Abstract
Colorectal cancer screening can contribute to reducing colorectal cancer incidence and mortality. Findings on disparities in the utilization of colorectal cancer screening between migrants and non-migrants have been inconsistent, with some studies reporting lower, and some higher utilization among migrants. The aim of the present study was to examine potential disparities in fecal occult blood testing and colonoscopy among migrants in Germany. Data from a population survey on 11,757 men and women aged ≥50 years is used. Using multivariable logistic regression, the utilization of fecal occult blood testing and colonoscopy was compared between non-migrants, migrants from EU countries and migrants from non-EU countries, adjusting for socio-economic factors and also taking into account intersectional differences by sex and age. The study shows that migrants from the EU (adjusted OR = 0.73; 95%-CI: 0.57, 0.94) and from non-EU countries (adjusted OR = 0.39; 95%-CI: 0.31, 0.50) were less likely to utilize fecal occult blood testing than non-migrants. No disparities for the use of colonoscopy were observed. The findings are in line with studies from other countries and can be indicative of different barriers migrants encounter in the health system. Adequate strategies taking into account the diversity of migrants are needed to support informed decision-making among this population group.
1. Introduction
In the last decade, there has been an increase in the incidence of colorectal cancer in most European countries [1]. Amongst others, this is attributed to demographic aging of the population [2,3] and changing lifestyle habits in younger age groups, such as poor nutrition and obesity [4]. Every year, more than 450,000 people across Europe develop colorectal cancer, and approximately 230,000 people die from the disease annually [5]. Studies show that colorectal cancer is the second leading cause of cancer death among men and the third leading cause among women in Europe [6]. Colorectal cancer is a serious and often life-changing disease. The 5-year survival rate is on average 65%, but varies considerably depending on the stage of the tumor [7]. Tumors detected early are associated with a significantly better survival rate [8]. If precancerous tumors are detected, they can be treated accordingly [9], thus interrupting the adenoma-carcinoma sequence and significantly reducing the risk of colorectal cancer [10].
Today, both non-invasive and invasive methods of colorectal cancer screening are available. Non-invasive stool tests, such as guaiac fecal occult blood tests (gFOBTs) and fecal immunochemical tests (FITs), are designed to detect microscopic amounts of blood in stool which can be indicators of tumors or polyps in the colon. Patients with a positive fecal occult blood test are usually advised to undergo colonoscopy for further examination. The purpose of colonoscopy, an invasive screening method which is also frequently used as a primary screening tool without prior fecal occult blood testing, is to examine the colon for polyps and other suspicious changes which have the potential to develop into malignant neoplasms. Polyps detected during colonoscopy are usually removed in the course of the procedure [11]. Both approaches to cancer screening have been demonstrated to reduce colorectal cancer mortality, and evidence shows that colonoscopy can also reduce the incidence of colorectal cancer [12,13,14].
Similar to other forms of preventive behavior, non-utilization of colorectal cancer screening is associated with a low socio-economic status [15,16], younger age [17], male gender [18] and the absence of a partner [19]. Lack of information about risk factors and symptoms of colorectal cancer as well as about colorectal cancer screening programs may contribute to a lower participation in colorectal cancer screening as well, because sufficient knowledge is an essential prerequisite for informed decision making [20,21]. In addition to deficits at the patient level, structural barriers in the healthcare system, such as distance to and office hours of screening facilities, organization of appointments, availability of translation services and lack of insurance coverage may affect participation in colorectal cancer screening [22].
Studies from several countries have also shown that even after adjusting for socio-demographic characteristics and the role of social determinants, migrants tend to utilize preventive health care services less frequently than non-migrants. This is also true for many types of cancer screenings such as regular Pap smears and mammography [23,24,25,26,27,28,29,30,31] as well as fecal occult blood testing and colonoscopy [28,32,33,34,35,36,37,38]. For example, a study from Denmark reported that non-Western migrants were 38% less likely to participate in fecal occult blood testing than the majority Danish population [28]. Similarly, survey data from a Swiss study show that individuals born in a country outside of Western Europe and North America have a 35% lower colorectal cancer screening rate compared to native Swiss individuals [32]. Comparable results have been reported from other European countries such as the Netherlands [33] and from other regions of the world such as Canada [34], Israel [35] and the United States [36,37,38].
In Germany, 26.7% of the population are migrants. This refers to individuals who have a non-German citizenship and/or who themselves or whose mother or father were born abroad [39]. Similar to evidence from other European and non-European countries, migrants in Germany have been reported to utilize cancer screening services less frequently than non-migrants. For cervical cancer screening, for example, studies show that non-EU migrant and EU migrant women are 33% and 20% less likely to participate in cervical cancer screening, respectively [26], even after adjusting for differences in social determinants. Disparities could be explained by different barriers migrants encounter in health care, comprising poor German language proficiency, a low health literacy or cultural needs not sufficiently addressed by health care facilities [40,41]. Several investigations from Germany and other countries have also shown that disparities may vary by sex or age [26,42,43,44], emphasizing the need for an intersectional perspective. However, findings are not consistent across the available data, as not all studies identified disparities between both population groups [45] and others reported a higher utilization among migrants than non-migrants [46].
