Abstract
Many bacterial communities display oscillations throughout the day; it has been shown that in humans, the healthy gut microbiome also shows fluctuations in the abundance of bacterial community, related with feeding times and sleep cycles. Likewise, in human milk there are beneficial bacteria that undergo changes in the same time interval and are transferred to the newborn by breastfeeding, colonizing the gastrointestinal tract. The aim of this work was to identify changes in the bacterial diversity of human milk throughout the day. Human milk samples were collected from a single donor three times during the day (morning, afternoon, night) for 5 consecutive days, and bacterial DNA was extracted. Bacterial diversity was characterized by high-throughput DNA sequencing of 16S rDNA libraries, and taxonomy was assigned by comparison of sequences against a database. Finally, the significant differences in relative abundance and alpha and beta diversity were determined. The analysis of human milk displayed changes in the bacterial diversity during the day, with significant changes in the Shannon diversity index. Our data show that human milk seems to be affected by the daytime change, and these changes could influence the infant gut microbiota.
1. Introduction
Circadian rhythms are present in almost every organism in response to continuous changes in light as a result of the rotation of the planet around its axis [1]. Some reports describe the presence of photosynthetic bacteria in this rhythmic process; however, the circadian clock operates in non-photosynthetic bacteria too, such as those of the intestinal microbiota [2]. The gut microbiota composition and diversity are sensitive to changes in the environment of the host, such as the circadian cycle [3]. Many of these bacteria display oscillations throughout the day, regulated by the host feeding times and sleep cycles, contributing to homeostasis, and participating in several physiological processes [4,5]. Likewise, in human milk there are beneficial bacteria that are transferred to the newborn by breastfeeding, colonizing the gastrointestinal tract [6]. In addition, other studies suggest that some components of human milk, such as proteins, nucleotides, and vitamins, among others, undergo diurnal variations in the same way the gut microbiota do in both mice and humans [4,7]. We believe that in human milk, these oscillations enhance the well-being of the breast-fed infant.
Besides the knowledge of gut microbiota and circadian rhythm, the relationship between the variation in the bacterial diversity in human milk and different hours of the day is not well understood; for this reason, the aim of this work is to characterize the bacterial community in human milk over the course of the day, and to identify the possible changes in the bacterial composition and diversity of the human milk microbiota.
2. Materials and Methods
2.1. Study Design
A longitudinal case report study was performed on a healthy 35-year-old multiparous volunteer Mexican woman, four months after the vaginal delivery of a male neonate without complications and no fetal distress.
2.2. Sample Collection
Human milk samples were collected from the donor three times a day, approximately once every 8 h (morning (7:00), afternoon (15:00) and night (22:00)) over five consecutive days. The samples were manually collected in aseptic conditions and immediately stored at −20 °C until processing for DNA extraction.
2.3. DNA Extraction and Preparation of V3 16S rDNA Libraries
Bacterial DNA was extracted using the FavorPrep™ Stool DNA Isolation Mini Kit (Cat No. FASTI 001-1) following the manufacturer’s instructions. For library preparation a ~281 bp amplicon containing the V3 hypervariable region of the 16S RNA gene in each sample was amplified and equimolar concentrations of amplicons were pooled. After that, bacterial communities were characterized by the high-throughput sequencing of V3-16S rDNA libraries in the Ion Torrent PGM System as previously reported [6].
2.4. Data Analysis
The sequencing data were analyzed using QIIME pipeline v1.9.0. [8] and taxonomy was assigned by comparison of the sequences against the Greengenes reference database. To detect significant differences in the relative abundances of bacterial taxa among groups, the linear discriminant analysis effect size program (LEfSe v1.0) was used. Finally, the alpha diversity was determined using the Phyloseq and ggplot2 packages in the R environment (v3.4.4). For beta diversity, the dissimilarity was estimated using Bray–Curtis analysis. A two-dimensional scatter plot was generated using Multidimensional scaling (MDS) in the R environment.
2.5. Statistical Analyses
The statistical comparison of groups was calculated with the non-parametric Friedman test for repeated measures using the Social Science Statistic software. p < 0.05 was considered statistically significant. The analysis of similarities (ANOSIM) was used for category comparisons of the phylogenetic distance metrics.
