Hidradenitis Suppurativa in Association with Ulcerative Proctitis: Surgical Management in a Refractory Case to Topical and Systemic Treatment
Abstract
:1. Introduction
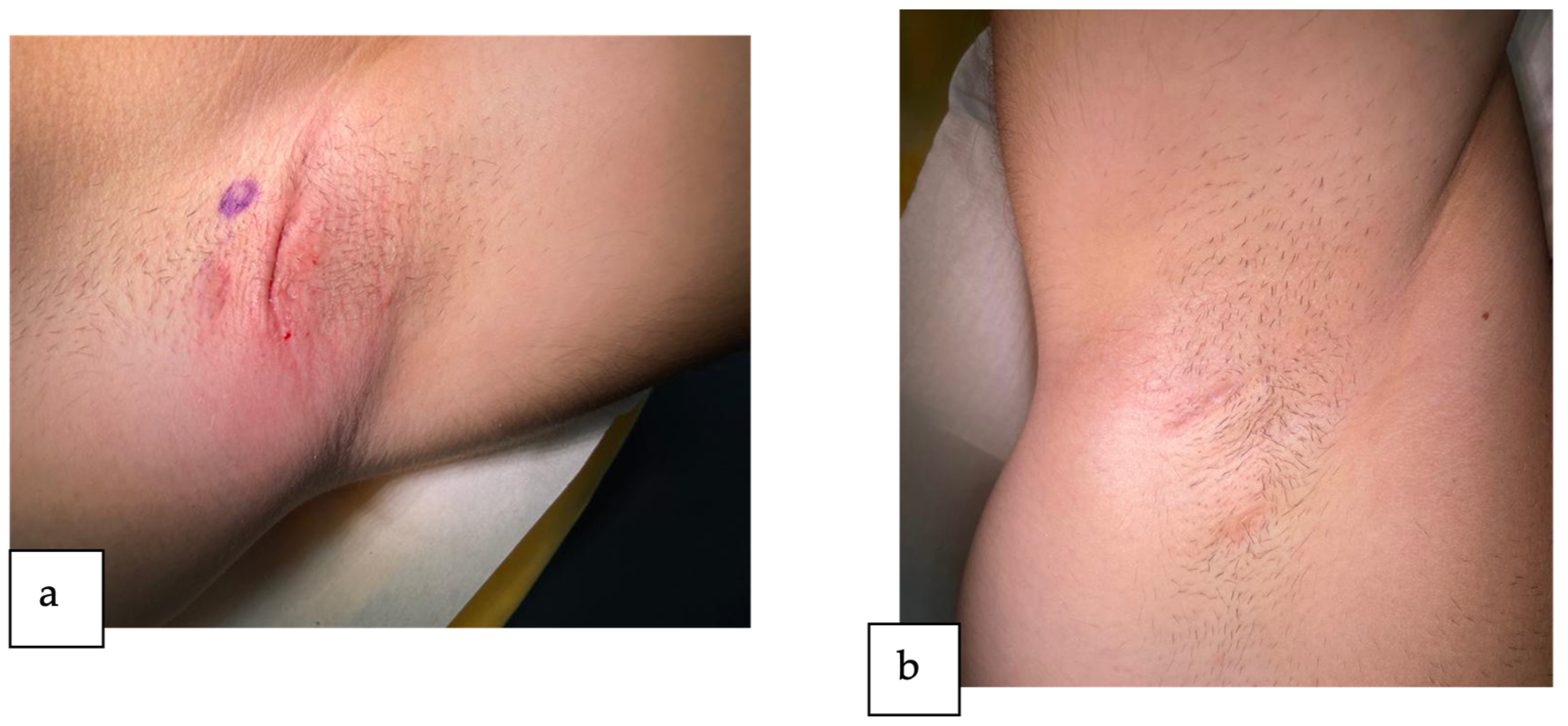
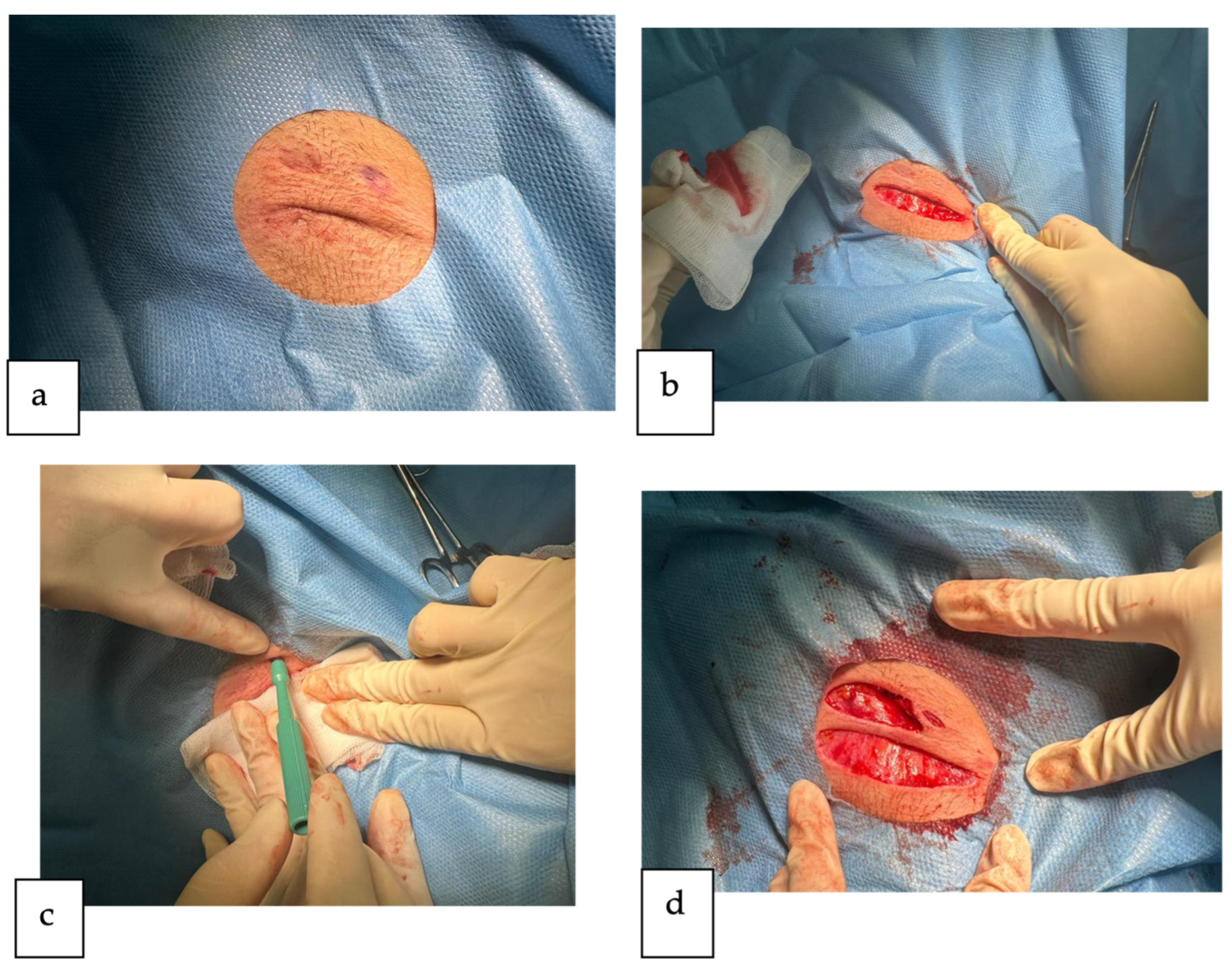
2. Detailed Case Description
3. Discussion
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| HS | Hidradenitis suppurativa |
| FPSUs | Folliculopilosebaceous units |
| IBD | Inflammatory bowel disease |
| CD | Crohn’s disease |
| UC | Ulcerative colitis |
| IHS4 | International Hidradenitis Suppurativa Severity Score System |
References
- Sellheyer, K.; Krahl, D. “Hidradenitis suppurativa” is acne inversa! An appeal to (finally) abandon a misnomer. Int. J. Dermatol. 2005, 44, 535–540. [Google Scholar] [CrossRef]
- Revuz, J. Hidradenitis suppurativa. J. Eur. Acad. Dermatol. Venereol. 2009, 23, 985–998. [Google Scholar] [CrossRef] [PubMed]
- Kouris, A.; Platsidaki, E.; Christodoulou, C.; Efstathiou, V.; Dessinioti, C.; Tzanetakou, V.; Korkoliakou, P.; Zisimou, C.; Antoniou, C.; Kontochristopoulos, G. Quality of Life and Psychosocial Implications in Patients with Hidradenitis Suppurativa. Dermatology 2016, 232, 687–691. [Google Scholar] [CrossRef] [PubMed]
- Ingram, J.R. The epidemiology of hidradenitis suppurativa. Br. J. Dermatol. 2020, 183, 990–998. [Google Scholar] [CrossRef] [PubMed]
- Garg, A.; Lavian, J.; Lin, G.; Strunk, A.; Alloo, A. Incidence of hidradenitis suppurativa in the United States: A sex- and age-adjusted population analysis. J. Am. Acad. Dermatol. 2017, 77, 118–122. [Google Scholar] [CrossRef] [PubMed]
- Kurzen, H.; Kurokawa, I.; Jemec, G.B.E.; Emtestam, L.; Sellheyer, K.; Giamarellos-Bourboulis, E.J.; Giamarellos-Bourboulis, E.J.; Nagy, I.; Bechara, F.G.; Sartorius, K.; et al. What causes hidradenitis suppurativa? Exp. Dermatol. 2008, 17, 455–456. [Google Scholar]
- Ingram, J.R.; Dellavalle, R.P.; Owen, C.; Ofori, A.O. Hidradenitis Suppurativa: Pathogenesis, Clinical Features, and Diagnosis; UpToDate; Wolters Kluwer: Alphen aan den Rijn, The Netherlands, 2023. [Google Scholar]