Little is known about potential disparities in colorectal cancer screening. For purposes of colorectal cancer screening, several screening tests are available in Germany, including fecal occult blood testing and colonoscopy. All individuals aged 50 and older, including migrants with a residence status, are eligible to participate in regular colorectal cancer screening free of charge as part of their social health insurance. Individuals aged between 50 and 54 years can utilize an immunological test for hidden blood in the stool once a year, and those 55 years of age or older can utilize the test every two years without any additional fees. Insured individuals who wish to undergo fecal occult blood testing can contact either their family doctor or specialists and request a stool test or obtain tests kits from pharmacies. The tests can be performed at home, with samples being submitted to a physician for further testing [47,48]. In addition, men aged 50 years and older and women aged 55 years and older are recommended to undergo colonoscopy every 10 years. As an alternative to colonoscopy, individuals aged 55 years and older can undergo a stool examination every two years free of charge [49].
Data from the German National Cohort (NAKO) show that fecal occult blood testing is utilized 18% (OR = 0.82, 95%-CI = 0.78–0.87) less frequently by migrants than non-migrants [50], corroborating the aforementioned findings from studies conducted on disparities in the utilization of cervical cancer screening and mammography. However, another study examining the utilization of colorectal cancer screening in migrant women by means of data from a large health insurance organization revealed higher odds of both fecal occult blood testing (OR = 1.34; 95%-CI: 1.15–1.55) and colonoscopy (OR = 1.81; 95%-CI: 1.71–1.91) for migrant women as compared to the women from the majority population. Being based on administrative data, the available information was limited and potential confounding by social determinants could not be taken into account [51]. Similarly, little is known about intersectional differences in colorectal cancer screening between migrants and non-migrants in Germany, which previous studies have pointed out for other types of cancer screening [24].
Based on data from a large-scale and nationwide population survey, the aim of the present study was to examine the utilization of fecal occult blood testing and colonoscopy among migrants and non-migrants in Germany, also considering intersectional differences by sex and age. The findings can contribute to the development of adequate strategies aiming to enable adequate access to colorectal cancer screening and informed decision making in a diverse population.
2. Methods
2.1. Data
The study uses data from the German Health Update 2014/2015, conducted by the Robert Koch-Institute on behalf of the German Federal Ministry of Health from November 2014 to July 2015 [52]. The German Health Update 2014/2015 is a cross-sectional survey of 24,016 individuals aged 18 years and older living in private household in Germany. Data collection was done through a mixed-mode design, using standardized self-administered online or postal questionnaires. Participation in the survey was voluntary and the response rate was 26.9% [53]. For the analysis of the present study, only respondents aged 50 years and older were included, since from this age onwards colorectal cancer screening is recommended in Germany [49]. The initial sample studied consisted of n = 12,244 individuals. Of these, about 4.0% (n = 487) did not provide responses to one of the covariates or outcomes studied and were therefore excluded from the analysis, resulting in a final sample of n = 11,757.
2.2. Study Variables
The primary dependent variables were the use of a fecal occult blood test and the utilization of colonoscopy. In the survey, respondents were asked when they last had a test for hidden blood in the stool (fecal occult blood test), with the response categories being ‘within the last 12 months’, ‘1 to less than 2 years ago’, ‘2 to less than 3 years ago’, ‘3 years ago or more‘ and ‘never‘. For the present study, we distinguished between respondents who have had a fecal occult blood test at least once in their life and those who never had one. In addition, participants were asked when they last had a colonoscopy. Participants were given five response options: ‘within the last 12 months’, ‘1 to less than 5 years ago’, ‘5 to less than 10 years ago’, ‘10 years ago or more’ and ‘never’. Again, for the present study, the variable was dichotomized into individuals who utilized colonoscopy at least once in their life and those who had never undergone colonoscopy.
The results were subjected to a comparison between the three populations of non-migrants, migrants from EU countries and migrants from non-EU countries. Respondents were considered migrants if they either had non-German citizenship or were born in another country [39]. Several control variables were taken into account. The socio-demographic covariates considered were sex (male, female), age (five-year age groups treated as a continuous measure), partnership status (living with a partner vs. not living with a partner) and socioeconomic status (low, middle, high), the latter of which was based on a standardized summary measure taking into account occupational status, vocational education and net equivalent income [54]. Furthermore, the region of residence of the respondents in Germany (Northwest, North Rhine-Westphalia, Middle, East, Bavaria/Baden-Wuerttemberg), the type of residential area (rural, small towns, medium-sized towns, cities) and the self-related health status (rated on a scale from 1 [“very good”] to 5 [“very poor”]) were also included to account for regional and health differences between the population groups, respectively.
2.3. Analysis
The distribution of the variables between the three different population groups was compared using chi-square tests and analysis of variance, with the significance level set to p < 0.05. Two multivariable logistic regression analyses were performed to investigate the association between the utilization of fecal occult blood testing or colonoscopy (dependent variables) and the three population groups (primary independent variable), respectively. In both models, the aforementioned confounding variables were accounted for. The results were reported as adjusted odds ratios (aOR) with 95% confidence intervals (95%-CI). To examine intersectional differences between the population groups related to sex and age, a three-way interaction term was included in each of the two models. The results were presented graphically as predicated probabilities. All analyses were conducted using Stata 15 [55].
