Arbuscular Mycorrhizal Fungi Inoculation Reduced the Growth of Pre-Rooted Olive Cuttings in a Greenhouse
Abstract
:1. Introduction
2. Materials and Methods
2.1. Establishment and Management of the Trials
2.2. Leaf Gas Exchange
2.3. Soil and Plant Sampling and Analyses
2.4. Data Analysis
3. Results
3.1. Plant Dry Matter Yield
3.2. Plant Morphological Traits
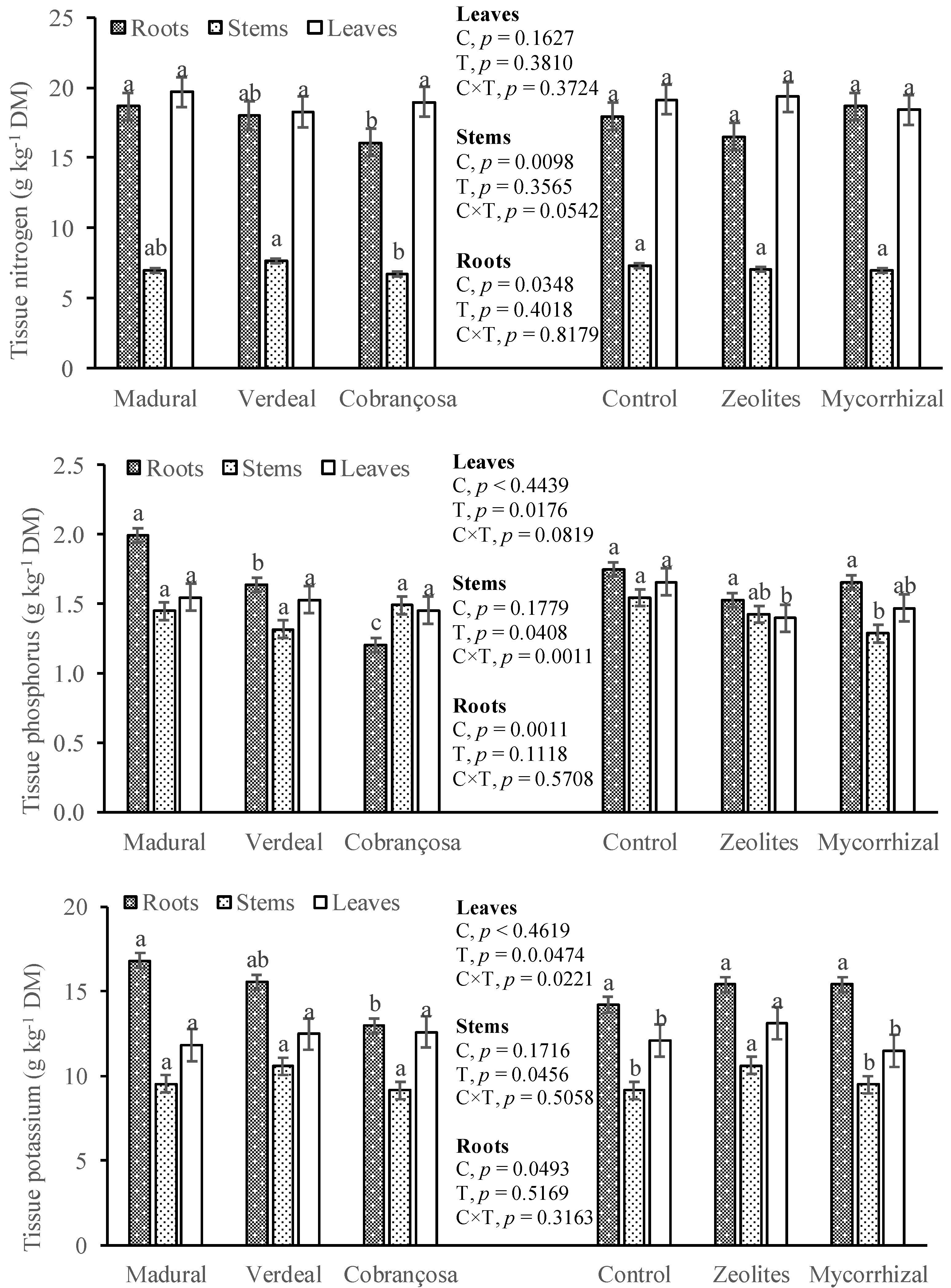
3.3. Tissue Nutrient Concentration
3.4. Leaf Gas Exchange
3.5. Soil Properties
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Bonfante, P.; Perotto, S. Strategies of arbuscular mycorrhizal fungi when infecting host plants. New Phytol. 1995, 130, 3–21. [Google Scholar] [CrossRef]
- Lanfranco, L.; Bonfante, P.; Genre, A. The mutualistic interaction between plants and arbuscular mycorrhizal fungi. Microbiol. Spectr. 2016, 4, 1–20. [Google Scholar] [CrossRef]
- López-Ráez, J.A.; Pozo, M.J. Chemical signalling in the arbuscular mycorrhizal symbiosis: Biotechnological applications. In Symbiotic Endophytes; Aroca, R., Ed.; Springer: Berlin/Heidelberg, Germany, 2013; pp. 215–232. [Google Scholar]
- Calvo-Polanco, M.; Sánchez-Romera, B.; Aroca, R. Arbuscular mycorrhizal fungi and the tolerance of plants to drought and salinity. In Symbiotic Endophytes; Aroca, R., Ed.; Springer: Berlin/Heidelberg, Germany, 2013; pp. 271–288. [Google Scholar]
- Luciani, E.; Palliotti, A.; Tombesi, S.; Gardi, T.; Michel, M.; Berrios, J.G.; Zadra, C.; Farinelli, D. Mitigation of multiple summer stresses on hazelnut (Corylus avellana L.): Effects of the new arbuscular mycorrhiza Glomus iranicum tenuihypharum sp. nova. Sci. Hortic. 2019, 257, 108659. [Google Scholar] [CrossRef]
- Dag, A.; Yermiyahu, U.; Ben-Gal, A.; Zipori, I.; Kapulnik, Y. Nursery and post-transplant field response of olive trees to arbuscular mycorrhizal fungi in an arid region. Crop Pasture Sci. 2009, 60, 427–433. [Google Scholar] [CrossRef]
- Bati, C.B.; Santilli, E.; Lombardo, L. Effect of arbuscular mycorrhizal fungi on growth and on micronutrient and macronutrient uptake and allocation in olive plantlets growing under high total Mn levels. Mycorrhiza 2015, 25, 97–108. [Google Scholar] [CrossRef]
- Ortas, I.; Bykova, A. The effect of mycorrhiza inoculation and phosphorus application on phosphorus efficiency of wheat plants. Commun. Soil Sci. Plant Anal. 2018, 49, 1199–1207. [Google Scholar] [CrossRef]
- Rodrigues, M.A.; Piroli, L.B.; Forcelini, D.; Raimundo, S.; Domingues, L.S.; Cassol, L.C.; Correia, C.M.; Arrobas, M. Use of commercial mycorrhizal fungi in stress-free growing conditions of potted olive cuttings. Sci. Hortic. 2021, 275, 109712. [Google Scholar] [CrossRef]