- Revuz, J.E.; Canoui-Poitrine, F.; Wolkenstein, P.; Viallette, C.; Gabison, G.; Pouget, F.; Poli, F.; Faye, O.; Roujeau, J.C.; Bonnelye, G.; et al. Prevalence and factors associated with hidradenitis suppurativa: Results from two case-control studies. J. Am. Acad. Dermatol. 2008, 59, 596–601. [Google Scholar] [CrossRef]
- Chen, W.-T.; Chi, C.-C. Association of Hidradenitis Suppurativa with Inflammatory Bowel Disease. JAMA Dermatol. 2019, 155, 1022–1027. [Google Scholar] [CrossRef]
- Van Der Zee, H.H.; Van Der Woude, C.J.; Florencia, E.F.; Prens, E.P. Hidradenitis suppurativa and inflammatory bowel disease: Are they associated? Results of a pilot study. Br. J. Dermatol. 2010, 162, 195–197. [Google Scholar] [CrossRef]
- Slade, D.E.M.; Powell, B.W.; Mortimer, P.S. Hidradenitis suppurativa: Pathogenesis and management. Br. J. Plast. Surg. 2003, 56, 451–461. [Google Scholar] [CrossRef]
- Deckers, I.E.; van der Zee, H.H.; Boer, J.; Prens, E.P. Correlation of early-onset hidradenitis suppurativa with stronger genetic susceptibility and more widespread involvement. J. Am. Acad. Dermatol. 2015, 72, 485–488. [Google Scholar] [CrossRef]
- Saunte, D.M.; Boer, J.; Stratigos, A.; Szepietowski, J.C.; Hamzavi, I.; Kim, K.H.; Zarchi, K.; Antoniou, C.; Matusiak, L.; Lim, H.W.; et al. Diagnostic delay in hidradenitis suppurativa is a global problem. Br. J. Dermatol. 2015, 173, 1546–1549. [Google Scholar] [CrossRef] [PubMed]
- Micheletti, R.G. Natural history, presentation, and diagnosis of hidradenitis suppurativa. Semin. Cutan. Med. Surg. 2014, 33, S51–S53. [Google Scholar] [CrossRef]
- Canoui-Poitrine, F.; Revuz, J.E.; Wolkenstein, P.; Viallette, C.; Gabison, G.; Pouget, F.; Poli, F.; Faye, O.; Bastuji-Garin, S. Clinical characteristics of a series of 302 French patients with hidradenitis suppurativa, with an analysis of factors associated with disease severity. J. Am. Acad. Dermatol. 2009, 61, 51–57. [Google Scholar] [CrossRef] [PubMed]
