Differences and Similarities in Breast and Colorectal Cancer Screening Uptake among Municipalities in Flanders, Belgium
Abstract
:1. Introduction
2. Methods
2.1. Study Setting and Data Source
2.2. Study Design and Objective
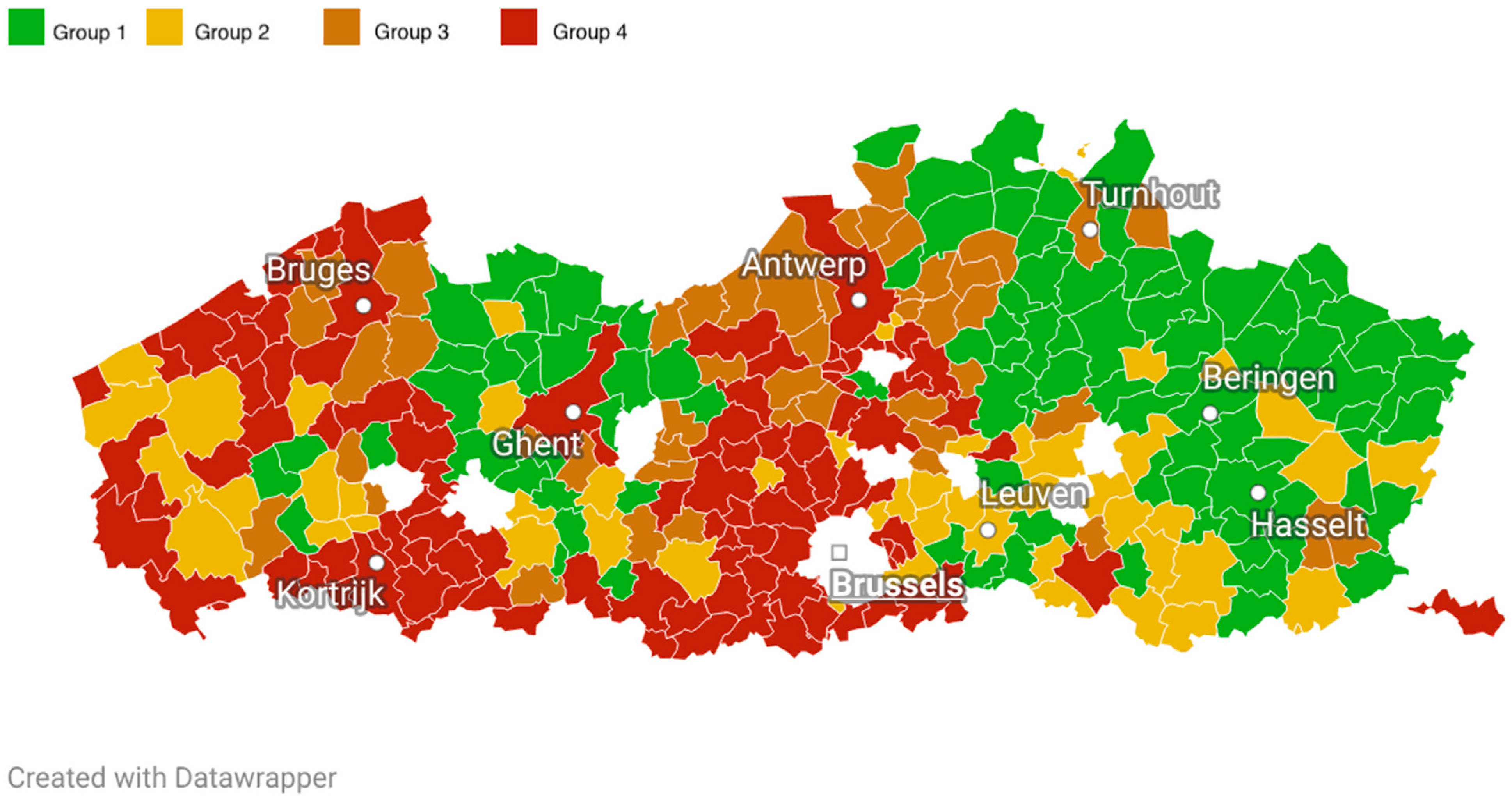
- Group 1: BC municipal uptake ≥ median uptake of Flemish municipalities, CRC municipal uptake ≥ median uptake of Flemish municipalities (“high BC, high CRC”);
- Group 2: BC municipal uptake ≥ median uptake of Flemish municipalities, CRC municipal uptake < median uptake of Flemish municipalities (“high BC, low CRC”);
- Group 3: BC municipal uptake < median uptake of Flemish municipalities, CRC municipal uptake ≥ median uptake of Flemish municipalities (“low BC, high CRC”);
- Group 4: BC municipal uptake < median uptake of Flemish municipalities, CRC municipal uptake < median uptake of Flemish municipalities (“low BC, low CRC”).
