Enhanced Cell Inactivation and Double-Strand Break Induction in V79 Chinese Hamster Cells by Monochromatic X-Rays at Phosphorus K-Shell Absorption Peak
Abstract
:1. Introduction
2. Materials and Methods
2.1. Cell Line and Culture Media
2.2. Irradiation Setup
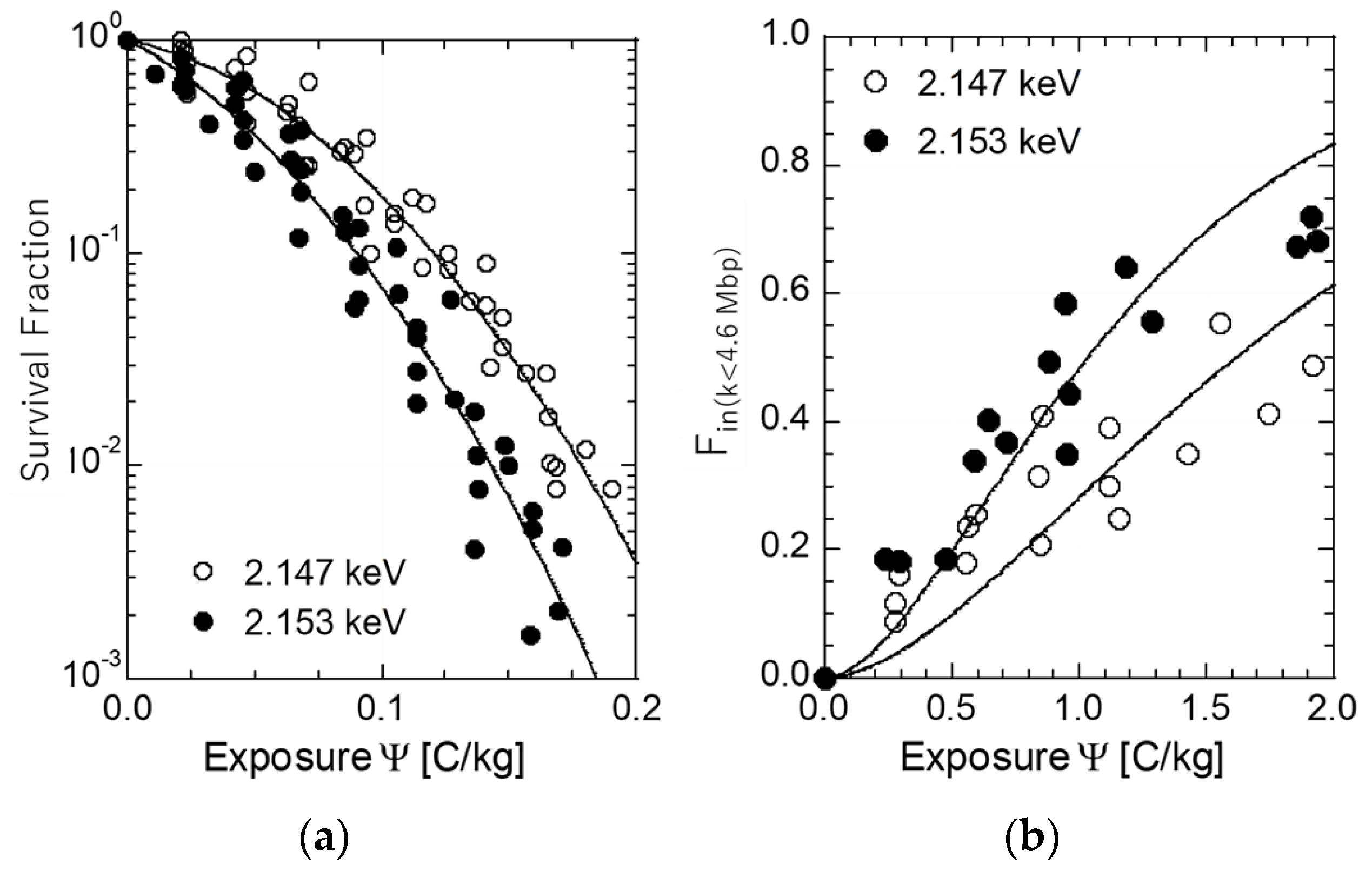
2.3. Colony Formation Assay for Survival Curves
2.4. Pulse Field Gel Electrophoresis (PFGE) and DSB Measurements
3. Results
3.1. Cell Inactivation on and Below the Phosphorous K-Shell Peak
3.2. Induction of DSBs
3.3. Cross-Section of DSB Induction and Quantum Yield
4. Discussion
4.1. Evaluation of Enhanced Cell Inactivation on Phosphorus K-Shell Peak from Linear-Quadratic Model
4.2. Enhance Induction of DNA DSBs
4.3. Conversion of Exposure ψ (C/kg) to Absorbed Dose D (Gy): Mass Energy-Absorption Coefficient μen/ρ
4.4. Cell Inactivation and DSB Induction per Absorbed Dose in Gy
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Attix, F.H. Introduction to Radiological Physics and Radiation Dosimetry; Wiley-VCH Verlag GmbH & Co, KGaA: Weinheim, Germany, 1986. [Google Scholar] [CrossRef]
- Humm, J.; Pomplun, E.; Booz, J.; Charlton, D.E. Energy and Number Distributions of Electrons and Photons Emitted After Photoelectric Interactions of X-rays With Phosphorus in DNA. In Radiation Protection: Eighth Symposium on Microdosimetry; Booz, J., Ebert, H., Eds.; Commission of the European Communities: Jülich, Germany, 1983; pp. 187–198. [Google Scholar]
- Watanabe, R.; Yokoya, A.; Fujii, K.; Saito, K. DNA strand breaks by direct energy deposition by Auger and photo-electrons ejected from DNA constituent atoms following K-shell photoabsorption. Int. J. Radiat. Biol. 2004, 80, 823–832. [Google Scholar] [CrossRef] [PubMed]
- Hieda, K.; Hirono, T.; Azami, A.; Suzuki, M.; Furusawa, Y.; Maezawa, H.; Usami, N.; Yokoya, A.; Kobayashi, K. Single- and double-strand breaks in pBR322 plasmid DNA by monochromatic X-rays on and off the K-absorption peak of phosphorus. Int. J. Radiat. Biol. 1996, 70, 437–445. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, K.; Hieda, K.; Maezawa, H.; Ando, M.; Ito, T. Monochromatic X-ray irradiation system (0.08–0.4 nm) for radiation biology studies using synchrotron radiation at the photon factory. J. Radiat. Res. 1987, 28, 243–253. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hieda, K.; Ito, T. Radiobiological Experiments in the X-ray Region with Synchrotron Radiation Ch 13. In Handbook on Synchrotron Radiation; Ebashi, S., Koch, M., Rubenstein, E., Eds.; North-Holland: Amsterdam, The Netherlands, 1991; Volume 4, pp. 431–465. [Google Scholar]