- Zouboulis, C.C.; Tzellos, T.; Kyrgidis, A.; Jemec, G.B.E.; Bechara, F.G.; Giamarellos-Bourboulis, E.J.; Ingram, J.R.; Kanni, T.; Karagiannidis, I.; Martorell, A.; et al. Development and validation of the International Hidradenitis Suppurativa Severity Score System (IHS4), a novel dynamic scoring system to assess HS severity. Br. J. Dermatol. 2017, 177, 1401–1409. [Google Scholar] [CrossRef] [PubMed]
- Simonart, T. Hidradenitis suppurativa and smoking. J. Am. Acad. Dermatol. 2010, 62, 149–150. [Google Scholar] [CrossRef] [PubMed]
- Nazary, M.; van der Zee, H.H.; Prens, E.P.; Folkerts, G.; Boer, J. Pathogenesis and pharmacotherapy of Hidradenitis suppurativa. Eur. J. Pharmacol. 2011, 672, 1–8. [Google Scholar] [CrossRef]
- Ingram, J.R.; Dellavalle, R.P.; Owen, C.; Ofori, A.O. Hidradenitis Suppurativa: Management; UpToDate; Wolters Kluwer: Alphen aan den Rijn, The Netherlands, 2023. [Google Scholar]
- Ellis, L.Z. Hidradenitis suppurativa: Surgical and other management techniques. Dermatol. Surg. 2012, 38, 517–536. [Google Scholar] [CrossRef]
- Deckers, I.E.; Benhadou, F.; Koldijk, M.J.; del Marmol, V.; Horváth, B.; Boer, J.; van der Zee, H.H.; Prens, E.P. Inflammatory bowel disease is associated with hidradenitis suppurativa: Results from a multicenter cross-sectional study. J. Am. Acad. Dermatol. 2017, 76, 49–53. [Google Scholar] [CrossRef]
- Zee HH van der Horvath, B.; Jemec, G.B.E.; Prens, E.P. The Association between Hidradenitis Suppurativa and Crohn’s Disease: In Search of the Missing Pathogenic Link. J. Investig. Dermatol. 2016, 136, 1747–1748. [Google Scholar] [CrossRef]
- Bao, B.; Zhu, C.; Shi, J.; Lu, C. Causal association between inflammatory bowel disease and hidradenitis suppurativa: A two-sample bidirectional Mendelian randomization study. Front. Immunol. 2023, 14, 1071616. Available online: https://www.frontiersin.org/articles/10.3389/fimmu.2023.1071616 (accessed on 27 December 2023). [CrossRef] [PubMed]
- Kamal, N.; Cohen, B.L.; Buche, S.; Delaporte, E.; Colombel, J.-F. Features of Patients with Crohn’s Disease and Hidradenitis Suppurativa. Clin. Gastroenterol. Hepatol. 2016, 14, 71–79. [Google Scholar] [CrossRef] [PubMed]