3. Results
3.1. Bacterial Diversity Changes According the Sampling Time in the Day
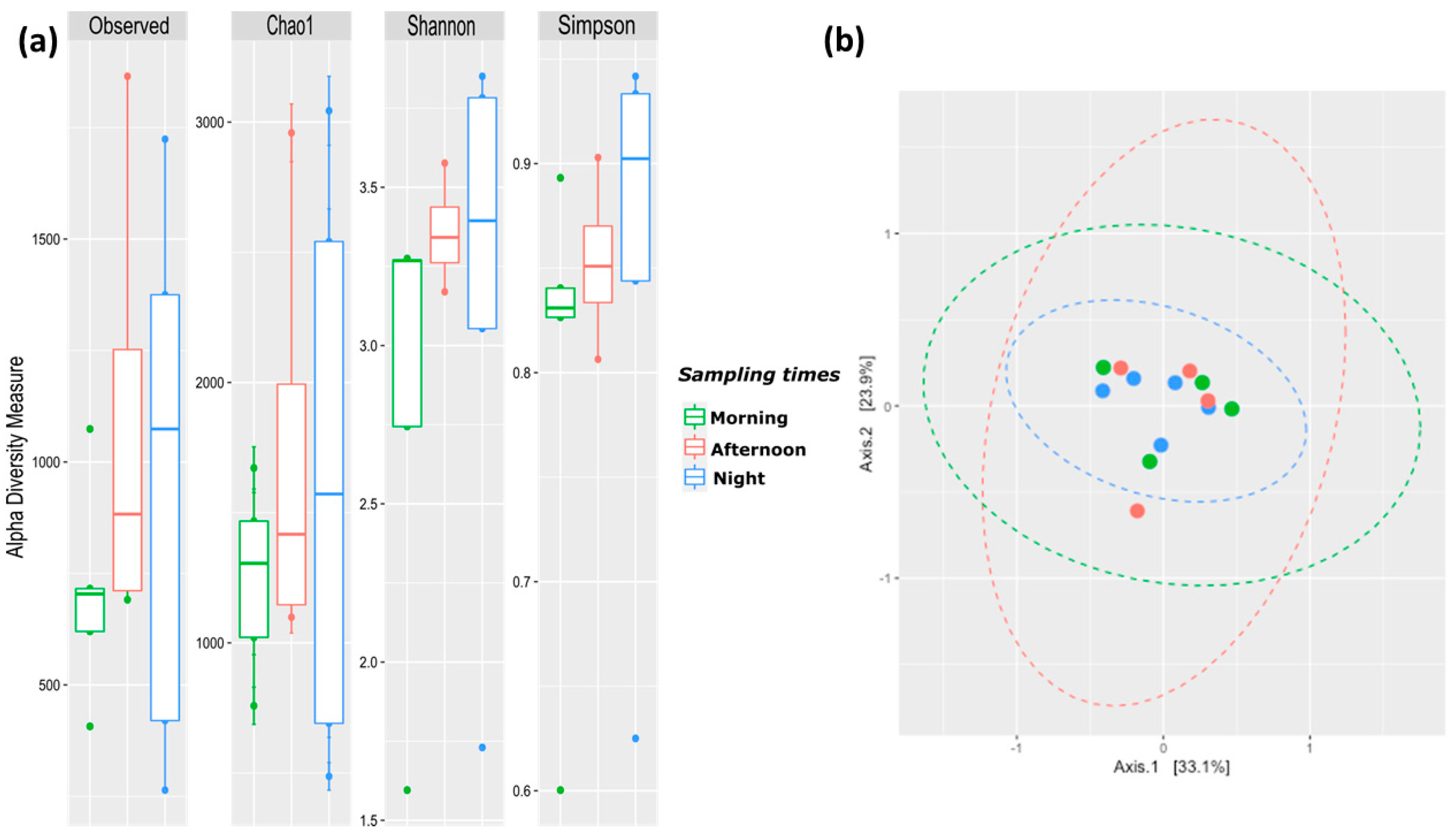
The characterization of the microbiota in human milk at different sampling times of the day showed significant changes in the bacterial alpha diversity according to the Shannon diversity index (p = 0.022); however, no significant changes were detected in the bacterial richness evaluated by observed number of species (p = 0.861), Chao 1 (0.846), or in the dominance by Simpson index (0.449) (Figure 1a). This difference in the diversity was detected between the morning sampling time and the night. Conversely, no differences were observed in the bacterial composition or diversity (alpha and beta diversity) of the afternoon group compared with the morning and night sampling time groups (Figure 1a,b).
