Abstract
Transition metal oxides (TMO) and their carbon composites have become a glittering upcoming material science candidate. Their interesting properties, such as their meticulous morphology, plentiful availability, flexible surface chemistry along with outstanding mechanical, thermal, and optical properties make them ideal for efficient photocatalytic dye degradation. An extensive range of TMO, and their carbon composites are reviewed highlighting the progression and opportunities for the photocatalytic degradation of dyes. Here, we concisely describe the numerous techniques to extend the optical absorption of these TMOs involving dye sensitization, metal doping, etc. Besides this, an overview of all aspects of dye degradation along with the prevailing challenges for future utilization and development of such nanocomposites towards highly efficient dye degradation system are also reported.
1. Introduction
With the increase in world population and rapid urbanization, natural resources are getting exhausted. Development of urban areas causes organic effluents from industries and agricultural activities, which not only cause threatens to human lives but also affected aquatic life very adversely [1]. Globally, 1.2 billion people are deficient in accessing safe drinking water, and millions die from uncontrolled spread of diseases and chronic illness transmitted via unsafe water [2,3]. Particularly, there is an explosive development in the dye and dyeing industries since last few decades. As effluents, many potentially hazardous compounds have been introduced into the environment, which are harmful for both flora and fauna on the planet. As per the World Bank’s estimation, textile dyeing and treatment effluents results in 17 to 20% of water pollution [4]. The affecting factors broadly comprise of poor biodegradable waste such as used dyes and of which ~10–15% enter the environment through wastes. These dyes can be cationic, anionic or non-ionic depending on their chemical structure. The release of dyes in effluent results in perturbations of the marine life leading to severe effects on exposed organisms owing to the reduction in photosynthetic rate.
Moreover, water quality is significantly influenced due to the color of the dye, causing esthetic pollution, which is associated with contamination [5,6]. Willmott et al. has reported that dye concentrations as small as 0.005 mg dm−3 is perceptible in river water [7]. Today, 20% of our population has no access to safe water, 40% suffers owing to improper sanitary conditions and millions of people die every year from diseases communicated through unsafe water [2,8]. Dye effluent commonly contains chemicals which are toxic, carcinogenic or mutagenic to aquatic species [9,10]. In addition, human disorders say nausea, hemorrhage, ulceration of the skin and mucous membranes [11] are caused due to the existence of dyes or their degradation products in water. Along with this, these dyes can also cause acute injury to the central nervous system and reproductive system [12]. This leads to deeper concern for colored wastewater discharges and convincing the dye users to espouse cleaner technology methodologies for benign dyeing strategies and upgrading dyes fatigue.
Consequently, significant efforts have been employed to initiate different suitable purification methods to destroy these pollutants. However, their incomplete removal during processing led to their presence in wastewater effluents in minute amount which is a matter of big concern towards the health and wealth of ecosystem, as they have endocrine disrupting potency [13].
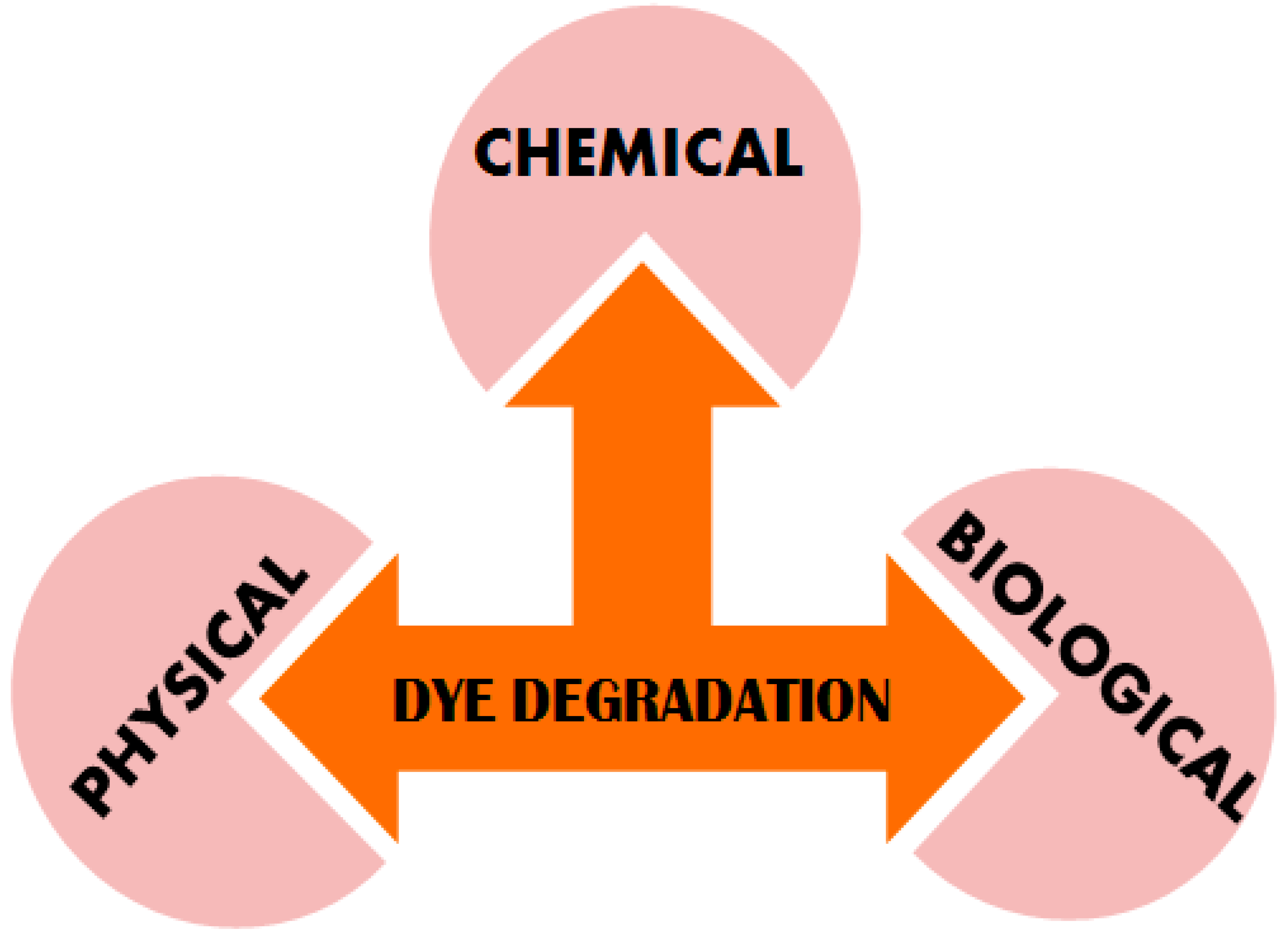
Modernized society demands for pure water have made degradation of the dyes a great challenge that needs to be addressed for the well-being of humans and aquatic life. Several physical, biological and chemical techniques have been utilized for water purification (Figure 1). Adsorption and ion exchange are the most widely used physical methods for dyes removal resulting in high quality water [14]. However, the most commonly used adsorbents such as activated carbon have a high cost and hence replaced by other alternatives which demonstrate good efficiency [15,16]. Further, this method transfers pollutants to other phases that need to be treated. Another method involves coagulation/flocculation for color removal from textile effluents is associated with high-cost reagents, which impedes their practicability. Separation processes by utilizing membranes, reverse osmosis and ultrafiltration with microfiltration help detach the dyes from larger molecules in the wastewater. These processes have limited applications due to the inherent limitations, such as high expenditure, limited versatility, elevated salt content in the waste matter, replacement of membranes and problems associated with the disposal.
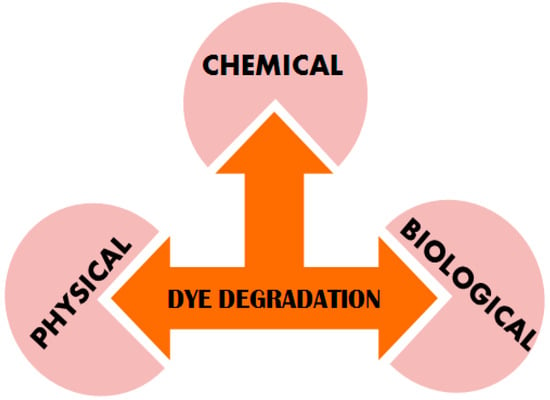
Figure 1.
Different ways of dye degradation constitute the Physical, Chemical and Biological approaches.
Utilization of biological methods proved to be an economical alternative since it requires minimal major processing costs, but numerous dyes are deliberated to defy microbial attack. Different bacteria and fungi have been used to treat textile effluents via various biological processes [17,18,19,20,21,22]. Two conditions, aerobic and anaerobic, have been combined for the proficient removal of azo dyes from wastewater, as azo dyes are not efficiently metabolized under aerobic conditions. In contrast, biological enzymes under anaerobic conditions utilize a high reduction rate of electrophilic azo-bond of the dye molecule to produce colorless aromatic amines [23]. Such amines are resilient to further anaerobic mineralization and are sometimes mutagenic to animals. Lourenco et al. suggested a sequential anaerobic–aerobic scheme utilized for wastewater treatment involving mineralization of the amines under aerobic environments via hydroxylation pathway with the ring-opening procedure [24]. Furthermore, the use of whole cells instead of enzymes has been given preference for the degradation of dyes in wastewater since cells can protect the enzymes from the severe environment. However, due to their complex structure with different chromophoric groups, dyes are enormously recalcitrant [23]. In addition, the frequent daily unpredictability of features of such wastewater enhances the difficulty of treatment [25]. Although several known physical and biological methodologies have been extensively utilized for decolorization [26], no one has materialized by way of complete remediation. Wastewater treatment via microbial biocatalysts offers potential advantages over different physical processes, but biological methods are rather inefficient, whereas physical methods are impeded in the operation scale.
Consequently, various chemical processing, such as chemical oxidation, solvent extraction, catalytic degradation, etc. for water purification, emerged. However, these are time consuming and high on cost hindering their practical utility. To provide a wholesome remediation, nanotechnology is the potential approach, focusing on unique attributes of materials emerging from nano dimensions that ensure the purification of water resources. This is possible through photocatalysis, a property exhibited by semiconducting oxides or more precisely transition metal oxides (TMO), whereby they absorb photons to undergo redox reactions, breaking complex organic molecules into simpler fragments [27]. The diffusion length of charged carriers (e−/h+) from bulk to surface is significantly reduced for nanoparticles facilitating these carriers’ migration rate and hence enhancing the photoactivity. TMOs such as titanium dioxide (TiO2), zinc oxide (ZnO), iron oxide (Fe2O3) and magnesium oxide (MgO) have been explored extensively in photocatalytic applications for water remediation [28,29]. Among them, the most widely utilized is TiO2 owing to photochemical stability, chemical inertness, and ecologically benign [30]. In addition, its competitive rival, ZnO, has better electron mobility [31]. Even though they show excellent properties, their application in photocatalytic degradation (UV light driven) is hindered owing to wide band gap. Hence, band gap modification is required for improving the photodegradation efficiency, which will be more comprehensively discussed in later section.
Currently, nanotechnology-based water purification involves photocatalytic dye degradation mainly focusing on three major areas for efficient performance:
- Visible light-driven photocatalyst
- High adsorption capacity
- Large electron/hole recombination lifetimes
Due to the wideband gaps of the transition metal oxides, extensive research has been employed to redshift their band gaps towards visible light-driven photocatalysis. Besides, the higher bandgap, rapid electron/hole pair recombination further impedes the photocatalytic activity. This may arise due to low adsorption capacity of existing adsorbent, causing a significant bottleneck for dye mineralization [32,33].
In order to achieve the above-mentioned goals, two approaches are typically employed which are the doping of existing transition metal oxides with foreign transition metal ion to tune the band gap and hence, altering the optical absorption of the resulting nanocomposite. Introduction of foreign transition metal ions results in formation of spinel or mixed bimetallic transition metal oxides. Furthermore, co-adsorbents serve the purpose to enhance the adsorption capacity with larger electron/hole recombination lifetime. High adsorption capacity is achieved by the co-adsorbent, which will facilitate the charge transfer processes resulting in higher efficiency [32,33].
Besides, the large volume of literature assembled by the researchers, here we offer a concise insight of the research on efficient photo dye degradation via transition metal oxide-based composites, along with the latest drift towards photocatalytic studies.
2. Dye Degradation
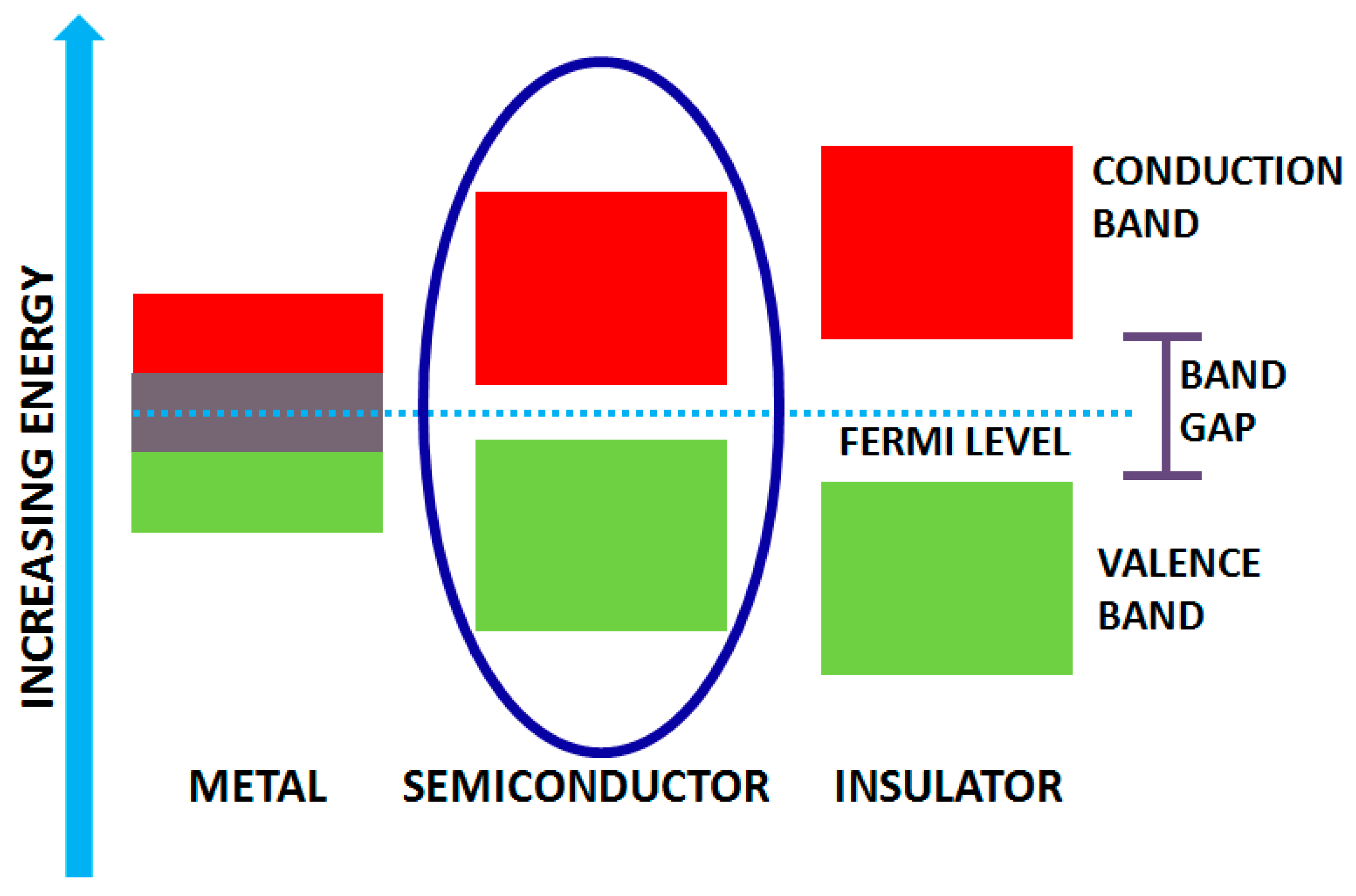
Solids, in general, possess different band gaps which result in diverse properties. Based on varying bandwidths, they are classified as metals, insulators and semiconductors (Figure 2). Solids (like metals), have overlapped band gap regardless of the temperature. The higher band is the conduction band whereas the lower one is the valence band and possesses gap for semiconductors and insulators. Bandgap in insulators is very high, and hence it is difficult to excite electrons from the valence to conduction band, and the gap is called the forbidden gap. However, in semiconductors, the bandgap is small enough for the electrons to promote from valence to conduction band at room temperature. Their bandgap lies in the range of 0 to 7 eV bounding energy levels in the visible and UV light [34,35]. Photocatalysis involves light irradiation on the catalyst which functions as thermal energy and hence excites the electron from the valence to conduction band of the catalyst. Therefore, the band gap’s width is the primary requisite for efficient photocatalyst, which strongly influences the electrical and optical characteristics of the material. To utilize the light from the solar spectrum, band gap engineering is the fundamental approach operated for semiconducting materials, and hence they are broadly utilized as photocatalysts.
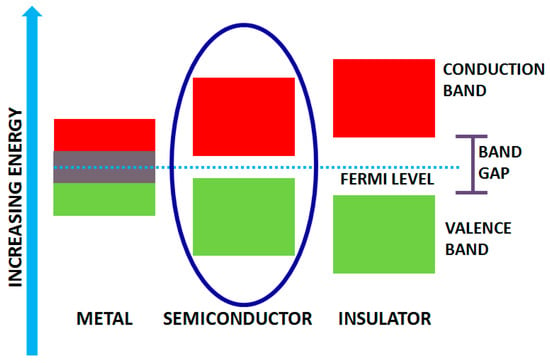
Figure 2.
Band gap structure for metals, semiconductors and insulators.
