, 0.2%, and 0.4% β-1,3-glucan, referred to as B0, B1, B2, B3, and B4, respectively (
Table 1).
In order to create experimental diets, all ingredients need to be processed individually. The initial step involved grinding the larger particles of the ingredients. Subsequently, a more homogeneous mixture of particles and powders was obtained using a 212 μm sieve screen. Following this, it was ensured that the individually bagged powders exhibited uniform and consistent quality. The individual ingredients were accurately weighed according to the list of diet ingredients (
Table 1) using a 0.1 mg electronic scale. The finished weighed solid ingredients were added to the mixing vessel with accurately weighed semi-solid and liquid ingredients (fish oil, soybean oil, and distilled water). The mix was produced in preliminary strip form using a twin-screw extruder (School of Chemical Engineering, South China University of Technology, Guangzhou, China) and finally dried in a forced-air oven at 40 °C. The dried feeds were stored in sealed bags at −20 °C.
The determination of moisture, crude lipid, and crude protein contents in each experimental diet was conducted by using standard procedures [
52]. To determine moisture, the sample was dried at 105 °C for 24 h to a constant weight. The Dumas nitrogen determination apparatus combustion method was utilized to determine the crude protein content. The ash content was determined by setting the parameter 550 °C in a Muffle furnace for 6 h and Soxhlet extraction method was employed to measure total lipid (
Table 1).
2.3. Experimental Animals and Feeding Trial
At the outset, the tanks used for the experiment were sterilized and dried. The prawns were acclimatized for seven days in a pre-raised environment. During this period, a commercial feed with 39% protein was consumed by them. Following the adaptation period, fifteen tanks (each with a volume of 300 L) were randomly chosen to stock uniformly sized, healthy prawns with an initial average weight of 0.10 g, with each tank housing seventy prawns. The prawns were fed twice a day at 8:00 a.m. and 5:00 p.m., depending on their average weight (about 4–6% of the body weight). They were fed β-1,3-glucan diets for eight weeks. Throughout the eight-week experimental period, it was ensured that the daily water exchange was one-third of the original volume. After the water exchange was completed, water quality parameters were monitored in real-time to maintain a temperature between 25 and 28 °C, a total ammonia and nitrate content of less than 0.1 mg/L, and a dissolved oxygen content in the water of more than 6.5 mg/L, with the following parameters being maintained. The cycle of light and darkness under natural daylight conditions is fourteen hours of light and ten hours of darkness.