3. Results
Responses from 11,757 individuals aged 50 years and older who provided information on all study variables were available. Of these, 3.9% were migrants from EU countries and 3.0% were migrants from non-EU countries. The three population groups showed differences in the distribution of some demographic and health factors (see Table 1). Non-EU migrants were younger, had a lower socioeconomic status, were more likely to live in cities and rated their health status slightly lower than non-migrants. Regarding the use of fecal occult blood testing, migrants from non-EU countries showed a significantly lower use than non-migrants (65.3% vs. 82.5%). Fewer disparities were observed between migrants from EU countries and non-migrants (78.9% vs. 82.5%). With regard to the utilization of colonoscopy, the three population groups did not differ significantly from each other, with percentages being 60.1% for non-migrants, 62.0% for migrants from EU- and 56.3% for migrants from non-EU countries.

Table 1.
Description of the study sample by population group (German Health Update 2014/2015, respondents aged ≥50 years, n = 11,757).
Differences with regard to fecal occult blood testing remained significant after adjusting for the role of demographic and socio-economic factors. Table 2 shows that migrants from EU countries (adjusted OR = 0.73; 95%-CI: 0.57, 0.94) and non-EU countries (adjusted OR = 0.39; 95%-CI: 0.31, 0.50) were 27% and 61%, respectively, less likely to participate in fecal occult blood testing than non-migrants. With the exception of the type of residential area, all control variables were significantly associated with the utilization of fecal occult blood testing. Confirming the results from the descriptive analysis, a second multivariable logistic regression model showed that the odds for the utilization of colonoscopy did not differ significantly between the three population groups.

Table 2.
Results of the multivariable logistic regression model with utilization of fecal occult blood testing and colonoscopy, respectively, as the dependent variables. Adjusted odds ratios (aOR) and 95% confidence intervals (95%-CI) (German Health Update 2014/2015, respondents aged ≥50 years, n = 11,757).
Whereas the respective three-way interaction terms included into the model were not significant at p < 0.05, a graphical inspection of the predicated probabilities indicates that, particularly for men, disparities between migrants and non-migrants with respect to the utilization of fecal occult blood testing tended to decrease with age, but increased for the utilization of colonoscopy (Figure 1).
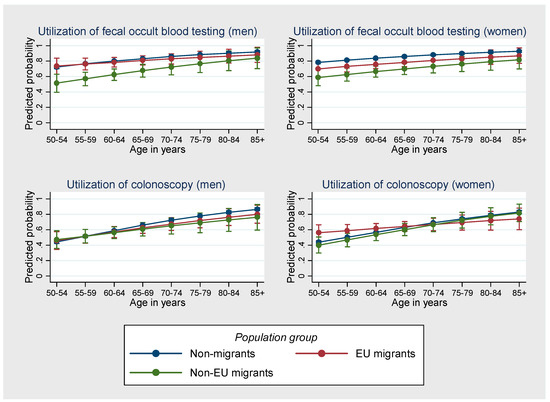
Figure 1.
Probability of the utilization of fecal occult blood testing and colonoscopy by population group, sex and age. Results of the multivariable logistic regression models with interaction effects between population group, sex and age. Predicted probabilities (German Health Update 2014/2015, respondents aged ≥50 years, n = 11,757).
4. Discussion
The present study used a nationwide population-based cross-sectional survey to investigate the level of participation in colorectal cancer screening among migrants and non-migrants in Germany. The study shows that migrants from EU and non-EU countries who reside in Germany have a significantly lower utilization of fecal occult blood testing than non-migrants. In contrast, there are no significant differences in colonoscopy participation.
With regard to fecal occult blood testing, the results are in line with a large number of studies from different countries, which also identified a considerably lower utilization of cancer screening among migrants than non-migrants [23,24,25,26,27,28,29,30,31,56]. The lower utilization of the fecal occult blood test may result from several types of barriers migrants encounter in health care. For instance, insufficient language proficiency and poor health literacy can constitute a significant obstacle to participation. Both can make it difficult for migrants to obtain necessary information about the rationale of screening programs, thus making it difficult to arrive at an informed decision about participation. Consequently, such information deficits and lack of knowledge related to colorectal cancer screening can create mistrust about its purpose [30,41]. Knowledge deficits of migrants in relation to colorectal cancer screening have been documented by different studies [41,57]. For example, a study from the United Kingdom shows that such deficits are more likely among ethnic minorities than among the British majority population. The same study also reported a pronounced sense of embarrassment and shame perceived by migrants with respect to attending colorectal cancer screening [57]. This emphasizes the need for migrants receiving culturally sensitive information on colorectal cancer screening in their mother tongue, informing them about the potential advantages and limitations [30]. Although colorectal cancer screening has been shown to reduce colorectal cancer morbidity and mortality, it is also associated with disadvantages, such as false negative or false positive results and the risk of complications during colonoscopy [13,58]. Therefore, utilizing or not utilizing colorectal cancer screening must be based on an informed decision [59,60]. Studies on informed decision making among migrants show that this population group, on average, makes less informed decisions than non-migrants [61]. Consequently, effective communication strategies, including accessible information material, need to be employed to promote informed decision making [41,62]. These strategies need to be tailored to the specifics of the language, to cultural beliefs and a potentially limited health literacy of migrants [41]. The low utilization of fecal occult blood testing by migrant communities may also be due to lower risk perceptions resulting from a low disease risks in their countries of origin [30,63]. The underestimation of cancer risks may thus contribute to a lower participation rate and needs to be addressed by communication strategies as well.