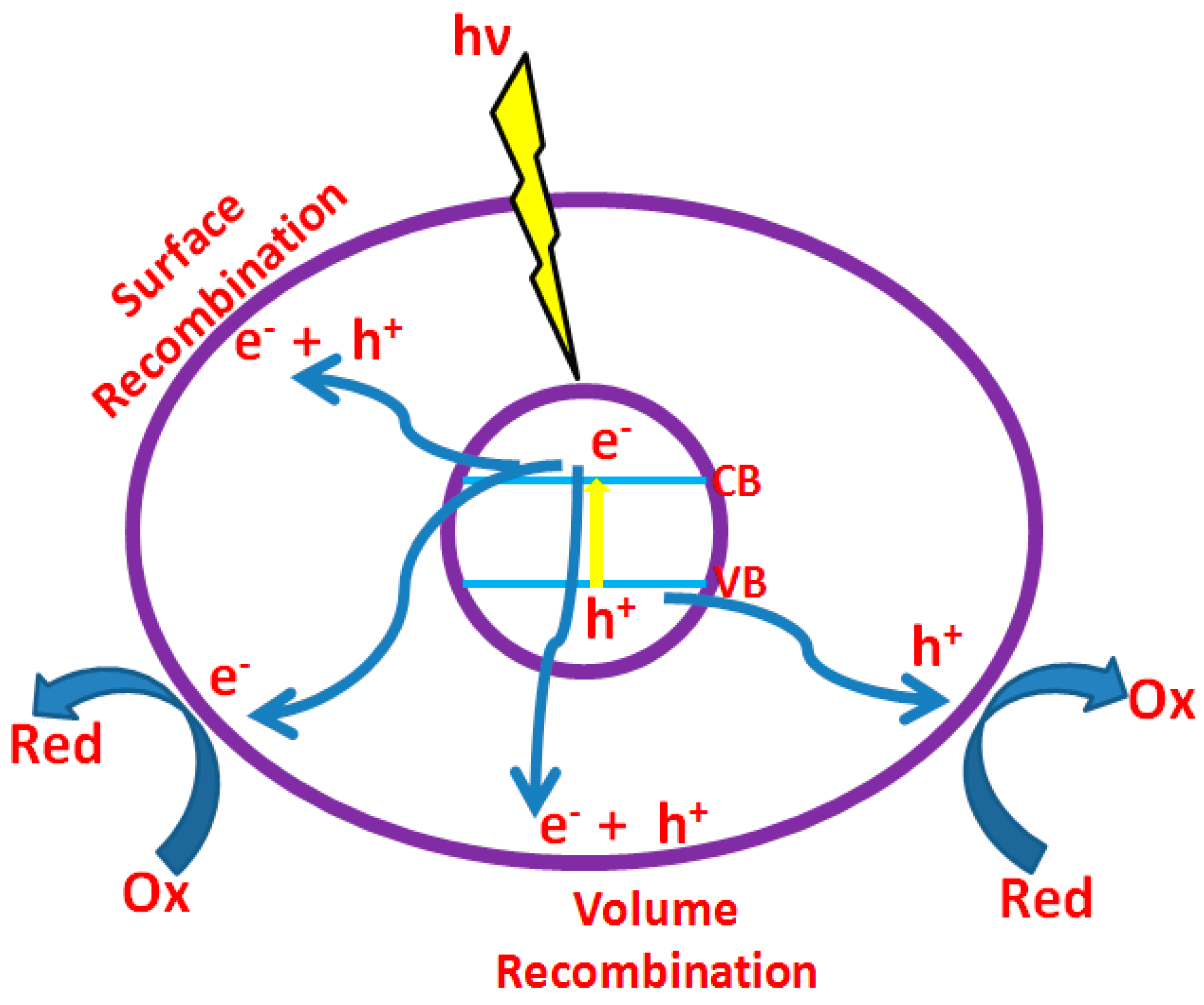
In the photocatalytic dye degradation, contaminants are destroyed utilizing transition metal oxides as efficient adsorbents, light source and oxidizing agent such as air/oxygen. Absorption of photons (with energy ≥ the band gap between valence and conduction band of transition metal oxide) results in electron transition from the valence band to the conduction band resulting in the formation of hole (h+) in the valence band and electron (e−) in the conduction band (Figure 3). Oxygen adsorbed on the photocatalyst is reduced by the electron in the conduction band, whereas the hole oxidizes the pollutant or sometimes it oxidizes water to produce •OH radicals. Here, h+ and e− act as oxidizing and reducing agents, respectively [32]. When reduction and oxidation do not proceed concurrently, electrons accumulate in the conduction band resulting in fast recombination of electrons and holes, reducing photonic efficiency. Hence, their consumption is a vital requirement to endorse photocatalytic performance. Various parameters such as light intensity, catalyst loading, pH, type of adsorbent, contaminant (dye) type and concentration etc. influences photocatalytic dye degradation [33]. Among them, this article concentrates on adsorbent properties, favorable for photo dye degradation with high efficiency.
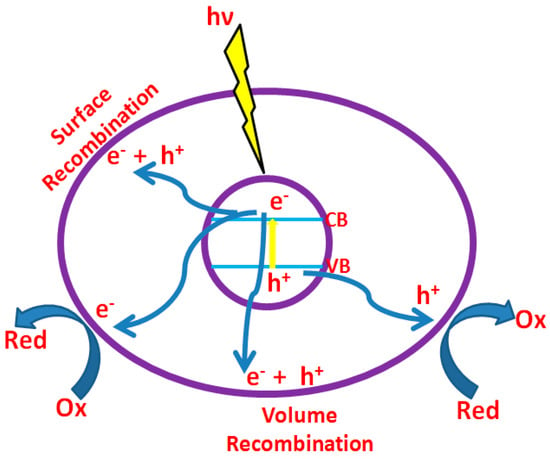
Figure 3.
Different processes (reduction, oxidation, surface recombination, etc.) takes place after irradiation of photocatalyst.
3. Nanocomposites for Photocatalytic Dye Degradation
3.1. Transition Metal Oxides as Photocatalyst
Chemical reactions in the water system are greatly influenced by the light (visible and UV light). Most of the semiconductors can strongly interact with the photons’ wavelengths as their band gaps lie in the same range. In addition to this, the large bandgap of these materials results in the absorption of UV light limiting their practical application because UV light only constitutes 5% of the solar spectrum. Besides suitable bandgap, potential adsorbents should be easy to produce, cost-effective, photostable and non-toxic for the environment [36,37]. The researchers have also explored numerous other techniques to extend the optical absorption of these transition metal oxides involving dye sensitization, metal doping, semiconductor coupling and non-metal deposition [38,39,40,41,42,43,44]. The purposes for utilizing these techniques are as follows:
- Enhance adsorption capacity at the catalyst surface
- Tuning the optical band gap or light absorption capability of the photocatalyst
- Preventing the recombination of charged carriers in combination with facilitated charge transfer
This section emphasizes the recent advances in various types of metal oxide nanoparticles and their composites for photocatalytic dye degradation from the wastewater.
3.1.1. Dye Degradation by Titanium Oxides
Photocatalytic reactions firstly reported by Fujishima and Honda et al. on the surface of TiO2 (photocatalyst) which are photolysis of water, as shown below [45]:
TiO2 + hυ → h+ + e−
½ H2O + h+ → ½ O2 + H+
H+ + e− → ½ H2
Here, H2 gas is produced from water and light interaction from solar spectrum (visible and UV), which can act as an alternative to fossil fuels [46,47]. It was then noted that the intermediate reactions resulted in formation of OH radicals, and hence, photocatalysis becomes important for the decomposition of water pollutants [48,49]. Researchers later focus on improving the efficiency of the involved photocatalysts (semiconducting oxides) for degradation of contaminants to purify water.
Earlier numerous studies have been reported for various photocatalyst, which produces photo excited e−/h+ pair for the decomposition of dyes. Among existing materials, TiO2 proved to be the most active photocatalyst for dye degradation [38,50].
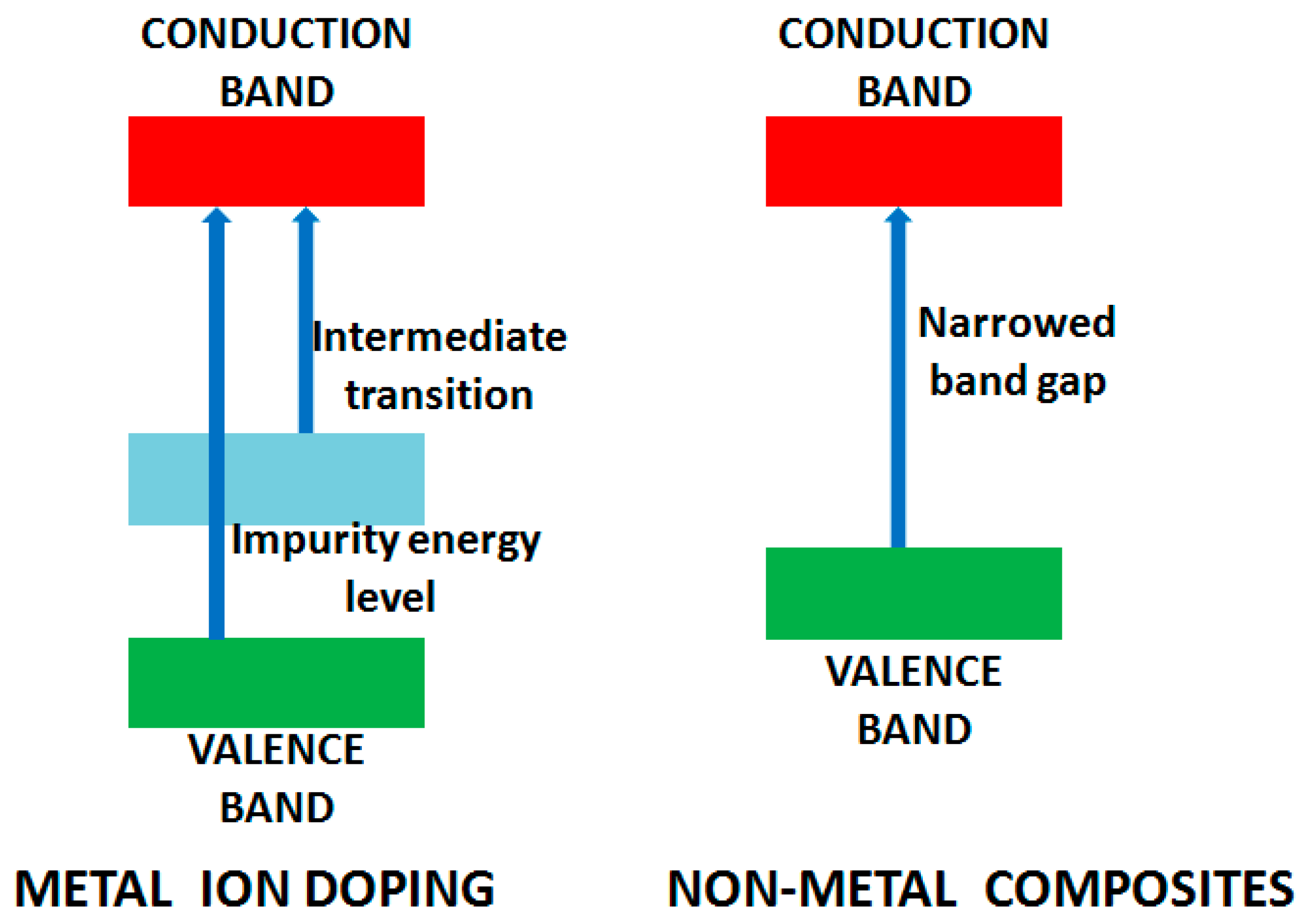
An outstanding review on metal doping for photocatalysis is reported by Carp et al. [37] with the main objective of reducing the band gap of the photocatalyst and the generation of intra-states between valence and conduction band resulting in UV to visible light absorption. The doped ion alters the recombination rate by acting as a trapping center for charge carrier and modifying the surface properties by enhancing adsorption capacity. Silver (Ag) nanoparticles loaded on different semiconductors have been illustrated as an effective approach for improving photocatalytic performance [51,52]. The metallic nanoparticle of Ag acts as a sink to accept the photoexcited electrons facilitating the reduction process and decelerating electron/hole recombination. Ag nanoparticles have also been utilized to tune metal oxide properties, i.e., TiO2, NiO and ZnO by surface plasmon resonance [42]. Excited by this, a number of research groups have explored the doping of TiO2 by different transition metal ions [53,54,55]. Vanadium doped TiO2 active under visible light illumination was reported by Klosek et al. After irradiation by visible light, excited vanadium donates an electron to TiO2 conduction band promoting the oxidation of surface adsorbed molecules [56]. Boron-doped photocatalyst has also been explored and displays high performance in mineralization of Methyl orange (MO) with decolorization degree of 90% in 2 min [57]. Alternatively, non-metal doping results in redshift of the energy which is absorbed by the photocatalyst, resulting from bandgap narrowing in which valence band is elevated (Figure 4), with no modification in the conduction band [34,49,58].
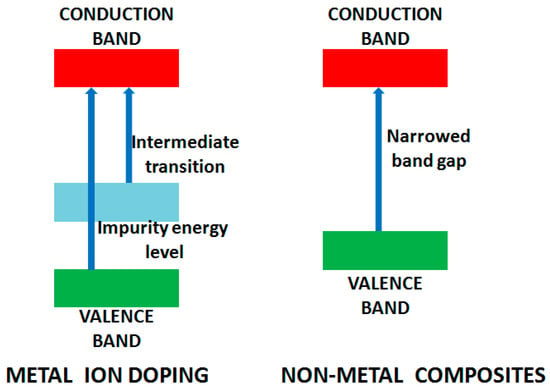
Figure 4.
Modification of band gaps on metal ion doping and for non-metal composite.
Doping of rare earth metal ions also enhances the potential of photocatalytic dye degradation owing to their distinctive chemical, physical and electronic properties. Associated f-orbital of the lanthanide ions has increased absorption of pollutants via Lewis base interaction and function as an efficient electron trapper. Du et al. suggested that doping TiO2 with rare earth metal ion enhances its photocatalytic performance [59]. Ce doping (1%) in TiO2 was reported to display best decolorization performance of Methylene Blue (MB). Zhu et al. reported 91.2% decolorization efficiency for MB by Ce+3 doped TiO2-SiO2 after 180 min irradiation [60].
In addition, there also exist reports on simultaneous deposition and doping, Ag/Ag(I)-TiO2 in which Ag(I) doping come up with the development of an isolated energy level of Ag 4d in TiO2 bandgap [61]. Visible light illumination resulted in the absorption of visible light by isolated energy level of 4d. In contrast, the surface metallic Ag facilitates the photogenerated carriers (electrons and holes) separation resulting in higher visible light activity than the TiO2 modified with Ag.
Another alternative to improve the visible light absorption or narrowing the bandgap is to substitute oxygen with inorganic ions (C+4, S−2, etc.). Different methods have been used to produce visible light active TiO2−xNx photocatalyst along with the shift of absorption edge [62,63,64,65,66,67]. It is predicted to lower the bandgap energy because N2p states of dopant mix with the O2p resulting in the narrow bandgap. Moreover, TiO2 oxygen sites replaced by nitrogen form isolated impurity energy levels above valence band, which will also narrow the bandgap. Strongly localized N2p states above the valence band (isolated empty states) incline to trap the photogenerated electrons, thus decreasing the photocurrent [68].
3.1.2. Dye Degradation by Zinc Oxides
In addition to TiO2, Zinc oxide is a promising adsorbent for wastewater dye degradation owing to its low cost, non-toxicity and tunable morphological features. Daneshvar et al. reported a suitable alternative, ZnO2 with similar photocatalysis mechanism as that of TiO2 [69]. Notably, tunable morphology of ZnO also played crucial role in wastewater remediation. The ZnO hierarchical mesoporous microspheres displayed remarkably high adsorption capacity (334 mg g−1) for Congo red dye in comparison with the commercially available ZnO powder [70]. Hollow microsphere of ZnO-ZIF-8 [Zn(MeIM2)] has also been explored for dye adsorption and reported with further enhanced efficiency by the core-shell structure of the resulting composite [71]. Recently, green synthesis of ZnO has attracted extensive attention as a feasible, highly efficient and eco-friendly protocol to eliminate the environmental impact of commonly utilized synthesis methods in laboratory. The Panos extract mediated quaker ladies type ZnO nano flower (ZnO/QNF) degraded the 15 mg L−1 MB, Eosin Y (EY) and Malachite green (MG) to >99% within 80, 90 and 110 min, respectively [72]. ZnO nanoparticles synthesized using the Calliandra hematocephala leaf extract shows 88% MB degradation, under solar radiation in 270 min [73], while ZnO nanoparticles obtained using Quince seed Mucilage as stabilizing agent displays 80% degradation of MB within 2 h [74]. Furthermore, ZnO nanoparticles synthesized via extracellular culture filtrates of Aspergillus niger degraded the Bismarck brown dye with 90% efficiency [75]. Additionally, Cr-doped ZnO with different amount of Cr3+ were reported for the removing anionic dyes by enhancing the surface defects on ZnO nanoparticles. This correspondingly resulted in formation of hydroxyl groups for the protonation leading to positive charge generation. This positively charged ZnO nanoparticles undergo strong electrostatic interaction and selectively eliminate the anionic dyes driven by photocatalysis [76].
3.1.3. Dye Degradation by Iron Oxides
Extensive availability of iron oxide is further reported with large photocatalytic activity. Advantageously, their recovery from the system owing to the inherent magnetic effect endow a promising adsorbent alternative. Photocatalytic degradation features of iron oxides (Fe+3) in different forms such as α-Fe2O3, γ-Fe2O3, α-FeOOH, β-FeOOH, and γ-FeOOH were explored for the visible range dye degradation. Further, azo dyes are demineralized by iron oxide powder, resulting in substituted benzene and naphthalene as degraded products [77]. Optimized crystallite size (35, 100, 150 nm) of the iron oxide nanoparticles were also demonstrated to successfully degrade Congo red. Notably, the Congo red was degraded in presence and absence of visible light using the 35 nm nanoparticles with no distinct effect on the photocatalytic decomposition [78]. The addition of Zn2+ and Al3+ to the hydrothermal reaction system resulted in the formation of iron oxides with cubic and disc structures, respectively, which successfully degraded Rhodamine B (RB) in the presence of 30% H2O2 solution. The photodegradation processes amplified significantly with increasing amount of H2O2 which facilitate diffusion onto the iron oxide surface enhancing the generation of reactive •OH [79]. Additionally, Shenoy et al. studied the effect of morphology-dependent surface charges of iron oxide for the visible light photocatalytic degradation of MB. The nanorods morphology with high negative surface charges (−20.8 mV) showed ~86% MB degradation, which outperformed other morphologies like hexagonal, oval and agglomerated nanoparticles [80]. Furthermore, iron oxide nanoparticles synthesized via various green routes also demonstrated high degradation efficiency. Teucrium polium leaf extract synthesized iron oxide nanoparticles (IONPs) degraded the MO dye with 73.6% efficiency in a 6 h reaction [81], while, magnetic α-Fe2O3 nanoparticles synthesized using Cynometra ramiflora fruit extract demonstrated 94% degradation of MB in 110 min, under sunlight irradiation [82]. Iron oxide nanoparticles synthesized using pomegranate (Punica granatum) seeds extract also possesses 95% reactive blue dye degradation under UV light irradiation for 56 min [83]. However, rod-like Fe2O3 magnetic nanoparticles synthesized using the Peltophorum pterocarpum leaf extract could achieve 92% MB degradation in minimum 27 min. This rapid degradation has been related to the “electron relay process” in which BH4− ions generated from NaBH4 transfer electrons to Fe2O3 nanoparticles generating a negatively charged environment to degrade the MB dye to harmless products [84]. Such green synthesized nanoparticles can potentially serve as an efficient catalyst for environmental remediation. Different metal doping in iron oxide alters the recombination process of charge carriers and augments the photocatalytic activity. Different Au loadings in Au-Fe2O3 aerogels were utilized as photocatalysts for mineralization of Disperse Blue 79 dye. This correspondingly promoted the accumulation of the photogenerated electrons and delayed charge carrier recombination, improving the efficiency [85].