Figure 1.
Human milk diversity according to sampling time in the day. (a) Alpha diversity indexes based on observed number of species, Chao1, Shannon and Simpson. (b) Beta diversity analysis two-dimensional plot calculated with Bray–Curtis test for the human milk samples. Human milk morning samples are plotted as green dots, afternoon samples are represented as red dots and night samples are denoted as blue dots.
3.2. Different Bacterial Taxa are Found between Morning and Night Sampling
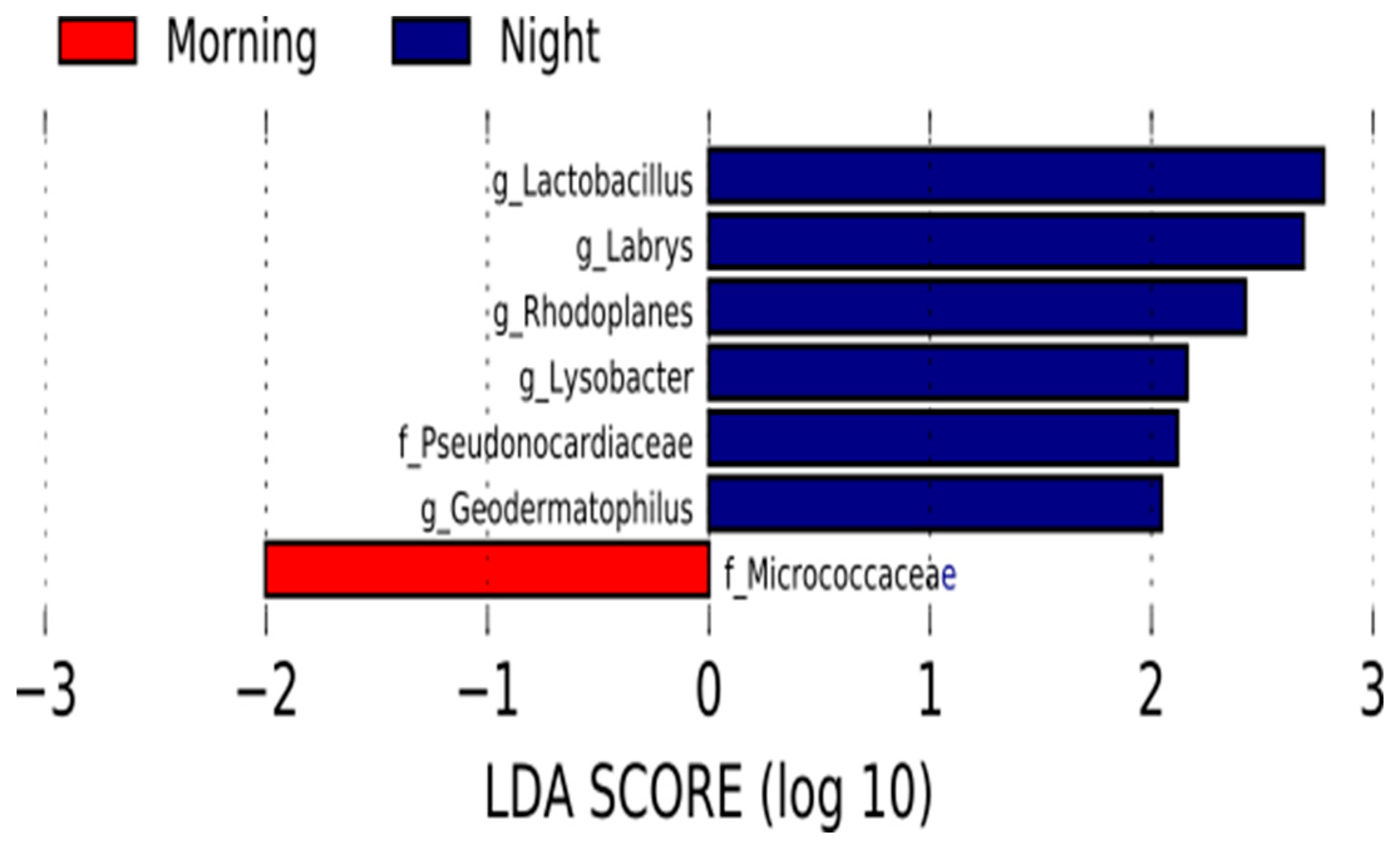
Likewise, variations in the relative abundance of some bacterial taxa were identified between the night and morning samples using linear discriminant effect size. Three proteobacteria, two actinobacteria taxa and the genus lactobacillus were more represented in the night milk samples set. On the other hand, the family Micrococcaceae was more abundant in the samples collected in the morning (Figure 2).
