Is the Interfacial Electrochemical Behavior of Quercetin the Same as That of Catechol Plus Resorcinol?
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals
2.2. Electrodes and Electrode Cleaning
2.3. Electrochemical Deposition and Electrochemical Behavior of the Obtained Films
2.4. Surface Properties of the Quercetin-Based Films
3. Results
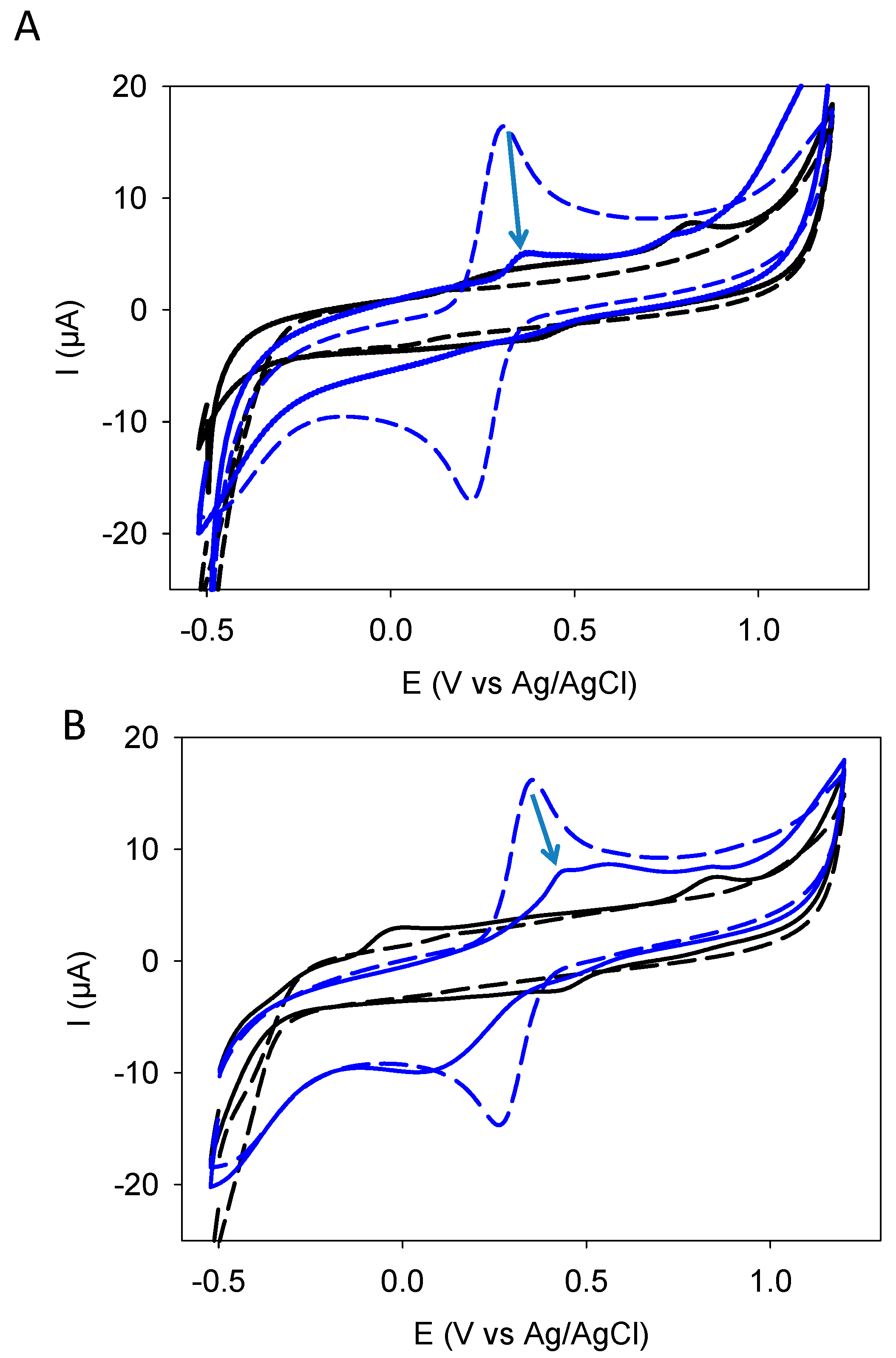
3.1. Deposition by CV
3.2. Permeability and Film Properties of the Quercetin Based Films
4. Discussion
5. Conclusions
Supplementary Materials
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Musiani, M.M.; Mengoli, G.; Furlanetto, F. Improved polyaniline coatings by electropolymerization. J. Appl. Polym. Sci. 1984, 29, 4433–4438. [Google Scholar] [CrossRef]
- Wang, X.; Shi, G.; Liang, Y. Low potential electropolymerization of thiophene at a copper oxide electrode. Electrochem. Comm. 1999, 1, 536–539. [Google Scholar] [CrossRef]