3.1.4. Dye Degradation by Other Oxides
Various photocatalysts like CdS, GaAs and PbS are not stable since they undergo photocorrosion besides their toxicity [36,86]. Additionally, the low charge separation efficiency of photoexcited carriers in TiO2 impeded its applicability [87]. So, several other TMOs involving MnO2, CuO2, Ag2O modified MoS2 etc. are explored as photocatalyst. MnO2 nanosheets modified with fiber glass was developed by Zhang et al. [88] and explored for mineralization of MB at different pH with efficiency ˃ 95%. Pandit et al. reported colloidal MnO2 catalyzed degradation of methyl red and MO dye with high efficiency [89]. Besides colloidal form, mesoporous MnO2 also exhibits superb performance for MB mineralization in H2O2 at neutral pH and it can be recycled with tiny decrement of catalytic activity at room temperature [90].
Dyes are also used as sensitizers in the visible region including erythrosine B, thionine, rhodamine and ruthenium trisbipyridine [91,92,93,94]. Dye sensitization process involves indirect electron injection from the excited dye rather than the photocatalyst. Firstly, dye gets excited to singlet and triplet states followed by conversion to dye radicals. Electrons are then injected to the photocatalyst conduction band and interact with the adsorbed oxygen [95]. In addition to this, photocatalytic activity can be enhanced by coupling two semiconductors with different bandgaps providing more efficient separation of charged carriers and extended light absorption range [96,97,98]. Ternary nanocomposite Cu2O-BiVO4-WO3 has also been explored and showed augmented photocatalytic activity compared to their pristine components due to better charge separation [99].
Ag2O nanorods modified MoS2 microflowers (Ag2O/MoS2) based heterostructures exhibited ten times larger photocatalytic efficiency. This is ascribed to consequent morphology and enhanced charge separation efficiency resulting from synergistic effects [100].
Effect of different oxidants like peroxymonosulphate (PMS), peroxydisulphate (PDS) and hydrogen peroxide (H2O2) on photocatalysis had been further explored by utilizing different adsorbents [38]. Peroxydisulphate (PDS) showed the off-putting effect on MB demineralization, whereas H2O2 and peroxymonosulphate (PMS) enhances the degradation rate.
3.2. Bimetallic Transition Metal Oxides as Photocatalyst
Visible light-driven photocatalyst are the ideal alternative as solar energy constitutes 43% of the visible light. Monometallic oxides, their reduced counterparts or noble metal doped derivatives have been utilized in MB degradation under visible light, however, their efficiency is impeded by light absorption characteristics [101,102,103,104]. This can be made in our favor by tailoring the band gap of the resulting composite, which will alter the composite’s light absorption feature. Hence, bandgap engineering is a vital requirement for high efficiency of the adsorbent in photo dye degradation. Doping with other transition metal oxide is a typical approach resulting in energy states’ generation between valence and conduction band for electronic transitions due to photoexcitation [105]. Mahmood et al. reported that optimum doping of Mn+2 in ZnO enhances its photocatalytic activity by narrowing its bandgap [106]. Further, Deka et al. reported the tuning of bandgap of ZnO with Mn content (Zn1−xMnxO with x = 0.02, 0.05) which resulted in varying performance [107]. The photocatalyst’s effective performance also depends on various other parameters say pH, dye concentration, light intensity etc. which have been explored by investigating the influence of these factors on the rate of photoactive dye degradation utilizing CeCr2O5 as photocatalyst [108]. With the increase in dye concentration, the photocatalytic rate first increases and then decreases. This phenomenon arises because more dye molecules are available for degradation with the increase in dye concentration, consequently enhancing the rate. After a certain dye concentration, rate declines. This is attributed to the dye’s filtering action where it acts as filter for incident light, inhibiting the desired light intensity to approach the semiconductor surface.
CeO2 acts as a promoter for Fe2O3/γ-Al2O3 has been explored by Liu et al. [109] for photocatalytic azo dye degradation. They concluded that doping of CeO2 results in efficient dispersion and smaller particle size of Fe2O3 presenting more active sites. Further, CeO2 presence increases the chemisorption of oxygen, thereby improving the redox properties of the photocatalyst. CeO2 is also doped in ZnO by Saravanan et al., and the resulting composite is found to exhibit improved photodegradation rate as compared to ZnO [110]. CeO2 nanoparticles, owing to their lower bandgap, show enhanced photoactivity of the composite on visible light irradiation. This enhancement on visible illumination is attributed to CeO2 nanoparticles, which capture the electrons with hydroxyl radical formation from the holes and, hence, prevent the electron/hole recombination. In addition, the composite shows efficient dispersion of ceria crystallites on ZnO [111,112] with higher surface area, presenting more and more active sites for the redox reactions. Magesh et al. [113] found that redshift is observed for TiO2 modified with Ce compared to TiO2 in UV-vis spectrophotometry and optimal Ce loading at 0.5–2.0% in the mixed oxide CeO2–TiO2 proved active under visible light for the MB photocatalytic decomposition. Photocatalytic activity of CuO/CeO2 was also explored and reported to exhibit high performance owing to redox behavior of CeO2 and its strong interaction between CuO and CeO2 [114]. Irradiation by different light on the photocatalyst tailors the photodriven catalytic performance. Tung et al. have reported that under UV light irradiation, iron-doped TiO2 reveal consideration increment in photoactivity than that of pure TiO2 or Iron oxide [115]. This enhancement may be ascribed to impedance of electron/hole recombination with the incorporation of Fe ion in TiO2 lattice. On the other hand, photoactive performance of the catalyst transforms under visible light state. It illustrates that pure iron (II, III) oxide exhibits excellent photoactive behavior which surpasses that of TiO2. Iron doping at 0.5 mol in TiO2 made it comparable and for 1 mol, no increment was observed. Hence, iron-doped TiO2 (photocatalyst) reacted differently under UV and visible light with no adverse effect on photoactivity in the former case. The formation of recombination sites was observed via iron doping in TiO2 under visible light, thereby reducing photodriven catalysis. Zhu et al. also supported the above finding suggesting that the rate of recombination was decreased by utilizing iron ions as hole or electron snare in TiO2 [116]. Photocatalytic studies of MnO2/Fe3O4 nanocomposite show high photocatalytic efficiency and high stability with recyclability towards the degradation of MB under UV-vis illumination [117].
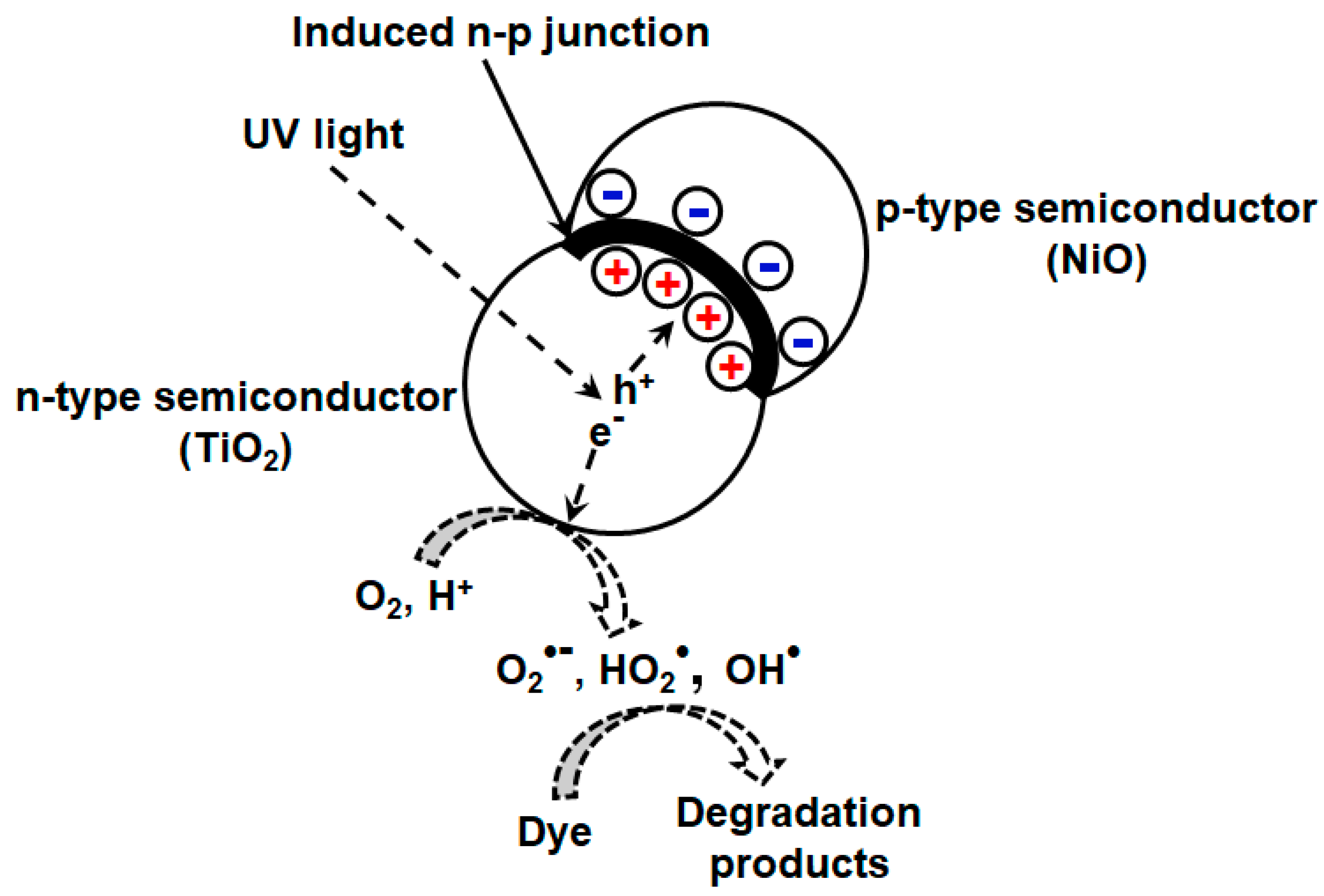
Different strategies have been reported to enhance the photocatalytic efficiency or retard the electron/hole recombination rate in mixed oxides. TiO2-NiO mixed oxide developed by sol gel method reportedly have high efficiency for photodriven MO removal [118]. This enhancement is attributed to the induced n-p junction at the interface, which prevents electron/hole recombination upon irradiation by light. This can be explained by (Figure 5) considering the existence of both TiO2 (n-type, electron rich) and NiO (p-type, hole rich) phases or n-p junction at the interfacial contact where diffusion of electrons and holes takes place to p-type and n-type semiconductors, respectively. At equilibrium, the internal electric field arises due to accumulation of electrons and holes, which act as potential barrier and prevent electron/hole recombination. They also investigated the variation of nickel content on catalytic efficiency of the synthesized composite. Highest efficiency was exhibited by Ti75–Ni25 composite, and with further increment in Ni content, specific surface area and total pore volume of the resulting composite diminishes, which is detrimental for photocatalytic reactions.
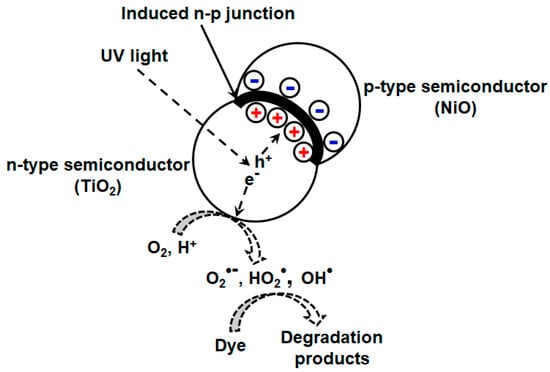
Figure 5.
Schematic showing p-n junction at the interface of p-type NiO and n-type TiO2 semiconductors for improved photocatalytic degradation of MO.
Sreethawong et al. loaded noble metal on the mixed oxides to enhance their photocatalytic rate [119]. Mesoporous assembled TiO2–SiO2 mixed oxides loaded with monometallic Pt and bimetallic Pt–Au were studied as adsorbents under visible light driven catalysis for different loadings of Au and Pt. Similarly, Fe was introduced to ZnO/SiO2 to improve their catalytic performance by shifting the absorption bandgap threshold from UV towards visible region, thereby modifying surface physical and chemical properties [120]. Various other factors say dye solution pH, initial dye concentration, illumination time, and catalyst loading effecting photo-mediated oxidation of MB were investigated and explored for the photocatalytic process’s optimal conditions. The efficiency of photocatalysis decreases with increase in pH with a value of ~99.9% when pH increases from 2 to 4. However, for negatively charged MB in acidic medium and positively charged ZnO below pH 9, increase in pH change the charge on ZnO by absorbing OH− ions favoring the formation of •OH radicals resulting in diminished photoactive efficiency. This is due to gradual increase in electrostatic repulsion between anionic dye and ZnO. Additionally, catalyst loading also has considerable effect on photoactivity of Fe/ZnO-SiO2. When loading increased above 0.1 g, efficiency decreases owing to agglomeration of catalyst particles as a consequence of increase in particle size and decrease in surface area, diminishing the number of surface-active sites. Further, the effect of different loading of MnO2 over TiO2 is explored by Xue et al. and reported to have high surface area and large pore size with enhanced adsorption properties over whole region of visible light [121]. The enlargement of the pore size is explored and attributed to the coating mechanism where dopant Mn+2 is coated onto the host in the synthesis procedure. During the synthesis, the formation of TiO2 nanoclusters takes place with the oxidation of Mn+2 to MnO2 in air which resist TiO2 nanoclusters to join together. The clusters were thus prevented to come close to form a small pore, subsequently affected the crystallization of TiO2. This led to remarkable absorption of visible light due to narrow band gap of MnO2. Wang et al. reported a SnO2–TiO2 photocatalyst based on TiO2 nanofibers by electrospinning followed by hydrothermal treatment [122]. Under UV light illumination, SnO2–TiO2 composite exhibits almost 2.5 times higher than TiO2 photoactivity for RB mineralization. Li et al. synthesized MnO2 loaded CuO nanosheets and reported to possess high catalytic activity due to its composite phases, large surface area and dispersibility [123]. Series of CuO/MnO2 compounds with different CuO loadings was synthesized and demonstrate the highly efficient CuO/MnO2 interfaces as active sites for photocatalysis [124]. MnO2 layer is also coated on CuO to form nanorod array which addresses the issues related with the powder catalysis say easy agglomeration, difficult separation and possible secondary pollution [125]. In spite of this, metal vanadates are also widely utilized for photocatalysis. Recently, bismuth vanadate, BiVO4 with layered structure and bandgap 2.4 eV was reported with varying molar ratio of Bi to V [126]. By alteration in the molar ratio, different crystal structures were obtained, tetragonal scheelite (ts), monoclinic scheelite (ms) and tetragonal zircon (tz). Amongst them, a higher photocatalytic activity under UV light has been exhibited by ms-BiVO4, despite the consequences of its morphology with different dyes say RB and MB. Moreover, it demonstrates the potential of ms-BiVO4 under 6h visible light illumination with degradation efficiency of 65.0% for MB and ~77.8% for RB. Shang et al. [127] also reported nanosized BiVO4 (dia 60 nm) with visible light-driven photocatalytic activity. They suggested that nano BiVO4 possessed excellent photocatalytic efficiency towards the degradation of RB in 40 min. Furthermore, Yin et al. [128] synthesized ms-BiVO4 by using cetyltrimethylammonium bromide (CTAB) and reported that CTAB transformed the mechanism of reaction with the formation of an intermediate BiOBr. Besides high photocatalytic efficiency, no considerable loss was witnessed after five cycles, signifying the high stability and absence of photocorrosion during RB degradation by ms-BiVO4.
Despite metal vanadates, different metal molybdenum oxides were reported as efficient photocatalysts for the treatment of wastewater using UV or visible light illumination [129,130,131]. Xu et al. demonstrated high photocatalytic degradation activity of 90% under visible light irradiation for MO treatment using zeolite supported Bi2MoO6 [131]. This enhanced performance is attributed to uniform distribution of Bi2MoO6 nanoparticles on zeolite. ZrMo2O8 was reported by solid state technique and possess ZrO8 and Mo2O8 polyhedra [132]. It has specific degradation properties towards cationic dyes, say malachite green (MG) and RB.
Various other bimetallic oxides such as NaTaO3, CoTaO6 and CeTaO4 were supposed to have extended conduction and valence band, increasing the lifetime of photogeneration transporters, promoting the redox reaction on the catalyst surface [133,134,135,136]. Application of Fe-Co mixed oxide with varying Fe/Co ratio for degradation of 2, 4-dichlorophenol was explored and proved effective than bulk Co3O4 [137]. Authors also examined CoxFe3−xO4 nanoparticles for RB heterogeneous degradation and revealed that higher cobalt content results inefficient removal of the dye. Another bimetallic oxide NaBiO3 examined under visible light irradiation for RB degradation via direct cleavage of the conjugated chromophore of dye molecule along with the degradation as dominating mechanism [138].
Besides this positive effect of bimetallic oxide interaction, the photocatalytic efficiency can also be poisoned via metal-metal ion interaction. Influence of different crystallographic forms of MnO2 on Degussa P-25 TiO2 resulted in photocatalytic inhibition or complete deactivation depending upon the amount of MnO2 [139]. This is due to the heterojunction formation at MnO2–TiO2 interface which alter the chemical state of Ti+4 and O−2 sites in TiO2. MnO2 also widen the bandgap of TiO2, thereby diminishing the UV absorption by creating impurity energy levels which serve as a recombination center for photoexcited charged carriers.
Among various mixed oxides formed via doping of foreign transition metal ion in host transition metal oxides, spinel oxides form a special class which have narrower bandgap with efficient catalytic properties. Spinel oxides synthesized by different methods possess high electrochemical activity with facilitated charge transfer [140,141,142,143,144,145]. Nano iron–cobalt spinel oxide was explored for photocatalytic activity via mineralization of monoazo-dye Acid Orange 7 with sulfate radicals in aqueous solution [146]. Quenching studies established that sulfate radicals are the primary species produced in the dye’s catalytic degradation, which enhances the efficiency of the photocatalyst. Further, 3D magnetic ZnFe2O4/MnO2 hybrid was explored for photocatalysis and found to possess better activity (owing to its high BET surface area) with easy recyclability from reaction solution [147]. Cobalt ferrite, CoFe2O4 was synthesized by co-precipitation on the TiO2 surface to achieve recyclability with efficient photocatalytic ability which is estimated by following the degradation kinetics of textile dye in absence and presence of different electron acceptors [148]. It displayed maximum increase in the decomposition of Reactive red 120 (dye) in peroxomonosulphate (PMS) as compared to other electron acceptors. Ordinary NiCo2O4 and their core ring structured nanoplatelets were developed by Cui et al., which reports that the core ring NiCo2O4 nanoplatelets display elevated photocatalytic activity MB demineralization under visible light [149]. This enhanced property is attributed to smaller size, high absorption capacity with active and efficient internal electronic transitions. In addition, the core ring structure provides larger surface area and active surface Co atoms which could be the primary reason for outstanding photoactivity. Various other composites of doped transition metal oxides or bimetallic oxides with improved photocatalytic activity are included in Table 1 [132,150,151,152,153,154,155,156,157,158,159,160,161].