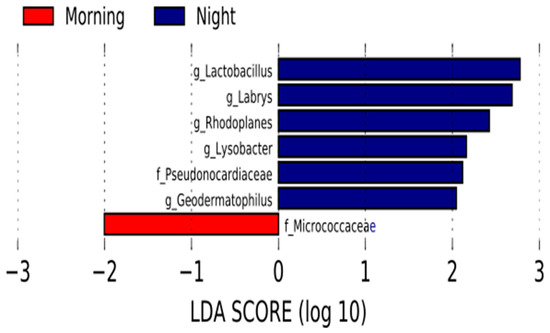
Figure 2.
Represented significant bacterial taxa for each time of the day. Linear discriminant analysis (LDA) effect size (LEfSe) comparison of bacterial taxa between human milk morning samples and night samples. Horizontal bars represent the effect size for each taxon: red indicates taxa enriched in the morning group and blue indicates taxa enriched in the night group. LDA score cutoff of 2.0.
4. Discussion
Circadian fluctuations in some bioactive components have been previously reported; these changes are suggested to transfer chronobiological information from mother to child to assist the development of the biological clock [7]. Our results on bacterial abundance showed that the members of the phyla Proteobacteria, Actinobacteria and Firmicutes are more abundant at night. This result agrees with other studies on changes in the abundances of members of the microbiota in some organisms. For instance, during the analysis of bacterial abundance in saliva in humans, a night-time predominance of the phylum Actinobacteria was observed in healthy volunteers [9]. In other studies, in mice, it has been determined that members of the phylum Proteobacteria presented oscillations in abundance through the day, with a greater abundance of these bacteria at night [1]. Additionally, Lactobacillus (Firmicutes) exhibited rhythmic oscillations in a 24 h cycle, associated with feeding time, showing in this manner phase shifts between dark and light in mice [4]. Finally, as has previously been reported for fecal microbiota, these changes could influence the composition of the infant gut microbiota, since breastfeeding is considered the most optimal mode of feeding for neonates.