2.3. Determinants
- Age group (group 1: females aged between 55 and 59 years old; group 2: females aged between 60 and 64 years old; group 3: females aged between 65 and 69 years old) (%)
- Average household size (n)
- Residential stability (same address as previous year) (%)
- Having a partner (%)
- Having child(ren) (%)
- Foreign nationality (%)
- Socio-economic variables:
- Average income (EUR)
- Position in the labor market (job seekers, wage-earners, self-employed, (early) retired) (%)
- Students in higher education (%)
- Health-related variables:
- Chronic conditions (%)
- Diabetes (%)
- Disabilities (%)
- General practitioner (GP) visits (%)
- Preventive dental visits (%)
2.4. Covariates for Adjustment
2.5. Statistical Analysis
3. Results
3.1. Municipal Characteristics
3.2. Factors Associated with BC/CRC Screening Uptake
3.2.1. Factors Associated with Higher BC/CRC Screening Uptake
3.2.2. Factors Associated with Lower BC/CRC Screening Uptake
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- World Health Organization (WHO); International Agency for Research on Cancer (IARC). Global Cancer Observatory. Available online: https://gco.iarc.fr/ (accessed on 2 January 2021).
- Bray, F.; Ferlay, J.; Soerjomataram, I.; Siegel, R.L.; Torre, L.A.; Jemal, A. Global Cancer Statistics 2018: Globocan Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J. Clin. 2018, 68, 394–424. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Belgian Cancer Registry. Cancer Fact Sheets. Available online: https://kankerregister.org/Cancer_Fact_Sheets (accessed on 2 January 2021).
- Center, M.M.; Jemal, A.; Smith, R.A.; Ward, E. Worldwide Variations in Colorectal Cancer. CA. Cancer J. Clin. 2009, 59, 366–378. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gøtzsche, P.C.; Olsen, O. Is Screening for Breast Cancer with Mammography Justifiable? Lancet 2000, 355, 129–134. [Google Scholar] [CrossRef]
- Mandel, J.S.; Bond, J.H.; Church, T.R.; Snover, D.C.; Bradley, G.M.; Schuman, L.M.; Ederer, F. Reducing Mortality from Colorectal Cancer by Screening for Fecal Occult Blood. Minnesota Colon Cancer Control Study. N. Engl. J. Med. 1993, 328, 1365–1371. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nelson, H.D.; Fu, R.; Cantor, A.; Pappas, M.; Daeges, M.; Humphrey, L. Effectiveness of Breast Cancer Screening: Systematic Review and Meta-Analysis to Update the 2009 U.S. Preventive Services Task Force Recommendation. Ann. Intern. Med. 2016, 164, 244–255. [Google Scholar] [CrossRef]
- Lin, J.S.; Piper, M.A.; Perdue, L.A.; Rutter, C.M.; Webber, E.M.; O’Connor, E.; Smith, N.; Whitlock, E.P. Screening for Colorectal Cancer: Updated Evidence Report and Systematic Review for the US Preventive Services Task Force. JAMA 2016, 315, 2576–2594. [Google Scholar] [CrossRef] [Green Version]
- Centre for Cancer Detection. Monitoring Report of the Flemish Colorectal Cancer Screening Programme 2019. Available online: https://www.bevolkingsonderzoek.be (accessed on 2 January 2021).
- Centre for Cancer Detection. Available online: https://bevolkingsonderzoek.incijfers.be/jive (accessed on 1 February 2021).
- Flemish Provincial Authorities. Available online: https://provincies.incijfers.be/databank (accessed on 1 February 2021).
- Goossens, M.; Van Hal, G.; Van der Burg, M.; Kellen, E.; Van Herck, K.; De Grève, J.; Martens, P.; Van Limbergen, E. Quantifying Independent Risk Factors for Failing to Rescreen in a Breast Cancer Screening Program in Flanders, Belgium. Prev. Med. 2014, 69, 280–286. [Google Scholar] [CrossRef]
- Hoeck, S.; Pringels, S.; Kellen, E.; Van Herck, K.; Martens, P.; Van Limbergen, E.; Francart, J.; Van Hal, G. First Results of the Flemish Colorectal Cancer Screening Program: Start-up-Period Late 2013. Acta Gastro-Enterol. Belg. 2016, 79, 421–428. [Google Scholar]
- Directorate-General for Health and Consumers (European Commission); Executive Agency for Health and Consumers (European Commission); World Health Organization; von Karsa, L.; Patnick, J.; Segnan, N. European Guidelines for Quality Assurance in Colorectal Cancer Screening and Diagnosis; Publications Office of the European Union: Luxembourg, 2010.