- Konishi, H.; Yokoya, A.; Shiwaku, H.; Motohashi, H.; Makita, T.; Kashihara, Y.; Hashimoto, S.; Harami, T.; Sasaki, T.A.; Maeta, H.; et al. Synchrotron radiation beamline to study radioactive materials at the Photon factory. Nucl. Instrum. Methods Phys. Res. Sect. A Accel. Spectrometers Detect. Assoc. Equip. 1996, 372, 322–332. [Google Scholar] [CrossRef]
- Kobayashi, K. Photon-Induced Biological Consequences. In Charged Particle and Photon Interactions with Matter: Chemical, Physicochemical, and Biological Consequences with Applications; Mozumder, A., Hatano, Y., Eds.; Taylar & Francis Group: London, UK, 2003; pp. 471–490. [Google Scholar] [CrossRef]
- Maezawa, H.; Furusawa, Y.; Kobayashi, K.; Hieda, K.; Suzuki, M.; Usami, N.; Yokoya, A.; Mori, T. Lethal effect of K-shell absorption of intracellular phosphorus on wild-type and radiation sensitive mutants of Escherichia coli. Acta Oncol. 1996, 35, 889–894. [Google Scholar] [CrossRef] [Green Version]
- Munakata, N.; Hieda, K.; Usami, N.; Yokoya, A.; Kobayashi, K. Inactivation action spectra of Bacillus subtilis spores with monochromatic soft X rays (0.1–0.6 nm) of synchrotron radiation. Radiat. Res. 1992, 131, 72–80. [Google Scholar] [CrossRef]
- Usami, N.; Yokoya, A.; Ishizaka, S.; Kobayashi, K. Reparability of lethal lesions produced by phosphorus photoabsorption in yeast cells. J. Radiat. Res. 2001, 42, 317–331. [Google Scholar] [CrossRef] [Green Version]
- Watanabe, M.; Suzuki, M.; Watanabe, K.; Suzuki, K.; Usami, N.; Yokoya, A.; Kobayashi, K. Mutagenic and transforming effects of soft-X-rays with resonance energy of phosphorus K-absorption edge. Int. J. Radiat. Biol. 1992, 61, 161–168. [Google Scholar] [CrossRef]
- Takahashi, N.; Hieda, K.; Morohoshi, F.; Munakata, N. Base substitution spectra of nalidixylate resistant mutations induced by monochromatic soft X and 60Co gamma-rays in Bacillus subtilis spores. J. Radiat. Res. 1999, 40, 115–124. [Google Scholar] [CrossRef] [Green Version]
- Kobayashi, K.; Hieda, K.; Maezawa, H.; Furusawa, Y.; Suzuki, M.; Ito, T. Effects of K-shell X-ray absorption of intracellular phosphorus on yeast cells. Int. J. Radiat. Biol. 1991, 59, 643–650. [Google Scholar] [CrossRef]
- Saigusa, S.; Ejima, Y.; Kobayashi, K.; Sasaki, M.S. Induction of chromosome aberrations by monochromatic X-rays with resonance energy of phosphorus K-shell absorption edge. Int. J. Radiat. Biol. 1992, 61, 785–790. [Google Scholar] [CrossRef] [PubMed]
- Tomita, M.; Maeda, M.; Usami, N.; Yokoya, A.; Watanabe, R.; Kobayashi, K. Enhancement of DNA double-strand break induction and cell killing by K-shell absorption of phosphorus in human cell lines. Int. J. Radiat. Biol. 2016, 92, 724–732. [Google Scholar] [CrossRef] [PubMed]
- Maeda, M.; Kobayashi, K.; Hieda, K. Efficiencies of induction of DNA double strand breaks in solution by photoabsorption at phosphorus and platinum. Int. J. Radiat. Biol. 2004, 80, 841–847. [Google Scholar] [CrossRef]