- Tanwar, A.; Aggarwal, A.; Kaushish, S.; Chauhan, S. Interactive effect of AM fungi with Trichoderma viride and Pseudomonas fluorescens on growth and yield of broccoli. Plant Protect. Sci. 2013, 49, 137–145. [Google Scholar] [CrossRef] [Green Version]
- Valentine, A.J.; Mortimer, P.E.; Kleinert, A.; Kang, Y.; Benedito, V.A. Carbon metabolism and costs of arbuscular mycorrhizal associations to host roots. In Symbiotic Endophytes; Aroca, R., Ed.; Springer: Berlin/Heidelberg, Germany, 2013; pp. 233–252. [Google Scholar]
- Marschner, H.; Dell, B. Nutrient uptake in mycorrhizai symbiosis. Plant Soil 1994, 159, 89–102. [Google Scholar] [CrossRef]
- Koller, R.; Rodriguez, A.; Robin, C.; Scheu, S.; Bonkowski, M. Protozoa enhance foraging efficiency of arbuscular mycorrhizal fungi for mineral nitrogen from organic matter in soil to the benefit of host plants. New Phytol. 2013, 199, 203–211. [Google Scholar] [CrossRef]
- Garcia, K.; Zimmermann, S.D. The role of mycorrhizal associations in plant potassium nutrition. Front. Plant Sci. 2014, 17. [Google Scholar] [CrossRef] [Green Version]
- Tekaya, M.; Mechri, B.; Mbarki, N.; Cheheb, H.; Hammami, M.; Attia, F. Arbuscular mycorrhizal fungus Rhizophagus irregularis influences key physiological parameters of olive trees (Olea europaea L.) and mineral nutrient profile. Photosynthetica 2017, 55, 308–316. [Google Scholar] [CrossRef]
- Ouledali, S.; Ennajeh, M.; Zrig, A.; Gianinazzi, S.; Khemira, H. Estimating the contribution of arbuscular mycorrhizal fungi to drought tolerance of potted olive trees (Olea europaea). Acta Physiol. Plant 2018, 40, 80–93. [Google Scholar] [CrossRef]
- Mechri, B.; Tekaya, M.; Attia, F.; Hammami, M.; Chehab, H. Drought stress improved the capacity of Rhizophagus irregularis for inducing the accumulation of oleuropein and mannitol in olive (Olea europaea) roots. Plant Physiol. Biochem. 2020, 156, 178–191. [Google Scholar] [CrossRef]
- Zhan, F.; Li, B.; Jiang, M.; Yue, X.; He, Y.; Xia, Y.; Wang, Y. Arbuscular mycorrhizal fungi enhance antioxidant defence in the leaves and the retention of heavy metals in the roots of maize. Environ. Sci. Pollut. Res. 2018, 25, 24338–24347. [Google Scholar] [CrossRef] [PubMed]
- García-Sánchez, M.; Silva-Castro, G.A.; Sanchez, A.; Arriagada, C.; García-Romera, I. Effect of arbuscular mycorrhizal fungi and mycoremediated dry olive residue in lead uptake in wheat plants. Appl. Soil Ecol. 2021, 159, 103838. [Google Scholar] [CrossRef]