- Orgill, D.P.; Jeschke, M.G.; Owen, C. Surgical Management of Hidradenitis Suppurativa; UpToDate; Wolters Kluwer: Alphen aan den Rijn, The Netherlands, 2023. [Google Scholar]
- Kohorst, J.J.; Baum, C.L.; Otley, C.C.; Roenigk, R.K.; Schenck, L.A.; Pemberton, J.H.; Dozois, E.J.; Tran, N.V.; Senchenkov, A.; Davis, M.D.P. Surgical Management of Hidradenitis Suppurativa: Outcomes of 590 Consecutive Patients. Dermatol. Surg. 2016, 42, 1030–1040. [Google Scholar] [CrossRef] [PubMed]
- Zouboulis, C.C.; Desai, N.; Emtestam, L.; Hunger, R.E.; Ioannides, D.; Juhász, I.; Lapins, J.; Matusiak, L.; Prens, E.P.; Revuz, J.; et al. European S1 guideline for the treatment of hidradenitis suppurativa/acne inversa. J. Eur. Acad. Dermatol. Venereol. 2015, 29, 619–644. [Google Scholar] [CrossRef] [PubMed]
- Nesbitt, E.; Clements, S.; Driscoll, M. A concise clinician’s guide to therapy for hidradenitis suppurativa. Int. J. Women’s Dermatol. 2020, 6, 80. [Google Scholar] [CrossRef] [PubMed]
- Riis, P.T.; Boer, J.; Prens, E.P.; Saunte, D.M.L.; Deckers, I.E.; Emtestam, L.; Sartorius, K.; Jemec, G.B.E. Intralesional triamcinolone for flares of hidradenitis suppurativa (HS): A case series. J. Am. Acad. Dermatol. 2016, 75, 1151–1155. [Google Scholar] [CrossRef] [PubMed]
- Danby, F.W.; Hazen, P.G.; Boer, J. New and traditional surgical approaches to hidradenitis suppurativa. J. Am. Acad. Dermatol. 2015, 73, S62–S65. [Google Scholar] [CrossRef] [PubMed]
- Boer, J.; Jemec, G.B.E. Resorcinol peels as a possible self-treatment of painful nodules in hidradenitis suppurativa. Clin. Exp. Dermatol. 2010, 35, 36–40. [Google Scholar] [CrossRef] [PubMed]
- Kimball, A.B.; Okun, M.M.; Williams, D.A.; Gottlieb, A.B.; Papp, K.A.; Zouboulis, C.C.; Armstrong, A.W.; Kerdel, F.; Gold, M.H.; Forman, S.B.; et al. Two Phase 3 Trials of Adalimumab for Hidradenitis Suppurativa. N. Engl. J. Med. 2016, 375, 422–434. [Google Scholar] [CrossRef]
- PubMed. Four-Weekly Infliximab in the Treatment of Severe Hidradenitis Suppurativa. Available online: https://pubmed.ncbi.nlm.nih.gov/24641123/ (accessed on 12 December 2023).