2.4. Analysis of Growth-Related Parameters
At the end of the eight-week feeding experiment, the entire tank stock of prawns was counted and weighed after 24 h of cessation of feeding, and the final body weights (FBW) and initial body weights (IBW) obtained were used to calculate the final growth performance involving the following equations:
2.5. Aeromonas Hydrophila Challenge
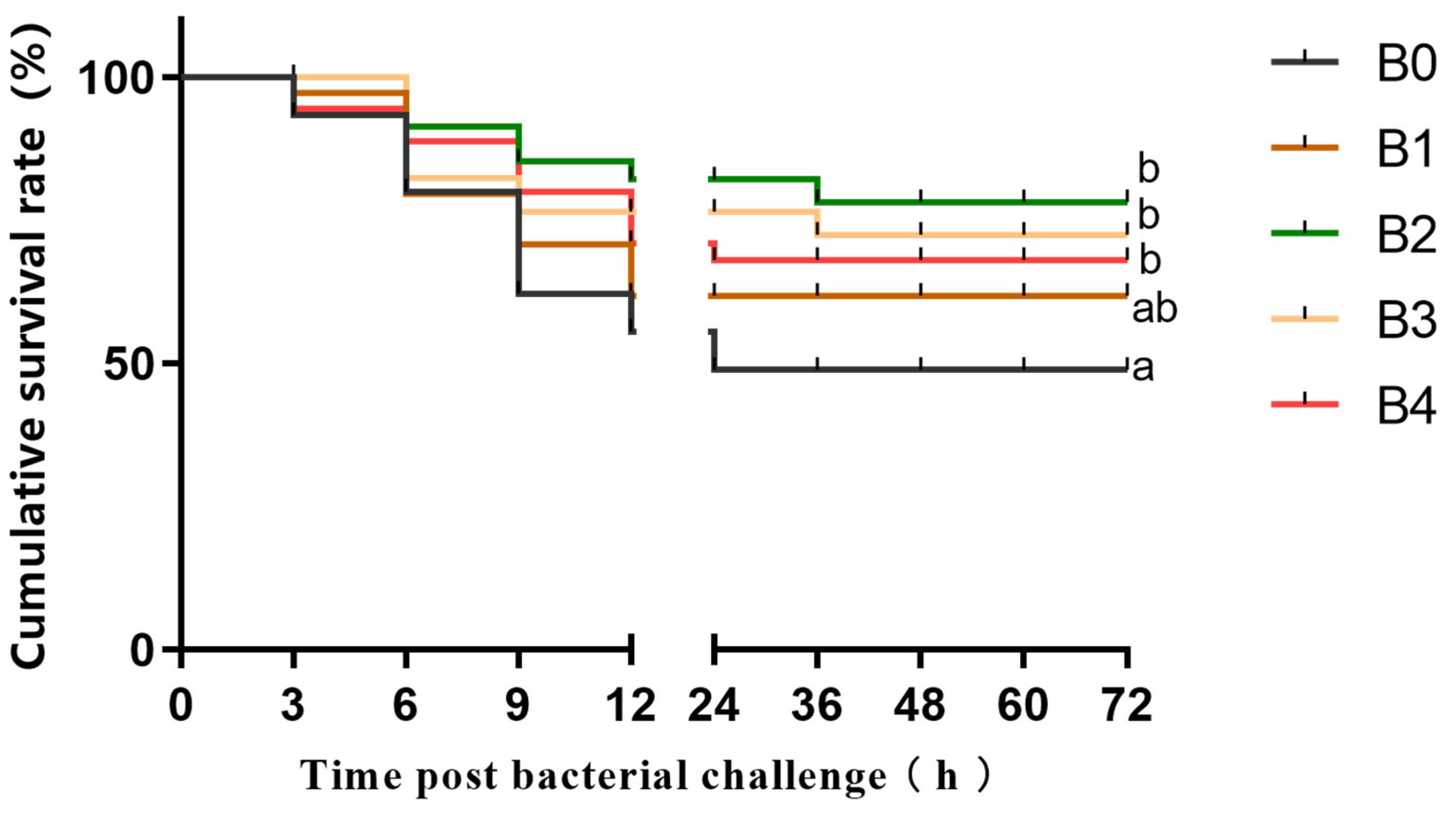
The strain of A. hydrophila used in this experiment was provided by Bei Na Biological Company. For the A. hydrophila challenge test, 12 prawns were randomly selected from each tank in the experimental group. The use of A. hydrophila solution at a concentration of 1.2 × 107 CFU/mL in a dose of 20 µL was determined based on the IFW and takedown pre-tests as a takedown reagent, and this reagent was injected into the cartilage of the basal joint of the prawns of the third maxillipeds. The cumulative survival rate of the prawns in each tank was recorded every three hours for a continuous period of three days (72 h) following injection with A. hydrophila.
2.6. Biochemical Parameters Analysis
Biochemical parameter analysis was performed by randomly sampling 10 prawns from each tank. Each prawn was anesthetized by immersion in 4 °C water. Subsequently, the hepatopancreas was dissected at the cephalothorax for sampling purposes. The ventral sinus was used to collect individual hemolymph and stored at −80 °C for future analysis. Prawn hepatopancreas samples were soaked in 0.86% saline and centrifuged at 1500× g for twenty minutes at 4 °C. The enzymatic activities of specific biochemical enzymes were analyzed using hepatopancreas samples. The superoxide dismutase (SOD) activity assay kit was purchased from Suzhou Comin Biotechnology Co., Ltd. (Suzhou, China, SOD-2-W). The kits (Nanjing Jianjian Bioengineering Institute, Nanjing, China) were used to detect acid phosphatase (ACP, A060-1-1), alkaline phosphatase (AKP, A059-1-1) activities, and malondialdehyde (MDA, A003-1-1) activities in hepatopancreas and serum. A full-wavelength microplate reader (Thermo Scientific Multiskan GO 1510, Shanghai, China) was used to measure absorptions during analysis procedures. The other part of the hepatopancreas sample was kept for the sole purpose of extracting total RNA (ribonucleic acid) for analyzing mRNA (messenger ribonucleic acid) expression.