- Hassena, A.B.; Zouari, M.; Trabelsi, L.; Decou, R.; Amar, F.B.; Chaari, A.; Soua, N.; Labrousse, P.; Khabou, W.; Zouari, N. Potential effects of arbuscular mycorrhizal fungi in mitigating the salinity of treated wastewater in young olive plants (Olea europaea L. cv. Chetoui). Agric. Water Manag. 2021, 245, 106635. [Google Scholar] [CrossRef]
- Berdeni, D.; Cotton, T.E.A.; Daniell, T.J.; Bidartondo, M.I.; Cameron, D.D.; Evans, K.L. The effects of arbuscular mycorrhizal fungal colonisation on nutrient status, growth, productivity, and canker resistance of apple (Malus pumila). Front. Microbiol. 2018, 9, 1461. [Google Scholar] [CrossRef] [PubMed]
- Godbold, D.L.; Hoosbeek, M.R.; Lukac, M.; Cotrufo, M.F.; Janssens, I.A.; Ceulemans, R.; Polle, A.; Velthorst, E.J.; Scarascia-Mugnozza, G.; Angelis, P.; et al. Mycorrhizal hyphal turnover as a dominant process for carbon input into soil organic matter. Plant Soil 2006, 281, 15–24. [Google Scholar] [CrossRef] [Green Version]
- Leifheit, E.F.; Verbruggen, E.; Rillig, M.C. Arbuscular mycorrhizal fungi reduce decomposition of woody plant litter while increasing soil aggregation. Soil Biol. Biochem. 2015, 81, 323–328. [Google Scholar] [CrossRef]
- Harrison, M.J. Signaling in the arbuscular mycorrhizal symbiosis. Annu. Rev. Microbiol. 2005, 59, 19–42. [Google Scholar] [CrossRef]
- García-Garrido, J.M.; Lendzemo, V.; Castellanos-Morales, V.; Steinkellner, S.; Vierheilig, H. Strigolactones, signals for parasitic plants and arbuscular mycorrhizal fungi. Mycorrhiza 2009, 19, 449–459. [Google Scholar]
- Miransari, M. Arbuscular mycorrhizal fungi and uptake of nutrients. In Symbiotic Endophytes; Aroca, R., Ed.; Springer: Berlin/Heidelberg, Germany, 2013; pp. 253–270. [Google Scholar]
- Ouledali, S.; Ennajeh, M.; Ferrandino, A.; Khemira, H.; Schubert, A.; Secchi, F. Influence of arbuscular mycorrhizal fungi inoculation on the control of stomata functioning by abscisic acid (ABA) in drought-stressed olive plants. S. Afr. J. Bot. 2019, 121, 152–158. [Google Scholar] [CrossRef]
- Seifi, E.; Teymoor, Y.S.; Alizadeh, M.; Fereydooni, H. Olive mycorrhization: Influences of genotype, mycorrhiza, and growing periods. Sci. Hortic. 2014, 180, 214–219. [Google Scholar] [CrossRef]
- Jiménez-Moreno, M.J.; Moreno-Márquez, M.C. Moreno-Alías, I.; Rapoport, H.; Fernández-Escobar, R. Interaction between mycorrhization with Glomus intraradices and phosphorus in nursery olive plants. Sci. Hortic. 2018, 233, 249–255. [Google Scholar] [CrossRef]
