Correlations Between Achilles Tendon Stiffness and Jumping Performance: A Comparative Study of Soccer and Basketball Athletes
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Protocol
2.3. Data Processing and Statistical Analyses
3. Results
3.1. Intra-Class Correlations (ICCs)
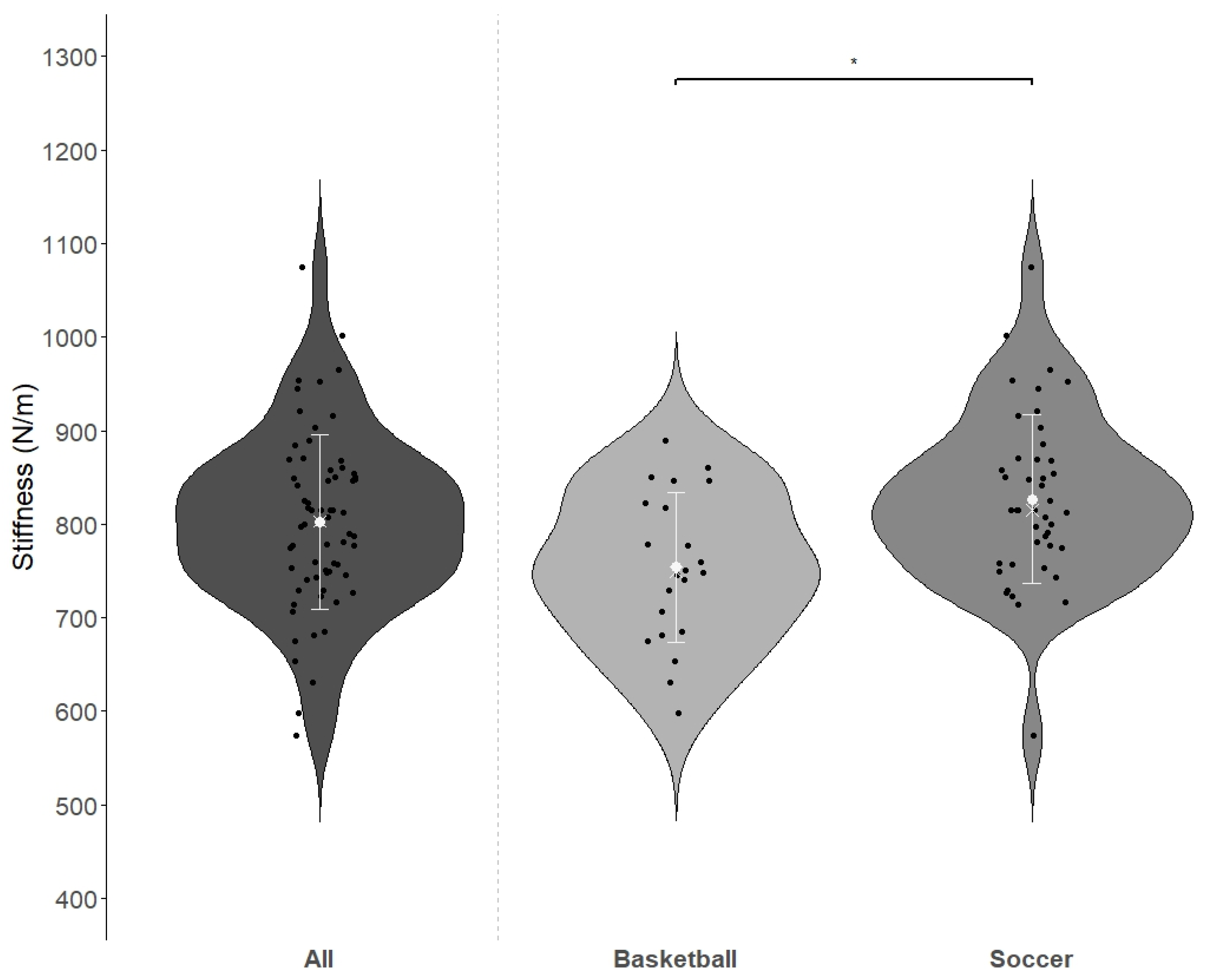
3.2. Analyses of Differences Between and Within the Two Sport Types
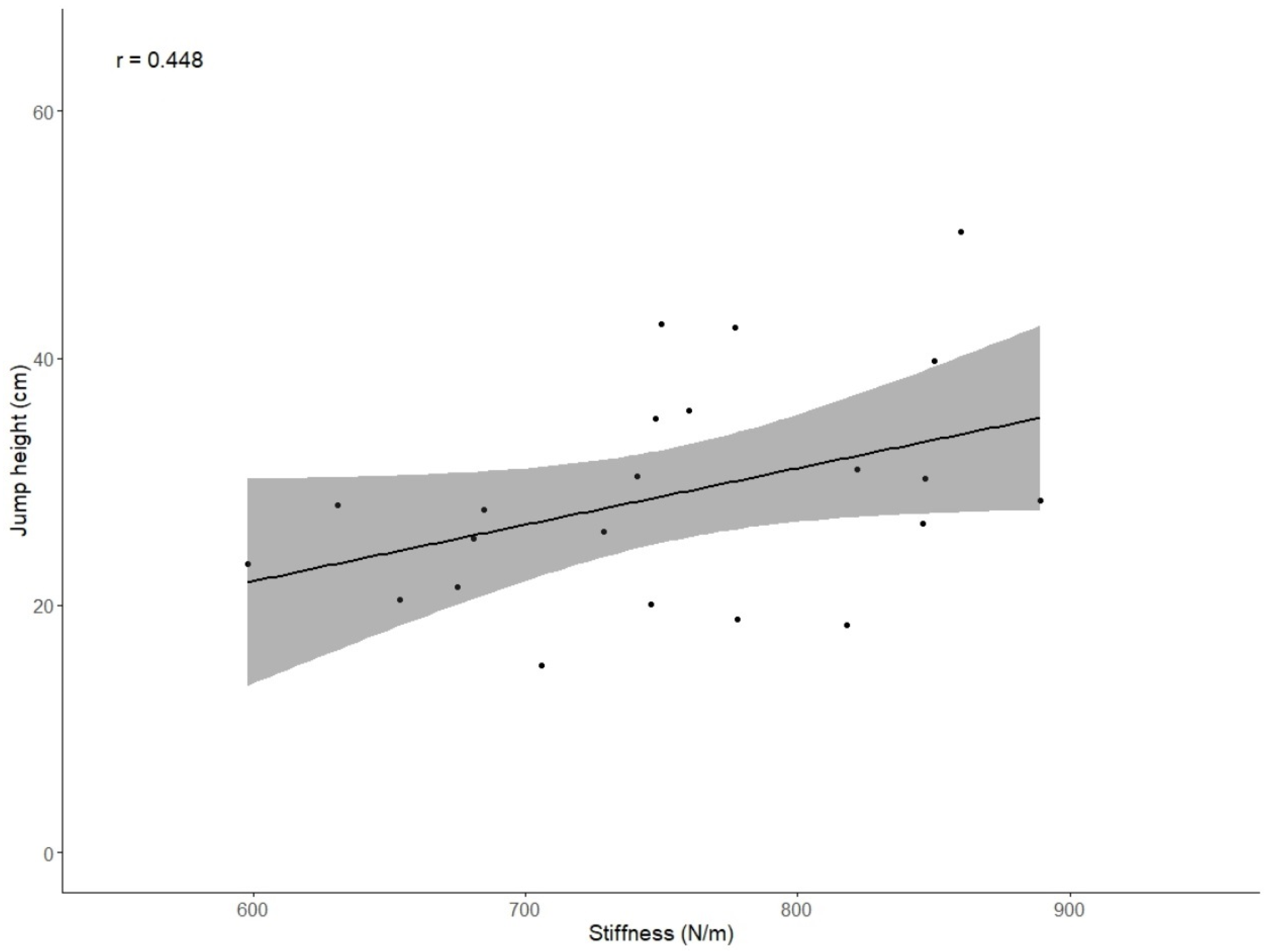
3.3. Analyses of Correlations
3.4. Multiple Linear Regression Analyses
4. Discussion
4.1. Introductory Considerations Regarding AT Stiffness
4.2. ICCs
4.3. Side Differences
4.4. Differences Between Soccer and Basketball Players
4.5. Correlation Analyses
4.6. Limitations
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Dvorak, J.; Junge, A.; Graf-Baumann, T.; Peterson, L. Football Is the Most Popular Sport Worldwide. Am. J. Sports Med. 2004, 32, 3S–4S. [Google Scholar] [CrossRef] [PubMed]
- Khaitovich, K.F. The Most Popular Sports in the World. Br. J. Glob. Ecol. Sustain. Dev. 2023, 17, 92–95. [Google Scholar]
- Gervasi, M.; Benelli, P.; Venerandi, R.; Fernández-Peña, E. Relationship between Muscle-Tendon Stiffness and Drop Jump Performance in Young Male Basketball Players during Developmental Stages. Int. J. Environ. Res. Public Health 2022, 19, 17017. [Google Scholar] [CrossRef]
- Szymski, D.; Achenbach, L.; Krutsch, V.; Alt, V.; Meffert, R.; Krutsch, W.; Fehske, K. Injury Epidemiology in Men’s Professional Team Sports: Is Media Analysis Helpful? Arch. Orthop. Trauma Surg. 2021, 141, 655–661. [Google Scholar] [CrossRef]
- Stovitz, S.D.; Shrier, I. Injury Rates in Team Sport Events: Tackling Challenges in Assessing Exposure Time. Br. J. Sports Med. 2012, 46, 960–963. [Google Scholar] [CrossRef]
- Florit, D.; Pedret, C.; Casals, M.; Malliaras, P.; Sugimoto, D.; Rodas, G. Incidence of Tendinopathy in Team Sports in a Multidisciplinary Sports Club over 8 Seasons. J. Sports Sci. Med. 2019, 18, 780–788. [Google Scholar]
- Hoenig, T.; Gronwald, T.; Hollander, K.; Klein, C.; Frosch, K.H.; Ueblacker, P.; Rolvien, T. Video Analysis of Achilles Tendon Ruptures in Professional Male Football (Soccer) Reveals Underlying Injury Patterns and Provides Strategies for Injury Prevention. Knee Surg. Sports Traumatol. Arthrosc. 2023, 31, 2236–2245. [Google Scholar] [CrossRef]