Regarding participation in colonoscopy, the present study does not indicate any significant disparities between migrants and non-migrants. This finding corresponds to results from a study from Italy which also reported a similar utilization of colonoscopy for migrants and non-migrants [64]. This could indicate that with respect to colonoscopy, health care providers are more successful in reducing barriers to access, thus avoiding discrimination against migrants. However, a similarly high utilization of colonoscopy among migrants and non-migrants may also result from a selection effect. Men and women in Germany, in addition to being eligible for colonoscopy from the age of 50 and 55, respectively, are also advised to undergo colonoscopy if they have a positive fecal occult blood test. In these cases, colonoscopy is the second stage of colorectal cancer screening. Migrants who have already participated in fecal occult blood testing may be individuals who are experiencing fewer barriers in the health care system and who are generally more compliant with screening examinations [64]. Finally, significant differences in fecal occult blood testing and lack of differences in colonoscopy may possibly be attributed to migrants questioning the efficacy of the former, whereas the efficacy of colonoscopy is accepted. Although differences in the perceived effectiveness have been reported for some population groups [65], to the best of the authors’ knowledge, no studies on that subject have been conducted for migrants in Germany and should be the focus of future investigations.
An examination of interaction effects has indicated that, with increasing age, disparities in the utilization of fecal occult blood testing between migrants and non-migrants tend to decrease, particularly for men. This counter-intuitive finding contradicts previous research which showed that older migrants are more disadvantaged than younger individuals [66]. However, similar findings were reported in other studies [42]. Given that the interaction effects in the present study were not significant, further investigations are needed.
The strengths of the present study include the large sample size and the high quality of the data. However, some limitations need to be considered as well. The results are not based on administrative data, but on self-reporting by the survey participants. Since the survey refers to previous screenings that may have occurred up to 10 or more years ago, a recall bias may be present in the analysis. Although inconsistent findings on the validity of self-reported information on cancer screenings have been reported [67,68], studies on the utilization of other preventive services suggest that self-reported data from the German Healthy Update survey corresponds to administrative data [69]. Another limitation is that the health survey was conducted only in German language. Consequently, individuals with poor German language skills, who presumably also encounter considerable barriers in the health care system, could not participate in the survey and are therefore underrepresented in the study. This likely also explains why the percentage of migrants in our sample is only 6.9%, whereas according to census data from the same year, 12.9% of the population aged 50 years or older in Germany were migrants [70]. Therefore, our results may underestimate the true differences in the utilization of colorectal cancer screening between migrants and non-migrants. Furthermore, the present study could not account for heterogeneity in terms of religion, culture, ethnicity and length of stay, which may also have an impact on the utilization of colorectal cancer screening [41,71,72]. Finally, it must be emphasized that our data refers to migrants in Germany and therefore the results may not be generalizable to other countries with other population profiles and other histories of migration.
5. Conclusions
The results of the study indicate a lower utilization of fecal occult blood testing among migrants from EU and non-EU countries compared to non-migrants in Germany. This can be explained by barriers migrant communities face in accessing health services. In order to ensure equal access to health care for this population group and to address knowledge deficits with respect to colorectal cancer screening, there is a need for comprehensive education about advantages and disadvantages, taking into account the specific cultural and language needs of migrants. This way, this population group can be supported in making informed decisions for or against participation in colorectal cancer screening. Further research, also employing qualitative methods, should examine the reasons for migrants’ lower participation in fecal occult blood testing. In 2019, a nationally organized colorectal cancer screening program was introduced in Germany. As part of that program, all individuals with statutory health insurance received a postal invitation from their statutory health organization for colorectal cancer screening at the age of 50, 55, 60 and 65 years, including information on advantages and disadvantages of screening and on the different screening methods and procedures [47]. Future studies need to evaluate how effective these measures are in reducing existing disparities in fecal occult blood testing and in promoting informed decision making in colorectal screening in general.
Author Contributions
Conceptualization, D.W. and P.B.; methodology, D.W. and P.B.; software, P.B.; validation, P.B., D.W. and Y.Y.-A.; formal analysis, P.B. and D.W.; investigation, P.B. and D.W.; resources, P.B.; data curation, P.B.; writing—original draft preparation, D.W.; writing—review and editing, P.B., D.W. and Y.Y.-A.; visualization, P.B. and D.W.; supervision, P.B. and Y.Y.-A.; project administration, P.B. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
The study uses secondary data from a national survey collected by a national institution in Germany. Participation in the survey was anonymous and voluntary following participants’ informed consent, and no experiments were involved. Considering national guidelines and recommendations for secondary data analysis, no further ethical endorsement was required for this analysis.
Informed Consent Statement
Informed consent was obtained from all subjects involved in the study.
Data Availability Statement
Data used in the present study can be obtained from the Robert Koch Institute, Germany (see https://www.rki.de/DE/Content/Gesundheitsmonitoring/Studien/Geda/Geda_2014_inhalt.html) (accessed on 1 June 2022).
Conflicts of Interest
The authors declare no conflict of interest.