5. Conclusions
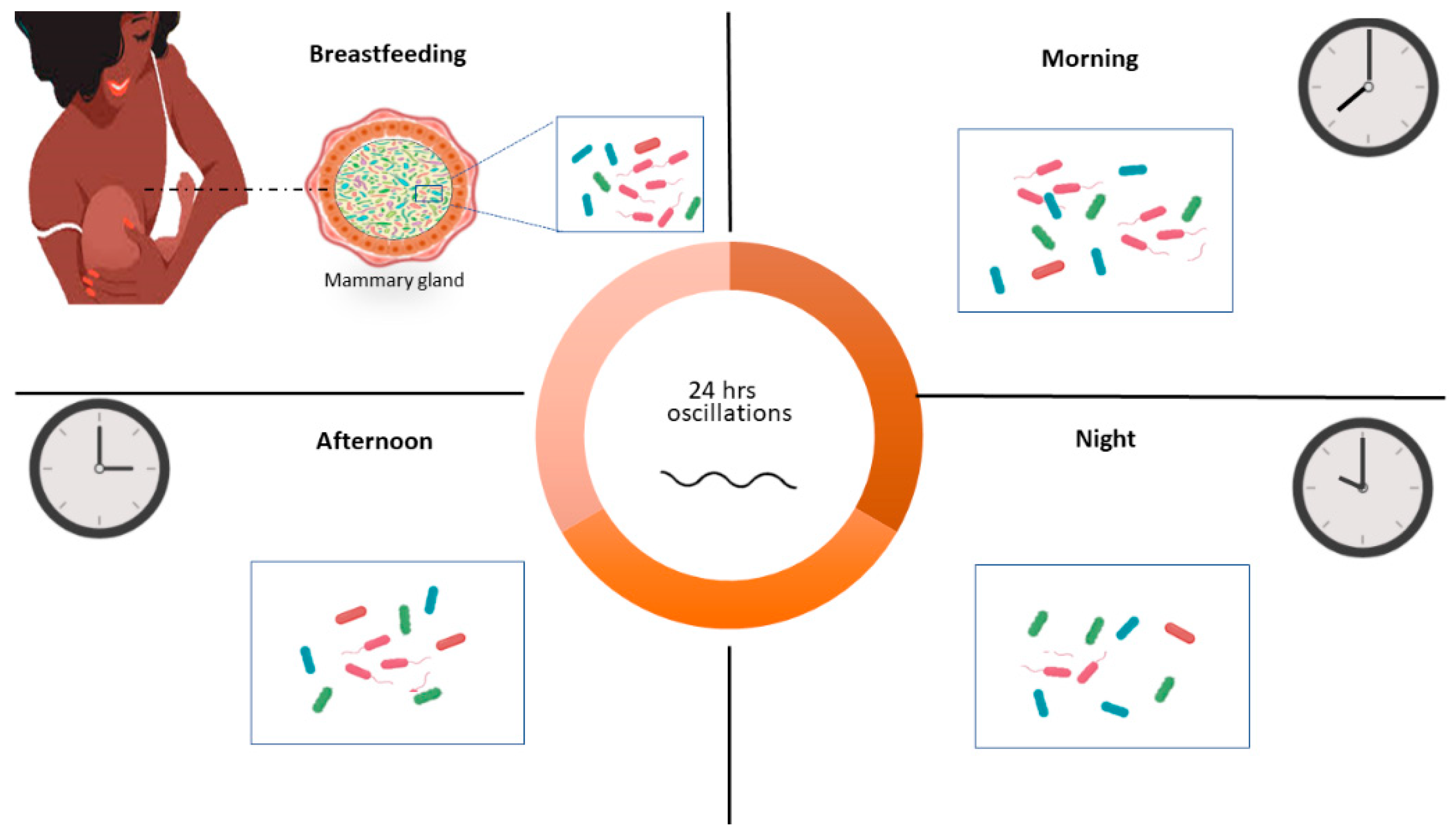
As some components of human milk, the human milk microbiota seem to be affected by circadian changes. The results presented in this work suggest that the milk microbiota exhibit diurnal fluctuations, resulting in specific bacterial arrangements depending on the time of day (Figure 3), at least in the studied case. However, since the possible conclusions from a case study are limited, it is necessary to study a larger number of samples to obtain more conclusive results.
Figure 3.
Model for the bacterial diversity change during the day. Bacterial microbiota show variations in diversity and composition between the morning and night. This could have an impact on the infant microbiota.
Author Contributions
Conceptualization, K.C.-C., and J.G.-M.; methodology, K.C.-C., N.G.Z.-T., F.H.-Q., A.P.-E. and R.G.-G.; software, K.C.-C. and F.H.-Q.; validation, K.C.-C., N.G.Z.-T., and J.G.-M.; formal analysis, K.C.-C., and F.H.-Q.; investigation, N.G.Z.-T., and K.C.-C.; resources, R.G.-G., and J.G.-M.; data curation, K.C.-C., and F.H.-Q.; writing—original draft preparation, K.C.-C., and N.G.Z.-T.; writing—review and editing K.C.-C., N.G.Z.-T., and J.G.-M.; visualization K.C.-C., F.H.-Q., and N.G.Z.-T.; supervision, J.G.-M.; project administration, J.G.-M.; funding acquisition, J.G.-M. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by Cinvestav, and Consejo Nacional de Ciencia y Tecnología (CONACyT) México, CONACyT-163235-INFR-2011-01.