- Directorate-General for Health and Consumers (European Commission); von Karsa, L.; Holland, R.; Broeders, M.; de Wolf, C.; Perry, N.; Törnberg, S. European Guidelines for Quality Assurance in Breast Cancer Screening and Diagnosis, 4th ed.; Publications Office of the European Union: Luxembourg, 2013.
- Velentgas, P.; Dreyer, N.A.; Nourjah, P.; Smith, S.R.; Torchia, M.M. (Eds.) Developing a Protocol for Observational Comparative Effectiveness Research: A User’s Guide; AHRQ Methods for Effective Health Care; Agency for Healthcare Research and Quality (US): Rockville, MD, USA, 2013. [Google Scholar]
- Tran, T.N.; Van Hal, G.; Peeters, M.; Jidkova, S.; De Schutter, H.; Hoeck, S. Population-Based Data Reveal Factors Associated with Organised and Non-Organised Colorectal Cancer Screening: An Important Step towards Improving Coverage. Int. J. Environ. Res. Public Health 2021, 18, 8373. [Google Scholar] [CrossRef]
- Ding, L.; Jidkova, S.; Greuter, M.J.W.; Van Herck, K.; Goossens, M.; Martens, P.; de Bock, G.H.; Van Hal, G. Coverage Determinants of Breast Cancer Screening in Flanders: An Evaluation of the Past Decade. Int. J. Equity Health 2020, 19, 212. [Google Scholar] [CrossRef]
- Textor, J.; van der Zander, B.; Gilthorpe, M.S.; Liskiewicz, M.; Ellison, G.T. Robust Causal Inference Using Directed Acyclic Graphs: The R Package “Dagitty”. Int. J. Epidemiol. 2016, 45, 1887–1894. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- de Jong, V.M.T.; Eijkemans, M.J.C.; van Calster, B.; Timmerman, D.; Moons, K.G.M.; Steyerberg, E.W.; van Smeden, M. Sample Size Considerations and Predictive Performance of Multinomial Logistic Prediction Models. Stat. Med. 2019, 38, 1601–1619. [Google Scholar] [CrossRef] [PubMed]
- R Foundation for Statistical Computing, Vienna, Austria. Available online: https://www.r-project.org/foundation/ (accessed on 20 November 2021).
- Giordano, L.; Von Karsa, L.; Tomatis, M.; Majek, O.; De Wolf, C.; Lancucki, L.; Hofvind, S.; Nystrom, L.; Segnan, N.; Ponti, A. Mammographic Screening Programmes in Europe: Organization, Coverage and Participation. J. Med. Screen. 2012, 19 (Suppl. S1), 72–82. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Navarro, M.; Nicolas, A.; Ferrandez, A.; Lanas, A. Colorectal Cancer Population Screening Programs Worldwide in 2016: An Update. World J. Gastroenterol. 2017, 23, 3632–3642. [Google Scholar] [CrossRef] [PubMed]
- Hoeck, S.; van de Veerdonk, W.; De Brabander, I.; Kellen, E. Does the Flemish Colorectal Cancer Screening Programme Reach Equity in FIT Uptake? Eur. J. Public Health 2019, 29, 1108–1114. [Google Scholar] [CrossRef] [PubMed]
- Ding, L.; Jidkova, S.; Greuter, M.J.W.; Van Herck, K.; Goossens, M.; De Schutter, H.; Martens, P.; Van Hal, G.; de Bock, G.H. The Role of Socio-Demographic Factors in the Coverage of Breast Cancer Screening: Insights from a Quantile Regression Analysis. Front. Public Health 2021, 9, 648278. [Google Scholar] [CrossRef] [PubMed]