- von Caemmerer, S.; Farquhar, G.D. Some relationships between the biochemistry of photosynthesis and the gas exchange of leaves. Planta 1981, 153, 376–387. [Google Scholar] [CrossRef] [PubMed]
- Van Reeuwijk, L.P. Procedures for Soil Analysis, 6th ed.; Technical Paper 9; International Soil Reference and Information Centre: Wageningen, The Netherlands, 2002. [Google Scholar]
- Balbino, L.R. La méthode Egner-Riehm et la détermination du phosfore et du potassium «assimilável» des sols du Portugal. In II Col. Medit Cont. Fert. Plantas Cultivadas; Facultad de Ciencias: Sevilla, Spain, 1968; pp. 55–65. [Google Scholar]
- Alef, K.; Nannipieri, P.; Trazar-Cepeda, C. Phosphatase activity. In Methods in Applied Soil Microbiology and Biochemistry; Alef, K., Nannipieri, P., Eds.; Academic Press: San Diego, CA, USA, 1995; pp. 214–215. [Google Scholar]
- Temminghoff, E.E.J.M.; Houba, V.G. Plant Analysis Porocedures, 2nd ed.; Klwuwer Academic Publishers: Dordrecht, The Netherlands, 2004. [Google Scholar]
- Jarrel, W.M.; Beverly, R.B. The dilution effect in plant nutrition studies. Adv. Agron. 1981, 34, 197–224. [Google Scholar]
- Arrobas, M.; Ferreira, I.Q.; Afonso, S.; Rodrigues, M.A. Sufficiency ranges and crop nutrient removals for peppermint (Mentha × piperita L.) established from field and pot fertilizer experiments. Comm. Soil Sci. Plant Anal. 2018, 49, 1719–1730. [Google Scholar] [CrossRef] [Green Version]
- Schroeder, M.S.; Janos, D.P. Phosphorus and intraspecific density alter plant responses to arbuscular mycorrhizas. Plant Soil 2004, 264, 335–348. [Google Scholar] [CrossRef]
- Konvalinková, T.; Püschel, D.; Řezáčová, V.; Gryndlerová, H.; Jansa, J. Carbon flow from plant to arbuscular mycorrhizal fungi is reduced under phosphorus fertilization. Plant Soil 2017, 419, 319–333. [Google Scholar] [CrossRef]
- Russelle, M.P. Biological dinitrogen fixation in agriculture. In Nitrogen in Agricultural Systems; Schepers, J.S., Raun, W.R., Eds.; Agronomy Monograph n◦ 49; ASA, CSSA, SSSA: Madison, WI, USA, 2008; pp. 281–359. [Google Scholar]
- Cooper, J.E.; Scherer, H.W. Nitrogen fixation. In Marschner’s Mineral Nutrition of Higher Plants; Marschner, P., Ed.; Elsevier: London, UK, 2012; pp. 389–408. [Google Scholar]
- Ortas, I. Role of mycorrhizae on mineral nutrition of fruit trees. Acta Hortic. 2017, 1217. [Google Scholar] [CrossRef]