References
- Vuik, F.E.; Nieuwenburg, S.A.; Bardou, M.; Lansdorp-Vogelaar, I.; Dinis-Ribeiro, M.; Bento, M.J.; Zadnik, V.; Pellisé, M.; Esteban, L.; Kaminski, M.F.; et al. Increasing incidence of colorectal cancer in young adults in Europe over the last 25 years. Gut 2019, 68, 1820–1826. [Google Scholar] [CrossRef] [PubMed]
- Pilleron, S.; Sarfati, D.; Janssen-Heijnen, M.; Vignat, J.; Ferlay, J.; Bray, F.; Soerjomataram, I. Global cancer incidence in older adults, 2012 and 2035: A population-based study. Int. J. Cancer 2019, 144, 49–58. [Google Scholar] [CrossRef] [PubMed]
- Cinar, D.; Tas, D. Cancer in the elderly. North Clin. Istanb. 2015, 2, 73–80. [Google Scholar] [CrossRef]
- Gutlic, I.; Schyman, T.; Lydrup, M.-L.; Buchwald, P. Increasing colorectal cancer incidence in individuals aged <50 years-a population-based study. Int. J. Colorectal Dis. 2019, 34, 1221–1226. [Google Scholar] [CrossRef] [PubMed]
- EuropaColon. Colorectal Cancer in Europe. A Framework for Improving Outcomes for Patients. Available online: https://webgate.ec.europa.eu/chafea_pdb/assets/files/pdb/20124301/20124301_d04-00_en_ps_ecwhitepaper.pdf (accessed on 31 May 2022).
- European Cancer Information System. Colorectal Cancer Burden in EU-27. Available online: https://ecis.jrc.ec.europa.eu/factsheets.php (accessed on 31 May 2022).
- Roche. Colorectal cancer Colorectal Cancer. A Guide for Journalists on Colorectal Cancer and Its Treatment. Available online: https://assets.cwp.roche.com/f/126832/x/6a772fdf01/med-colorectal-cancer.pdf (accessed on 31 May 2022).
- Majek, O.; Gondos, A.; Jansen, L.; Emrich, K.; Holleczek, B.; Katalinic, A.; Nennecke, A.; Eberle, A.; Brenner, H. Survival from colorectal cancer in Germany in the early 21st century. Br. J. Cancer 2012, 106, 1875–1880. [Google Scholar] [CrossRef] [PubMed]
- Robert Koch-Institut. Inanspruchnahme der Darmspiegelung in Deutschland; Robert Koch Institute: Berlin, Germany, 2017. [Google Scholar]
- Weigl, K.; Hoffmeister, M. Darmkrebs-Screening. TumorDiagn. Ther. 2019, 40, 360–363. [Google Scholar] [CrossRef]
- Schreuders, E.H.; Ruco, A.; Rabeneck, L.; Schoen, R.E.; Sung, J.J.Y.; Young, G.P.; Kuipers, E.J. Colorectal cancer screening: A global overview of existing programmes. Gut 2015, 64, 1637–1649. [Google Scholar] [CrossRef]
- Brenner, H.; Stock, C.; Hoffmeister, M. Effect of screening sigmoidoscopy and screening colonoscopy on colorectal cancer incidence and mortality: Systematic review and meta-analysis of randomised controlled trials and observational studies. BMJ 2014, 348, g2467. [Google Scholar] [CrossRef]
- Hewitson, P.; Glasziou, P.; Watson, E.; Towler, B.; Irwig, L. Cochrane systematic review of colorectal cancer screening using the fecal occult blood test (hemoccult): An update. Am. J. Gastroenterol. 2008, 103, 1541–1549. [Google Scholar] [CrossRef]
- Shaukat, A.; Mongin, S.J.; Geisser, M.S.; Lederle, F.A.; Bond, J.H.; Mandel, J.S.; Church, T.R. Long-term mortality after screening for colorectal cancer. N. Engl. J. Med. 2013, 369, 1106–1114. [Google Scholar] [CrossRef]
- Zimmerman, R.K.; Nowalk, M.P.; Tabbarah, M.; Grufferman, S. Predictors of colorectal cancer screening in diverse primary care practices. BMC Health Serv. Res. 2006, 6, 116. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Shapiro, J.A.; Seeff, L.; Nadel, M. Colorectal cancer-screening tests and associated health behaviors. Am. J. Prev. Med. 2001, 21, 132–137. [Google Scholar] [CrossRef]
- Wools, A.; Dapper, E.A.; de Leeuw, J.R.J. Colorectal cancer screening participation: A systematic review. Eur. J. Public Health 2016, 26, 158–168. [Google Scholar] [CrossRef] [PubMed]
- Mosquera, I.; Mendizabal, N.; Martín, U.; Bacigalupe, A.; Aldasoro, E.; Portillo, I. Inequalities in participation in colorectal cancer screening programmes: A systematic review. Eur. J. Public Health 2020, 30, 416–425. [Google Scholar] [CrossRef]
- van Jaarsveld, C.H.M.; Miles, A.; Edwards, R.; Wardle, J. Marriage and cancer prevention: Does marital status and inviting both spouses together influence colorectal cancer screening participation? J. Med. Screen. 2006, 13, 172–176. [Google Scholar] [CrossRef]