- Hoeck, S.; Van Roy, K.; Willems, S. Barriers and Facilitators to Participate in the Colorectal Cancer Screening Programme in Flanders (Belgium): A Focus Group Study. Acta Clin. Belg. 2020, 77, 37–44. [Google Scholar] [CrossRef]
- Baumeister, A.; Aldin, A.; Chakraverty, D.; Monsef, I.; Jakob, T.; Seven, Ü.S.; Anapa, G.; Kalbe, E.; Skoetz, N.; Woopen, C. Interventions for Improving Health Literacy in Migrants. Cochrane Database Syst. Rev. 2019, 65, 54–64. [Google Scholar] [CrossRef]
- Rao Kondapally Seshasai, S.; Kaptoge, S.; Thompson, A.; Di Angelantonio, E.; Gao, P.; Sarwar, N.; Whincup, P.H.; Mukamal, K.J.; Gillum, R.F.; Holme, I.; et al. Emerging Risk Factors Collaboration. Diabetes Mellitus, Fasting Glucose, and Risk of Cause-Specific Death. N. Engl. J. Med. 2011, 364, 829–841. [Google Scholar] [CrossRef] [Green Version]
- Kellen, E.; Nuyens, C.; Molleman, C.; Hoeck, S. Uptake of Cancer Screening among Adults with Disabilities in Flanders (Belgium). J. Med. Screen. 2020, 27, 48–51. [Google Scholar] [CrossRef]
- Gofine, M.; Mielenz, T.J.; Vasan, S.; Lebwohl, B. Use of Colorectal Cancer Screening among People with Mobility Disability. J. Clin. Gastroent. 2018, 52, 789–795. [Google Scholar] [CrossRef] [PubMed]
- Steele, C.B.; Townsend, J.S.; Courtney-Long, E.A.; Young, M. Prevalence of Cancer Screening among Adults with Disabilities, United States, 2013. Prev. Chronic Dis. 2017, 14, 160312. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tran, T.N.; Ferrari, A.; Hoeck, S.; Peeters, M.; Van Hal, G. Colorectal Cancer Screening: Have We Addressed Concerns and Needs of the Target Population? Gastrointest. Disord. 2021, 3, 173–203. [Google Scholar] [CrossRef]
- Jensen, L.F.; Mukai, T.O.; Andersen, B.; Vedsted, P. The Association between General Practitioners Attitudes towards Breast Cancer Screening and Women’s Screening Participation. BMC Cancer 2012, 12, 254. [Google Scholar] [CrossRef] [Green Version]
- Cancer Screening in the European Union (2017). Report on the Implementation of the Council Recommendation on Cancer Screening (Second Report). Available online: https://screening.iarc.fr/EUreport.php (accessed on 21 January 2022).
- Kobayashi, L.C.; von Wagner, C.; Wardle, J. Perceived Life Expectancy Is Associated with Colorectal Cancer Screening in England. Ann. Behav. Med. Publ. Soc. Behav. Med. 2017, 51, 327–336. [Google Scholar] [CrossRef] [Green Version]
- Data from: Surveillance, Epidemiology, and End Results (SEER) Program, 2014–2018. Available online: https://seer.cancer.gov/explorer/application.html?site=20&data_type=1&graph_type=3&compareBy=sex&chk_sex_1=1&rate_type=2&race=1&advopt_precision=1&advopt_show_ci=on&advopt_display=2 (accessed on 21 January 2022).
- Achat, H.; Close, G.; Taylor, R. Who Has Regular Mammograms? Effects of Knowledge, Beliefs, Socioeconomic Status, and Health-Related Factors. Prev. Med. 2005, 41, 312–320. [Google Scholar] [CrossRef]
- Damiani, G.; Federico, B.; Basso, D.; Ronconi, A.; Bianchi, C.B.N.A.; Anzellotti, G.M.; Nasi, G.; Sassi, F.; Ricciardi, W. Socioeconomic Disparities in the Uptake of Breast and Cervical Cancer Screening in Italy: A Cross Sectional Study. BMC Public Health 2012, 12, 99. [Google Scholar] [CrossRef]