Table 1.
Photocatalytic performance of bimetallic transition metal oxides. N.A. = data not available.
3.3. Carbon Based Composites as Photocatalyst
Another approach is to enhance the adsorption capacity with larger electron/hole recombination lifetime. This can be achieved by supporting the transition metal oxides over co-adsorbent (support) which should have high adsorption capacity. The co-adsorbent should have high surface area and network conductivity for efficient adsorption and facilitated charge transfer processes, respectively. Carbonaceous materials such as activated carbons, carbon nanotubes and graphene are interesting and potential material for co-adsorbents owing to their higher surface area, unique pore structure and high conductivity [162,163,164,165,166,167,168]. Predominantly, the functions of co-adsorbent or support are as follows:
- Reduce agglomeration of the particles
- Enhance surface area of the resulting composite
- Govern the life span of the photocatalyst via reducing the recombination rate of charged carriers
Activated carbon–TiO2 composites have been reported to exhibit improvement in photocatalytic activity attributed to the high adsorption capacity of the support and ready passage of charged carriers to TiO2 particles [169,170,171,172,173]. However, under-exploitation of nanotexture and nanoscale phenomena with untackled bandgap engineering needs researchers to explore other alternatives [174,175]. Carbon nanotubes (CNTs) are reported to possess effective synergism with TiO2 to enhance photocatalysis by diminishing the electron/hole recombination [176,177,178]. Hoffmann et al. reported TiO2–CNT composite with transference of photogenerated electrons to CNTs and holes to TiO2, thus impeding the reblending of electrons and holes [179]. MWCNT TiO2 composites have been extensively reported to improve their photocatalytic activity [180,181]. Magnetic photocatalyst, NiFe2O4/MWNT was synthesized by hydrothermal method and exhibited high photoactivity [182]. The poorly active NiFe2O4 nanocrystals are significantly converted into active one in the presence of MWNT, and this is attributed to synergism between the two with effective charge transfer and declined rate of recombination of photogenerated carriers. In addition to this, their remarkable magnetic property made it efficiently recyclable using an external magnet.
Among various carbonaceous material, graphene, two-dimensional sheets of sp2 hybridized carbon, possess high charge carrier mobility with excellent mechanical strength [183]. Graphene can tailor the particle size and microstructure of the guest material and promote electron transfer in photocatalytic reactions [184]. Direct growth of transition metal salt on graphene oxide (GO) has been widely used for graphene/metal oxide composites [185]. In spite of this, various other graphene-based composites are in-situ synthesis and hydrothermal or solvothermal approach.
3.3.1. In-Situ Synthesis
During this, metal salts and graphene oxide (GO) are used as substrates. The salt is then added to GO (dispersed solution) followed by conversion to metal oxide in the oxidizing agent’s presence, followed by the reduction of GO to graphene [141,142,143,167]. Graphene–ZnO composite was synthesized by adsorbing Zn+2 ions on graphene oxide sheets in the presence of NaOH and NaBF4 followed by drying at 150 °C [186]. Similarly, SnO2 and TiO2 were grown over graphene by taking SnCl2 and TiCl3 as substrates [187]. Different morphological structures were obtained by reducing Sn and Ti salts in presence of GO by the same method. This can be attributed to different hydrolysis rate of the salt with their reduction abilities. Researchers have utilized different methods for dispersing graphene oxide while synthesizing different graphene-based composites via in-situ method [188,189]. Anionic surfactants have also been used for stabilizing graphene in aqueous solution for in situ growth of TiO2 over graphene [190]. Whereas, Park et al. reported graphite TiO2 composite via exfoliation–reorganization of graphite oxide without using any chemical reagents in aqueous slurry [191].
3.3.2. Hydrothermal Approach
During hydrothermal growth, precursors (semiconductor oxide) are loaded on graphene oxide in a dispersed medium followed by a higher temperature reduction. ZnFe2O4/graphene composite was synthesized by the same method with Zn(NO3)2.6H2O and Fe(NO3)2.9H2O as precursors [192]. During growth, metal salts are oxidized with a reduction of graphene oxide. In-situ hydrothermal growth was utilized for graphene-BiWO6 by Mukherji et al. in the presence of graphene oxide [193]. Shen et al. synthesized graphene TiO2 composite via hydrothermal growth with scalable methods by utilizing water and glucose (reducing agent) [194]. Graphene TiO2 composite was dispersed by controlling the hydrolysis via one-step solvothermal approach to achieve a uniformly dispersed metal oxide over graphene [37].
3.3.3. Photocatalytic Activity
Graphene TiO2 nanocomposite, obtained via chemical oxidation of graphene suggested “on demand” graphene–metal oxide mechanism [195]. Functionalized GO nanosheets have efficient binding sites for metal oxide particles (TiO2, ZnO2) and remain in solution without aggregation [196]. After photo-irradiation, the electrons interact with GO nanosheets to remove various oxygen functionalities yielding reduced graphene oxide (RGO) [197,198]. Some electrons are used for reduction purpose, and others are stored in RGO. The graphene TiO2 can be recovered after solvent evaporation. Photocatalytic degradation of MB using TiO2 nanorods GO composite exhibits anticharge recombination mechanism [199]. Authors found that the electrons at GO react with adsorbed oxygen to form •OH radicals reducing the recombination of charge carriers (e−/h+). Further, graphene presence in the composite influences the morphology and particle size distribution in the composite and accordingly alters the photocatalytic performance. Zhou et al. showed the uniform dispersion of TiO2 nanoparticles on graphene sheets with narrow particle size distribution resulting in superior photocatalytic activity under simulated sunlight irradiation [200]. The electron/hole pairs are produced in TiO2 upon illumination with UV light and the photoexcited electrons transfer to graphene sheets, scavenged by dissolved oxygen and promote the electron/hole separation. Additionally, Du et al. demonstrated the improved photocatalysis by ordered macro-mesoporous TiO2–graphene composite towards decomposition of MB in water than bare mesoporous TiO2 film [188]. All the above results of graphene-based composites displayed that graphene can promote charge separation in the composites and consequently results in high photocatalytic activity.
Graphene–ZnO composite was reported by adsorbing Zn+2 ions on GO sheets followed by addition of NaOH and NaBH4 [186]. Various graphene composites (ZnO–graphene, Co3O4–graphene, Mn3O4–graphene, etc.) have been explored for their photocatalytic application to purify wastewater [186,201,202]. These composites showed high adsorption capacity and large charge separation facilitating overall performance. The significant role of graphene in the graphene TiO2 composite is demonstrated by Zhang et al. [203] during the degradation of MB in water (Figure 6). Role of graphene and CNT were further explored by making their nanocomposites CuO.ZnO.Fe2O3/rGO and CuO.ZnO.Fe2O3/CNTs by utilizing against MB dye [204]. The resulting nanocomposites engendered stress in their lattice, resulting in some defects. These consequently resulted in enhanced chemical reactivity with improved efficiency owing to the newly generated electronic state resulting from varied interactions such as metal–metal, metal–oxygen–metal.
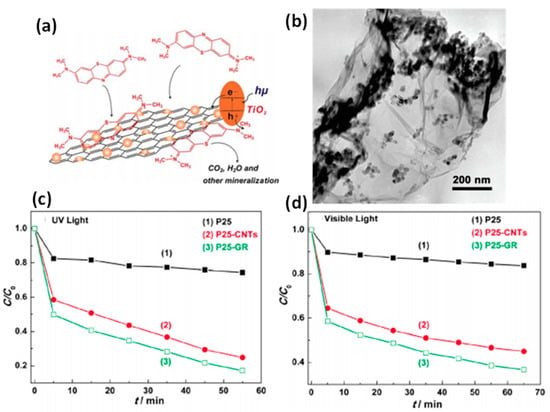
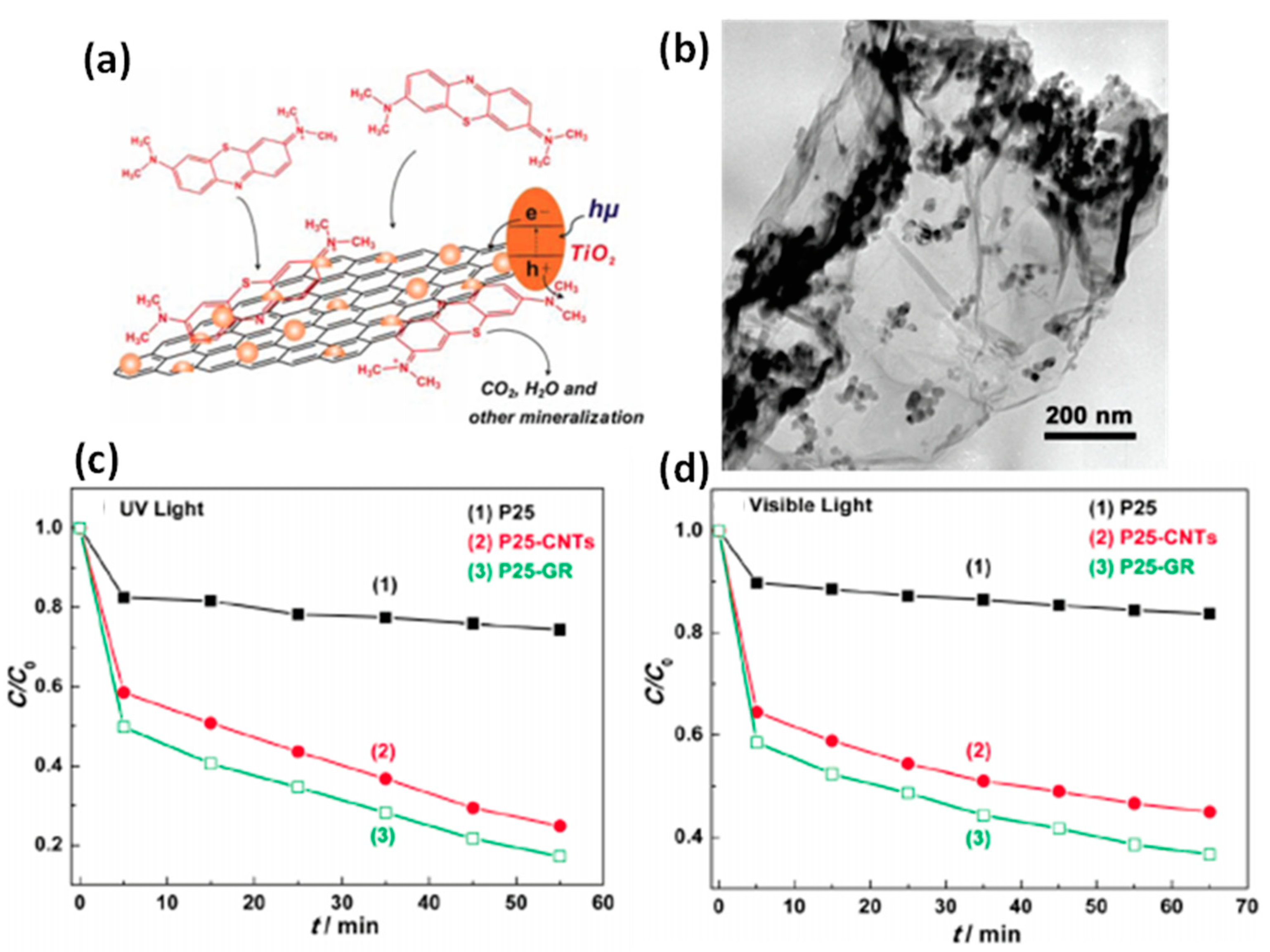
Figure 6.
(a) Schematic of P25-GR and probable process of photodriven degradation of the dye (MB) over P-25-GR with TEM micrograph (b) of the composite. Comparison of photocatalytic activity in the degradation of MB under (c) UV light (λ = 365 nm) and (d) visible light (λ = 400 nm). Adapted with permission form Ref. [203]. Copyright 2010, American Chemical Society.
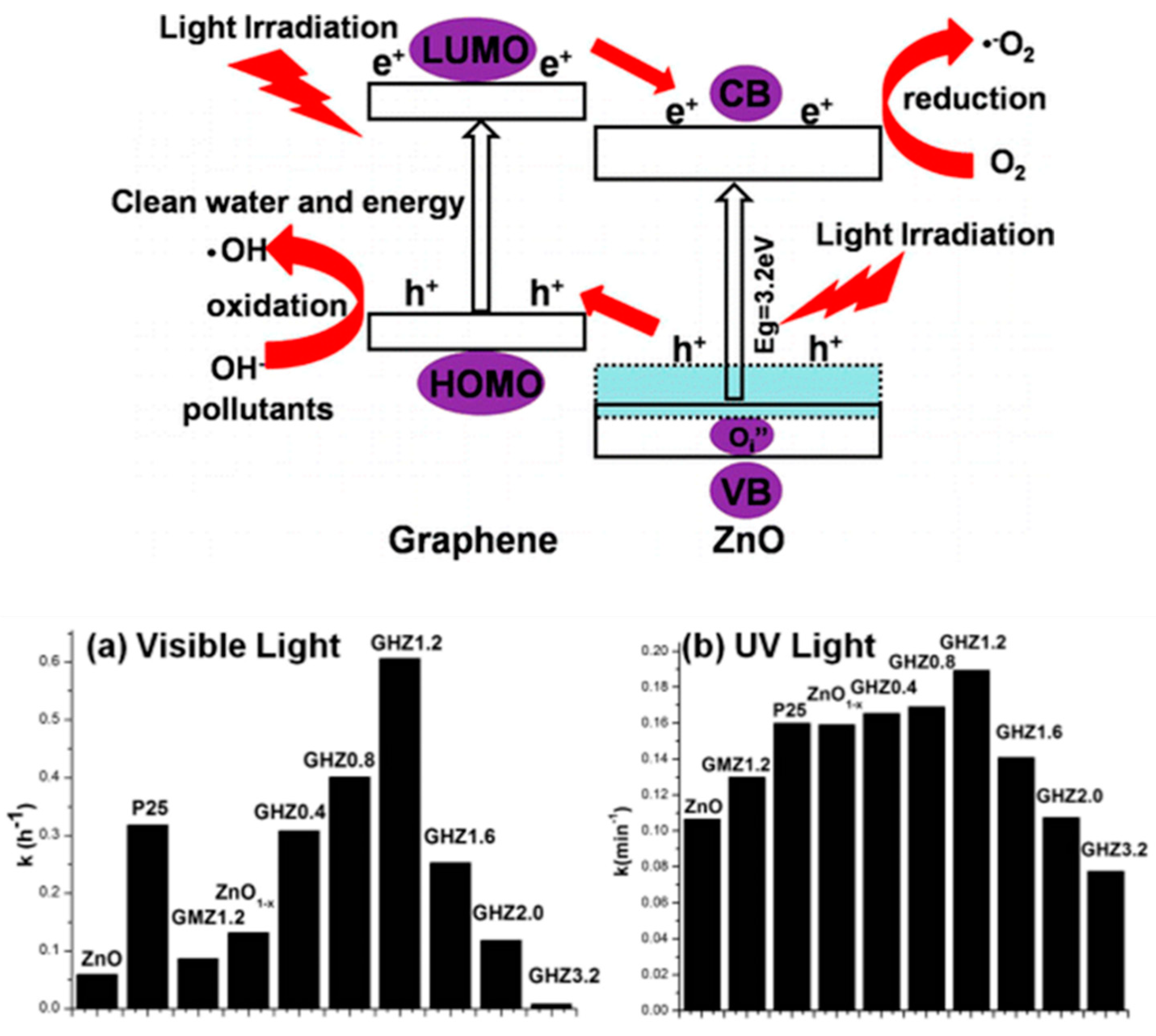
In addition, the graphene content in the composites significantly influences the photocatalytic efficiency. The effect of composites with different graphene content has been studied via photocatalytic degradation of MB in water [205]. Bai et al. utilized Zn1−x/graphene composite and provide insights for the development of graphene-based defect type material [206]. Superior photocatalytic activity of the synthesized composite corresponds to synergic effect of graphene and Zn1−x surface defect layer. The electron/hole separation results due to the elevation of highest occupied molecular orbital (HOMO) of graphene and Oi″ ZnO1−x defect level in Zn1−x/graphene composite (Figure 7). Non-stoichiometric W18O49 has been reported as a potential visible-driven photocatalyst due to its unusual defective structure [207].
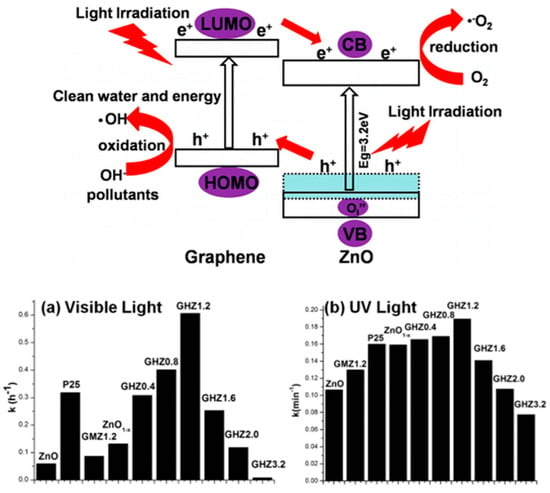
Figure 7.
Schematic demonstrating the photocatalytic process over ZnO1−x/graphene. Photocatalytic degradation of MB over ZnO1−x and ZnO1−x/graphene under illumination of (a) visible light and (b) UV light. Adapted with permission from [206]. Copyright 2013, American Chemical Society.