- Bitterlich, M.; Franken, P.; Graefe, J. Atmospheric drought and low light impede mycorrhizal effects on leaf photosynthesis—A glasshouse study on tomato under naturally fluctuating environmental conditions. Mycorrhiza 2019, 29, 13–28. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chandrasekaran, M.; Chanratana, M.; Kim, K.; Seshadri, S.; Sa, T. Impact of arbuscular mycorrhizal fungi on photosynthesis, water status, and gas exchange of plants under salt stress: A meta-analysis. Front. Plant Sci. 2019, 10, 457. [Google Scholar] [CrossRef] [PubMed]
- Ryakhovskaya, N.I.; Gainatulina, V.V. Potato and oat yield in short-cycle crop rotation with zeolite application. Russ. Agric. Sci. 2009, 35, 153–155. [Google Scholar] [CrossRef]
- Bernardi, A.C.D.C.; Bezerra, M.; Monte, D.M.; Renato, P.; Paiva, P.; Werneck, C.G. Dry matter production and nutrient accumulation after successive crops of lettuce, tomato, rice, and andropogon grass in a substrate with zeolite. Rev. Bras. Ciênc. Solo 2010, 34, 435–442. [Google Scholar] [CrossRef]
- Liator, M.I.; Katz, L.; Shenker, M. The influence of compost and zeolite co-addition on the nutrients status and plant growth in intensively cultivated Mediterranean soils. Soil Use Manag. 2017, 33, 72–80. [Google Scholar] [CrossRef] [Green Version]
- Guaya, D.; Mendoza, A.; Valderrama, C.; Farran, A.; Sauras-Year, T.; Cortina, J.L. Use of nutrient-enriched zeolite (NEZ) from urban wastewaters in amended soils: Evaluation of plant availability of mineral elements. Sci. Total Environ. 2020, 727, 138646. [Google Scholar] [CrossRef]
- Kieliszewska-Rokicka, B. Phosphate status and acid phosphatase activity in soil and ectomycorrhizas in two mature stands of scots pine (Pinus sylvestris L.) exposed to different levels of anthropogenic pollution. Acta Soc. Bot. Pol. 1999, 68, 311–317. [Google Scholar] [CrossRef] [Green Version]
- Egerton-Warburton, L. Aluminum-tolerant Pisolithus ectomycorrhizas confer increased growth, mineral nutrition, and metal tolerance to Eucalyptus in acidic mine soil. Appl. Environ. Soil Sci. 2015, 9, 803821. [Google Scholar] [CrossRef] [Green Version]



| Soil Properties | Value | Soil Properties (Cont.) | Value |
|---|---|---|---|
| 1 Clay (%) | 23.9 | 5 Extractable P (mg P2O5 kg−1) | 33.1 ± 2.82 |
| 1 Silt (%) | 21.8 | 5 Extractable K (mg K2O kg−1) | 55.4 ± 6.13 |
| 1 Sand (%) | 54.3 | 6 Exch. Ca (cmolc kg−1) | 21.7 ± 0.31 |
| 2 pH (H2O) | 6.2 ± 0.06 | 6 Exch. Mg (cmolc kg−1) | 5.3 ± 0.15 |
| 2 pH (KCl) | 5.4 ± 0.12 | 6 Exch. K (cmolc kg−1) | 0.3 ± 0.06 |
| 3 Organic C (g kg−1) | 8.5 ± 0.21 | 6 Exch. Na (cmolc kg−1) | 0.6 ± 0.10 |