- Hoffman, J.R.; Fry, A.; Howard, R.; Maresh, C.M.; Kraemer, W.J. Strength, Speed and Endurance Changes During the Course of a Division I Basketball Season. J. Strength Cond. Res. 1991, 5, 144–149. [Google Scholar]
- Hoffman, J.R.; Tenenbaum, G.; Maresh, C.M.; Kraemer, W.J. Relationship Between Athletic Performance Tests and Playing Time in Elite College Basketball Players. J. Strength Cond. Res. 1996, 10, 67–71. [Google Scholar]
- Khlifa, R.; Aouadi, R.; Hermassi, S.; Chelly, M.S.; Jlid, M.C.; Hbacha, H.; Castagna, C. Effects of a Plyometric Training Program with and without Added Load on Jumping Ability in Basketball Players. J. Strength Cond. Res. 2010, 24, 2955–2961. [Google Scholar]
- Makaraci, Y.; Soslu, R. Relationship Between Jumping Performance in Various Tasks, Sprint and Agility in Basketball Players. Gazi J. Phys. Educ. Sport. Sci. 2022, 27, 357–374. [Google Scholar] [CrossRef]
- Burkett, L.N.; Phillips, W.T.; Ziuraitis, J. The Best Warm-up for the Vertical Jump in College-Age Athletic Men. J. Strength Cond. Res. 2005, 19, 673–676. [Google Scholar] [PubMed]
- Manouras, N.; Papanikolaou, Z.; Karatrantou, K.; Kouvarakis, P.; Gerodimos, V. The Efficacy of Vertical vs. Horizontal: Plyometric Training on Speed, Jumping Performance and Agility in Soccer Players. Int. J. Sports Sci. Coach. 2016, 11, 702–709. [Google Scholar] [CrossRef]
- McFarland, I.T.; Dawes, J.J.; Elder, C.L.; Lockie, R.G. Relationship of Two Vertical Jumping Tests to Sprint and Change of Direction Speed among Male and Female Collegiate Soccer Players. Sports 2016, 4, 11. [Google Scholar] [CrossRef] [PubMed]
- Ferrauti, A.; Fett, J.; Frytz, A.; Götz, J.K.; Hanakam, F.; Hottenrott, L.; Kappenstein, J.; Kittel, T.; Krombholz, A.; Möllmann, J.; et al. Trainingswissenschaft für die Sportpraxis—Lehrbuch für Studium, Ausbildung und Unterricht im Sport; Ferrauti, A., Ed.; Springer Spektrum: Berlin, Germany, 2020; ISBN 978-3-662-58227-5. [Google Scholar]