2.7. 16S Illumina High-Throughput Sequencing Analysis Process
Intestinal flora and growth performance are often closely linked. In order to explore the effect of different levels of β-1,3-glucan (low, medium, and high) on prawns, 15 prawns from each tank in B0, B2, and B4 were selected as a representative reference for measuring intestinal flora. A TIANamp Micro DNA Purification Kit (Tiangen, Beijing, China, GDP316) was used to isolate the intestinal contents of ten prawns and extract the total bacterial community DNA. DNA samples were analyzed using a NanoDrop spectrophotometer (Thermo Fisher Scientific, Waltham, MA, USA) for their quality and quantity. In the following step, the DNA samples were mailed to Origingene Biological Medicine Technology Co. (Shanghai, China) for 16S intestinal flora analysis.
Sequence data from the V3-V4 region of the bacterial 16S rRNA gene were amplified with prokaryotic primers F341/R806. Each primer had unique 8-bp barcodes added to distinguish the products of polymerase chain reaction (PCR). A different 8bp barcode is added to each primer in order to differentiate the final product of the PCR. FastPfu Buffer, FastPfu Polymerase, primer, dNTPs, and template DNA are added and mixed proportionally in the reaction system, and amplification was accomplished under the following PCR setup program: denaturation of DNA at 95 °C for 5 min, followed by denaturation at 95 °C for 30 s, annealing at 55 °C for 30 s, extension at 72 °C for 45 s, and finally extension at 72 °C for 10 min, for a total of 27 cycles. Illumina high-throughput sequencing allows purification of PCR products, the results of which have been accepted at GenBank, and since the former experiment was submitted together with the samples from this experiment, the package can now be accessed via
https://www.ncbi.nlm.nih.gov/bioproject/954951 (accessed on 12 April 2023) and the SRA search number PRJNA954951 expanded. Quality-filtered raw reads using QIIME (version 1.17) analysis were carried out. The reads were clustered in high quality using USEARCH version 7.1, with a similarity threshold of 97% as the basis for generating OUT (Operational Units of Classification). The SILVA rRNA database was used as data support and the 16S rRNA sequences were correlated and analyzed using the RDP Classifier, which sets a confidence threshold of 70%. Finally, community diversity and species richness estimates were created in Mothur in version v.1.30.1.
2.8. mRNA Expression Analysis
The trizol method was used to extract the total RNA from the hepatopancreas samples of 5 prawns from each tank. The Thermo NanoDrop 2000 nucleic acid and protein analyzer (Thermo Fisher Scientific, Waltham, MA, USA) was used to determine the concentration and purity of RNA. Total RNA was reverse transcribed to cDNA under the instructions of the Reverse Transcription Kit (Takara, Japan) and stored in a refrigerator at −20 °C for future use. Quantitative real-time polymerase chain reaction (qRT-PCR) was used to examine the mRNA expression of immune-related genes in the hepatopancreas. The analyzed genes include lipopolysaccharide and
LGBP,
lectin,
LBP, tumor
p53, and cysteine-aspartic acid protease 3 (
caspase 3). The mixture reaction system was 20 L, including 7.6 μL H
2O, primers (10 μM) of 0.2 L each, 10 μL 2× SYBR Green Premix Ex Taq, and 2 μL cDNA. The qRT-PCR instruments were as follows: 10 min at 95 °C, followed by 40 cycles of 10 s at 94 °C, 30 s at 58 °C, and 32 s at 72 °C. The mixture reaction system was used to generate the appropriate melting curves after the qRT-PCR reaction to determine the correctness of the results. Three replicate assays were completed each time including a negative control lacking cDNA, and amplification efficiencies were guaranteed to be between 95% and 103%, with correlation coefficients greater than 0.98 for each gene. 18S ribosomal RNA (18S rRNA) was used as an internal reference gene The primer sequences of the genes used can be seen in
Table 2. Gene mRNA expression was calculated using the 2
−ΔΔCT [
53] quantification method. The cDNA sequences of the oriental river prawn from Genbank were used to design the primers and then synthesized by Sangon Biotech Co., Ltd. (Shanghai, China).