| 4 Extractable B (mg kg−1) | 0.8 ± 0.06 | 7 Exch. acidity (cmolc kg−1) | 0.1 ± 0.00 |
| LN | ILA (cm2) | TLA (cm2 plant−1) | LMA (g m−2) | LWR (g g−1) | SWR (g g−1) | RWR (g g−1) | |
|---|---|---|---|---|---|---|---|
| Cultivar (C) | |||||||
| Madural | 66.7 a * | 3.5 b | 231.4 b | 137.7 ab | 0.42 c | 0.43 a | 0.15 a |
| Verdeal | 74.7 a | 3.5 b | 257.1 b | 133.1 b | 0.45 b | 0.41 a | 0.14 a |
| Cobrançosa | 76.9 a | 4.7 a | 350.8 a | 147.8 a | 0.51 a | 0.33 a | 0.16 a |
| Soil treatment (T) | |||||||
| Control | 78.4 a | 4.3 a | 338.2 a | 138.9 a | 0.47 a | 0.39 a | 0.15 a |
| Zeolites | 80.1 a | 3.5 b | 273.8 ab | 135.9 a | 0.45 a | 0.40 a | 0.15 a |
| Mycorrhizal | 59.8 b | 3.9 ab | 227.2 b | 143.7 a | 0.46 a | 0.39 a | 0.15 a |
| Prob. (C) | 0.3276 | <0.0001 | 0.0002 | 0.01 | <0.0001 | <0.0001 | 0.1211 |
| Prob. (T) | 0.0114 | 0.026 | 0.0010 | 0.2557 | 0.5524 | 0.7062 | 0.818 |
| Prob. (C × T) | 0.2962 | 0.0129 | 0.4404 | 0.1197 | 0.8832 | 0.6633 | 0.7298 |
| A (μmolm2s1) | gs (mmolm2s1) | A/gs (µmol mol1) | Ci/Ca | |
|---|---|---|---|---|
| Cultivar (C) | ||||
| Madural | 8.8 b * | 127.5 b | 69.9 a | 0.684 a |
| Verdeal | 9.9 ab | 166.5 ab | 60.3 a | 0.717 a |
| Cobrançosa | 11.0 a | 188.0 a | 63.9 a | 0.699 a |
| Soil treatment (T) | ||||
| Control | 8.7 b | 137.3 b | 64.8 a | 0.700 a |
| Zeolites | 8.9 b | 139.5 b | 68.4 a | 0.687 a |
| Mycorrhizal | 12.1 a | 204.9 a | 60.9 a | 0.712 a |
| Prob. (C) | 0.025 | 0.005 | 0.067 | 0.113 |
| Prob. (T) | <0.0001 | <0.0001 | 0.185 | 0.267 |
| Prob. (C × T) | <0.0001 | <0.0001 | 0.026 | 0.039 |
| A (μmolm−2s−1) | gs (mmolm−2s−1) | A/gs (µmol mol−1) | Ci/Ca | |
|---|---|---|---|---|
| Control | 10.3 b * | 180.1 b | 58.7 a | 0.720 a |
| Sterilised soil | 11.2 ab | 184.9 b | 58.8 a | 0.719 a |
| Mycorrhizal | 14.2 a | 272.2 a | 54.1 a | 0.732 a |
| Prob. | 0.039 | 0.029 | 0.659 | 0.699 |
| Organic C (g kg−1) | pH (H2O) | Extrac. P (mg kg−1) | Extrac. K (mg kg−1) | Exch. Ca (cmolc kg−1) | Exch. Mg (cmolc kg−1) | Exch. K (cmolc kg−1) | Exch. Na (cmolc kg−1) | CEC (cmolc kg−1) | APA (µg g−1 h−1) | |
|---|---|---|---|---|---|---|---|---|---|---|
| Cultivar (C) | ||||||||||
| Madural | 13.4 a * | 6.20 a | 63.4 a | 140.1 a | 9.86 ab | 1.94 a | 0.56 a | 0.44 a | 12.81 ab | 412.5 a |
| Verdeal | 14.3 a | 6.21 a | 70.4 a | 142.8 a | 9.99 a | 1.95 a | 0.50 a | 0.39 a | 12.84 a | 328.8 a |
| Cobrançosa | 13.9 a | 6.21 a | 69.5 a | 151.7 a | 8.91 b | 1.85 a | 0.52 a | 0.45 a | 11.74 b | 356.1 a |
| Soil treatment (T) | ||||||||||