- Sekiyama, H. P K Absorption Spectra of PO43−, PHO32−, PH2O2−, P2O74− and P3O105−. In Photon Factory Activity Report 1984/1985, Part B Users’ Reports, National Laboratory for High Energy Physics; KEK: Hong Kong, 1986; p. 198. [Google Scholar]
- White, D.R.; Booz, J.; Griffith, R.V.; Spokas, J.J.; Wilson, I.J. Report 44. J. Int. Comm. Radiat. Units Meas. 2016, 22. [Google Scholar] [CrossRef]
- Hubbell, J.H. Photon mass attenuation and energy-absorption coefficients. Int. J. Appl. Radiat. Isot. 1982, 33, 1269–1290. [Google Scholar] [CrossRef]
- Konishi, T.; Takeyasu, A.; Yasuda, N.; Natsume, T.; Nakajima, H.; Matsumoto, K.; Asuka, T.; Sato, Y.; Furusawa, Y.; Hieda, K. Number of Fe ion traversals through a cell nucleus for mammalian cell inactivation near the bragg peak. J. Radiat. Res. 2005, 46, 415–424. [Google Scholar] [CrossRef] [Green Version]
- Blocher, D. In CHEF electrophoresis a linear induction of dsb corresponds to a nonlinear fraction of extracted DNA with dose. Int. J. Radiat. Biol. 1990, 57, 7–12. [Google Scholar] [CrossRef]
- Fox, M.; Boyle, J.M. Factors affecting the growth of Chinese hamster cells in halt selection media. Mutat. Res. Mol. Mech. Mutagen. 1976, 35, 445–464. [Google Scholar] [CrossRef]
- Kellerer, A.M.; Rossi, H.H. The Theory of Dual Radiation Action. In Current Topics in Radiation Research; Ebert, M., Howard, A., Eds.; North-Holland Publishing Company: Amsterdam, The Netherlands, 1974; Volume 8, pp. 85–158. [Google Scholar]
- Tobias, C. The Repair-Misrepair Model of Cell Survival. In Radiation Biology and Cancer Research; Meyn, R., Withers, H., Eds.; Raven Press: New York, NY, USA, 1980; pp. 195–230. [Google Scholar]
- Chadwick, K.; Leenhouts, H. The Molecular Theory of Radiation Biology; Springer: Berlin, Germany, 1981. [Google Scholar] [CrossRef]
- Curtis, S.B. Lethal and potentially lethal lesions induced by radiation--a unified repair model. Radiat. Res. 1986, 106, 252–270. [Google Scholar] [CrossRef]
- Curtis, S.B. The Lethal and Potentially Lethal Model—A Review and Recent Development. In Quantitative Mathematical Models in Radiation Biology; Springer: Berlin/Heidelberg, Germany, 1988; pp. 137–146. [Google Scholar] [CrossRef]
- Alpen, E. Radiation Biophysics; Elsevier: Amsterdam, The Netherlands, 1998. [Google Scholar] [CrossRef]
- Barendsen, G.W. Mechanisms of Cell Reproductive Death and Shapes of Radiation Dose-survival Curves of Mammalian Cells. Int. J. Radiat. Biol. 1990, 57, 885–896. [Google Scholar] [CrossRef]