- Gorostiaga, E.M.; Llodio, I.; Ibáñez, J.; Granados, C.; Navarro, I.; Ruesta, M.; Bonnabu, H.; Izquierdo, M. Differences in Physical Fitness among Indoor and Outdoor Elite Male Soccer Players. Eur. J. Appl. Physiol. 2009, 106, 483–491. [Google Scholar] [CrossRef]
- Kongsgaard, M.; Reitelseder, S.; Pedersen, T.G.; Holm, L.; Aagaard, P.; Kjaer, M.; Magnusson, S.P. Region Specific Patellar Tendon Hypertrophy in Humans Following Resistance Training. Acta Physiol. 2007, 191, 111–121. [Google Scholar] [CrossRef]
- Heinemeier, K.M.; Kjaer, M. In Vivo Investigation of Tendon Responses to Mechanical Loading. J. Musculoskelet. Neuronal Interact. 2011, 11, 115–123. [Google Scholar]
- Fouré, A.; Nordez, A.; Cornu, C. Effects of Eccentric Training on Mechanical Properties of the Plantar Flexor Muscle-Tendon Complex. J. Appl. Physiol. 2013, 114, 523–537. [Google Scholar] [CrossRef]
- Park, D.Y.; Chou, L. Stretching for Prevention of Achilles Tendon Injuries: A Review of the Literature. Foot Ankle Int. 2006, 27, 1086–1095. [Google Scholar] [CrossRef]
- Orner, S.; Kratzer, W.; Schmidberger, J.; Grüner, B. Quantitative Tissue Parameters of Achilles Tendon and Plantar Fascia in Healthy Subjects Using a Handheld Myotonometer. J. Bodyw. Mov. Ther. 2018, 22, 105–111. [Google Scholar] [CrossRef]
- Kawakami, Y.; Muraoka, T.; Ito, S.; Kanehisa, H.; Fukunaga, T. In Vivo Muscle Fibre Behaviour during Counter-Movement Exercise in Humans Reveals a Significant Role for Tendon Elasticity. J. Physiol. 2002, 540, 635–646. [Google Scholar] [CrossRef] [PubMed]
- Lichtwark, G.A.; Wilson, A.M. In Vivo Mechanical Properties of the Human Achilles Tendon during One-Legged Hopping. J. Exp. Biol. 2005, 208, 4715–4725. [Google Scholar] [CrossRef] [PubMed]
- Burgess, K.E.; Connick, M.J.; Graham-Smith, P.; Pearson, S.J. Plyometric vs. Isometric Training Influences on Tendon Properties and Muscle Output. J. Strength Cond. Res. 2007, 21, 986–989. [Google Scholar] [CrossRef]
- Arampatzis, A.; Karamanidis, K.; Morey-Klapsing, G.; De Monte, G.; Stafilidis, S. Mechanical Properties of the Triceps Surae Tendon and Aponeurosis in Relation to Intensity of Sport Activity. J. Biomech. 2007, 40, 1946–1952. [Google Scholar] [CrossRef]