- Saidman, S.B.; Vela, M.E. Electroplymerization of pyrrole onto aluminium from alkaline solutions containing a surfactant. Thin Solid Films 2005, 493, 96–103. [Google Scholar] [CrossRef]
- Bejerano, T.; Forgacs, C.; Gileadi, E. Selective inhibition of electrode reactions by organic compounds. Electroanal. Chem. Interf. Electrochem. 1970, 2, 69–79. [Google Scholar] [CrossRef]
- Li, S.; Wang, H.; Young, M.; Xu, F.; Cheng, G.; Cong, H. Properties of electropolymerized dopamine and its analogues. Langmuir 2019, 35, 1119–1125. [Google Scholar] [CrossRef] [PubMed]
- Eom, T.; Lee, J.; Lee, S.; Ozlu, B.; Kim, S.; Martin, D.C.; Shim, B.S. Highly conductive polydopamine coatings by direct electrochemical synthesis on Au. ACS Appl. Polym. Mater. 2022, 4, 5319–5329. [Google Scholar] [CrossRef]
- Szewczyk, J.; Aguilar-Ferrer, D.; Coy, E. Polydopamine films: Electrochemical growth and sensing applications. Eur. Polym. J. 2022, 174, 111346. [Google Scholar] [CrossRef]
- Schindler, S.; Bechtold, T. Mechanistic insights into the electrochemical oxidation of dopamine by cyclic voltammetry. J. Electroanal. Chem. 2019, 836, 94–101. [Google Scholar] [CrossRef]
- Huynh, M.T.; Anson, C.W.; Cavell, A.C.; Stahl, S.S.; Hammes-Schiffer, S. Quinone 1e− and 2e−/2H+ reduction potentials: Identification and analysis of deviations from systematic scalling relationships. J. Am. Chem. Soc. 2016, 138, 15903–15910. [Google Scholar] [CrossRef] [PubMed]
- Enache, T.A.; Oliveira-Brett, A.M. Phenol and para-substituted phenols electrochemical oxidation pathways. J. Electroanal. Chem. 2011, 655, 9–16. [Google Scholar] [CrossRef]
- Barham, A.S.; Kennedy, B.M.; Cunnane, V.J.; Daous, M.A. The electrochemical polymerization of 1,2-dihydroxybenzee and 1,2-dihydroxybenzene alcohol prepared in different solution media. Electrochim. Acta 2014, 147, 19–24. [Google Scholar] [CrossRef]