- Kimball, A.B.; Jemec, G.B.E.; Alavi, A.; Reguiai, Z.; Gottlieb, A.B.; Bechara, F.G.; Paul, C.; Bourboulis, E.J.G.; Villani, A.P.; Schwinn, A.; et al. Secukinumab in moderate-to-severe hidradenitis suppurativa (SUNSHINE and SUNRISE): Week 16 and week 52 results of two identical, multicentre, randomised, placebo-controlled, double-blind phase 3 trials. Lancet 2023, 401, 747–761. [Google Scholar] [CrossRef]
- van der Zee, H.H.; Prens, E.P.; Boer, J. Deroofing: A tissue-saving surgical technique for the treatment of mild to moderate hidradenitis suppurativa lesions. J. Am. Acad. Dermatol. 2010, 63, 475–480. [Google Scholar] [CrossRef] [PubMed]
- Taylor, E.M.; Hamaguchi, R.; Kramer, K.M.; Kimball, A.B.; Orgill, D.P. Plastic Surgical Management of Hidradenitis Suppurativa. Plast. Reconstr. Surg. 2021, 147, 479. [Google Scholar] [CrossRef] [PubMed]
- Janse, I.; Bieniek, A.; Horváth, B.; Matusiak, Ł. Surgical Procedures in Hidradenitis Suppurativa. Dermatol. Clin. 2016, 34, 97–109. [Google Scholar] [CrossRef] [PubMed]


Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Stoenică, I.-V.; Dumitrașcu, M.C.; Petca, A.; Petca, R.-C.; Șandru, F. Hidradenitis Suppurativa in Association with Ulcerative Proctitis: Surgical Management in a Refractory Case to Topical and Systemic Treatment. Reports 2024, 7, 13. https://doi.org/10.3390/reports7010013
Stoenică I-V, Dumitrașcu MC, Petca A, Petca R-C, Șandru F. Hidradenitis Suppurativa in Association with Ulcerative Proctitis: Surgical Management in a Refractory Case to Topical and Systemic Treatment. Reports. 2024; 7(1):13. https://doi.org/10.3390/reports7010013
Chicago/Turabian StyleStoenică, Ioana-Valentina, Mihai Cristian Dumitrașcu, Aida Petca, Răzvan-Cosmin Petca, and Florica Șandru. 2024. "Hidradenitis Suppurativa in Association with Ulcerative Proctitis: Surgical Management in a Refractory Case to Topical and Systemic Treatment" Reports 7, no. 1: 13. https://doi.org/10.3390/reports7010013