- Bohm, S.; Mersmann, F.; Arampatzis, A. Human Tendon Adaptation in Response to Mechanical Loading: A Systematic Review and Meta-Analysis of Exercise Intervention Studies on Healthy Adults. Sports Med.-Open 2015, 1, 7. [Google Scholar] [CrossRef]
- Couppé, C.; Suetta, C.; Kongsgaard, M.; Justesen, L.; Hvid, L.G.; Aagaard, P.; Kjær, M.; Magnusson, S.P. The Effects of Immobilization on the Mechanical Properties of the Patellar Tendon in Younger and Older Men. Clin. Biomech. 2012, 27, 949–954. [Google Scholar] [CrossRef]
- Geremia, J.M.; Baroni, B.M.; Bobbert, M.F.; Bini, R.R.; Lanferdini, F.J.; Vaz, M.A. Effects of High Loading by Eccentric Triceps Surae Training on Achilles Tendon Properties in Humans. Eur. J. Appl. Physiol. 2018, 118, 1725–1736. [Google Scholar] [CrossRef]
- Feng, Y.N.; Li, Y.P.; Liu, C.L.; Zhang, Z.J. Assessing the Elastic Properties of Skeletal Muscle and Tendon Using Shearwave Ultrasound Elastography and MyotonPRO. Sci. Rep. 2018, 8, 17064. [Google Scholar] [CrossRef]
- Schneebeli, A.; Falla, D.; Clijsen, R.; Barbero, M. Myotonometry for the Evaluation of Achilles Tendon Mechanical Properties: A Reliability and Construct Validity Study. BMJ Open Sport Exerc. Med. 2020, 6, e000726. [Google Scholar] [CrossRef]
- Waugh, C.M.; Korff, T.; Fath, F.; Blazevich, A.J. Effects of Resistance Training on Tendon Mechanical Properties and Rapid Force Production in Prepubertal Children. J. Appl. Physiol. 2014, 117, 257–266. [Google Scholar] [CrossRef]
- Bojsen-Møller, J.; Magnusson, S.P.; Rasmussen, L.R.; Kjaer, M.; Aagaard, P. Muscle Performance during Maximal Isometric and Dynamic Contractions Is Influenced by the Stiffness of the Tendinous Structures. J. Appl. Physiol. 2005, 99, 986–994. [Google Scholar] [CrossRef] [PubMed]
- Seon, S.Y.; Park, B.J.; Lee, K.J.; An, K.O. The Effect of Stiffness of Lower Extremity Soft Tissue on the Athletic Performance of K-7 League Soccer Players: A Pilot Study. Exerc. Sci. 2022, 31, 452–458. [Google Scholar] [CrossRef]
- Pożarowszczyk, B.; Gołaś, A.; Chen, A.; Zając, A.; Kawczyński, A. The Impact of Post Activation Potentiation on Achilles Tendon Stiffness, Elasticity and Thickness among Basketball Players. Sports 2018, 6, 117. [Google Scholar] [CrossRef]