2.9. Data Analysis and Processing
The Kolmogorov–Smirnov test was used to determine data normality, while Levene’s test was used to test for variance homogeneity. After confirming the parametric assumptions, the effect of different diets that contain β-1,3-glucans on the measured parameters of M. nipponense was evaluated using an analysis of variance (ANOVA).
After the prerequisite ANOVA showed significant differences, Tukey’s multiple range test was used to compare means between groups. Third orthogonal polynomials were used to analyze the survival rate of bioinfestation experiments to correctly estimate the optimal amount of β-1,3-glucans to be added. The cubic equation in this experiment had the highest R2 value and the lowest p-value when analyzed as a mapping exercise, so polynomial regression analyses were used to determine the optimal ratio of β-1,3-glucan required for optimal survival. The Mantle–Cox test was employed to evaluate survival rates for prawns infected with A. hydrophila. Statistical analyses were performed using SPSS 25.0 (Chicago, IL, USA). The mean ± standard deviation (Mean ± SD) is the way results are expressed.
Alpha diversity indexes including Chao1 reflected community richness while Shannon and Simpson indices represented community diversity levels. Coverage analysis determined whether the identified 16S rRNA gene sequences accounted for a majority presence of bacteria in samples.
4. Discussion
In the present study, the inclusion of dietary β-1,3-glucan enhanced the weight gain and survival rate of
M. nipponense. The improved growth performance and survival rate observed in
M. nipponense may be attributed to enhanced absorption and accumulation of nutrients as well as improved innate immune response facilitated by β-1,3-glucan supplementation. Despite being underutilized in practical agriculture due to a lack of scientific strategies for its application, our study demonstrates that incorporating β-1,3-glucan into feed rations can effectively enhance growth performance and survival rates, highlighting its potential for superior production applications. Similar findings have been reported with
L. vannamei where the addition of 0.02% or 0.04% β-glucan resulted in increased weight gain [
35,
54]. Furthermore, β-glucan has also shown significant improvements in weight gain and survival rate of
Penaeus monodon [
55]. Notably, the present study revealed a trend wherein the survival rate of
M. nipponense initially increased but then decreased with increasing levels of added β-1,3-glucan. Therefore, we recommend 0.18% β-1,3-glucan in