Du et al. further developed an approach to improve the charge carriers’ diffusivity by employing hierarchical porous systems [188]. Macro-mesoporous titania graphene composite reduces the mesopore channel’s diffusion length and enhances the effective surface area, resulting in diminished charge recombination rate. High photocatalytic efficiency of MnO2 graphene composite is attributed to synergism between graphene and MnO2 via efficient electron transfer channels with the high surface area for photoactive mineralization of MB [208]. Here, the adsorption–oxidation–desorption mechanism is involved for MB degradation. Firstly, MnO2 are adsorbed on graphene surface via π-π conjugation, which effectively increases the MB concentration near the surface, enhancing the catalytic rate. Therefore, the coupling between adsorption and catalytic reaction for MnO2/RGO composite results in efficient decomposition of MB compared with bare components. ZrO2–graphene composites are also analyzed for photocatalysis and observed that degradation time decreases with enhanced degradation factor due to the inclusion of graphene in ZrO2 [209]. This is attributed to the immediate shifting of electrons to graphene followed by the generation of electrons and holes on irradiation. The conjugated (sp2) carbon lattice increases light absorption range thereby promoting charge separation. In allusion to this, various other composites of graphene and transition metal oxides resulted with improved photocatalytic activity are presented in Table 2 [210,211,212,213,214,215,216,217,218,219,220].

Table 2.
Photocatalytic properties of graphene-based composites (photocatalysts).
Besides graphene-based composites of mono transition metal oxides, their bimetallic composites have also been extensively explored with the benefits of visible light driven photocatalysis. As the major portion of solar energy falls under visible light, it also improves the composite’s efficiency [221]. Enhanced diffusion coefficient associated with graphene-based spinel oxides composite may prove beneficial for photocatalysis application [141,143]. Fu and Wang et al. reported visible light-driven MB degradation by ZnFe2O4 graphene composite [192]. Here, the magnetic property of ZnFe2O4 makes the composite magnetically discrete from the suspension system negating the requirement of additional magnetic components. Visible light irradiation of ZnFe2O4 graphene composite results in effective charge separation and generates strong oxidants via photoactive degradation of H2O2 with enhanced stability. Enhanced charge transfer of the composite is also demonstrated by a small radius of the semicircle in high frequency region of impedance plot compared to that of bare GO and ZnFe2O4 (Figure 8). Further, MnFe2O4–graphene sand composite was used as photocatalyst against 100% MB degradation under sunlight irradiation [220]. The high performance resulted from simultaneous participation of Mn and Fe on the catalyst surface for the decomposing H2O2 and in the production of •OH favoring photogenerated carriers’ separation in MnFe2O4. Hydrothermal growth of graphene–BiWO6, reported by Gao et al. [222], shows enhanced photocatalytic degradation activity under RB’s visible light. The interaction of graphene with BiWO6 shifts the Fermi level and decrease the conduction band potential. Fu et al. reported the synergistic effect of CuFe2O4 nanoflakes and graphene sheets, resulting in superior photocatalytic performance. The possible mechanism of photocatalytic degradation takes place via the following steps:
CuFe2O4 + hν → CuFe2O4 (h+ + e−)
CuFe2O4 (e−) + graphene → CuFe2O4 + graphene (e−)
Graphene (e−) + O2 → O2●− + graphene
CuFe2O4 (h+) + OH− → CuFe2O4 + ●OH
CuFe2O4 (h+) + OH + O2●− + MB → CO2 + H2O
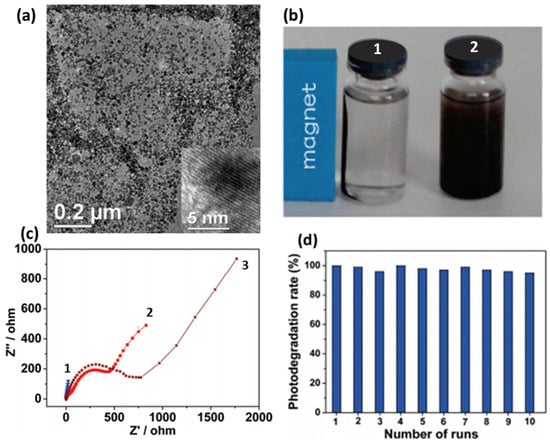
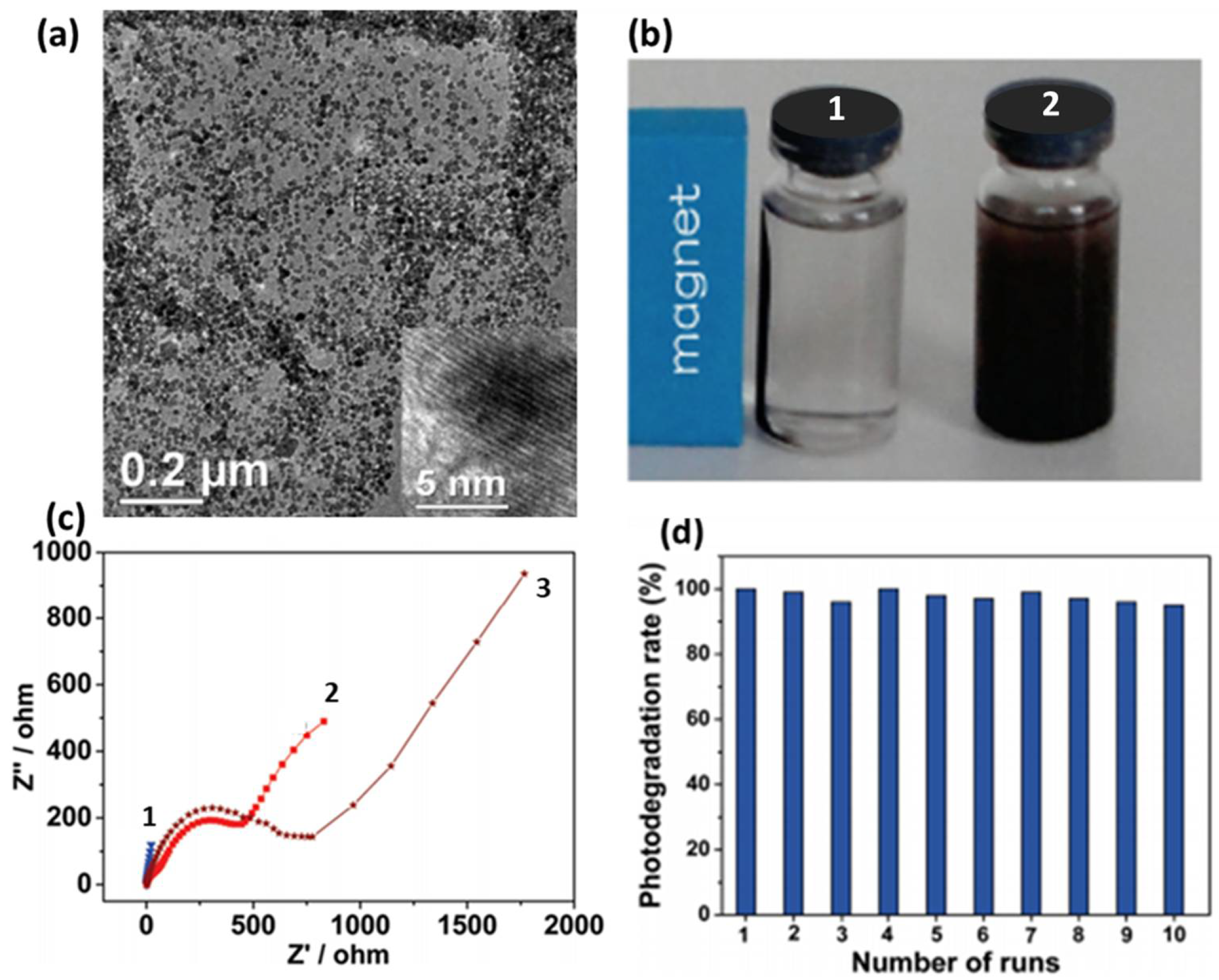
Figure 8.
(a) Uniform dispersion of ZnFe2O4 nanoparticles over graphene (b) Images of ZnFe2O4 graphene suspension with (1) and without (2) magnetic field, (c) EIS of (1) ZnFe2O4 graphene, (2) pure ZnFe2O4 and (3) GO and (d) stability test shown by photodegradation rate of MB using ZnFe2O4-G for 10 cycles. Adapted with permission from [192]. Copyright 2011, American Chemical Society.
Here, the generated active species oxidize MB molecules which are adsorbed on CuFe2O4 graphene composite via π-π or electrostatic interaction. CuBi2O4/G was also evaluated for their photocatalytic performance after their synthesis by solvothermal method [223]. They possess 95% and 85% degradation efficiency towards degradation of MB and MO compared to pristine CuBi2O4. Visible light irradiation of CuBi2O4 result in the generation of electrons and holes on the surface followed by transference of photoexcited electrons to graphene sheets by percolation mechanism. Then, negatively charged graphene activates oxygen to form superoxide anion radicals and holes, which are adsorbed by water to produce hydroxyl radicals. Active species generated by graphene then oxidizes the dye molecule (adsorbed on the CuBi2O4–graphene active sites) and hence, increases the photocatalytic degradation rate of dyes from industrial effluents. These new adsorbents ensure visible light-driven dye degradation with large adsorption capacity and electron/hole recombination lifetime.
4. Conclusions and Perspectives
Photocatalysis is an imperative route for greener decipher of energy and environmental issues of modern era. Catalytic efficiency of various photocatalysts has been altered by employing different techniques by the researchers. This review has given stress over different adsorbents based on transition metal oxides, bimetallic oxides and their carbon composites for photocatalysis. Different approaches regarding synthetic technique, heterostructure, etc. led to augmented adsorptivity, light absorption intensity, electron/hole pair lifetime with extended light adsorption range which may unlock various breaks for the next generation photocatalysts. These approaches have been proved good but no one proved a panacea. However, the mechanistic description is still lacking and vigilant designs of the functional materials are vital for rational progress. It is worthy to note that although great improvements have been accomplished by utilizing the “rising star: graphene” in photocatalysis, still there are some challenges for full exploitation of graphene-based composites.
In addition to the above tactics to enhance photocatalysis, the deficiency in analogous results among various studies of photocatalysis led to a great challenge, in spite of a large amount of literature. Recently, the progress has been made on the utilization of nanocarbons for existing photocatalyst which improves the photocatalytic activity in a broad spectrum. Further, the photocatalysts’ development and endorsement depend on the harmonic growth of academia and industrial revolution, particularly for the optimal testing condition and methods for dye degradation. The intensive efforts from systematic amalgamation of academician research worldwide would bring revolt in developing potential photocatalysts for dye degradation.
Author Contributions
P.A. (Preety Ahuja) and S.K.U.; writing original draft preparation, S.K.U., R.K. and P.A. (Pankaj Attri); review and editing. All authors have read and agreed to the published version of the manuscript.
Funding
Preety Ahuja acknowledges NPDF (project no. PDF/2015/000470) from the Department of Science and Technology, India; Pankaj Attri is thankful to JSPS-KAKENHI grant number 20K14454.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Wang, Y.; Sun, X.; Xian, T.; Liu, G.; Yang, H. Photocatlytic degradation of simulated dye wastewater in diferent pH environments by using BaTiO3/Bi2WO6 heterojunction photocatalysts. Opt. Mat. 2021, 113, 110853. [Google Scholar] [CrossRef]
- Ning, L.; Xukai, L.; Mengting, H.; Xiaoguang, D.; Beibei, Y.; Guanyi, C.; Shaobin, W. Catalytic membrane-based oxidation-filtration systems for organic wastewater purification: A review. J. Hazard. Mater. 2021, 414, 125478. [Google Scholar]
- Jaspal, D.; Malviya, A. Composites for wastewater purification: A review. Chemosphere 2020, 246, 125788. [Google Scholar] [CrossRef]
- Chan, S.H.S.; Wu, T.Y.; Juan, J.C.; Teh, C.Y. Recent developments of metal oxide semiconductors as photocatalysts in advanced oxidation processes (AOPs) for treatment of dye wastewater. J. Chem. Technol. Biotechnol. 2011, 86, 1130–1158. [Google Scholar] [CrossRef]
- Waters, B.D. The Regulator’s View. In Colour in Dye House Effluent; Cooper, P., Ed.; The Society of Dyers and Colourists: Bradford, UK, 1995; pp. 22–30. [Google Scholar]
- Pereira, A.G.B.; Rodrigues, F.H.A.; Paulino, A.T.; Martins, A.F.; Fajardo, A.R. Recent advances on composite hydrogels designed for the remediation of dye-contaminated water and wastewater: A review. J. Clean. Prod. 2021, 284, 124703. [Google Scholar] [CrossRef]
- Banat, I.M.; Nigam, P.; Singh, D.; Marchant, R. Microbial decolorization of textile-dye-containing effluents: A review. Bioresour. Technol. 1996, 58, 217–227. [Google Scholar] [CrossRef]
- Schwarzenbach, R.P.; Escher, B.I.; Fenner, K.; Hofstettere, T.B.; Johnson, C.A.; Gunten, U.V.; Wehrli, B. The Challenge of Micropollutants in Aquatic Systems. Science 2006, 313, 1072–1077. [Google Scholar] [CrossRef] [PubMed]
- Daneshvar, N.; Ashassi-Sorkhabi, H.; Tizpar, A. Decolorization of orange II by electrocoagulation method. Sep. Purif. Technol. 2003, 31, 153–162. [Google Scholar] [CrossRef]
- Donkadokula, N.Y.; Kola, A.K.; Naz, I.; Saroj, D. A review on advanced physico-chemical and biological textile dye wastewater treatment techniques. Rev. Environ. Sci. BioTechnol. 2020, 19, 543–560. [Google Scholar] [CrossRef]
- Kishor, R.; Purchase, D.; Saratale, G.D.; Saratale, R.G.; Ferreira, L.F.R.; Bilal, M.; Chandra, R.; Bhargava, R.N. Ecotoxicological and health concerns of persistent colouring pollutantsof textile industry wastewater and treatement approaches for environmental safety. J. Environ. Chem. Eng. 2021, 9, 105012. [Google Scholar] [CrossRef]
- Kadirvelu, K.; Kavipriya, M.; Karthika, C.; Radhika, M.; Vennilamani, N.; Pattabhi, S. Utilization of various agricultural wastes for activated carbon preparation and application for the removal of dyes and metal ions from aqueous solutions. Bioresour. Technol. 2003, 87, 129–132. [Google Scholar] [CrossRef]
- Baruah, S.; Dutta, J. Nanotechnology applications in pollution sensing and degradation in agriculture: A review. Environ. Chem. Lett. 2009, 7, 191–204. [Google Scholar] [CrossRef]
- Choy, K.K.H.; McKay, G.; Porter, J.F. Resources, Sorption of acid dyes from effluents using activated carbon. Resour. Conserv. Recycl. 1999, 27, 57–71. [Google Scholar] [CrossRef]
- Crini, G. Non-conventional low-cost adsorbents for dye removal: A review. Bioresour. Technol. 2006, 97, 1061–1085. [Google Scholar] [CrossRef]
- Gupta, V.K.; Suhas. Application of low-cost adsorbents for dye removal—A review. J. Environ. Manag. 2009, 90, 2313–2342. [Google Scholar] [CrossRef]
- Carvalho, M.C.; Pereira, C.; Gonçalves, I.C.; Pinheiro, H.M.; Santos, A.R.; Lopes, A.; Ferra, M.I. Assessment of the biodegradability of a monosulfonated azo dye and aromatic amines. Int. Biodeterior. Biodegrad. 2008, 62, 96–103. [Google Scholar] [CrossRef]
- Lin, J.; Zhang, X.; Li, Z.; Lei, L. Biodegradation of Reactive blue 13 in a two-stage anaerobic/aerobic fluidized beds system with a Pseudomonas sp. isolate. Bioresour. Technol. 2010, 101, 34–40. [Google Scholar] [CrossRef] [PubMed]
- Wafaa, M.A.E.-R.; Hassan, M.; Khalafallah, M. Microflora involved in textile dye waste removal. J. Basic Microbiol. 2003, 43, 167–174. [Google Scholar]
- Ambrósio, S.T.; Campos-Takaki, G.M. Decolorization of reactive azo dyes by Cunninghamella elegans UCP 542 under co-metabolic conditions. Bioresour. Technol. 2004, 91, 69–75. [Google Scholar] [CrossRef]
- Chander, M.; Arora, D.S. Evaluation of some white-rot fungi for their potential to decolourize industrial dyes. Dyes Pigm. 2007, 72, 192–198. [Google Scholar] [CrossRef]
- Wesenberg, D.; Kyriakides, I.; Agathos, S.N. White-rot fungi and their enzymes for the treatment of industrial dye effluents. Biotechnol. Adv. 2003, 22, 161–187. [Google Scholar] [CrossRef]
- Robinson, T.; McMullan, G.; Marchant, R.; Nigam, P. Remediation of dyes in textile effluent: A critical review on current treatment technologies with a proposed alternative. Bioresour. Technol. 2001, 77, 247–255. [Google Scholar] [CrossRef]
- Lourenco, N.D.; Novais, J.M.; Pinheiro, H.M. Reactive textile dye colour removal in a sequencing batch reactor. Water Sci. Technol. 2000, 42, 321–328. [Google Scholar] [CrossRef]
- Gürses, A.; Yalçina, M.; Dogar, C. Electrocoagulation of some reactive dyes: A statistical investigation of some electrochemical variables. Waste Manag. 2002, 22, 491–499. [Google Scholar] [CrossRef]
- Hao, O.J.; Kim, H.; Chiang, P.C. Decolorization of Wastewater. Crit. Rev. Environ. Sci. Technol. 2000, 30, 449–505. [Google Scholar] [CrossRef]
- Ullah, R.; Dutta, J. Photocatalytic degradation of organic dyes with manganese-doped ZnO nanoparticles. J. Hazard. Mater. 2008, 156, 194–200. [Google Scholar] [CrossRef] [PubMed]
- Danish, M.S.S.; Estrella, L.L.; Alemaida, I.M.A.; Lisin, A.; Moiseev, N.; Ahmadi, M.; Nazari, M.; Wali, M.; Zaheb, H.; Senjyu, T. Photocatalytic Applications of Metal Oxides for Sustainable Environmental Remediation. Metals 2021, 11, 80. [Google Scholar] [CrossRef]
- Gautam, S.; Agrawal, H.; Thakur, M.; Akbari, A.; Sharda, H.; Kaur, R.; Amini, M. Metal oxides and metal organic frameworks for the photocatalytic degradation: A review. J. Environ. Chem. Eng. 2020, 8, 103726. [Google Scholar] [CrossRef]
- Zhu, J.; Jiang, Z. Electrochemical Photocatalytic Degradation of Eriochrome Black T Dye Using Synthesized TiO2@CNTs Nanofibers. Int. J. Electrochem. Sci. 2021, 16, 210318. [Google Scholar] [CrossRef]
- Sukri, S.N.A.M.; Shameli, K.; Isa, E.D.M.; Ismail, N.A. Green Synthesis of Zinc Oxide-Based Nanomaterials for Photocatalytic Studies: A Mini Review. IOP Conf. Ser. Mater. Sci. Eng. 2021, 1051, 012083. [Google Scholar] [CrossRef]
- Thiruvenkatachari, R.; Vigneswaran, S.; Moon, I.S. A review on UV/TiO2 photocatalytic oxidation process. Korean J. Chem. Eng. 2008, 25, 64–72. [Google Scholar] [CrossRef]