- Sakalauskaite, R.; Satkunskiene, D. The Foot Arch and Viscoelastic Properties of Plantar Fascia and Achilles Tendon. J. Vibroengineering 2012, 14, 1751–1759. [Google Scholar]
- Abdelsattar, M.; Konrad, A.; Tilp, M. Relationship between Achilles Tendon Stiffness and Ground Contact Time during Drop Jumps. J. Sports Sci. Med. 2018, 17, 223–228. [Google Scholar]
- Konrad, A.; Paternoster, F.K. No Association between Jump Parameters and Tissue Stiffness in the Quadriceps and Triceps Surae Muscles in Recreationally Active Young Adult Males. Appl. Sci. 2022, 12, 1596. [Google Scholar] [CrossRef]
- Dittrich, H.; Schimmack, M.; Siemsen, C.-H. (Eds.) Orthopädische Biomechanik: Einführung in die Endoprothetik der Gelenke der Unteren Extremitäten; Springer: Berlin/Heidelberg, Germany, 2019. [Google Scholar]
- Pożarowszczyk, B.; Pawlaczyk, W.; Smoter, M.; Zarzycki, A.; Mroczek, D.; Kumorek, M.; Witkowski, K.; Adam, K. Effects of Karate Fights on Achilles Tendon Stiffness Measured by Myotonometry. J. Hum. Kinet. 2017, 56, 93–97. [Google Scholar] [CrossRef]
- Nitzsche, N.; Siebert, T.; Schulz, H.; Stutzig, N. Effect of Plyometric Training on Dynamic Leg Strength and Jumping Performance in Rhythmic Gymnastics: A Preliminary Study. Isokinet. Exerc. Sci. 2022, 30, 79–87. [Google Scholar] [CrossRef]
- Weir, J. Quantifying Test-Retest Reliability Using the Intraclass Correlation Coefficient and the SEM. J. Strength Cond. Res. 2005, 19, 231–240. [Google Scholar]
- Cook, R.D.; Weisberg, S. Criticism and Influence Analysis in Regression. Sociol. Methodol. 1982, 13, 313–361. [Google Scholar]
- Cristi-Sánchez, I.; Danes-Daetz, C.; Neira, A.; Ferrada, W.; Yáñez Díaz, R.; Silvestre Aguirre, R. Patellar and Achilles Tendon Stiffness in Elite Soccer Players Assessed Using Myotonometric Measurements. Sports Health 2019, 11, 157–162. [Google Scholar] [CrossRef] [PubMed]
- Atkinson, G.; Nevill, A. Statistical Methods for Assessing Measurement Error (Reliability) in Variables Relevant to Sports Medicine. Sports Med. 1998, 26, 217–238. [Google Scholar]