- Zackrisson, S.; Andersson, I.; Manjer, J.; Janzon, L. Non-Attendance in Breast Cancer Screening Is Associated with Unfavourable Socio-Economic Circumstances and Advanced Carcinoma. Int. J. Cancer 2004, 108, 754–760. [Google Scholar] [CrossRef]
- Hanske, J.; Meyer, C.P.; Sammon, J.D.; Choueiri, T.K.; Menon, M.; Lipsitz, S.R.; Noldus, J.; Nguyen, P.L.; Sun, M.; Trinh, Q.-D. The Influence of Marital Status on the Use of Breast, Cervical, and Colorectal Cancer Screening. Prev. Med. 2016, 89, 140–145. [Google Scholar] [CrossRef]
- Willems, B.; Bracke, P. The Impact of Regional Screening Policies on the Diffusion of Cancer Screening Participation in Belgium: Time Trends in Educational Inequalities in Flanders and Wallonia. BMC Health Serv. Res. 2018, 18, 943. [Google Scholar] [CrossRef]
- Sedgwick, P. Ecological Studies: Advantages and Disadvantages. BMJ 2014, 348, g2979. [Google Scholar] [CrossRef] [PubMed]

| Median (Range) | |||||
|---|---|---|---|---|---|
| 2014 | 2015 | 2016 | 2017 | ||
| Demographic variables | |||||
| Age group 1 (%) | high BC, high CRC | 38.0 (32.3–42.2) | 37.4 (27.6–42.7) | 37.6 (25.9–42.4) | 37.2 (29.9–42.8) |
| high BC, low CRC | 36.9 (27.3–43.2) | 37.5 (33.7–42.2) | 37.6 (26.1–42.2) | 37.7 (26.5–42.4) | |
| low BC, high CRC | 37.3 (33.9–41.5) | 37.1 (32.7–40.5) | 37.1 (32.6–41.5) | 37.6 (33.5–41.2) | |
| low BC, low CRC | 37.1 (29.2–43.0) | 37.1 (26.8–43.9) | 37.7 (29.7–42.8) | 37.3 (28.1–42.4) | |
| Age group 2 (%) | high BC, high CRC | 32.9 (29.1–37.1) | 33.1 (28.8–37.4) | 32.9 (28.2–37.3) | 33.1 (29.0–36.7) |
| high BC, low CRC | 32.5 (27.7–36.1) | 32.7 (29.7–36.5) | 33.2 (29.0–40.2) | 33.5 (28.5–35.9) | |
| low BC, high CRC | 32.7 (29.8–36.2) | 32.7 (30.1–36.4) | 33.1 (30.4–35.9) | 33.2 (29.7–37.1) | |
| low BC, low CRC | 32.6 (28.9–41.1) | 32.6 (28.3–44.0) | 33.1 (29.0–37.3) | 33.3 (24.7–38.1) | |
| Age group 3 (%) | high BC, high CRC | 29.2 (24.9–35.8) | 29.4 (24.9–35.8) | 29.4 (24.9–36.8) | 29.2 (25.9–39.0) |
| high BC, low CRC | 30.8 (25.1–37.9) | 29.4 (26.1–34.2) | 29.3 (25.1–38.7) | 29.0 (25.0–39.3) | |
| low BC, high CRC | 29.1 (25.9–33.3) | 30.1 (26.0–35.6) | 29.7 (25.2–33.3) | 29.2 (26.4–33.0) | |
| low BC, low CRC | 30.5 (24.9–38.4) | 30.2 (25.0–38.9) | 29.6 (25.6–37.0) | 29.5 (24.7–38.6) | |
| Average household size (n) | high BC, high CRC | 2.44 (2.18–2.61) | 2.42 (2.17–2.61) | 2.41 (2.17–2.62) | 2.42 (2.17–2.59) |
| high BC, low CRC | 2.41 (1.93–2.61) | 2.44 (2.01–2.65) | 2.41 (1.93–2.60) | 2.44 (1.94–2.70) | |
| low BC, high CRC | 2.45 (2.31–2.67) | 2.41 (2.24–2.55) | 2.41 (2.22–2.61) | 2.39 (2.22–2.52) | |
| low BC, low CRC | 2.41 (1.94–2.61) | 2.40 (1.93–2.66) | 2.41 (1.92–2.66) | 2.39 (1.91–2.63) | |
| Same address (%) | high BC, high CRC | 93.0 (89.5–96.5) | 92.7 (87.6–95.4) | 93.0 (89.1–97.8) | 92.8 (90.4–94.8) |
| high BC, low CRC | 92.6 (85.3–95.3) | 92.1 (84.5–96.5) | 92.5 (84.6–95.5) | 92.3 (83.7–98.9) | |
| low BC, high CRC | 92.7 (91.1–94.7) | 92.3 (89.7–94.2) | 92.7 (88.2–95.0) | 92.3 (88.1–94.3) | |