- Ahmed, S.; Rasul, M.G.; Martens, W.N.; Brown, R.; Hashib, M.A. Advances in heterogeneous photocatalytic degradation of phenols and dyes in wastewater: A review. Water Air Soil Pollut. 2011, 215, 3–29. [Google Scholar] [CrossRef]
- Malato, S.; Fernández-Ibáñez, P.; Maldonado, M.I.; Blanco, J.; Gernjak, W. Decontamination and disinfection of water by solar photocatalysis: Recent overview and trends. Catal. Today 2009, 147, 1–59. [Google Scholar] [CrossRef]
- Woods-Robinson, R.; Han, Y.; Zhang, H.; Ablekim, T.; Khan, I.; Persson, K.A.; Zakutayev, A. Wide Band Gap Chalcogenide Semiconductors. Chem. Rev. 2020, 120, 4007–4055. [Google Scholar] [CrossRef] [PubMed]
- Gupta, S.M.; Tripathi, M. A review of TiO2 nanoparticles. Chi. Sci. Bull. 2011, 56, 1639–1657. [Google Scholar] [CrossRef]
- Carp, O.; Huisman, C.L.; Reller, A. Photoinduced reactivity of titanium dioxide. Prog. Solid State Chem. 2004, 32, 33–177. [Google Scholar] [CrossRef]
- Sun, H.; Liu, S.; Liu, S.; Wang, S. A comparative study of reduced graphene oxide modified TiO2, ZnO and Ta2O5 in visible light photocatalytic/photochemical oxidation of methylene blue. Appl. Catal. B 2014, 146, 162–168. [Google Scholar] [CrossRef]
- Haneda, D.L.H. Synthesis of nitrogen-containing ZnO powders by spray pyrolysis and their visible-light photocatalysis in gas-phase acetaldehyde decomposition. J. Photochem. Photobiol. A 2003, 155, 171–178. [Google Scholar]
- Yang, G.C.C.; Chan, S.W. Photocatalytic reduction of chromium(VI) in aqueous solution using dye-sensitized nanoscale ZnO under visible light irradiation. J. Nanopart. Res. 2009, 11, 221–230. [Google Scholar] [CrossRef]
- Huang, Y.; Wei, Y.; Wu, J.; Guo, C.; Wang, M.; Yin, S.; Sato, T. Low temperature synthesis and photocatalytic properties of highly oriented ZnO/TiO2−xNy coupled photocatalysts. Appl. Catal. B 2012, 123–124, 9–17. [Google Scholar] [CrossRef]
- Maleh, H.K.; Kumar, B.G.; Rajendran, S.; Qin, J.; Vadivel, S.; Durgalakshmi, D.; Gracia, F.; Soto-Moscoso, M.; Orooji, Y.; Karimi, F. Tuning of metal oxides photocatalytic performance using Ag nanoparticles integration. J. Mol. Liq. 2020, 314, 113588. [Google Scholar] [CrossRef]
- Karthikeyan, C.; Arunachalam, P.; Ramachandran, K.; Al-Mayouf, A.M.; Karuppuchamy, S. Recent advances in semiconductor metal oxides with enhanced methods for solar photocatalytic applications. J. Alloys Compd. 2020, 828, 154281. [Google Scholar] [CrossRef]
- Oyewo, O.A.; Elemike, E.E.; Onwudiwe, D.C.; Onyango, M.S. Metal oxide-cellulose nanocomposites for the removal of toxic metals and dyes from wastewater. Int. J. Biol. Macromol. 2020, 164, 2477–2496. [Google Scholar] [CrossRef] [PubMed]
- Fujishima, A.; Honda, K. Electrochemical Photolysis of Water at a Semiconductor Electrode. Nature 1972, 238, 37–38. [Google Scholar] [CrossRef]
- Li, C.; Yuan, J.; Han, B.; Shangguan, W. Synthesis and photochemical performance of morphology-controlled CdS photocatalysts for hydrogen evolution under visible light. Int. J. Hydrogen Energy 2011, 36, 4271–4279. [Google Scholar] [CrossRef]
- Fujita, E.; Muckerman, J.T.; Domen, K. Editorial: A current perspective on photocatalysis. Chemsuschem 2011, 4, 155–157. [Google Scholar] [CrossRef]
- Hermann, J.M. Fundamentals and misconceptions in photocatalysis. J. Photochem. Photobio A 2010, 216, 85–93. [Google Scholar] [CrossRef]
- Fujishima, A.; Zhang, X. Titanium dioxide photocatalysis: Present situation and future approaches. Comptes Rendus Chim. 2006, 9, 750–760. [Google Scholar] [CrossRef]
- Zhao, D.; Chen, C.; Wang, Y.; Ma, W.; Zhao, J.; Rajh, T.; Zang, L. Enhanced photocatalytic degradation of dye pollutants under visible irradiation on Al(III)-modified TiO2: Structure, interaction, and interfacial electron transfer. Environ. Sci. Technol. 2008, 42, 308–314. [Google Scholar] [CrossRef]
- Yu, J.; Xiong, J.; Cheng, B.; Liu, S. Fabrication and characterization of Ag–TiO2 multiphase nanocomposite thin films with enhanced photocatalytic activity. Appl. Catal. B 2005, 60, 211–221. [Google Scholar] [CrossRef]
- Sung-Suh, H.M.; Choi, J.R.; Hah, H.J.; Koo, S.M.; Bae, Y.C. Comparison of Ag deposition effects on the photocatalytic activity of nanoparticulate TiO2 under visible and UV light irradiation. J. Photochem. Photobiol. A 2004, 163, 37–44. [Google Scholar] [CrossRef]
- Wilke, K.; Breuer, H.D. The influence of transition metal doping on the physical and photocatalytic properties of titania. J. Photochem. Photobiol. A 1999, 121, 49–53. [Google Scholar] [CrossRef]
- Martin, S.T.; Morrison, C.L.; Hoffmann, M.R. Photochemical Mechanism of Size-Quantized Vanadium-Doped TiO2 Particles. J. Phys. Chem. 1994, 98, 13695–13704. [Google Scholar] [CrossRef]
- Choi, W.; Termin, A.; Hoffmann, M.R. The Role of Metal Ion Dopants in Quantum-Sized TiO2: Correlation between Photoreactivity and Charge Carrier Recombination Dynamics. J. Phys. Chem. 1994, 98, 13669–13679. [Google Scholar] [CrossRef]
- Klosek, S.; Raftery, D. Visible Light Driven V-Doped TiO2 Photocatalyst and Its Photooxidation of Ethanol. J. Phys. Chem. B 2001, 105, 2815–2819. [Google Scholar] [CrossRef]
- Sun, M.; Lin, T.; Cheng, G.; Ye, F.; Yu, L. Hydrothermal Synthesis of Boron-Doped MnO2 and Its Decolorization Performance. J. Nanomater. 2014, 2014, 175924. [Google Scholar] [CrossRef]
- Ksibi, M.; Rossignol, S.; Tatibouët, J.-M.; Trapalis, C. Synthesis and solid characterization of nitrogen and sulfur-doped TiO2 photocatalysts active under near visible light. Mater. Lett. 2008, 62, 4204–4206. [Google Scholar] [CrossRef]
- Du, P.; Bueno-Lopez, A.; Verbaas, M.; Almeida, A.R.; Makkee, M.; Moulijin, J.A.; Mul, G. The effect of surface OH-population on the photocatalytic activity of rare earth-doped P25-TiO2 in methylene blue degradation. J. Catal. 2008, 260, 75–80. [Google Scholar] [CrossRef]
- Zhu, J.; Xie, J.; Chen, M.; Jiang, D.; Wu, D. Low temperature synthesis of anatase rare earth doped titania-silica photocatalyst and its photocatalytic activity under solar-light. Colloids Surf. A Physicochem. Eng. Asp. 2010, 355, 178–182. [Google Scholar] [CrossRef]
- Liu, R.; Wang, P.; Wang, X.; Yu, H.; Yu, J. UV- and Visible-Light Photocatalytic Activity of Simultaneously Deposited and Doped Ag/Ag(I)-TiO2 Photocatalyst. J. Phys. Chem. C 2012, 116, 17721–17728. [Google Scholar] [CrossRef]
- Suda, Y.; Kawasaki, H.; Ueda, T.; Ohshima, T. Preparation of high quality nitrogen doped TiO2 thin film as a photocatalyst using a pulsed laser deposition method. Thin Solid Films 2004, 453–454, 162–166. [Google Scholar] [CrossRef]
- Gole, J.L.; Stout, J.D.; Burda, C.; Lou, Y.; Chen, X. Highly Efficient Formation of Visible Light Tunable TiO2-xNx Photocatalysts and Their Transformation at the Nanoscale. J. Phys. Chem. B 2004, 108, 1230–1240. [Google Scholar] [CrossRef]
- Miyauchi, M.; Ikezawa, A.; Tobimatsu, H.; Iriea, H.; Hashimoto, K. Zeta potential and photocatalytic activity of nitrogen doped TiO2 thin films. Phys. Chem. Chem. Phys. 2004, 6, 865–870. [Google Scholar] [CrossRef]
- Hiromasa, T.; Masahiro, M. N-doped TiO2 Nanotube with Visible Light Activity. Chem. Lett. 2004, 33, 1108–1109. [Google Scholar]
- Wang, J.; Tafen, D.N.; Lewis, J.P.; Hong, Z.; Manivannan, A.; Zhi, M.; Li, M.; Wu, N. Origin of Photocatalytic Activity of Nitrogen-Doped TiO2 Nanobelts. J. Am. Chem. Soc. 2009, 131, 12290–12297. [Google Scholar] [CrossRef]
- Emeline, A.V.; Kuznetsov, V.N.; Rubchuk, V.K.; Serpone, N. Visible-Light-Active Titania Photocatalysts: The Case of N-Doped TiO2 S—Properties and Some Fundamental Issues. Int. J. Photoenergy 2008, 2008, 258394. [Google Scholar] [CrossRef]
- Wu, Z.; Dong, F.; Liu, Y.; Wang, H. Enhancement of the visible light photocatalytic performance of C-doped TiO2 by loading with V2O5. Catal. Commum. 2009, 11, 82–86. [Google Scholar] [CrossRef]
- Daneshvar, N.; Salari, D.; Khataee, A.R. Photocatalytic degradation of azo dye acid red 14 in water on ZnO as an alternative catalyst to TiO2. J. Photochem. Photobiol. A 2004, 162, 317–322. [Google Scholar] [CrossRef]
- Moussavi, G.; Mahmoudi, M. Removal of azo and anthraquinone reactive dyes from industrial wastewaters using MgO nanoparticles. J. Hazard. Mater. 2009, 168, 806–812. [Google Scholar] [CrossRef]
- El-Hankari, S.; Aguilera-Sigalat, J.; Bradshaw, D. Surfactant-assisted ZnO processing as a versatile route to ZIF composites and hollow architectures with enhanced dye adsorption. J. Mater. Chem. A 2016, 4, 13509–13518. [Google Scholar] [CrossRef]
- Kaliraj, L.; Ahn, J.C.; Rupa, E.J.; Abid, S.; Lu, J.; Yang, D.C. Synthesis of panos extract mediated ZnO nano-flowers as photocatalyst for industrial dye degradation by UV illumination. J. Photochem. Photobiol. B 2019, 199, 111588. [Google Scholar] [CrossRef]
- Vinayagam, R.; Selvaraj, R.; Arivalagan, P.; Varadavenkatesan, T. Synthesis, characterization and photocatalytic dye degradation capability of Calliandra haematocephala-mediated zinc oxide nanoflowers. J. Photochem. Photobiol. B 2020, 203, 111760. [Google Scholar] [CrossRef]
- Moghaddas, S.M.T.H.; Elahi, B.; Javanbakht, V. Biosynthesis of pure zinc oxide nanoparticles using Quince seed mucilage for photocatalytic dye degradation. J. Alloys Compd. 2020, 821, 153519. [Google Scholar] [CrossRef]
- Kalpana, V.N.; Kataru, B.A.S.; Sravani, N.; Vigneshwari, T.; Panneerselvam, A.; Rajeswari, V.D. Biosynthesis of zinc oxide nanoparticles using culture filtrates of Aspergillus niger: Antimicrobial textiles and dye degradation studies. Open Nano 2018, 3, 48–55. [Google Scholar] [CrossRef]
- Meng, A.; Xing, J.; Li, Z.; Li, Q. Cr-doped ZnO nanoparticles: Synthesis, characterization, adsorption property, and recyclability. ACS Appl. Mater. Interfaces 2015, 7, 27449–27457. [Google Scholar] [CrossRef] [PubMed]
- Feng, W.; Nansheng, D.; Helin, H. Degradation mechanism of azo dye CI reactive red 2 by iron powder reduction and photooxidation in aqueous solutions. Chemosphere 2000, 41, 1233–1238. [Google Scholar] [CrossRef]
- Khedr, M.H.; Abdel Halim, K.S.; Soliman, N.K. Synthesis and photocatalytic activity of nano-sized iron oxides. Mater. Lett. 2009, 63, 598–601. [Google Scholar] [CrossRef]
- Kusior, A.; Michalec, K.; Jelen, P.; Radecka, M. Shaped Fe2O3 nanoparticles-Synthesis and enhanced photocatalytic degradation towards RhB. Appl. Surf. Sci. 2019, 476, 342–352. [Google Scholar] [CrossRef]
- Shenoy, M.R.; Ayyasamy, S.; Reddy, M.V.V.; Kadarkarai, G.; Suryakanth, J.; Tamilarasan, S.; Thangavelu, S.; Jeyaramane, A.C. The effect of morphology-dependent surface charges of iron oxide on the visible light photocatalytic degradation of methylene blue dye. J. Mater. Sci. Mater. Electron. 2020, 31, 17703–17717. [Google Scholar] [CrossRef]
- Kouhbanani, M.A.J.; Beheshtkhoo, N.; Taghizadeh, S.; Amani, A.M.; Alimardani, V. One-step green synthesis and characterization of iron oxide nanoparticles using aqueous leaf extract of Teucrium polium and their catalytic application in dye degradation. Adv. Nat. Sci. Nanosci. Nanotechnol. 2019, 10, 15007. [Google Scholar] [CrossRef]
- Bishnoi, S.; Kumar, A.; Selvaraj, R. Facile synthesis of magnetic iron oxide nanoparticles using inedible Cynometra ramiflora fruit extract waste and their photocatalytic degradation of methylene blue dye. Mater. Res. Bull. 2018, 97, 121–127. [Google Scholar] [CrossRef]
- Bibi, I.; Nazar, N.; Ata, S.; Sultan, M.; Ali, A.; Abbas, A.; Jilani, K.; Kamal, S.; Sarim, F.M.; Khan, M.I.; et al. Green synthesis of iron oxide nanoparticles using pomegranate seeds extract and photocatalytic activity evaluation for the degradation of textile dye. J. Mater. Res. Technol. 2019, 8, 6115–6124. [Google Scholar] [CrossRef]
- Anchan, S.; Pai, S.; Sridevi, H.; Varadavenkatesan, T.; Vinayagam, R.; Selvaraj, R. Biogenic synthesis of ferric oxide nanoparticles using the leaf extract of Peltophorum pterocarpum and their catalytic dye degradation potential. Biocatal. Agric. Biotechnol. 2019, 20, 101251. [Google Scholar] [CrossRef]
- Wang, C.-T. Photocatalytic activity of nanoparticle gold/iron oxide aerogels for azo dye degradation. J. Non-Cryst. Solids 2007, 353, 1126–1133. [Google Scholar] [CrossRef]
- Mills, A.; Hunte, S.L. An overview of semiconductor photocatalysis. J. Photochem. Photobiol. A Chem. 1997, 108, 1–35. [Google Scholar] [CrossRef]
- Guo, S.Y.; Han, S.; Chi, B.; Pu, J.; Li, J. A facile low-temperature approach to designing controlled amorphous-based titania composite photocatalysts with excellent noble-metal-free photocatalytic hydrogen production. ACS Appl. Mater. Interfaces 2014, 6, 4743–4751. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.X.; Guo, X.L.; Huang, M.; Hao, X.D.; Yuan, Y.; Hua, C. Engineering birnessite-type MnO2 nanosheets on fiberglass for pH-dependent degradation of methylene blue. J. Phys. Chem. Solids 2015, 83, 40–46. [Google Scholar] [CrossRef]
- Pandit, A.A.; Pawar, R.A.; Shinde, D.R. Colloidal MnO2 catalyzed degradation of two azo dyes methyl red and methyl orange from aqueous medium. Int. J. Eng. Res. 2013, 4, 1119–1122. [Google Scholar]
- Liu, Y.; Chen, Z.; Shek, C.H.; Wu, C.M.L.; Lai, J.K.L. Hierarchical Mesoporous MnO2 Superstructures Synthesized by Soft-Interface Method and Their Catalytic Performances. ACS Appl. Mater. Interfaces 2014, 6, 9776–9784. [Google Scholar] [CrossRef]
- Ranjit, K.T.; Willner, I.; Bossmann, S.; Braun, A. Iron(III) Phthalocyanine-Modified Titanium Dioxide: A Novel Photocatalyst for the Enhanced Photodegradation of Organic Pollutants. J. Phys. Chem. B 1998, 102, 9397–9403. [Google Scholar] [CrossRef]
- Iliev, V. Phthalocyanine-modified titania—Catalyst for photooxidation of phenols by irradiation with visible light. J. Photochem. Photobiol. A: Chem. 2002, 151, 195–199. [Google Scholar] [CrossRef]
- Mele, G.; Sole, R.D.; Vasapollo, G.; García-López, E.; Palmisano, L.; Schiavello, M. Photocatalytic degradation of 4-nitrophenol in aqueous suspension by using polycrystalline TiO2 impregnated with functionalized Cu(II)–porphyrin or Cu(II)–phthalocyanine. J. Catal. 2003, 217, 334–342. [Google Scholar] [CrossRef]
- Yu, J.C.; Xie, Y.; Tang, H.Y.; Zhang, L.; Chan, H.C.; Zhao, J. Visible light-assisted bactericidal effect of metalphthalocyanine-sensitized titanium dioxide films. J. Photochem. Photobiol. A Chem. 2003, 156, 235–241. [Google Scholar] [CrossRef]
- Kumar, S.G.; Devi, L.G. Review on Modified TiO2 Photocatalysis under UV/Visible Light: Selected Results and Related Mechanisms on Interfacial Charge Carrier Transfer Dynamics. J. Phys. Chem. A 2011, 115, 13211–13241. [Google Scholar] [CrossRef] [PubMed]