| Control | 12.8 b | 6.19 b | 49.3 c | 57.4 c | 9.07 b | 1.78 b | 0.18 c | 0.31 b | 11.34 b | 344.9 a |
| Zeolites | 13.7 ab | 6.15 b | 68.9 b | 261.6 a | 10.13 a | 2.02 a | 1.05 a | 0.63 a | 13.84 a | 401.2 a |
| Mycorrhizal | 15.1 a | 6.28 a | 85.1 a | 115.7 b | 9.57 b | 1.94 ab | 0.36 b | 0.34 b | 12.22 b | 351.3 a |
| Prob (C) | 0.0833 | 0.9760 | 0.1817 | 0.6900 | 0.0155 | 0.4649 | 0.6025 | 0.6801 | 0.0275 | 0.2774 |
| Prob (T) | 0.0201 | 0.0004 | <0.0001 | <0.0001 | 0.0314 | 0.0270 | <0.0001 | <0.0001 | <0.0001 | 0.1538 |
| Prob (C×T) | 0.0068 | 0.0035 | 0.0791 | 0.7186 | 0.0625 | 0.0267 | 0.5758 | 0.9758 | 0.0593 | 0.7096 |
| Organic C (g kg−1) | pH (H2O) | Extrac. P (mg kg−1) | Extrac. K (mg kg−1) | Exch. Ca (cmolc kg−1) | Exch. Mg (cmolc kg−1) | Exch. K (cmolc kg−1) | Exch. Na (cmolc kg−1) | CTC (cmolc kg−1) | APA (µg g−1 h−1) | |
|---|---|---|---|---|---|---|---|---|---|---|
| Control | 12.9 a * | 6.09 b | 46.1 b | 56.0 b | 9.21 ab | 1.82 ab | 0.16 b | 0.28 a | 11.49 b | 348.8 a |
| Sterilised soil | 13.3 a | 6.18 ab | 51.4 b | 69.0 b | 8.16 b | 1.61 b | 0.18 b | 0.31 a | 10.26 b | 136.8 b |
| Mycorrhizal | 14.6 a | 6.29 a | 83.4 a | 100.7 a | 10.55 a | 2.15 a | 0.38 a | 0.36 a | 13.45 a | 418.3 a |
| Prob. | 0.1208 | 0.0107 | 0.0172 | <0.0001 | 0.0006 | 0.0011 | <0.0001 | 0.0433 | 0.0002 | 0.0005 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lopes, J.I.; Correia, C.M.; Gonçalves, A.; Silva, E.; Martins, S.; Arrobas, M.; Rodrigues, M.Â. Arbuscular Mycorrhizal Fungi Inoculation Reduced the Growth of Pre-Rooted Olive Cuttings in a Greenhouse. Soil Syst. 2021, 5, 30. https://doi.org/10.3390/soilsystems5020030
Lopes JI, Correia CM, Gonçalves A, Silva E, Martins S, Arrobas M, Rodrigues MÂ. Arbuscular Mycorrhizal Fungi Inoculation Reduced the Growth of Pre-Rooted Olive Cuttings in a Greenhouse. Soil Systems. 2021; 5(2):30. https://doi.org/10.3390/soilsystems5020030
Chicago/Turabian StyleLopes, João I., Carlos M. Correia, Alexandre Gonçalves, Ermelinda Silva, Sandra Martins, Margarida Arrobas, and Manuel Ângelo Rodrigues. 2021. "Arbuscular Mycorrhizal Fungi Inoculation Reduced the Growth of Pre-Rooted Olive Cuttings in a Greenhouse" Soil Systems 5, no. 2: 30. https://doi.org/10.3390/soilsystems5020030
APA StyleLopes, J. I., Correia, C. M., Gonçalves, A., Silva, E., Martins, S., Arrobas, M., & Rodrigues, M. Â. (2021). Arbuscular Mycorrhizal Fungi Inoculation Reduced the Growth of Pre-Rooted Olive Cuttings in a Greenhouse. Soil Systems, 5(2), 30. https://doi.org/10.3390/soilsystems5020030