Acknowledgments
We are grateful to the participant mother who provide the samples. We thank CONACyT Doctoral Fellowships for K.C.-C. (777953), and F.H.-Q. (291236), and Master Fellowship for N.G.Z.-T. (997494). We are grateful to Ms. Viridiana Rosas Ocegueda for administrative assistance. J.G.-M. (19815) is a Fellow from the Sistema Nacional de Investigadores, Mexico.
Conflicts of Interest
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, or in the decision to publish the results.
References
- Liang, X.; Bushman, F.D.; FitzGerald, G.A. Rhythmicity of the intestinal microbiota is regulated by gender and the host circadian clock. Proc. Natl. Acad. Sci. USA 2015, 112, 10479–10484. [Google Scholar] [CrossRef] [PubMed]
- Costantini, C.; Renga, G.; Sellitto, F.; Borghi, M.; Stincardini, C.; Pariano, M.; Zelante, T.; Chiarotti, F.; Bartoli, A.; Mosci, P.; et al. Microbes in the era of circadian medicine. Front. Cell. Infect. Microbiol. 2020, 10, 30. [Google Scholar] [CrossRef] [PubMed]
- Voigt, R.M.; Forsyth, C.B.; Green, S.J.; Mutlu, E.; Engen, P.; Vitaterna, M.H.; Turek, F.W.; Keshavarzian, A. Circadian disorganization alters intestinal microbiota. PLoS ONE 2014, 9, e97500. [Google Scholar] [CrossRef] [PubMed]
- Thaiss, C.A.; Zeevi, D.; Levy, M.; Zilberman-Schapira, G.; Suez, J.; Tengeler, A.C.; Abramson, L.; Katz, M.N.; Korem, T.; Zmora, N.; et al. Transkingdom control of microbiota diurnal oscillations promotes metabolic homeostasis. Cell 2014, 159, 514–529. [Google Scholar] [CrossRef] [PubMed]
- Trinder, M.; Bisanz, J.E.; Burton, J.P.; Reid, G. Bacteria Need Sleep Too?: Microbiome Circadian Rhythmicity, Metabolic Disease, and Beyond. Univ. Tor. Med. J. 2015, 92, 52–55. [Google Scholar]
- Corona-Cervantes, K.; García-González, I.; Villalobos-Flores, L.E.; Hernández-Quiroz, F.; Piña-Escobedo, A.; Hoyo-Vadillo, C.; Rangel-Calvillo, M.N.; García-Mena, J. Human milk microbiota associated with early colonization of the neonatal gut in Mexican newborns. PeerJ 2020, 8, e9205. [Google Scholar] [CrossRef] [PubMed]
- Italianer, M.F.; Naninck, E.F.; Roelants, J.A.; van der Horst, G.T.; Reiss, I.K.; Goudoever, J.B.; Joosten, K.F.; Chaves, I.; Vermeulen, M.J. Circadian Variation in Human Milk Composition, a Systematic Review. Nutrients 2020, 12, 2328. [Google Scholar] [CrossRef] [PubMed]
- Caporaso, J.G.; Kuczynski, J.; Stombaugh, J.; Bittinger, K.; Bushman, F.D.; Costello, E.K.; Fierer, N.; Pena, A.G.; Goodrich, J.K.; Gordon, J.I.; et al. QIIME allows analysis of high-throughput community sequencing data. Nat. Methods 2010, 7, 335–336. [Google Scholar] [CrossRef] [PubMed]
- Johnson, C.H.; Zhao, C.; Xu, Y.; Mori, T. Timing the day: What makes bacterial clocks tick? Nat. Rev. Microbiol. 2017, 15, 232–242. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).