- Linsebigler, A.L.; Lu, G.; Yates, J.T., Jr. Photocatalysis on TiO2 Surfaces: Principles, Mechanisms, and Selected Results. Chem. Rev. 1995, 95, 735–758. [Google Scholar] [CrossRef]
- Shang, J.; Yao, W.; Zhu, Y.; Wu, N. Structure and photocatalytic performances of glass/SnO2/TiO2 interface composite film. Appl. Catal. A Gen. 2004, 257, 25–32. [Google Scholar] [CrossRef]
- Shi, L.; Li, C.; Gu, H.; Fang, D. Morphology and properties of ultrafine SnO2–TiO2 coupled semiconductor particles. Mater. Chem. Phys. 2000, 62, 62–67. [Google Scholar] [CrossRef]
- Omrani, N.; Nezamzadeh-Ejhieh, A. A comprehensive study on the enhanced photocatalytic activity of Cu2O/BiVO4/WO3 nanoparticles. J. Photochem. Photobiol. A 2020, 389, 112223. [Google Scholar] [CrossRef]
- Khalid, N.R.; Bilal, M.; Tahir, M.B.; Shakil, M.; Iqbal, T.; Rafique, M.; Yousaf, N.; Gillani, S.S.A. Interfacial coupling effect of Ag2O nanorods over MoS2 microflowers for improved photocatalytic activity. Ceram. Int. 2020, 46, 6856–6859. [Google Scholar] [CrossRef]
- Irie, H.; Watanabe, Y.; Hashimoto, K. Nitrogen-Concentration Dependence on Photocatalytic Activity of TiO2-xNx Powders. J. Phys. Chem. B 2003, 107, 5483–5486. [Google Scholar] [CrossRef]
- Asahi, R.; Morikawa, T.; Ohwaki, T.; Aoki, K.; Taga, Y. Visible-light photocatalysis in nitrogen-doped titanium oxides. Science 2001, 293, 269–271. [Google Scholar] [CrossRef]
- Wu, T.; Liu, G.; Zhao, J.; Hidaka, H.; Serpone, N. Evidence for H2O2 Generation during the TiO2-Assisted Photodegradation of Dyes in Aqueous Dispersions under Visible Light Illumination. J. Phys. Chem. B 1999, 103, 4862–4867. [Google Scholar] [CrossRef]
- Belhekar, A.A.; Awate, S.V.; Anand, R. Photocatalytic activity of titania modified mesoporous silica for pollution control. Catal. Commun. 2002, 3, 453–458. [Google Scholar] [CrossRef]
- Scaria, J.; Nidheesh, P.V.; Kumar, M.S. Synthesis and applications of various bimetallic nanomaterials in water and wastewater treatment. J. Environ. Manag. 2020, 259, 110011. [Google Scholar] [CrossRef]
- Mahmood, M.A.; Baruah, S.; Dutta, J. Enhanced visible light photocatalysis by manganese doping or rapid crystallization with ZnO nanoparticles. Mater. Chem. Phys. 2011, 130, 531–535. [Google Scholar] [CrossRef]
- Deka, S.; Joy, P.A. Synthesis and magnetic properties of Mn doped ZnO nanowires. Solid State Commun. 2007, 142, 190–194. [Google Scholar] [CrossRef]
- Bhati, I.; Sharma, J.; Ameta, A.; Ameta, S.C. Synthesis and characterization of nanosized CeCr2O5 and its use as a photocatalyst. Int. J. Chem. Sci. 2011, 9, 1787–1796. [Google Scholar]
- Liu, Y.; Sun, D. Effect of CeO2 doping on catalytic activity of Fe2O3/γ-Al2O3 catalyst for catalytic wet peroxide oxidation of azo dyes. J. Hazard. Mater. 2007, 143, 448–454. [Google Scholar] [CrossRef]
- Saravanan, R.; Karthikeyan, N.; Govindan, S.; Narayanan, V.; Stephen, A. Photocatalytic degradation of organic dyes using ZnO/CeO2 nanocomposite material under visible light. Adv. Mat. Res. 2012, 584, 381–385. [Google Scholar]
- Yang, X.; Wang, Y.; Xu, L.; Yu, X.; Guo, Y. Silver and Indium Oxide Codoped TiO 2 Nanocomposites with Enhanced Photocatalytic Activity. J. Phys. Chem. C 2008, 112, 11481–11489. [Google Scholar] [CrossRef]
- Georgekutty, R.; Seery, M.K.; Pillai, S.C. A Highly Efficient Ag-ZnO Photocatalyst: Synthesis, Properties, and Mechanism. J. Phys. Chem. C 2008, 112, 13563–13570. [Google Scholar] [CrossRef]
- Ganesan, M.; Viswanathan, B.; Viswanath, R.P.; Varadarajan, T.K. Photocatalytic behavior of CeO2-TiO2 system for the degradation of methylene blue. Indian J. Chem. 2009, 48, 480–488. [Google Scholar]
- Xu, D.; Cheng, F.; Lu, Q.; Dai, P. Microwave Enhanced Catalytic Degradation of Methyl Orange in Aqueous Solution over CuO/CeO2 Catalyst in the Absence and Presence of H2O2. Ind. Eng. Chem. Res. 2014, 53, 2625–2632. [Google Scholar] [CrossRef]
- Tung, W.S.; Daoud, W.A. New approach toward nanosized ferrous ferric oxide and Fe3O4-doped titanium dioxide photocatalysts. ACS Appl. Mater. Interfaces 2009, 1, 2453–2461. [Google Scholar] [CrossRef] [PubMed]
- Zhu, J.; Zheng, W.; Hea, B.; Jhang, J.; Anpo, M. Characterization of Fe–TiO2 photocatalysts synthesized by hydrothermal method and their photocatalytic reactivity for photodegradation of XRG dye diluted in water. J. Mol. Catal. A Chem. 2004, 216, 35–43. [Google Scholar] [CrossRef]
- Zhang, L.; Lian, J.; Wu, L.; Duan, Z.; Jiang, J.; Zhao, L. Synthesis of a Thin-Layer MnO2 Nanosheet-Coated Fe3O4 Nanocomposite as a Magnetically Separable Photocatalyst. Langmuir 2014, 30, 7006–7013. [Google Scholar] [CrossRef] [PubMed]
- Sreethawong, T.; Ngamsinlapasathian, S.; Yoshikawa, S. Surfactant-aided sol–gel synthesis of mesoporous-assembled TiO2–NiO mixed oxide nanocrystals and their photocatalytic azo dye degradation activity. Chem. Eng. J. 2012, 192, 292–300. [Google Scholar] [CrossRef]
- Sreethawong, T.; Yoshikawa, S. Impact of photochemically deposited monometallic Pt and bimetallic Pt–Au nanoparticles on photocatalytic dye-sensitized H2 production activity of mesoporous-assembled TiO2–SiO2 mixed oxide nanocrystal. Chem. Eng. J. 2012, 197, 272–282. [Google Scholar] [CrossRef]
- Mohamed, R.M.; Mkhalid, I.A.; Baeissa, E.S.; Al-Rayyani, M.A. Photocatalytic Degradation of Methylene Blue by Fe/ZnO/SiO2 Nanoparticles under Visible light. J. Nanotechnol. 2012, 2012, 329082. [Google Scholar]
- Xue, M.; Huang, L.; Wang, J.Q.; Wang, Y.; Gao, L.; Zhu, J.H.; Zhou, Z.G. The direct synthesis of mesoporous structured MnO2/TiO2 nanocomposite: A novel visible-light active photocatalyst with large pore size. Nanotechnology 2008, 19, 185604. [Google Scholar] [CrossRef]
- Wang, C.; Shao, C.; Zhang, X.; Liu, Y. SnO2 Nanostructures-TiO2 Nanofibers Heterostructures: Controlled Fabrication and High Photocatalytic Properties. Inorg. Chem. 2009, 48, 7261–7268. [Google Scholar] [CrossRef] [PubMed]
- Li, H.B.; Song, Y.D.; Chen, S.G. Preparation and Characterization of MnO2 Loaded CuO Nanosheets. Adv. Mat. Res. 2013, 652–654, 241–244. [Google Scholar]
- Qian, K.; Qian, Z.X.; Hua, Q.; Jiang, Z.Q.; Huang, W.X. Structure–activity relationship of CuO/MnO2 catalysts in CO oxidation. Appl. Surf. Sci. 2013, 273, 357–363. [Google Scholar] [CrossRef]
- Yang, Z.; Yang, Y.; Zhu, X.; Chen, G.; Zhang, W. An Outward Coating Route to CuO/MnO2 Nanorod Array Films and Their Efficient Catalytic Oxidation of Acid Fuchsin Dye. Ind. Eng. Chem. Res. 2014, 53, 9608–9615. [Google Scholar] [CrossRef]
- Guo, Y.; Yang, X.; Ma, F.; Li, K.; Xu, L.; Yuan, X.; Guo, Y. Additive-free controllable fabrication of bismuth vanadates and their photocatalytic activity toward dye degradation. Appl. Surf. Sci. 2010, 256, 2215–2222. [Google Scholar] [CrossRef]
- Shang, M.; Wang, W.; Zhou, L.; Sun, S.; Yin, W. Nanosized BiVO4 with high visible-light-induced photocatalytic activity: Ultrasonic-assisted synthesis and protective effect of surfactant. J. Hazard. Mater. 2009, 172, 338–344. [Google Scholar] [CrossRef]
- Yin, W.; Wang, W.; Zhou, L.; Sun, S.; Zhang, L. CTAB-assisted synthesis of monoclinic BiVO4 photocatalyst and its highly efficient degradation of organic dye under visible-light irradiation. J. Hazard. Mater. 2009, 173, 194–199. [Google Scholar] [CrossRef]
- Ghorai, T.K.; Dhak, D.; Dalai, S.; Pramanik, P. Preparation and photocatalytic activity of nano-sized nickel molybdate (NiMoO4) doped bismuth titanate (Bi2Ti4O11) (NMBT) composite. J. Alloys Compd. 2008, 463, 390–397. [Google Scholar] [CrossRef]
- Cruz, A.M.L.; Alfaro, S.O. Synthesis and characterization of γ-Bi2MoO6 prepared by co-precipitation: Photoassisted degradation of organic dyes under vis-irradiation. J. Mol. Catal. A Chem. 2010, 320, 85–91. [Google Scholar] [CrossRef]
- Xu, X.; Ni, Q. Synthesis and characterization of novel Bi2MoO6/NaY materials and photocatalytic activities under visible light irradiation. Catal. Commun. 2010, 11, 359–363. [Google Scholar] [CrossRef]
- Sahoo, P.P.; Sumithra, S.; Madras, G.; Row, T.N.G. Synthesis, structure and photocatalytic properties of β-ZrMo2O8. Bull. Mater. Sci. 2009, 32, 337–342. [Google Scholar] [CrossRef]
- Mulla, I.S.; Natarajan, N.; Gaikwad, A.B.; Samuel, V.; Guptha, U.N.; Ravi, V. A coprecipitation technique to prepare CoTa2O6 and CoNb2O6. Mater. Lett. 2007, 61, 2127–2129. [Google Scholar] [CrossRef]
- Machida, M.; Yabunaka, J.; Kijima, T. Synthesis and Photocatalytic Property of Layered Perovskite Tantalates, RbLnTa2O7 (Ln = La, Pr, Nd, and Sm). Chem. Mater. 2000, 12, 812–817. [Google Scholar] [CrossRef]
- Eng, H.W.; Barnes, P.W.; Auer, B.M.; Woodward, P.M. Investigations of the electronic structure of d0 transition metal oxides belonging to the perovskite family. J. Solid State Chem. 2003, 175, 94–109. [Google Scholar] [CrossRef]
- Ullah, R.; Sun, H.; Ang, H.M.; Tadé, M.O.; Wang, S. Visible light photocatalytic degradation of organics on nanoparticles of bi-metallic oxides. Sep. Purify. Technol. 2012, 89, 98–106. [Google Scholar] [CrossRef]
- Su, S.; Guo, W.; Leng, Y.; Yi, C.; Ma, Z. Heterogeneous activation of Oxone by CoxFe3−xO4 nanocatalysts for degradation of rhodamine B. J. Hazard. Mater. 2013, 244–245, 736–742. [Google Scholar] [CrossRef] [PubMed]
- Yu, K.; Yang, S.; He, H.; Sun, C.; Gu, C.; Ju, Y. Visible light-driven photocatalytic degradation of rhodamine B over NaBiO3: Pathways and mechanism. J. Phys. Chem. A 2009, 113, 10024–10032. [Google Scholar] [CrossRef]
- Li, S.J.; Ma, Z.; Wang, L.; Liu, J. Influence of MnO2 on the photocatalytic activity of P-25 TiO2 in the degradation of methyl orange. Sci. China Ser. B Chem. 2008, 51, 179–185. [Google Scholar] [CrossRef]
- Ahuja, P.; Ujjain, S.K.; Sharma, R.K.; Singh, G. Enhanced supercapacitor performance by incorporating nickel in manganese oxide. RSC Adv. 2014, 4, 57192–57199. [Google Scholar] [CrossRef]
- Ahuja, P.; Sharma, R.K.; Singh, G. Solid-state, high-performance supercapacitor using graphene nanoribbons embedded with zinc manganite. J. Mater. Chem. A 2015, 3, 4931–4937. [Google Scholar] [CrossRef]
- Ujjain, S.K.; Ahuja, P.; Sharma, R.K. Graphene nanoribbon wrapped cobalt manganite nanocubes for high performance all-solid-state flexible supercapacitors. J. Mater. Chem. A 2015, 3, 9925–9931. [Google Scholar] [CrossRef]
- Ahuja, P.; Sahu, V.; Ujjain, S.K.; Sharma, R.K.; Singh, G. Performance evaluation of Asymmetric Supercapacitor based on Cobalt manganite modified graphene nanoribbons. Electrochim. Acta 2014, 146, 429–436. [Google Scholar] [CrossRef]
- Ahuja, P.; Ujjain, S.K.; Kanojia, R. Electrochemical behaviour of manganese & ruthenium mixed oxide@ reduced graphene oxide nanoribbon composite in symmetric and asymmetric supercapacitor. Appl. Surf. Sci. 2018, 427, 102–111. [Google Scholar]
- Deori, K.; Ujjain, S.K.; Sharma, R.K.; Deka, S. Morphology Controlled Synthesis of Nanoporous Co3O4 Nanostructures and Their Charge Storage Characteristics in Supercapacitors. ACS Appl. Mater. Interfaces 2013, 5, 10665–10672. [Google Scholar] [CrossRef]
- Stoyanova, M.; Slavova, I.; Christoskova, S.; Ivanova, V. Catalytic performance of supported nanosized cobalt and iron–cobalt mixed oxides on MgO in oxidative degradation of Acid Orange 7 azo dye with peroxymonosulfate. Appl. Catal. A Gen. 2014, 476, 121–132. [Google Scholar] [CrossRef]
- Wang, Y.; Sun, H.; Ang, H.M.; Tadé, M.O.; Wang, S. Facile Synthesis of Hierarchically Structured Magnetic MnO2/ZnFe2O4 Hybrid Materials and Their Performance in Heterogeneous Activation of Peroxymonosulfate. ACS Appl. Mater. Interfaces 2014, 6, 19914–19923. [Google Scholar] [CrossRef]
- Sathishkumar, P.; Mangalaraja, R.V.; Anandan, S.; Ashokkumar, M. CoFe2O4/TiO2 nanocatalysts for the photocatalytic degradation of Reactive Red 120 in aqueous solutions in the presence and absence of electron acceptors. Chem. Eng. J. 2013, 220, 302–310. [Google Scholar] [CrossRef]
- Cui, B.; Lin, H.; Liu, Y.; Li, J.; Sun, P.; Zhao, X.; Liu, C. Photophysical and Photocatalytic Properties of Core-Ring Structured NiCo2O4 Nanoplatelets. J. Phys. Chem. C 2009, 113, 14083–14087. [Google Scholar] [CrossRef]
- Singh, M.K.; Patra, A. Highly efficient and Reusable ZnO microflower photocatalyst on stainless steel mesh under UV–Vis and natural sunlight. Opt. Mater. 2020, 107, 110000. [Google Scholar] [CrossRef]
- Basnet, P.; Samanta, D.; Chanu, T.I.; Chatterjee, S. Visible light facilitated degradation of alternate dye solutions by highly reusable Mn-ZnO nano-photocatalyst. J. Alloys Compd. 2021, 867, 158870. [Google Scholar] [CrossRef]
- Sharma, S.; Basu, S. Highly reusable visible light active hierarchical porous WO3/SiO2 monolith in centimeter length scale for enhanced photocatalytic degradation of toxic pollutants. Sep. Purif. Technol. 2020, 231, 115916. [Google Scholar] [CrossRef]
- Janitabar-Darzi, S.; Mahjoub, A.R. Investigation of phase transformations and photocatalytic properties of sol–gel prepared nanostructured ZnO/TiO2 composites. J. Alloys Compd. 2009, 486, 805–808. [Google Scholar] [CrossRef]
- Sajjad, A.K.L.; Shamaila, S.; Tian, B.; Chen, F.; Zhang, J. Comparative studies of operational parameters of degradation of azo dyes in visible light by highly efficient WOx/TiO2 photocatalyst. J. Hazard. Mater. 2010, 177, 781–791. [Google Scholar] [CrossRef]
- Ghasemi, S.; Rahimnejad, S.; Setayesh, S.R.; Rohani, S.; Golami, M.R. Transition metal ions effect on the properties and photocatalytic activity of nanocrystalline TiO2 prepared in an ionic liquid. J. Hazard. Mater. 2009, 172, 1573–1578. [Google Scholar] [CrossRef]
- Shen, W.; Li, Z.; Wang, H.; Liu, Y.; Guo, Q.; Zhang, Y. Photocatalytic degradation for methylene blue using zinc oxide prepared by codeposition and sol–gel methods. J. Hazard. Mater. 2008, 152, 172–175. [Google Scholar] [CrossRef]
- Singh, J.; Rathi, A.; Rawat, M.; Kumar, V.; Kim, K.-H. The effect of manganese doping on structural, optical, and photocatalytic activity of zinc oxide nanoparticles. Compos. B. Eng. 2019, 166, 361–370. [Google Scholar] [CrossRef]
- Xu, C.; Wei, X.; Guo, Y.; Wu, H.; Ren, Z.; Xu, G.; Shen, G.; Han, G. Surfactant-free synthesis of Bi2WO6 multilayered disks with visible-light-induced photocatalytic activity. Mater. Res. Bull. 2009, 44, 1635–1641. [Google Scholar] [CrossRef]
- Lin, S.; Chen, J.; Weng, X.; Yang, L.; Chen, X. Fabrication and photocatalysis of mesoporous ZnWO4 with PAMAM as a template. Mater. Res. Bull. 2009, 44, 1102–1105. [Google Scholar] [CrossRef]