- Gimeno Garcia, A.Z.; Buylla, N.H.A.; Nicolas-Perez, D.; Quintero, E. Public awareness of colorectal cancer screening: Knowledge, attitudes, and interventions for increasing screening uptake. ISRN Oncol. 2014, 2014, 425787. [Google Scholar] [CrossRef]
- Gede, N.; Reményi Kiss, D.; Kiss, I. Colorectal cancer and screening awareness and sources of information in the Hungarian population. BMC Fam. Pract. 2018, 19, 106. [Google Scholar] [CrossRef]
- Baron, R.C.; Rimer, B.K.; Coates, R.J.; Kerner, J.; Kalra, G.P.; Melillo, S.; Habarta, N.; Wilson, K.M.; Chattopadhyay, S.; Leeks, K. Client-directed interventions to increase community access to breast, cervical, and colorectal cancer screening a systematic review. Am. J. Prev. Med. 2008, 35, S56–S66. [Google Scholar] [CrossRef]
- Azerkan, F.; Sparén, P.; Sandin, S.; Tillgren, P.; Faxelid, E.; Zendehdel, K. Cervical screening participation and risk among Swedish-born and immigrant women in Sweden. Int. J. Cancer 2012, 130, 937–947. [Google Scholar] [CrossRef]
- Brunner-Ziegler, S.; Rieder, A.; Stein, K.V.; Koppensteiner, R.; Hoffmann, K.; Dorner, T.E. Predictors of participation in preventive health examinations in Austria. BMC Public Health 2013, 13, 1138. [Google Scholar] [CrossRef]
- Brzoska, P.; Aksakal, T.; Yilmaz-Aslan, Y. Disparities in the use of regular pap smears among migrant and non-migrant women in Austria: A population-based survey of 7633 women. J. Med. Screen. 2021, 28, 372–376. [Google Scholar] [CrossRef] [PubMed]
- Brzoska, P.; Aksakal, T.; Yilmaz-Aslan, Y. Utilization of cervical cancer screening among migrants and non-migrants in Germany: Results from a large-scale population survey. BMC Public Health 2020, 20, 5. [Google Scholar] [CrossRef]
- Carrasco-Garrido, P.; Hernandez-Barrera, V.; Lopez de Andres, A.; Jimenez-Trujillo, I.; Gallardo Pino, C.; Jimenez-Garcıa, R. Awareness and uptake of colorectal, breast, cervical and prostate cancer screening tests in Spain. Eur. J. Public Health 2014, 24, 264–270. [Google Scholar] [CrossRef]
- Frederiksen, B.L.; Jørgensen, T.; Brasso, K.; Holten, I.; Osler, M. Socioeconomic position and participation in colorectal cancer screening. Br. J. Cancer 2010, 103, 1496–1501. [Google Scholar] [CrossRef] [PubMed]
- Jensen, L.F.; Pedersen, A.F.; Andersen, B.; Vedsted, P. Identifying specific non-attending groups in breast cancer screening--population-based registry study of participation and socio-demography. BMC Cancer 2012, 12, 518. [Google Scholar] [CrossRef]
- Kristiansen, M.; Thorsted, B.L.; Krasnik, A.; von Euler-Chelpin, M. Participation in mammography screening among migrants and non-migrants in Denmark. Acta Oncol. 2012, 51, 28–36. [Google Scholar] [CrossRef]
- Rodríguez-Salés, V.; Roura, E.; Ibañez, R.; Peris, M.; Bosch, F.X.; de Sanjosé, S. Coverage of Cervical Cancer Screening in Catalonia for the Period 2008-2011 among Immigrants and Spanish-Born Women. Front. Oncol. 2013, 3, 297. [Google Scholar] [CrossRef]
- Fischer, R.; Collet, T.-H.; Zeller, A.; Zimmerli, L.; Gaspoz, J.-M.; Giraudon, K.; Rodondi, N.; Cornuz, J. Obesity and overweight associated with lower rates of colorectal cancer screening in Switzerland. Eur. J. Cancer Prev. 2013, 22, 425–430. [Google Scholar] [CrossRef]
- Deutekom, M.; van Rijn, A.F.; Dekker, E.; Blaauwgeers, H.; Stronks, K.; Fockens, P.; Essink-Bot, M.-L. Uptake of faecal occult blood test colorectal cancer screening by different ethnic groups in the Netherlands. Eur. J. Public Health 2009, 19, 400–402. [Google Scholar] [CrossRef]
- Sewitch, M.J.; Fournier, C.; Ciampi, A.; Dyachenko, A. Adherence to colorectal cancer screening guidelines in Canada. BMC Gastroenterol. 2007, 7, 39. [Google Scholar] [CrossRef]
- Wilf-Miron, R.; Peled, R.; Yaari, E.; Vainer, A.; Porath, A.; Kokia, E. The association between socio-demographic characteristics and adherence to breast and colorectal cancer screening: Analysis of large sub populations. BMC Cancer 2011, 11, 376. [Google Scholar] [CrossRef] [PubMed]
- Pollack, L.A.; Blackman, D.K.; Wilson, K.M.; Seeff, L.C.; Nadel, M.R. Colorectal cancer test use among Hispanic and non-Hispanic U.S. populations. Prev. Chronic Dis. 2006, 3, A50. [Google Scholar] [PubMed]