- Vincent, J. Statistics in Kinesiology; Human Kinetics Books: Champaign, IL, USA, 1994. [Google Scholar]
- Bédard, M.; Martin, N.J.; Krueger, P.; Brazil, K. Assessing Reproducibility of Data Obtained with Instruments Based on Continuous Measurements. Exp. Aging Res. 2000, 26, 353–365. [Google Scholar] [CrossRef] [PubMed]
- Wiesinger, H.P.; Kösters, A.; Müller, E.; Seynnes, O.R. Effects of Increased Loading on In Vivo Tendon Properties: A Systematic Review. Med. Sci. Sports Exerc. 2015, 47, 1885–1895. [Google Scholar] [CrossRef]
- Ando, R.; Sato, S.; Hirata, N.; Tanimoto, H.; Imaizumi, N.; Suzuki, Y.; Hirata, K.; Akagi, R. Relationship between Resting Medial Gastrocnemius Stiffness and Drop Jump Performance. J. Electromyogr. Kinesiol. 2021, 58, 102549. [Google Scholar] [CrossRef]
- Cruz-Montecinos, C.; Besomi, M.; Acevedo-Valenzuela, N.; Cares-Marambio, K.; Bustamante, A.; Guzmán-González, B.; Tapia-Malebrán, C.; Sanzana-Cuche, R.; Calatayud, J.; Méndez-Rebolledo, G. Soleus Muscle and Achilles Tendon Compressive Stiffness Is Related to Knee and Ankle Positioning. J. Electromyogr. Kinesiol. 2022, 66, 102698. [Google Scholar] [CrossRef]
- Hess, G.W. Achilles Tendon Rupture—A Review of Etiology, Population, Anatomy, Risk Factors, and Injury Prevention. Foot Ankle Spec. 2010, 3, 29–32. [Google Scholar] [CrossRef]
- Pruyn, E.C.; Watsford, M.L.; Murphy, A.J. Validity and Reliability of Three Methods of Stiffness Assessment. J. Sports Health Sci. 2016, 5, 476–483. [Google Scholar] [CrossRef]
- Kurashina, W.; Takahashi, T.; Sasanuma, H.; Saitsu, A.; Takeshita, K. Relationship Between Achilles Tendon Stiffness Using Myoton PRO and Translation Using a Tensile Testing Machine: A Biomechanical Study of a Porcine Model. Cureus 2023, 15, e49359. [Google Scholar] [CrossRef]
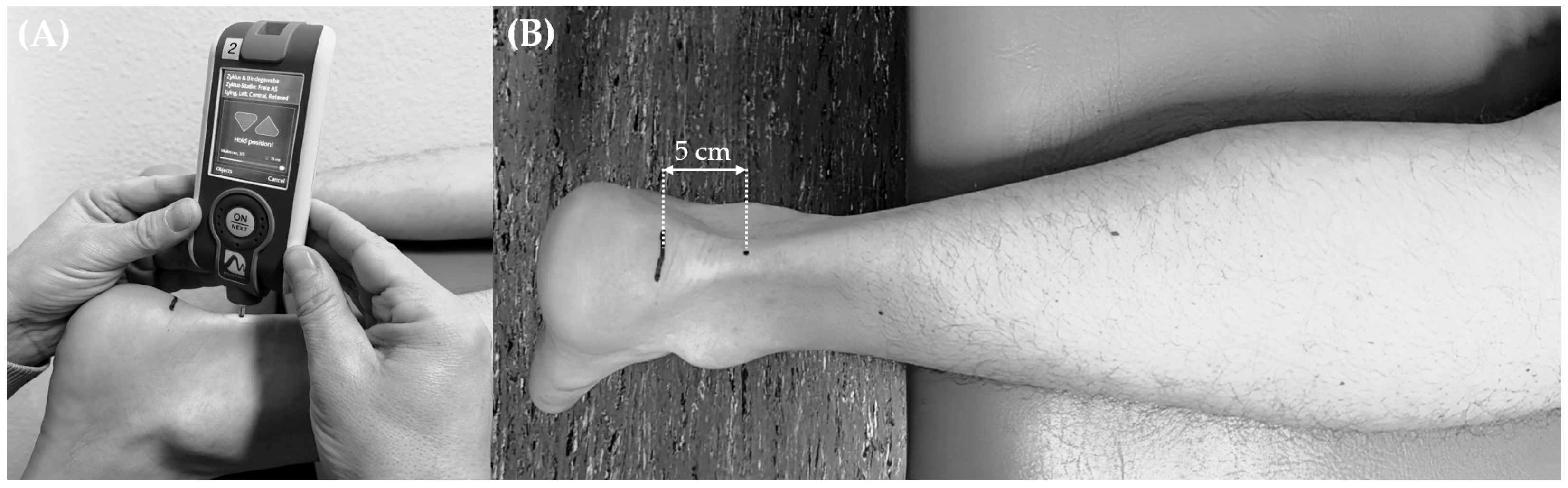

| Age (years) | Height (m) | Mass (kg) | BMI (kg/m2) | |
|---|---|---|---|---|
| Basketball players (n = 22) | 22.0 ± 4.1 * | 1.91 ± 0.1 * | 84.7 ± 13.7 * | 23.1 ± 1.9 |
| Soccer players (n = 44) | 26.3 ± 4.4 * | 1.79 ± 0.1 * | 75.5 ± 7.5 * | 23.5 ± 1.8 |
| p-value | * ˂0.001 | * ˂0.001 | * 0.007 | 0.417 |
| Effect size | δ 0.512 | δ −0.649 | D −0.925 | D 0.219 |