- Seddigi, Z.S. Removal of Alizarin Yellow Dye from Water Using Zinc Doped WO3 Catalyst. Bull. Environ. Contam. Toxicol. 2010, 84, 564–567. [Google Scholar] [CrossRef] [PubMed]
- Cho, I.; Kwak, C.H.; Kim, D.W.; Lee, S.; Hong, K.S. Photophysical, Photoelectrochemical, and Photocatalytic Properties of Novel SnWO4 Oxide Semiconductors with Narrow Band Gaps. J. Phys. Chem. C 2009, 113, 10647–10653. [Google Scholar] [CrossRef]
- Ahmad, M.; Ahmed, E.; Ahmed, W.; Elhissi, A.; Hong, Z.L.; Khalid, N.R. Enhancing visible light responsive photocatalytic activity by decorating Mn-doped ZnO nanoparticles on graphene. Ceram. Int. 2014, 40, 10085–10097. [Google Scholar] [CrossRef]
- Ahuja, P.; Ujjain, S.K.; Kanojia, R. MnOx/C nanocomposite: An insight on high-performance supercapacitor and non-enzymatic hydrogen peroxide detection. Appl. Surf. Sci. 2017, 404, 197–205. [Google Scholar] [CrossRef]
- Ujjain, S.K.; Sahu, V.; Sharma, R.K.; Singh, G. High performance, all solid state, flexible supercapacitor based on ionic liquid functionalized graphene. Electrochim. Acta 2015, 157, 245–251. [Google Scholar] [CrossRef]
- Ujjain, S.K.; Ahuja, P.; Sharma, R.K. Facile preparation of graphene nanoribbon/cobalt coordination polymer nanohybrid for non-enzymatic H2O2 sensing by dual transduction: Electrochemical and fluorescence. J. Mater. Chem. B 2015, 3, 7614–7622. [Google Scholar] [CrossRef]
- Putri, A.D.; Chotimah, N.; Ujjain, S.K.; Wang, S.; Futamura, R.; Vallejos-Burgos, F.; Khoerunnisa, F.; Morimoto, M.; Wang, Z.; Hattori, Y.; et al. Charge-transfer mediated nanopore-controlled pyrene derivatives/graphene colloids. Carbon 2018, 139, 512–521. [Google Scholar] [CrossRef]
- Ujjain, S.K.; Sharma, R.K.; Singh, G. Co3O4@reduced graphene oxide nanoribbon for high performance asymmetric supercapacitor. Electrochim. Acta 2015, 169, 276–282. [Google Scholar] [CrossRef]
- Ujjain, S.K.; Bagusetty, A.; Matsuda, Y.; Tanaka, H.; Ahuja, P.; De Tomas, C.; Sakai, M.; Vallejos-Burgos, F.; Futamura, R.; Suarez-Martinez, I.; et al. Adsorption separation of heavier isotope gases in subnanometer carbon pores. Nat. Commun. 2021, 12, 546. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Zhang, S.; Yu, Q.; Yin, W. The effects of activated carbon support on the structure and properties of TiO2 nanoparticles prepared by a sol–gel method. Appl. Surf. Sci. 2007, 253, 9254–9258. [Google Scholar] [CrossRef]
- Wang, X.; Hu, Z.; Chen, Y.; Zhao, G.; Liu, Y.; Wen, Z. A novel approach towards high-performance composite photocatalyst of TiO2 deposited on activated carbon. Appl. Surf. Sci. 2009, 255, 3953–3958. [Google Scholar] [CrossRef]
- Zhang, X.; Zhou, M.; Lei, L. Preparation of photocatalytic TiO2 coatings of nanosized particles on activated carbon by AP-MOCVD. Carbon 2005, 43, 1700–1708. [Google Scholar] [CrossRef]
- Zhang, X.; Zhou, M.; Lei, L. TiO2 photocatalyst deposition by MOCVD on activated carbon. Carbon 2006, 44, 325–333. [Google Scholar] [CrossRef]
- Ao, Y.; Xu, J.; Fu, D.; Yuan, C. A simple route for the preparation of anatase titania-coated magnetic porous carbons with enhanced photocatalytic activity. Carbon 2008, 46, 596–603. [Google Scholar] [CrossRef]
- Velasco, L.F.; Ania, C.O. Role of activated carbon features on the photocatalytic degradation of phenol. Appl. Surf. Sci. 2010, 256, 5254–5258. [Google Scholar] [CrossRef]
- Lee, D.K.; Kim, S.C.; Kim, S.J.; Chung, I.S.; Kim, S.W. Photocatalytic oxidation of microcystin-LR with TiO2-coated activated carbon. Chem. Eng. J. 2004, 102, 93–98. [Google Scholar] [CrossRef]
- Yao, Y.; Li, G.; Ciston, S.; Lueptow, R.M.; Gray, K.A. Photoreactive TiO2/carbon nanotube composites: Synthesis and reactivity. Environ. Sci. Technol. 2008, 42, 4952–4957. [Google Scholar] [CrossRef]
- Ahmmad, B.; Kusumoto, Y.; Somekawa, S.; Ikeda, M. Carbon nanotubes synergistically enhance photocatalytic activity of TiO2. Catal. Commun. 2008, 9, 1410–1413. [Google Scholar] [CrossRef]
- Xia, X.H.; Jia, Z.J.; Yu, Y.; Liang, Y.; Wang, Z.; Ma, L.L. Preparation of multi-walled carbon nanotube supported TiO2 and its photocatalytic activity in the reduction of CO2 with H2O. Carbon 2007, 45, 717–721. [Google Scholar] [CrossRef]
- Hoffmann, M.R.; Martin, S.T.; Choi, W.; Bahnemann, D.W. Environmental Applications of Semiconductor Photocatalysis. Chem. Rev. 1995, 95, 69–96. [Google Scholar] [CrossRef]
- Liu, B.; Zeng, H.C. Carbon Nanotubes Supported Mesoporous Mesocrystals of Anatase TiO2. Chem. Mater. 2008, 20, 2711–2718. [Google Scholar] [CrossRef]
- Woan, K.; Pyrgiotakis, G.; Sigmund, W. Photocatalytic Carbon-Nanotube-TiO2 Composites. Adv. Mater. 2009, 21, 2233–2239. [Google Scholar] [CrossRef]
- Xiong, P.; Fu, Y.; Wang, L.; Wang, X. Multi-walled carbon nanotubes supported nickel ferrite: A magnetically recyclable photocatalyst with high photocatalytic activity on degradation of phenols. Chem. Eng. J. 2012, 195–196, 149–157. [Google Scholar] [CrossRef]
- Fan, W.; Lai, Q.; Zhang, Q.; Wang, Y. Nanocomposites of TiO2 and Reduced Graphene Oxide as Efficient Photocatalysts for Hydrogen Evolution. J. Phys. Chem. C 2011, 115, 10694–10701. [Google Scholar] [CrossRef]
- Li, N.; Liu, C.; Zhen, C.; Li, F.; Zhang, L.; Cheng, H.M. Battery Performance and Photocatalytic Activity of Mesoporous Anatase TiO2 Nanospheres/Graphene Composites by Template-Free Self-Assembly. Adv. Funct. Mater. 2011, 21, 1717–1722. [Google Scholar] [CrossRef]
- Xiang, Q.; Yu, J.; Jaroniec, M. Graphene-based semiconductor photocatalysts. Chem. Soc. Rev. 2012, 41, 782–796. [Google Scholar] [CrossRef] [PubMed]
- Li, B.; Cao, H. ZnO@graphene composite with enhanced performance for the removal of dye from water. J. Mater. Chem. 2011, 21, 3346–3349. [Google Scholar] [CrossRef]
- Zhang, J.; Xiong, Z.; Zhao, X.S. Graphene–metal–oxide composites for the degradation of dyes under visible light irradiation. J. Mater. Chem. 2011, 21, 3634–3640. [Google Scholar] [CrossRef]
- Du, J.; Lai, X.; Yang, N.; Zhai, J.; Kisailus, D.; Su, F.; Wang, D.; Jiang, L. Hierarchically ordered macro-mesoporous TiO₂-graphene composite films: Improved mass transfer, reduced charge recombination, and their enhanced photocatalytic activities. ACS Nano 2011, 5, 590–596. [Google Scholar] [CrossRef]
- Chen, J.S.; Wang, Z.; Dong, X.C.; Chena, P.; Lou, X.W.D. Graphene-wrapped TiO2 hollow structures with enhanced lithium storage capabilities. Nanoscale 2011, 3, 2158–2161. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.; Hoi, D.; Li, J.; Yang, Z.; Nie, Z.; Kou, R.; Hu, D.; Wang, C.; Saraf, L.V.; Zhang, J.; et al. Self-Assembled TiO2–Graphene Hybrid Nanostructures for Enhanced Li-Ion Insertion. ACS Nano 2009, 3, 907–914. [Google Scholar] [CrossRef]
- Park, Y.; Kang, S.; Choi, W. Exfoliated and reorganized graphite oxide on titania nanoparticles as an auxiliary co-catalyst for photocatalytic solar conversion. Phys. Chem. Chem. Phys. 2011, 13, 9425–9431. [Google Scholar] [CrossRef]
- Fu, Y.; Wang, X. Magnetically Separable ZnFe2O4–Graphene Catalyst and its High Photocatalytic Performance under Visible Light Irradiation. Ind. Eng. Chem. Res. 2011, 50, 7210–7218. [Google Scholar] [CrossRef]
- Mukherji, A.; Seger, B.; Lu, G.Q.M.; Wang, L. Nitrogen Doped Sr2Ta2O7 Coupled with Graphene Sheets as Photocatalysts for Increased Photocatalytic Hydrogen Production. ACS Nano 2011, 5, 3483–3492. [Google Scholar] [CrossRef]
- Shen, J.; Yan, B.; Shi, M.; Ma, H.; Lia, N.; Ye, M. One step hydrothermal synthesis of TiO2-reduced graphene oxide sheets. J. Mater. Chem. 2011, 21, 3415–3421. [Google Scholar] [CrossRef]
- Williams, G.; Seger, B.; Kamat, P.V. TiO2-Graphene Nanocomposites. UV-Assisted Photocatalytic Reduction of Graphene Oxide. ACS Nano 2008, 2, 1487–1491. [Google Scholar] [CrossRef]
- Williams, G.; Kamat, P.V. Graphene-semiconductor nanocomposites: Excited-state interactions between ZnO nanoparticles and graphene oxide. Langmuir 2009, 25, 13869–13873. [Google Scholar] [CrossRef] [PubMed]
- Kamat, P.V. Graphene-Based Nanoarchitectures. Anchoring Semiconductor and Metal Nanoparticles on a Two-Dimensional Carbon Support. J. Phys. Chem. Lett. 2010, 1, 520–527. [Google Scholar] [CrossRef]
- Lightcap, I.V.; Kosel, T.H.; Kamat, P.V. Anchoring Semiconductor and Metal Nanoparticles on a Two-Dimensional Catalyst Mat. Storing and Shuttling Electrons with Reduced Graphene Oxide. Nano Lett. 2010, 10, 577–583. [Google Scholar] [CrossRef]
- Liu, J.; Bai, H.; Wang, Y.; Liu, Z.; Zhang, X.; Sun, D.D. Self-Assembling TiO2 Nanorods on Large Graphene Oxide Sheets at a Two-Phase Interface and Their Anti-Recombination in Photocatalytic Applications. Adv. Funct. Mater. 2010, 23, 4175–4181. [Google Scholar] [CrossRef]
- Zhou, K.; Zhu, Y.; Yang, X.; Jianga, X.; Lia, C. Preparation of graphene–TiO2 composites with enhanced photocatalytic activity. New J. Chem. 2011, 35, 353–359. [Google Scholar] [CrossRef]
- Yao, Y.; Yang, Z.; Sun, H.; Wang, S. Hydrothermal Synthesis of Co3O4–Graphene for Heterogeneous Activation of Peroxymonosulfate for Decomposition of Phenol. Ind. Eng. Chem. Res. 2012, 51, 14958–14965. [Google Scholar] [CrossRef]
- Yao, Y.; Xu, C.; Yu, S.; Zhang, D.; Wang, S. Facile Synthesis of Mn3O4–Reduced Graphene Oxide Hybrids for Catalytic Decomposition of Aqueous Organics. Ind. Eng. Chem. Res. 2013, 52, 3637–3645. [Google Scholar] [CrossRef]
- Zhang, H.; Lv, X.; Li, Y.; Wang, Y.; Li, J. P25-Graphene Composite as a High Performance Photocatalyst. ACS Nano 2010, 4, 380–386. [Google Scholar] [CrossRef] [PubMed]
- Fatima, R.; Warsi, M.F.; Zulfiqar, S.; Ragab, S.A.; Shakir, I.; Sarwar, M.I. Nanocrystalline transition metal oxides and their composites with reduced graphene oxide and carbon nanotubes for photocatalytic applications. Ceram. Int. 2020, 46, 16480–16492. [Google Scholar] [CrossRef]
- Zhang, Y.; Tang, Z.; Fu, X.; Xu, Y. TiO2−Graphene Nanocomposites for Gas-Phase Photocatalytic Degradation of Volatile Aromatic Pollutant: Is TiO2−Graphene Truly Different from Other TiO2−Carbon Composite Materials. ACS Nano 2010, 4, 7303–7314. [Google Scholar] [CrossRef] [PubMed]
- Bai, X.; Wang, L.; Zong, R.; Lv, Y.; Sun, Y.; Zhu, Y. Performance Enhancement of ZnO Photocatalyst via Synergic Effect of Surface Oxygen Defect and Graphene Hybridization. Langmuir 2013, 29, 3097–3105. [Google Scholar] [CrossRef]
- Li, X.; Yang, S.; Sun, J.; He, P.; Xu, X.; Ding, G. Tungsten oxide nanowire-reduced graphene oxide aerogel for high-efficiency visible light photocatalysis. Carbon 2014, 78, 38–48. [Google Scholar] [CrossRef]
- Qu, J.; Shi, L.; He, C.; Gao, F.; Li, B.; Zhou, Q.; Hu, H.; Shao, G.; Wang, X.; Qiu, J. Highly efficient synthesis of graphene/MnO2 hybrids and their application for ultrafast oxidative decomposition of methylene blue. Carbon 2014, 66, 485–492. [Google Scholar] [CrossRef]
- Rani, S.; Kumar, M.; Sharma, S.; Kumar, D.; Tyagi, S. Effect of Graphene in Enhancing the Photo Catalytic Activity of Zirconium Oxide. Catal. Lett. 2014, 144, 301–307. [Google Scholar] [CrossRef]
- Alamelu, K.; Raja, V.; Shiamala, L.; Jaffar Ali, B.M. Biphasic TiO2 nanoparticles decorated graphene nanosheets for visible light driven photocatalytic degradation of organic dyes. Appl. Surf. Sci. 2018, 430, 145–154. [Google Scholar] [CrossRef]
- Zhang, R.; Ma, Y.; Lan, W.; Sameen, D.E.; Ahmed, S.; Dai, J.; Qin, W.; Li, S.; Liu, Y. Enhanced photocatalytic degradation of organic dyes by ultrasonic-assisted electrospray TiO2/graphene oxide on polyacrylonitrile/β-cyclodextrin nanofibrous membranes. Ultrason. Sonochem. 2021, 70, 105343. [Google Scholar] [CrossRef]
- Nguyen, C.H.; Tran, M.L.; Tran, T.T.V.; Juang, R.-S. Enhanced removal of various dyes from aqueous solutions by UV and simulated solar photocatalysis over TiO2/ZnO/rGO composites. Sep. Purif. Technol. 2020, 232, 115962. [Google Scholar] [CrossRef]
- Truong, N.T.; Thi, H.P.N.; Ninh, H.D.; Phung, X.T.; Tran, C.V.; Nguyen, T.T.; Pham, T.D.; Dang, T.D.; Chang, S.W.; Rene, E.R.; et al. Facile fabrication of graphene@Fe-Ti binary oxide nanocomposite from ilmenite ore: An effective photocatalyst for dye degradation under visible light irradiation. J. Water Process. Eng. 2020, 37, 101474. [Google Scholar] [CrossRef]
- Chen, C.; Cai, W.; Long, M.; Zhou, B.; Wu, Y.; Wu, D.; Feng, Y. Synthesis of Visible-Light Responsive Graphene Oxide/TiO2 Composites with p/n Heterojunction. ACS Nano 2010, 4, 6425–6432. [Google Scholar] [CrossRef] [PubMed]
- Xu, T.G.; Zhang, L.W.; Cheng, H.Y.; Zhu, Y.F. Significantly enhanced photocatalytic performance of ZnO via graphene hybridization and the mechanism study. Appl. Catal. B 2011, 101, 382–387. [Google Scholar] [CrossRef]
- Zhu, M.; Chen, P.; Liu, M. Graphene Oxide Enwrapped Ag/AgX (X = Br, Cl) Nanocomposite as a Highly Efficient Visible-Light Plasmonic Photocatalyst. ACS Nano 2011, 5, 4529–4536. [Google Scholar] [CrossRef] [PubMed]
- Ng, Y.H.; Iwase, A.; Kudo, A.; Amal, R. Reducing Graphene Oxide on a Visible-Light BiVO4 Photocatalyst for an Enhanced Photoelectrochemical Water Splitting. J. Phys. Chem. Lett. 2010, 1, 2607–2612. [Google Scholar] [CrossRef]
- Zhang, X.Y.; Li, H.P.; Cui, X.L.; Lin, Y.H. Graphene/TiO2 nanocomposites: Synthesis, characterization and application in hydrogen evolution from water photocatalytic splitting. J. Mater. Chem. 2010, 20, 2801–2806. [Google Scholar] [CrossRef]
- Zhang, X.; Quan, X.; Chen, S.; Yu, H. Constructing graphene/InNbO4 composite with excellent adsorptivity and charge separation performance for enhanced visible-light-driven photocatalytic ability. Appl. Catal. B 2011, 105, 237–242. [Google Scholar] [CrossRef]
- Luciano, A.J.R.; Soletti, L.D.S.; Ferreira, M.E.C.; Cusioli, L.F.; Andrade, M.B.D.; Bergamasco, R.; Yamaguchi, N.U. Manganese ferrite dispersed over graphene sand composite for methylene blue photocatalytic degradation. J. Environ. Chem. Eng. 2020, 8, 104191. [Google Scholar] [CrossRef]
- Gratzel, M. Conversion of sunlight to electric power by nanocrystalline dye-sensitized solar cells. J. Photochem. Photobiol. A 2004, 164, 3–14. [Google Scholar] [CrossRef]
- Gao, E.; Wang, W.; Shanga, M.; Xua, J. Synthesis and enhanced photocatalytic performance of graphene-Bi2WO6 composite. Phys. Chem. Chem. Phys. 2011, 13, 2887–2893. [Google Scholar] [CrossRef] [PubMed]
- Fu, Y.; Chen, Q.; He, M.; Wan, Y.; Sun, X.; Xia, H.; Wang, X. Copper Ferrite-Graphene Hybrid: A Multifunctional Heteroarchitecture for Photocatalysis and Energy Storage. Ind. Eng. Chem. Res. 2012, 51, 11700–11709. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).