- Barendsen, G.W. Parameters of linear-quadratic radiation dose-effect relationships: Dependence on LET and mechanisms of reproductive cell death. Int. J. Radiat. Biol. 1997, 71, 649–655. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chapman, J.D. Single-hit mechanism of tumour cell killing by radiation. Int. J. Radiat. Biol. 2003, 79, 71–81. [Google Scholar] [CrossRef] [PubMed]
- Sech, C.L.; Frohlich, H.; Saint-Marc, C.; Charlier, M. DNA Breakage upon K-Shell Excitation of Phosphorus as a Model for Direct Effects in Radiation Biology. Radiat. Res. 1996, 145. [Google Scholar] [CrossRef]
- Yokoya, A.; Cunniffe, S.M.; Watanabe, R.; Kobayashi, K.; O’Neill, P. Induction of DNA strand breaks, base lesions and clustered damage sites in hydrated plasmid DNA films by ultrasoft X rays around the phosphorus K edge. Radiat. Res. 2009, 172, 296–305. [Google Scholar] [CrossRef]
- Maezawa, H.; Indo, H.P.; Usami, N.; Majima, H.J.; Ito, H.; Ohnishi, K.; Kobayashi, K. Enhancement of membrane lipid peroxidation in lung cancer cells irradiated with monoenergetic X-rays at the K-shell resonance absorption peak of phosphorus. J. Radiat. Res. 2020, 61, 237–242. [Google Scholar] [CrossRef]
- Bigelow, A.W.; Randers-Pehrson, G.; Garty, G.; Geard, C.R.; Xu, Y.; Harken, A.D.; Johnson, G.W.; Brenner, D.J. Ion, X-ray, UV and Neutron Microbeam Systems for Cell Irradiation. AIP Conf. Proc. 2010, 1336, 351–355. [Google Scholar] [CrossRef] [Green Version]
- Konishi, T.; Oikawa, M.; Suya, N.; Ishikawa, T.; Maeda, T.; Kobayashi, A.; Shiomi, N.; Kodama, K.; Hamano, T.; Homma-Takeda, S.; et al. SPICE-NIRS microbeam: A focused vertical system for proton irradiation of a single cell for radiobiological research. J. Radiat. Res. 2013, 54, 736–747. [Google Scholar] [CrossRef] [Green Version]
- Tomita, M.; Maeda, M. Mechanisms and biological importance of photon-induced bystander responses: Do they have an impact on low-dose radiation responses. J. Radiat. Res. 2015, 56, 205–219. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wu, L.J.; Randers-Pehrson, G.; Xu, A.; Waldren, C.A.; Geard, C.R.; Yu, Z.; Hei, T.K. Targeted cytoplasmic irradiation with alpha particles induces mutations in mammalian cells. Proc. Natl. Acad. Sci. USA 1999, 96, 4959–4964. [Google Scholar] [CrossRef] [Green Version]
- Maeda, M.; Usami, N.; Kobayashi, K. Low-dose hypersensitivity in nucleus-irradiated V79 cells studied with synchrotron X-ray microbeam. J. Radiat. Res. 2008, 49, 171–180. [Google Scholar] [CrossRef] [Green Version]
- Konishi, T.; Kobayashi, A.; Tengku Ahmad, T.F.; Wang, J. Enhanced DNA double strand break repair triggered by microbeam irradiation induced cytoplasmic damage. J. Radiat. Cancer Res. 2018, 9, 183–189. [Google Scholar] [CrossRef]