| low BC, low CRC | 91.8 (87.2–95.0) | 91.6 (85.4–95.7) | 91.9 (87.0–94.9) | 91.6 (86.7–94.4) | |
| Having partner (%) | high BC, high CRC | 53.4 (46.8–56.2) | 53.5 (48.0–55.9) | 53.4 (47.1–57.1) | 53.5 (48.8–55.8) |
| high BC, low CRC | 51.7 (42.3–55.9) | 51.8 (42.1–55.9) | 51.8 (42.2–56.6) | 51.6 (42.0–55.9) | |
| low BC, high CRC | 53.3 (49.2–56.1) | 53.3 (50.3–56.3) | 53.3 (47.5–56.1) | 53.2 (47.6–56.7) | |
| low BC, low CRC | 51.3 (41.2–55.4) | 51.3 (41.1–56.4) | 51.1 (41.0–55.1) | 51.1 (40.9–56.9) | |
| Having children (%) | high BC, high CRC | 29.9 (25.9–32.8) | 29.4 (26.3–32.7) | 29.1 (20.2–32.3) | 29.1 (26.7–32.0) |
| high BC, low CRC | 29.3 (21.9–31.3) | 29.4 (23.3–31.9) | 28.8 (21.2–31.7) | 28.8 (18.0–31.4) | |
| low BC, high CRC | 29.6 (28.1–32.3) | 28.9 (26.8–31.0) | 28.8 (26.6–31.0) | 28.4 (26.4–30.5) | |
| low BC, low CRC | 29.0 (22.5–32.9) | 28.7 (21.6–32.4) | 28.6 (21.8–31.6) | 28.3 (21.5–30.7) | |
| Foreign nationality (%) | high BC, high CRC | 2.5 (1.1–10.8) | 2.5 (0.6–8.7) | 2.6 (0.6–7.2) | 2.7 (0.7–7.8) |
| high BC, low CRC | 2.4 (0.8–19.9) | 3.5 (1.0–29.5) | 3.1 (0.9–20.8) | 3.6 (0.8–22.0) | |
| low BC, high CRC | 2.1 (0.7–10.8) | 2.1 (1.1–7.5) | 2.4 (0.8–10.1) | 3.0 (1.4–10.3) | |
| low BC, low CRC | 3.5 (0.9–29.0) | 3.4 (0.8–23.0) | 3.9 (1.1–29.6) | 3.9 (1.4–29.4) | |
| Socioeconomic variables | |||||
| Average income (×1000 €) | high BC, high CRC | 18.5 (0.2–25.0) | 18.6 (0.2–25.4) | 18.9 (0.2–24.2) | 19.2 (0.2–25.9) |
| high BC, low CRC | 19.2 (0.2–24.5) | 18.8 (1.6–26.9) | 18.4 (0.2–25.7) | 20.1 (1.8–25.3) | |
| low BC, high CRC | 20.0 (0.2–25.8) | 19.6 (0.0–27.6) | 19.7 (1.7–25.2) | 20.3 (0.2–30.7) | |
| low BC, low CRC | 20.6 (0.2–27.7) | 18.7 (1.7–24.9) | 19.0 (0.2–28.3) | 19.3 (0.2–25.6) | |
| Jobseeker rate (%) | high BC, high CRC | 2.2 (1.0–4.2) | 2.1 (0.7–3.9) | 1.8 (0.8–2.7) | 1.7 (0.7–2.4) |
| high BC, low CRC | 1.8 (1.0–3.8) | 1.7 (1.0–4.2) | 1.7 (0.7–3.8) | 1.4 (0.9–3.3) | |
| low BC, high CRC | 1.7 (1.0–4.4) | 1.7 (1.1–2.8) | 1.6 (1.1–3.6) | 1.5 (0.8–3.5) | |
| low BC, low CRC | 1.8 (1.1–4.3) | 1.8 (1.0–4.3) | 1.7 (1.0–3.9) | 1.6 (0.6–3.8) | |
| Wage earners (%) | high BC, high CRC | 36.8 (23.7–40.6) | 36.2 (24.1–40.6) | 36.7 (17.2–40.5) | 36.8 (24.2–40.3) |
| high BC, low CRC | 36.3 (17.1–39.0) | 36.9 (24.3–39.8) | 36.7 (23.6–39.7) | 36.7 (17.6–40.0) | |
| low BC, high CRC | 37.1 (26.0–40.9) | 37.3 (24.4–40.5) | 36.8 (26.3–40.4) | 37.0 (23.5–40.7) | |
| low BC, low CRC | 36.1 (23.9–40.5) | 36.1 (17.4–40.4) | 36.3 (23.5–40.5) | 36.6 (23.3–40.6) | |
| Self-employed (%) | high BC, high CRC | 7.8 (4.1–15.3) | 7.8 (4.0–16.9) | 8.0 (4.1–15.8) | 8.1 (6.0–14.4) |
| high BC, low CRC | 8.7 (5.4–16.8) | 7.8 (4.8–16.3) | 8.2 (5.0–17.2) | 7.8 (4.0–17.1) | |
| low BC, high CRC | 7.5 (4.7–16.4) | 7.6 (4.9–18.8) | 8.1 (5.1–15.3) | 8.4 (5.1–19.7) | |
| low BC, low CRC | 7.8 (4.0–18.5) | 8.3 (4.1–14.7) | 7.8 (4.2–19.1) | 8.0 (4.3–14.7) | |
| Early retired (%) | high BC, high CRC | 18.9 (10.6–23.1) | 19.2 (14.6–27.0) | 19.8 (14.4–26.9) | 19.9 (15.4–26.7) |