- James, T.M.; Greiner, K.A.; Ellerbeck, E.F.; Feng, C.; Ahluwalia, J.S. Disparities in colorectal cancer screening: A guideline-based analysis of adherence. Ethn. Dis. 2006, 16, 228–233. [Google Scholar] [PubMed]
- Ananthakrishnan, A.N.; Schellhase, K.G.; Sparapani, R.A.; Laud, P.W.; Neuner, J.M. Disparities in colon cancer screening in the Medicare population. Arch. Intern. Med. 2007, 167, 258–264. [Google Scholar] [CrossRef] [PubMed]
- Statistisches Bundesamt. Bevölkerung und Erwerbstätigkeit. Bevölkerung mit Migrationshintergrund. Ergebnisse des Mikrozensus 2020 (Fachserie 1 Reihe 2.2); Statistisches Bundesamt: Wiesbaden, Germany, 2021. [Google Scholar]
- Berens, E.-M.; Klinger, J.; Mensing, M.; Carol, S.; Schaeffer, D. Gesundheitskompetenz von Menschen mit Migrationshintergrund in Deutschland; Universität Bielefeld: Bielefeld, Germany, 2022. [Google Scholar]
- Woudstra, A.J.; Dekker, E.; Essink-Bot, M.-L.; Suurmond, J. Knowledge, attitudes and beliefs regarding colorectal cancer screening among ethnic minority groups in the Netherlands—A qualitative study. Health Expect. 2016, 19, 1312–1323. [Google Scholar] [CrossRef]
- Brzoska, P.; Wahidie, D.; Yilmaz-Aslan, Y. An Intersectional Perspective on the Utilization of Cervical Cancer Screening among Migrants. A Cross-Sectional Analysis of Survey Data from Austria. Cancers 2021, 13, 6082. [Google Scholar] [CrossRef]
- Cofie, L.E.; Hirth, J.M.; Cuevas, A.G.; Farr, D. A national study of gender and racial differences in colorectal cancer screening among foreign-born older adults living in the US. J. Behav. Med. 2020, 43, 460–467. [Google Scholar] [CrossRef]
- Negi, J.; Nambiar, D. Intersectional social-economic inequalities in breast cancer screening in India: Analysis of the National Family Health Survey. BMC Women’s Health 2021, 21, 324. [Google Scholar] [CrossRef]
- Berens, E.-M.; Mohwinkel, L.-M.; van Eckert, S.; Reder, M.; Kolip, P.; Spallek, J. Uptake of Gynecological Cancer Screening and Performance of Breast Self-Examination Among 50-Year-Old Migrant and Non-migrant Women in Germany: Results of a Cross-Sectional Study (InEMa). J. Immigr. Minority Health 2019, 21, 674–677. [Google Scholar] [CrossRef]
- Berens, E.-M.; Stahl, L.; Yilmaz-Aslan, Y.; Sauzet, O.; Spallek, J.; Razum, O. Participation in breast cancer screening among women of Turkish origin in Germany—A register-based study. BMC Women’s Health 2014, 14, 24. [Google Scholar] [CrossRef][Green Version]
- Kassenärztliche Bundesvereinigung. Organisiertes Darmkrebsscreening: Was Sich in der Praxis Ändert—Ein Überblick. Available online: https://www.kbv.de/media/sp/PraxisInfo_Darmkrebsscreening.pdf (accessed on 31 May 2022).
- AOK Gesundheitsmagazin. Darmkrebsfrüherkennung: Wie zuverlässig sind Stuhltests? Available online: https://www.aok.de/pk/magazin/koerper-psyche/krebs/stuhltest-wie-er-funktioniert-und-was-er-ueber-darmkrebs-aussagt/ (accessed on 31 May 2022).
- Riemann, J.F.; Hüppe, D. Darmkrebsvorsorge—Bewährtes und Neues. Gynäk Praxis 2021, 47, 391–396. [Google Scholar]
- Wiessner, C.; Keil, T.; Krist, L.; Zeeb, H.; Dragano, N.; Schmidt, B.; Ahrens, W.; Berger, K.; Castell, S.; Fricke, J.; et al. Personen mit Migrationshintergrund in der NAKO Gesundheitsstudie–soziodemografische Merkmale und Vergleiche mit der autochthonen deutschen Bevölkerung. Bundesgesundheitsblatt-Gesundh. -Gesundh. 2020, 63, 279–289. [Google Scholar] [CrossRef]
- Yildirim, T. Inanspruchnahme von Präventionsangeboten in der GKV durch türkischstämmige Migranten am Beispiel von Früherkennungs-und U- Untersuchungen; Bielefeld University: Bielefeld, Germany, 2017. [Google Scholar]
- Lange, C.; Jentsch, F.; Allen, J.; Hoebel, J.; Kratz, A.L.; von der Lippe, E.; Müters, S.; Schmich, P.; Thelen, J.; Wetzstein, M.; et al. Data Resource Profile: German Health Update (GEDA)—The health interview survey for adults in Germany. Int. J. Epidemiol. 2015, 44, 442–450. [Google Scholar] [CrossRef] [PubMed]
- Robert Koch-Institut. German Health Update: New Data for Germany and Europe the Background to and Methodology Applied in GEDA 2014/2015-EHIS; Robert Koch Institute: Berlin, Germany, 2017. [Google Scholar]
- Lampert, T.; Kroll, L.E.; Müters, S.; Stolzenberg, H. Messung des sozioökonomischen Status in der Studie “Gesundheit in Deutschland aktuell” (GEDA). Bundesgesundheitsblatt-Gesundh. Gesundh. 2013, 56, 131–143. [Google Scholar] [CrossRef]