M. nipponense diets to ensure optimal survival rate.
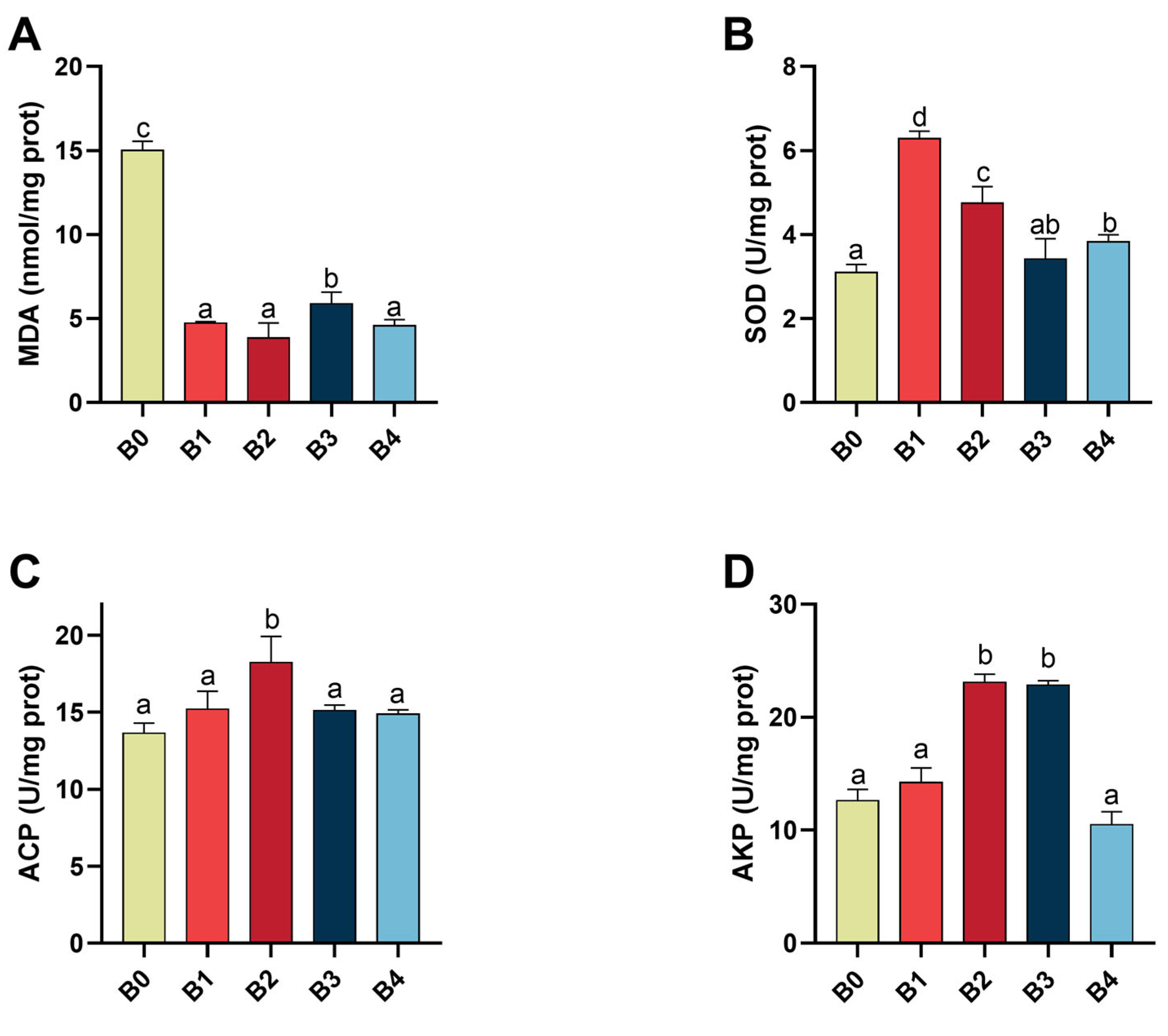
Oxygen is known to produce ROS, which are harmful to living organisms. [
56]. Lipid peroxidation is a common injury phenomenon induced by ROS, resulting from the oxidation of polyunsaturated fatty acids that are susceptible to attack by free radicals [
57]. Therefore, organisms possess enzymatic antioxidant defenses to minimize the detrimental effects of ROS [
58]. SOD plays a crucial role as an important component of the antioxidant enzyme system in biological systems and acts as a key enzyme in the elimination of ROS [
59]. MDA serves as an end product of lipid peroxidation and indirectly reflects the tissue damage caused by peroxidation [
60]. Hence, MDA content and SOD activity in the hepatopancreas can serve as an indicator for assessing the antioxidant capacity of prawns. In this study, feeding prawns with a β-1,3-glucan diet resulted in increased SOD activity but decreased MDA levels in
M. nipponense. These findings demonstrate that β-1,3-glucan enhances the antioxidant capacity of
M. nipponense. Similar results regarding enhanced antioxidant capacity after β-1,3-glucan supplementation have also been reported in swimming crabs (
Portunus trituberculatus) [
61] and
Apostichopus japonicus [
62]. Additionally, our experiment results indicate that β-1,3-glucan can increase AKP and ACP activities in the hepatopancreas of
M. nipponense. The activity level initially increases with increasing amounts of added β-1,3-glucan but eventually decreases thereafter; with 0.2% being associated with the highest observed activity level among all tested concentrations. This observation can be attributed to crustaceans possessing a large and complex innate immune system [
63], wherein AKP and ACP play vital roles as phosphatases involved in non-specific immunity responses [
64]. Previous studies have shown increased ACP and AKP activities when red swamp crayfish (
Procambarus clarkii) [
36] and Ussuri catfish (
Pseudobagrus ussuriensis) [
65] were fed diets supplemented with β-glucan. These findings demonstrate that the inclusion of appropriate β-1,3-glucan enhances both antioxidant capacity and nonspecific immunity in
M. nipponense. Notably, the improved non-specific immunity holds promise for reducing antibiotic usage during infectious disease outbreaks in prawn farming, thereby promoting sustainable development of prawn aquaculture.