| high BC, low CRC | 20.4 (13.1–34.4) | 19.2 (11.6–23.7) | 20.0 (15.7–36.1) | 19.8 (15.0–36.7) | |
| low BC, high CRC | 19.3 (14.9–22.8) | 20.1 (17.0–23.8) | 20.4 (16.2–23.1) | 20.6 (18.1–24.2) | |
| low BC, low CRC | 19.9 (14.4–31.2) | 20.2 (13.8–35.6) | 19.9 (14.4–32.5) | 20.2 (14.5–33.1) | |
| Higher education (%) | high BC, high CRC | 44.3 (11.1–71.5) | 44.6 (31.7–72.8) | 44.3 (0.0–65.6) | 45.6 (34.2–71.9) |
| high BC, low CRC | 41.2 (23.8–98.3) | 43.8 (0.0–99.8) | 41.8 (8.0–96.2) | 46.7 (8.2–84.4) | |
| low BC, high CRC | 45.4 (32.3–67.0) | 45.9 (36.6–70.5) | 46.9 (38.0–66.9) | 49.0 (38.6–69.6) | |
| low BC, low CRC | 44.5 (6.5–68.9) | 41.5 (6.5–61.8) | 45.0 (6.9–66.9) | 42.9 (7.0–62.1) | |
| Health-related variables | |||||
| GP visits (%) | high BC, high CRC | 84.8 (77.4–88.9) | 85.4 (78.2–88.9) | 86.3 (77.0–90.0) | 86.1 (74.3–89.3) |
| high BC, low CRC | 84.0 (72.4–89.1) | 83.8 (67.2–88.7) | 85.4 (68.2–89.3) | 84.1 (67.6–89.0) | |
| low BC, high CRC | 82.5 (78.6–88.5) | 84.5 (77.7–89.4) | 84.9 (77.3–89.0) | 84.1 (77.7–88.7) | |
| low BC, low CRC | 81.3 (65.9–86.9) | 83.0 (68.0–86.9) | 82.9 (67.8–88.6) | 83.3 (66.2–89.9) | |
| Preventive dental visits (%) | high BC, high CRC | 32.7 (19.0–44.5) | 36.2 (20.0–51.9) | 38.5 (21.6–53.7) | 42.0 (22.6–55.6) |
| high BC, low CRC | 30.7 (17.9–42.3) | 34.4 (23.9–43.6) | 36.5 (19.3–43.9) | 39.6 (26.8–50.9) | |
| low BC, high CRC | 32.2 (17.4–49.9) | 34.7 (19.5–45.8) | 37.7 (22.2–54.1) | 41.2 (31.9–50.8) | |
| low BC, low CRC | 30.9 (16.6–49.5) | 32.6 (18.7–46.6) | 35.8 (23.0–49.5) | 37.1 (20.7–47.7) | |
| Chronic conditions (%) | high BC, high CRC | 9.1 (6.6–12.3) | 9.7 (7.4–12.9) | 10.5 (7.8–13.9) | 11.1 (8.4–14.2) |
| high BC, low CRC | 10.1 (6.5–13.1) | 9.9 (6.8–13.5) | 11.2 (7.3–15.2) | 11.2 (7.8–14.9) | |
| low BC, high CRC | 8.8 (6.0–12.4) | 9.4 (6.5–13.0) | 10.2 (8.2–13.3) | 10.8 (8.6–13.8) | |
| low BC, low CRC | 8.7 (6.3–13.2) | 9.8 (7.1–14.3) | 10.0 (7.2–13.8) | 11.1 (7.5–16.4) | |
| Diabetes (%) | high BC, high CRC | 5.1 (3.9–7.6) | 5.2 (4.2–7.5) | 5.3 (4.2–10.3) | 5.1 (4.1–7.2) |
| high BC, low CRC | 5.9 (4.5–7.5) | 5.6 (3.8–8.0) | 5.8 (4.6–8.7) | 5.7 (3.8–9.1) | |
| low BC, high CRC | 5.3 (4.0–7.2) | 5.3 (4.2–6.9) | 5.6 (4.3–7.2) | 5.2 (4.1–6.7) | |
| low BC, low CRC | 5.5 (3.8–8.5) | 5.9 (4.0–8.5) | 5.8 (3.9–7.9) | 5.7 (3.9–8.4) | |
| Disabilities (%) | high BC, high CRC | 6.2 (3.0–12.2) | 6.5 (3.1–11.5) | 6.6 (3.9–13.3) | 6.5 (2.9–14.4) |
| high BC, low CRC | 8.2 (2.8–14.6) | 6.8 (2.2–13.8) | 7.7 (3.0–15.1) | 6.7 (2.4–13.3) | |
| low BC, high CRC | 6.0 (3.0–10.7) | 5.7 (3.0–10.6) | 6.1 (3.0–10.5) | 5.8 (2.9–10.7) | |
| low BC, low CRC | 5.8 (2.2–13.9) | 6.9 (2.5–14.7) | 6.0 (2.3–10.9) | 6.9 (2.3–15.2) | |
| OR (95% CI); p Value | |||
|---|---|---|---|
| Uptake a | Group 2 High BC, Low CRC | Group 3 Low BC, High CRC | Group 4 Low BC, Low CRC |
| Demographic variables | |||
| Age group 1 (%) | 0.93 (0.85–1.03); 0.16 | 0.97 (0.90–1.06); 0.54 | 0.90 (0.82–0.99); 0.04 * |
| Age group 2 (%) | 0.99 (0.88–1.122); 0.90 | 0.97 (0.86–1.09); 0.60 | 0.94 (0.82–1.07); 0.34 |