- Wang, J.; Konishi, T. Nuclear factor (erythroid-derived 2)-like 2 antioxidative response mitigates cytoplasmic radiation-induced DNA double-strand breaks. Cancer Sci. 2019, 110, 686–696. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, J.; Kobayashi, A.; Ohsawa, D.; Oikawa, M.; Konishi, T. Cytoplasmic Radiation Induced Radio-Adaptive Response in Human Lung Fibroblast WI-38 Cells. Radiat. Res. 2020, 194, 288–297. [Google Scholar] [CrossRef] [PubMed]


| Materials | μen/ρ [m2/kg] | |
|---|---|---|
| 2.147 keV | 2.153 keV | |
| Air (1) | 42.79 | 42.45 |
| Water (1) | 50.17 | 49.77 |
| Soft tissue (2) | 45.66 | 48.75 |
| Cell nucleus (2) | 46.11 | 71.05 |
| DNA (3) | 41.8 | 140.4 |
| Energy [keV] | α [C/kg]−1 | β [C/kg]−2 | Ψ37 [C/kg] | Ψ10 [C/kg] | Ψ1 [C/kg] |
|---|---|---|---|---|---|
| 2.147 | 5.54 ± 1.7 | 113.5 ± 11.9 | 0.0723 | 0.120 | 0.179 |
| 2.153 | 14.4 ± 2.5 | 124.2 ± 19.2 | 0.0486 | 0.090 | 0.143 |
| f1 | 2.60 ± 0.93 | 1.09 ± 0.2 | 1.49 | 1.33 | 1.25 |
| Energy [keV] | α [Gy]−1 | β [Gy]−2 | D37 [Gy] | D10 [Gy] | D1 [Gy] |
|---|---|---|---|---|---|
| 2.147 | 0.152 ± 0.047 | 0.085 ± 0.009 | 2.64 | 4.38 | 6.51 |
| 2.153 | 0.254 ± 0.045 | 0.039 ± 0.006 | 2.75 | 5.10 | 8.11 |
| f1 | 1.67 ± 0.59 | 0.45 ± 0.07 | 0.96 | 0.86 | 0.80 |
| Energy [keV] | R DSB/Mbp /Ψ | R′ 10−3 DSB/Mbp /Gy | R″ DSB/Cell /Gy | DSB/Cell/Ψi (or Di) | ||
|---|---|---|---|---|---|---|
| Ψ37 (D37) | Ψ10 (D10) | Ψ1 (D1) | ||||
| 2.147 | 0.224 ± 0.015 | 6.10 ± 0.40 | 31.4 ± 2.1 | 20.0 | 46.3 | 90.7 |
| 2.153 | 0.350 ± 0.020 | 6.20 ± 0.40 | 31.9 ± 2.1 | 22.1 | 61.5 | 135.8 |
| f1 | 1.56 ± 0.13 | 1.02 ± 0.26 | 1.02 ± 0.26 | 1.10 | 1.33 | 1.50 |
| Energy [keV] | Cross-Section 10−13 [m2] | Quantum Yield *** | |
|---|---|---|---|
| σDSB | σabsorption * | ||
| 2.147 | 5.02 ± 0.33 | 2.42 ± 0.46 | - |
| 2.153 | 7.79 ± 0.33 | 8.13 ± 0.16 | - |
| phosphorus K-shell ** | 2.77 ± 0.55 | 5.71 ± 0.49 | 0.49 ± 0.18 # |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Konishi, T.; Yoshihara, D.; Maeda, M.; Ohsawa, D.; Usami, N.; Hieda, K. Enhanced Cell Inactivation and Double-Strand Break Induction in V79 Chinese Hamster Cells by Monochromatic X-Rays at Phosphorus K-Shell Absorption Peak. Quantum Beam Sci. 2020, 4, 38. https://doi.org/10.3390/qubs4040038
Konishi T, Yoshihara D, Maeda M, Ohsawa D, Usami N, Hieda K. Enhanced Cell Inactivation and Double-Strand Break Induction in V79 Chinese Hamster Cells by Monochromatic X-Rays at Phosphorus K-Shell Absorption Peak. Quantum Beam Science. 2020; 4(4):38. https://doi.org/10.3390/qubs4040038
Chicago/Turabian StyleKonishi, Teruaki, Daichi Yoshihara, Munetoshi Maeda, Daisuke Ohsawa, Noriko Usami, and Kotaro Hieda. 2020. "Enhanced Cell Inactivation and Double-Strand Break Induction in V79 Chinese Hamster Cells by Monochromatic X-Rays at Phosphorus K-Shell Absorption Peak" Quantum Beam Science 4, no. 4: 38. https://doi.org/10.3390/qubs4040038