Cumulative survival following bacterial infection serves as a straightforward indicator to assess disease resistance in aquatic animals and evaluate the efficacy of cultivating biological health and enhancing immunity [
12]. Importantly,
M. nipponense fed with β-1,3-glucan diets exhibited a distinct increase in cumulative survival after
A. hydrophila infection; moreover, cumulative survival displayed an increasing trend followed by a decrease upon the addition of β-1,3-glucan. These observations suggest that appropriate amounts of addition of β-1,3-glucan can enhance the disease resistance of
M. nipponense. Similarly,
Micropterus salmoides showed higher survival after
Aeromonas schubertii infection when fed diets supplemented with β- glucan [
66], while juvenile tiger shrimp demonstrated increased survivability after WSSV infection when fed diets enriched with β-glucan [
67]. Collectively, these results indicated that to some extent β-1,3-glucan has the potential to mitigate aquatic disease outbreaks caused by pathogenic microorganisms. The fundamental role of the immune system is to distinguish between “self” and “non-self”. Crustaceans, known for their innate immunity, heavily rely on the recognition of “non-self” [
25]. Recent findings indicate that this recognition is facilitated by certain PRRs, which can be soluble or membrane-bound [
68]. A critical step in initiating the innate immune response involves the mutual recognition and interaction between PRRs and PAMPs present on the surface of pathogens and absent in the host [
69]. β-1,3-glucan found on fungal cell walls represents an important class of PAMPs [
70], capable of binding to common crustaceans PRRs such as
LBP [
11],
bGBP [
14],
LGBP [
12], and
lectin [