| All (n = 66) | 24.9 ± 4.7 | 1.83 ± 0.1 | 78.6 ± 10.8 | 23.4 ± 1.9 |
| JH (cm) | GCT (ms) | RSI | Stiffness (N/m) | |
|---|---|---|---|---|
| Basketball players (n = 22) | 29.0 ± 9.0 | 211.9 ± 41.8 | 1.4 ± 0.5 | 754 ± 80 * |
| Soccer players (n = 44) | 32.4 ± 5.7 | 235.0 ± 46.5 | 1.4 ± 0.4 | 827 ± 91 * |
| p-value | 0.114 | 0.074 | 0.935 | * 0.002 |
| Effect size | D 0.494 | δ 0.273 | D 0.023 | D 0.833 |
| All (n = 66) | 31.3 ± 7.1 | 227.3 ± 46.0 | 1.4 ± 0.4 | 803 ± 93 |
| Spearman r (p-Value) | ||
|---|---|---|
| Comparison | Soccer | Basketball |
| Stiffness vs. Jump height | −0.147 (0.341) | 0.448 (0.037) * |
| Stiffness vs. GCT | 0.060 (0.699) | 0.319 (0.148) |
| Stiffness vs. RSI | −0.104 (0.500) | 0.148 (0.511) |
| Jump height vs. GCT | −0.321 (0.003) * | −0.133 (0.554) |
| Jump height vs. RSI | 0.768 (0.500) | 0.838 (<0.001) * |
| GCT vs. RSI | −0.816 (<0.001) * | −0.624 (0.002) * |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Schmidt, D.; Verderber, L.; Germano, A.M.C.; Nitzsche, N. Correlations Between Achilles Tendon Stiffness and Jumping Performance: A Comparative Study of Soccer and Basketball Athletes. J. Funct. Morphol. Kinesiol. 2025, 10, 112. https://doi.org/10.3390/jfmk10020112
Schmidt D, Verderber L, Germano AMC, Nitzsche N. Correlations Between Achilles Tendon Stiffness and Jumping Performance: A Comparative Study of Soccer and Basketball Athletes. Journal of Functional Morphology and Kinesiology. 2025; 10(2):112. https://doi.org/10.3390/jfmk10020112
Chicago/Turabian StyleSchmidt, Daniel, Lukas Verderber, Andresa M. C. Germano, and Nico Nitzsche. 2025. "Correlations Between Achilles Tendon Stiffness and Jumping Performance: A Comparative Study of Soccer and Basketball Athletes" Journal of Functional Morphology and Kinesiology 10, no. 2: 112. https://doi.org/10.3390/jfmk10020112
APA StyleSchmidt, D., Verderber, L., Germano, A. M. C., & Nitzsche, N. (2025). Correlations Between Achilles Tendon Stiffness and Jumping Performance: A Comparative Study of Soccer and Basketball Athletes. Journal of Functional Morphology and Kinesiology, 10(2), 112. https://doi.org/10.3390/jfmk10020112