- Ortiz Peña, N.; Ihiawakrim, D.; Ball, V.; Stanescu, S.; Rastei, M.; Sanchez, C.; Portehault, D.; Ersen, O. Correlative microscopy insight on electrodeposited ultrathin graphite oxide films. Phys. Chem. Lett. 2020, 11, 9117–9122. [Google Scholar] [CrossRef]
- Ball, V.; Alfieri, M.L.; Ziegler, K.; Arntz, Y.; d’Ischia, M. Structure-controlled electrodeposition and electrochemical behavior of films from isomeric diphenols at the solid-liquid interface. Surf. Interf. 2022, 30, 101841. [Google Scholar] [CrossRef]
- Quideau, S.; Deffieux, D.; Douat-Casassus, C.; Pouységu, L. Plant polyphenols: Chemical properties, biological activities, and synthesis. Angew. Chem. Int. Ed. 2011, 50, 586–621. [Google Scholar] [CrossRef]
- Ball, V. Electrodeposition of antioxidant films from resveratrol. Colloids Surf. A Physicochem. Eng. Asp. 2024, 686, 133374. [Google Scholar] [CrossRef]
- Kissinger, P.T.; Heineman, W.R. Cyclic voltammetry. J. Chem. Educ. 1983, 60, 702–706. [Google Scholar] [CrossRef]
- Blois, M.S. Antioxydant determinations by the use of a stable free radical. Nature 1958, 181, 1199–1200. [Google Scholar] [CrossRef]
- Gileadi, E. Physical Electrochemistry: Fundamentals, Techniques and Applications; Wiley-VCH: Weinheim, Germany, 2011; Chapters 15 and 16. [Google Scholar]
- Nady, H.; El-Rabiei, M.M.; Abd El-Hafez, G.M. Electrochemical Oxidation Behavior of Some Hazardous Phenolic Compounds in Acidic Solution. Egypt. J. Pet. 2017, 26, 669–678. [Google Scholar] [CrossRef]


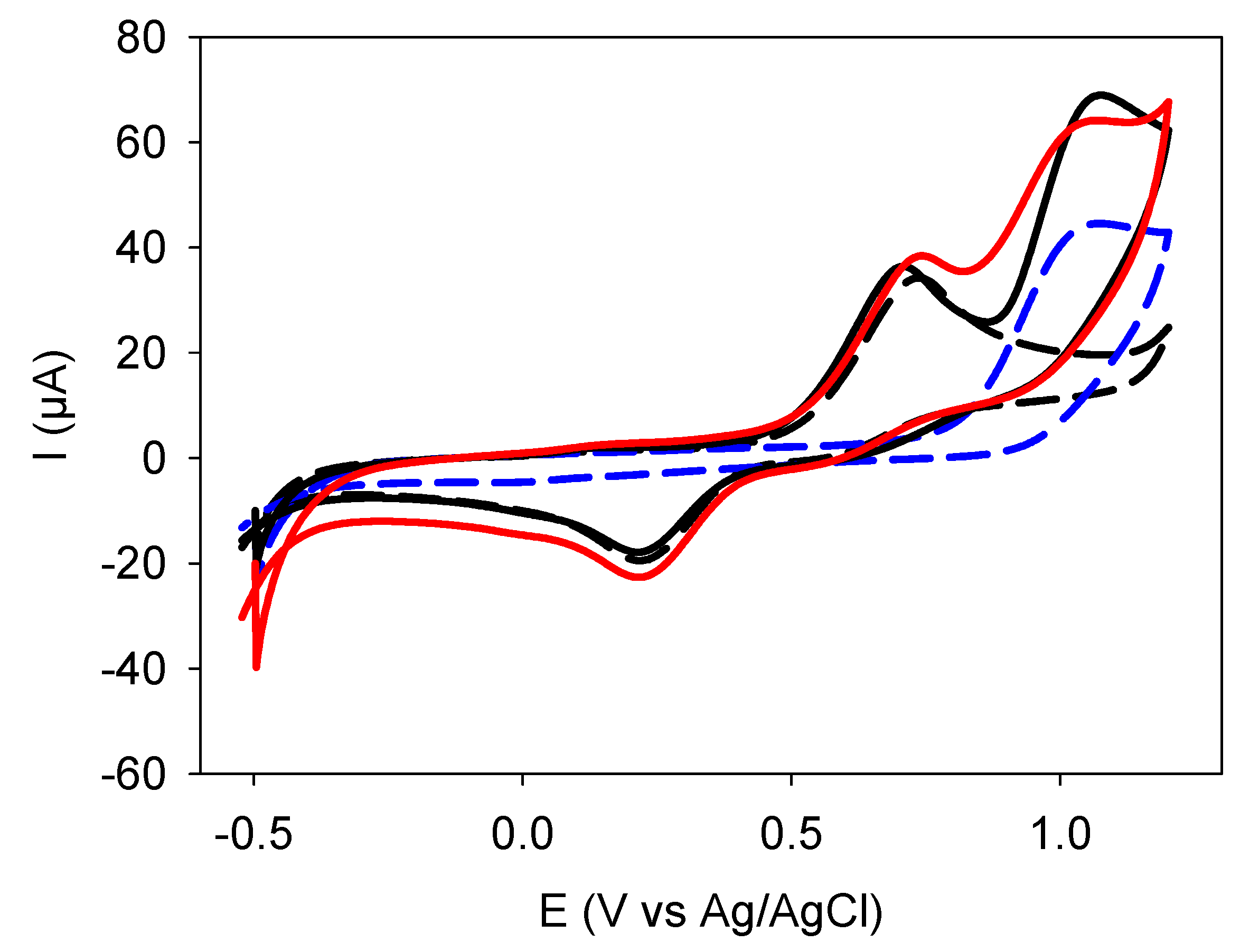
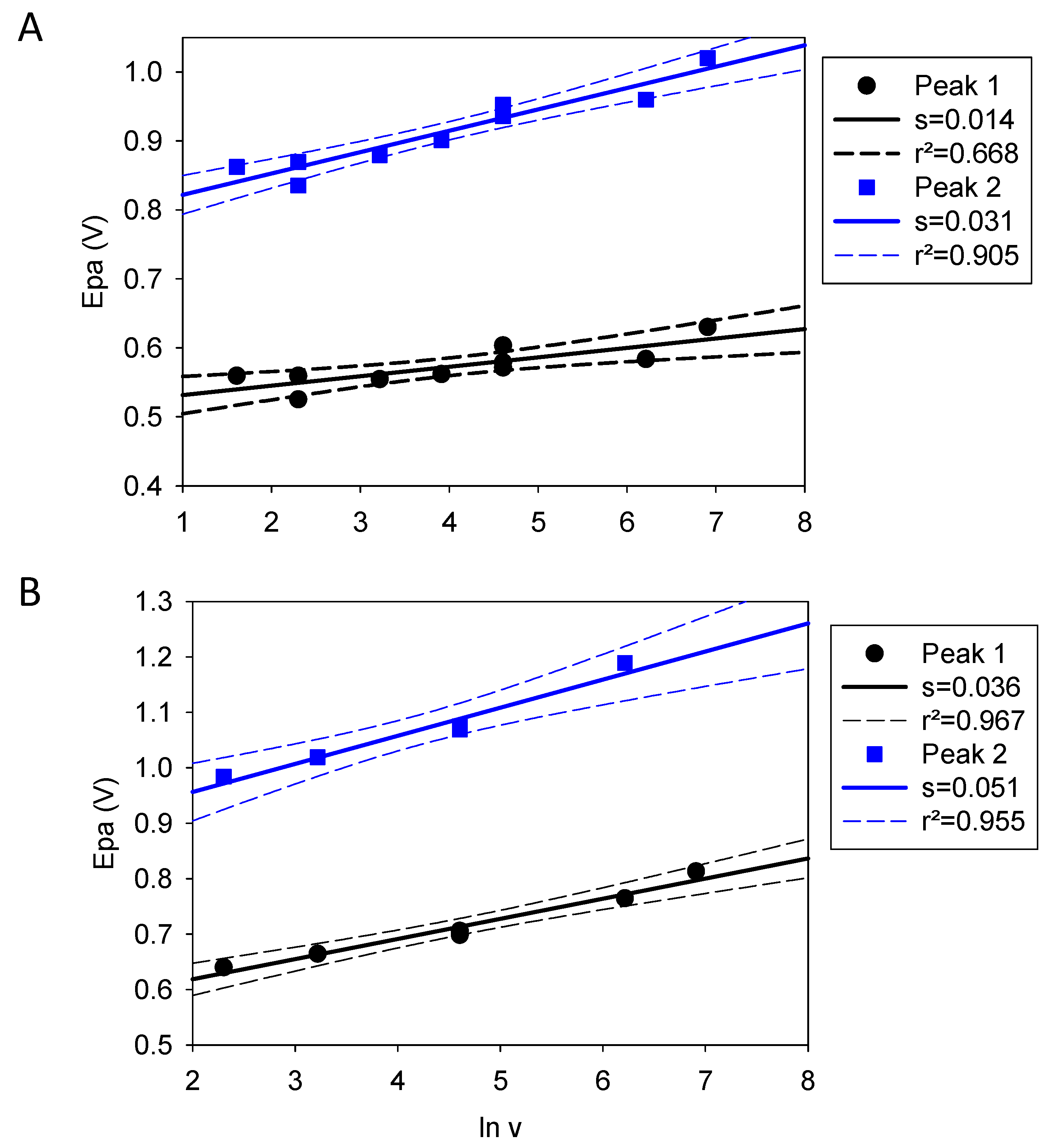
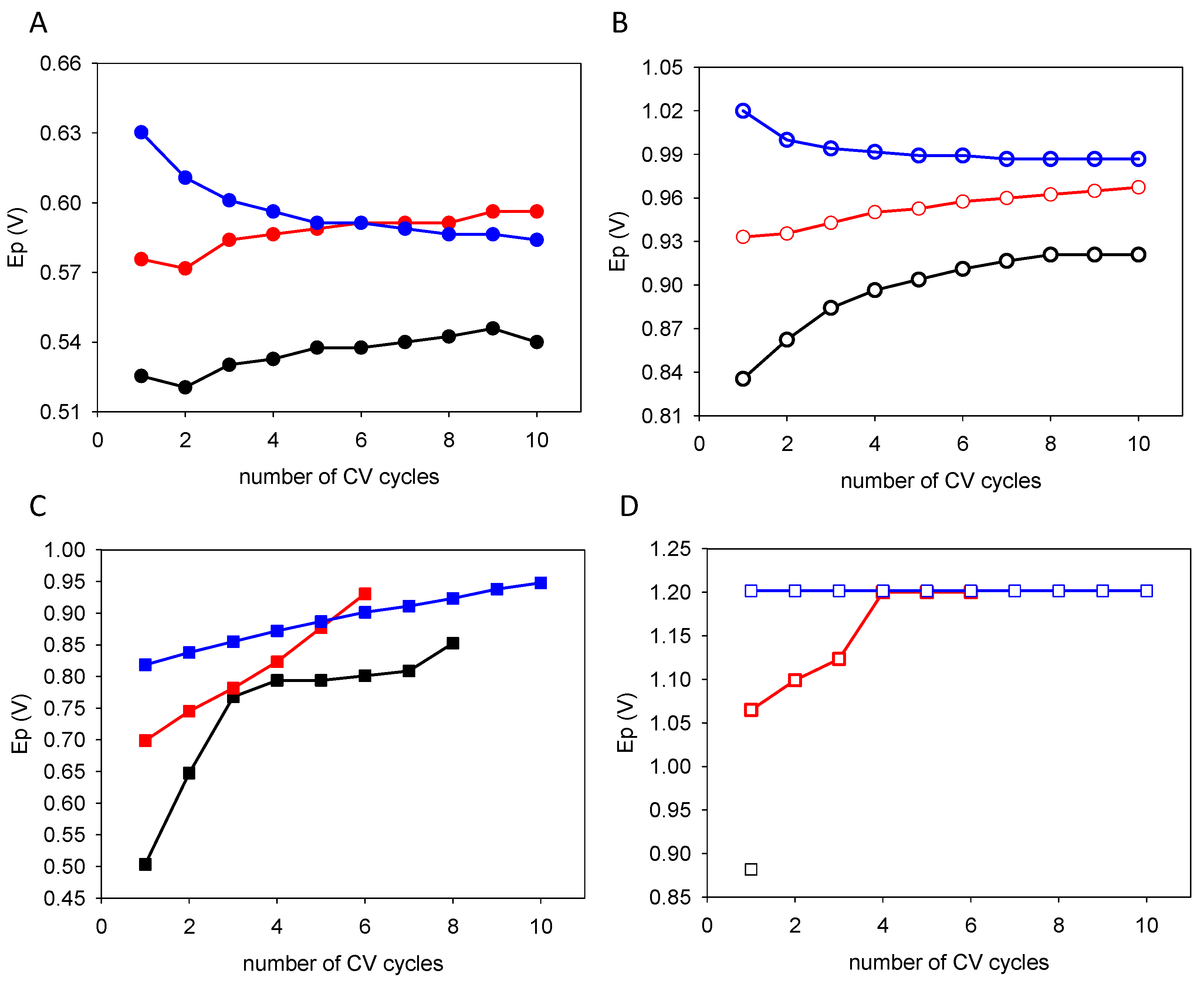
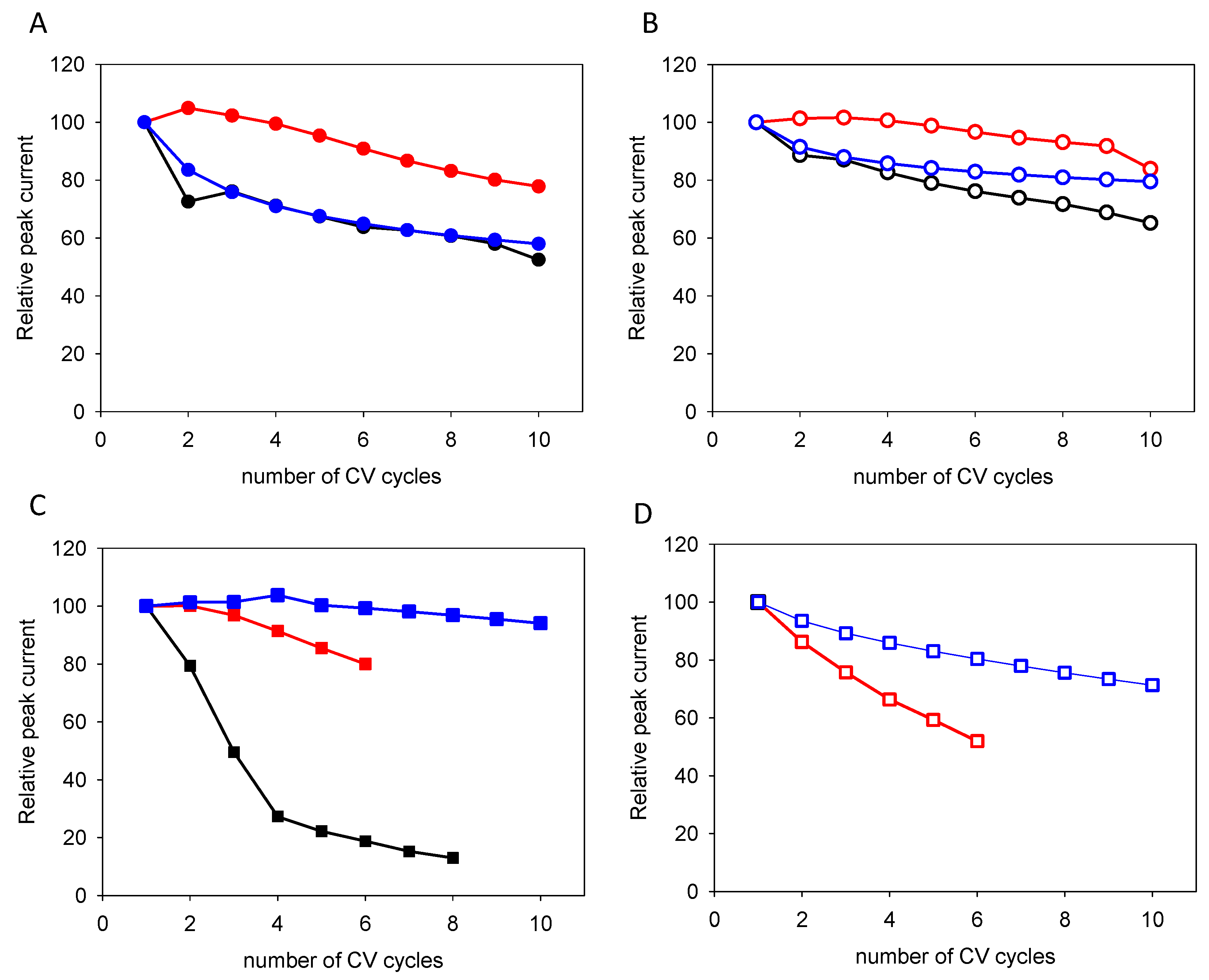

| Quercetin | Catechol + Resorcinol | |
|---|---|---|
| Peak 1 | ||
| Slope | 0.014 | 0.036 |
| Value of a | 0.93 | 0.36 |
| Peak 2 | ||
| Slope | 0.031 | 0.051 |
| Value of a | 0.42 | 0.25 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ball, V. Is the Interfacial Electrochemical Behavior of Quercetin the Same as That of Catechol Plus Resorcinol? Surfaces 2024, 7, 653-665. https://doi.org/10.3390/surfaces7030042
Ball V. Is the Interfacial Electrochemical Behavior of Quercetin the Same as That of Catechol Plus Resorcinol? Surfaces. 2024; 7(3):653-665. https://doi.org/10.3390/surfaces7030042
Chicago/Turabian StyleBall, Vincent. 2024. "Is the Interfacial Electrochemical Behavior of Quercetin the Same as That of Catechol Plus Resorcinol?" Surfaces 7, no. 3: 653-665. https://doi.org/10.3390/surfaces7030042
APA StyleBall, V. (2024). Is the Interfacial Electrochemical Behavior of Quercetin the Same as That of Catechol Plus Resorcinol? Surfaces, 7(3), 653-665. https://doi.org/10.3390/surfaces7030042