71]; this interaction subsequently triggers proPO (Prophenoloxidase), a widely distributed enzyme in blood and inner tissues of carapace, leading to activation of proPO-AS [
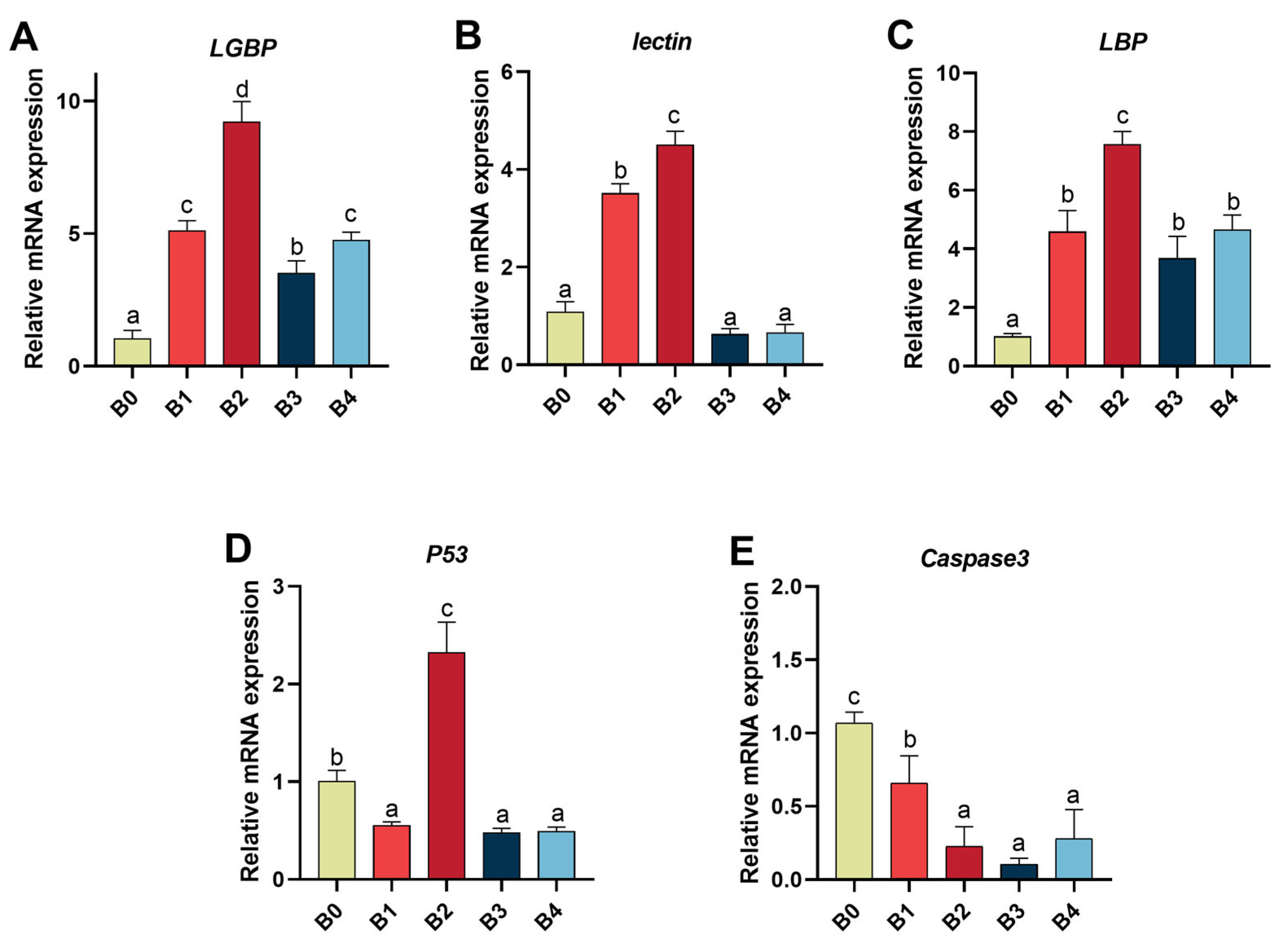
41]. Therefore, quantitatively analyzing mRNA expression levels of
LBP,
LGBP and
lectin in prawn hepatopancreas can serve as an effective tool for assessing prawn immune response status. Our results further demonstrate that feeding the prawns with β-1,3-glucan upregulates mRNA expression levels of
LBP,
LGBP, and
lectin genes. Interestingly, numerous studies have investigated the relationship between β-glucan and aquatic animals, with PO activity often being associated with certain PRRs-related genes. Therefore, we hypothesized that upregulating the expression of
LBP,
LGBP, and
lectin genes could enhance the immunity of
M. nipponense by increasing PO activity. For instance,
L. vannamei fed a diet supplemented with β-glucan exhibited simultaneous enhancements in both PO activity and
LGBP gene expression [
12], while
Macrobrachium rosenbergii fed a similar diet showed concurrent increases in both PO activity and
lectin gene expression [
72]. Furthermore, our study found that the β-1,3-glucan group had a significantly higher cumulative survival rate 72 h after
A. hydrophila infection compared to the control group. Hence, we postulated that
M. nipponense could activate proPO-AS by enhancing the expression of
LBP,
LGBP, and
lectin genes through β-1,3-glucan supplementation to resist
A. hydrophila infestation. Previous research has also demonstrated significant induction of crustacean
LGBP transcripts following challenges with bacteria such as
Vibrio harveyi [
73],
Vibrio alginolyticus [
74], and
Vibrio anguillarum [
75]. According to the results of the present study, the addition of moderate amounts of β-1,3-glucan to the diet improved the non-specific immunity of