- StataCorp. Stata Statistical Software: Release 15; StataCorp LP.: College Station, TX, USA, 2017. [Google Scholar]
- Gesink, D.; Mihic, A.; Antal, J.; Filsinger, B.; Racey, C.S.; Perez, D.F.; Norwood, T.; Ahmad, F.; Kreiger, N.; Ritvo, P. Who are the under- and never-screened for cancer in Ontario: A qualitative investigation. BMC Public Health 2014, 14, 495. [Google Scholar] [CrossRef] [PubMed]
- Robb, K.A.; Solarin, I.; Power, E.; Atkin, W.; Wardle, J. Attitudes to colorectal cancer screening among ethnic minority groups in the UK. BMC Public Health 2008, 8, 34. [Google Scholar] [CrossRef]
- Mikkelsen, E.M.; Thomsen, M.K.; Tybjerg, J.; Friis-Hansen, L.; Andersen, B.; Jørgensen, J.C.R.; Baatrup, G.; Njor, S.H.; Mehnert, F.; Rasmussen, M. Colonoscopy-related complications in a nationwide immunochemical fecal occult blood test-based colorectal cancer screening program. Clin. Epidemiol. 2018, 10, 1649–1655. [Google Scholar] [CrossRef]
- Michie, S.; Dormandy, E.; Marteau, T.M. The multi-dimensional measure of informed choice: A validation study. Patient Educ. Couns. 2002, 48, 87–91. [Google Scholar] [CrossRef]
- Marteau, T.M.; Dormandy, E.; Michie, S. A measure of informed choice. Health Expect. 2001, 4, 99–108. [Google Scholar] [CrossRef]
- Berens, E.-M.; Reder, M.; Razum, O.; Kolip, P.; Spallek, J. Informed Choice in the German Mammography Screening Program by Education and Migrant Status: Survey among First-Time Invitees. PLoS ONE 2015, 10, e0142316. [Google Scholar] [CrossRef]
- Gabel, P.; Larsen, M.B.; Edwards, A.; Kirkegaard, P.; Andersen, B. Effectiveness of a decision aid for colorectal cancer screening on components of informed choice according to educational attainment: A randomised controlled trial. PLoS ONE 2020, 15, e0241703. [Google Scholar] [CrossRef]
- Christou, A.; Thompson, S.C. Colorectal cancer screening knowledge, attitudes and behavioural intention among Indigenous Western Australians. BMC Public Health 2012, 12, 528. [Google Scholar] [CrossRef] [PubMed]
- Turrin, A.; Zorzi, M.; Giorgi Rossi, P.; Senore, C.; Campari, C.; Fedato, C.; Naldoni, C.; Anghinoni, E.; Carrozzi, G.; De’ Bianchi, P.S.; et al. Colorectal cancer screening of immigrants to Italy. Figures from the 2013 National Survey. Prev. Med. 2015, 81, 132–137. [Google Scholar] [CrossRef] [PubMed]
- Palmer, R.C.; Midgette, L.A.; Mullan, I.D. Colorectal cancer screening preferences among African Americans: Which screening test is preferred? J. Cancer Educ. 2010, 25, 577–581. [Google Scholar] [CrossRef] [PubMed]
- Brzoska, P.; Razum, O. Migration and occupational health: High work-related burden. Public Health Forum 2015, 23, 113–115. [Google Scholar] [CrossRef]
- Baier, M.; Calonge, N.; Cutter, G.; McClatchey, M.; Schoentgen, S.; Hines, S.; Marcus, A.; Ahnen, D. Validity of self-reported colorectal cancer screening behavior. Cancer Epidemiol. Biomark. Prev. 2000, 9, 229–232. [Google Scholar]
- Rauscher, G.H.; Johnson, T.P.; Cho, Y.I.; Walk, J.A. Accuracy of self-reported cancer-screening histories: A meta-analysis. Cancer Epidemiol. Biomark. Prev. 2008, 17, 748–757. [Google Scholar] [CrossRef] [PubMed]
- Hoebel, J.; Richter, M.; Lampert, T. Social status and participation in health checks in men and women in Germany: Results from the German Health Update (GEDA), 2009 and 2010. Dtsch. Ärzteblatt Int. 2013, 110, 679–685. [Google Scholar] [CrossRef]
- Statistisches Bundesamt. Bevölkerung und Erwerbstätigkeit. Bevölkerung mit Migrationshintergrund. Ergebnisse des Mikrozensus 2015 (Fachserie 1 Reihe 2.2); Statistisches Bundesamt: Wiesbaden, Germany, 2016. [Google Scholar]
- Wang, A.M.Q.; Yung, E.M.; Nitti, N.; Shakya, Y.; Alamgir, A.K.M.; Lofters, A.K. Breast and Colorectal Cancer Screening Barriers Among Immigrants and Refugees: A Mixed-Methods Study at Three Community Health Centres in Toronto, Canada. J. Immigr. Minority Health 2019, 21, 473–482. [Google Scholar] [CrossRef]
- Shapiro, E. Barriers to Colorectal Cancer Screening Among Russian-speaking Immigrants: The Importance of Culture and Home Country Experiences. J. Immigr. Minority Health 2021. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).