| Age group 3 (%) | 1.08 (0.97–1.21); 0.14 | 1.05 (0.95–1.15); 0.32 | 1.16 (1.04–1.29); 0.005 * |
| Average household size (n) | 0.51 (0.01–2.84); 0.74 | 16.44 (0.43–62.5); 0.13 | 5.58 (0.09–34.5); 0.41 |
| Same address (%) | 0.71 (0.59–0.87); 0.001 * | 0.88 (0.77–1.02); 0.09 | 0.60 (0.50–0.71); <0.001 * |
| Having partner (%) | 0.66 (0.57–0.75); <0.001 * | 0.96 (0.86–1.09); 0.58 | 0.58 (0.51–0.67); <0.001 * |
| Having children (%) | 0.81 (0.58–0.77); 0.046 * | 0.76 (0.63–0.92); 0.005 * | 0.72 (0.59–0.87); <0.001 * |
| Foreign nationality (%) | 1.30 (1.19–1.42); <0.001 * | 0.91 (0.79–1.05); 0.21 | 1.36 (1.24–1.49); <0.001 * |
| Socioeconomic variables | |||
| Average income (×1000) (€) | 1.00 (0.97–1.03); 0.72 | 1.00 (0.97–1.02); 0.81 | 1.00 (0.98–1.03); 0.63 |
| Jobseeker rate (%) | 0.23 (0.13–0.39); <0.001 * | 0.73 (0.41–1.27); 0.27 | 0.28 (0.16–0.50); <0.001 * |
| Wage earners (%) | 1.05 (0.96–1.16); 0.25 | 1.05 (0.98–1.12); 0.19 | 1.09 (1.00–1.19); 0.04 * |
| Self-employed (%) | 1.17 (1.06–1.30); 0.002 * | 1.09 (0.97–1.23); 0.13 | 1.14 (1.01–1.28); 0.03 * |
| Early retired (%) | 1.29 (1.11–1.51); 0.001 * | 1.20 (1.04–1.37); 0.01 * | 1.3 (1.17–1.55); <0.001 * |
| Higher education (%) | 0.99 (0.94–1.03); 0.58 | 1.00 (0.96–1.04); 0.85 | 0.94 (0.90–0.99); 0.02 * |
| Health-related variables | |||
| GP visits (%) | 0.84 (0.71–0.98); 0.032 * | 0.76 (0.65–0.89); <0.001 * | 0.72 (0.60–0.86); <0.001 * |
| Preventive dental visits (%) | 0.90 (0.86–0.95); <0.001 * | 0.99 (0.95–1.03); 0.54 | 0.93 (0.88–0.97); 0.002 * |
| Chronic conditions (%) | 1.02 (0.86–1.21); 0.78 | 0.85 (0.72–1.01); 0.06 | 0.94 (0.80–1.10); 0.43 |
| Diabetes (%) | 2.59 (1.80–3.72); <0.001 * | 1.27 (0.90–1.79); 0.1652 | 2.72 (1.87–3.96); <0.001 * |
| Disabilities (%) | 1.14 (1.03–1.27); 0.01 * | 0.93 (0.83–1.03); 0.15 | 0.98 (0.88–1.09); 0.75 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ferrari, A.; Tran, T.N.; Hoeck, S.; Peeters, M.; Goossens, M.; Van Hal, G. Differences and Similarities in Breast and Colorectal Cancer Screening Uptake among Municipalities in Flanders, Belgium. Gastrointest. Disord. 2022, 4, 84-96. https://doi.org/10.3390/gidisord4020010
Ferrari A, Tran TN, Hoeck S, Peeters M, Goossens M, Van Hal G. Differences and Similarities in Breast and Colorectal Cancer Screening Uptake among Municipalities in Flanders, Belgium. Gastrointestinal Disorders. 2022; 4(2):84-96. https://doi.org/10.3390/gidisord4020010
Chicago/Turabian StyleFerrari, Allegra, Thuy Ngan Tran, Sarah Hoeck, Marc Peeters, Mathieu Goossens, and Guido Van Hal. 2022. "Differences and Similarities in Breast and Colorectal Cancer Screening Uptake among Municipalities in Flanders, Belgium" Gastrointestinal Disorders 4, no. 2: 84-96. https://doi.org/10.3390/gidisord4020010
APA StyleFerrari, A., Tran, T. N., Hoeck, S., Peeters, M., Goossens, M., & Van Hal, G. (2022). Differences and Similarities in Breast and Colorectal Cancer Screening Uptake among Municipalities in Flanders, Belgium. Gastrointestinal Disorders, 4(2), 84-96. https://doi.org/10.3390/gidisord4020010