M. nipponense.
Caspases are closely associated with eukaryotic apoptosis and play a role in regulating cell growth, differentiation, and apoptosis [
76]. Apoptosis is considered to be a form of programmed cell death that primarily functions to eliminate damaged, dangerous, and non-functional cells in order to maintain organismal health [
77,
78]. The caspase protease family consists of numerous members, among which downstream
caspase3 acts as the executors of apoptosis by enzymatically cleaving specific proteins [
79]. In this study, the gene expression of
M. nipponense caspsae3 was significantly reduced in the group fed β-1,3-glucan compared to the control group. Therefore, it can be inferred that the addition of β-1,3-glucan to feed can attenuate apoptosis.
P53 is a protein involved in inhibiting cell cycle progression in DNA-damaged cells [
80], which is regulated by activating cellular repair mechanisms for DNA damage repair or inducing apoptosis if repair is not feasible. It plays a pivotal role in gene expression related to cell cycle, genetic stability maintenance, and apoptosis [
81]. In our experiment, the inclusion of 0.05%, 0.2%, and 0.4% β-1,3-glucan significantly downregulated
P53 gene expression in prawns consistently with
caspase3 trend. We speculate that the addition of β-1,3-glucan to the diet could attenuate apoptosis in
M. nipponense by enhancing the nonspecific immunity characteristic of crustaceans, which is consistent with the findings shown above for ACP and AKP. Furthermore, a correlation between the enhancement of specific immunity and the reduction in apoptosis, as indicated by ACP and AKP levels, has been observed in several other studies [
82,
83]. Interestingly, the inclusion of 0.1% β-1,3-glucan significantly up-regulated
P53 gene expression possibly due to its upstream position relative to
caspase3, as well as its involvement in antioxidant defense regulation, DNA repair processes, and apoptotic pathways [
84].
The intestine is an important organ for the digestion and absorption of nutrients in shrimp [
85,
86]. Gut microorganisms are essential for nutritional digestion and absorption, as well as immune function and disease resistance [
87]. Feeding probiotics to
Oreochromis niloticus has been shown to enhance intestinal villi development, growth performance, and mucosal immunity [
88]. Similarly, improving gut flora has been found to enhance immune responses and disease resistance in various finfish species [
89]. Identifying up- and down-regulated microorganisms is an important step in understanding how β-glucan affects the gut microbiota and how this change in the microbiota affects the host by altering nutrients, the immune system, and compounds absorbed through the epithelial barrier [
90]. In this study, the addition of β-1,3-glucan did not affect the gut flora abundance at phylum level, we speculate that the addition of β-1,3-glucan at the phylum level maintains the stability and diversity of the intestinal flora of
M. nipponense. At the genus level, this experiment demonstrated that the addition of β-1,3-glucan significantly increased the abundance of
Rhodobacteraceae_unclassified in the intestinal tract of
M. nipponense. Consistent with previous studies on β-glucan, it has been consistently observed that supplementation with β-glucan enhances the abundance of
Rhodobacteraceae, which may represent a key characteristic associated with competitive advantage induced by β-glucan [
91,
92]. Moreover, a prior investigation on
N. californicus revealed that
Rhodobacteraceae can serve as a probiotic in the diet of
N. californicus and improve resistance to acute low-salinity challenge in shrimp
Penaeus vannamei through higher survival rates and elevated levels of T-AOC activity as well as SOD, HSP70, and Relish gene transcripts [
93]. Therefore, dietary supplementation with β-1,3-glucan could enhance
Rhodobacteraceae abundance for gut health improvement in
M. nipponense. However, further exploration is required to fully understand this potential mutualistic relationship between
M. nipponense and specific taxa within its intestinal flora.