Abstract
In this study, the treatment of a winery wastewater (WW) by a photo-Fenton process, employing a combination of (S,S)-ethylenediamine-N,N’-disuccinic acid (EDDS) as a quelating agent and hydroxylamine (HA) to accelerate the Fe2+ recovery, was presented for the first time. The aim of this study was to improve the photo-Fenton process under UV-C, UV-A and solar radiation. The results show that under the best operational conditions—pH = 6.0, [Fe2+] = 5.0 mM, [H2O2] = 175 mM, [EDDS] = 1.0 mM, [HA] = 1.0 mM, agitation 350 rpm, time 240 min, temperature 298 K—a chemical oxygen demand (COD) removal of 93.2, 81.6 and 60.6% was achieved for UV-C, solar and UV-A radiation, respectively. EDDS-photo-Fenton is an excellent process for WW treatment.
1. Introduction
Wineries and other grape processing industries annually generate a large volume of wastewater. In addition, wineries wastewater treatment plants (WWTPs) are normally designed for the vintage period. Thus, they are oversized during the rest of the year, leading to an increase in foot implantation and high investment costs [1]. Winery wastewaters generally have a high organic load and phytotoxicity, constituting an environmental danger if directly disposed of in natural water courses or soils. Therefore, suitable treatment processes must be applied to reduce their pollutant load [2]. Advanced oxidation processes (AOPs) with the generation of hydroxyl radical (), the second-strongest known oxidant to fluorine, have been proposed as a complementary technology for the degradation of organic matter [3]. However, at higher pH levels, iron oxohydroxides are formed and ferric hydroxide is precipitated, which results in a decrease in the efficiency of the photo-Fenton process [4]. The application of a chelating agent such as Ethylenediamine-N,N′-disuccinic acid (EDDS) can be applied to decrease the precipitation of iron and increase photo-Fenton’s efficiency at a neutral pH [5]. Solar radiation was observed to be effective in the treatment of WW [6]; however, the application of the Fe2+-EDDS complex was never applied in the solar photo-Fenton process for the treatment of WW. Therefore, the aims of this study were to (1) optimize the photo-Fenton process and (2) evaluate the application of different types of radiation at pH 3.0 and 6.0.
2. Material and Methods
2.1. Reagents and Winery Wastewater Applied
Iron (II) sulfate heptahydrate (FeSO4·7H2O) was acquired by Panreac, and hydrogen peroxide (H2O2 30% w/w), ethylenediamine-N,N’-disuccinic acid (C10H16N2O8, EDDS) and hydroxylamine hydrochloride (NH2OH·HCl, HA) were acquired by Sigma-Aldrich. NaOH and H2SO4 (95%) were both obtained from Analar Normapur. Deionized water was used to prepare the respective solutions. The WW was collected from a Portuguese winery production unity located in the Douro region (northeastern Portugal), placed in small containers, and cooled at −40 °C until use.
2.2. Analytical Technics
Different physical–chemical parameters were determined in order to characterize the WW, including turbidity, total suspended solids (TSS), chemical oxygen demand (COD), biological oxygen demand (BOD5), total organic carbon (TOC) and total polyphenols. The main wastewater characteristics are shown in Table 1.

Table 1.
Characterization of winery wastewater.
2.3. Photo-Fenton Experimental Set-Up
In the photo-Fenton process, three reactors were used: (1) a batch cylindrical photoreactor equipped with a UV-C low-pressure mercury vapor lamp (TNN 15/32)—working power = 15 W (795.8 W/m2) and λmax = 254 nm (Heraeus, Hanau, Germany); (2) a UV-A LED photo system with 12 InGaN LED lamps (Roithner APG2C1-365E LEDS) with a maximum emission wavelength at 365 nm; (3) a PYREX glass batch reactor under natural solar radiation, which was used at the Laboratory Block building (4°17′15.2″ N 7°44′18.2″ W) at the University of Trás-os-Montes e Alto Douro (Portugal). In all cases, experiments were performed in a 500 mL stirred glass reactor under radiation. The temperature was maintained constant at 298 K for 240 min, and the photo-Fenton process was optimized under the following steps: (1) variation in the hydrogen peroxide dosage (8–349 mM), (2) variation in the Fe2+ dosage (1.0–10 mM), (3) variation in the EDDS dosage (1.0–10 mM), (4) variation in the HA dosage (1.0–10 mM) and (5) variation in radiation type (UV-C, UV-A and solar light) vs. pH (3.0 and 6.0). After the reaction has started, 2.5 mL of solution was withdrawn for COD measurements at different reaction times, completing a total period of 240 min.
2.4. Kinetic Modeling
In all the experiments, the COD degradation followed pseudo first-order kinetics, as follows (Equation (1)):
where the slope is km and COD0 and COD are the chemical oxygen demand at times t = 0 and t = t.
To determine the percentage of COD removal, Equation (2) was used as follows [7,8]:
where, C0 and Ct are the initial and final concentrations, respectively.
2.5. Statistical Analysis
All the COD removal experiments were performed in triplicate and the observed standard deviation was always less than 5% of the reported values. The statistical analysis was performed by OriginLab 2019 software (Northampton, MA, USA).
3. Results and Discussion
3.1. Chemical Treatment of WW
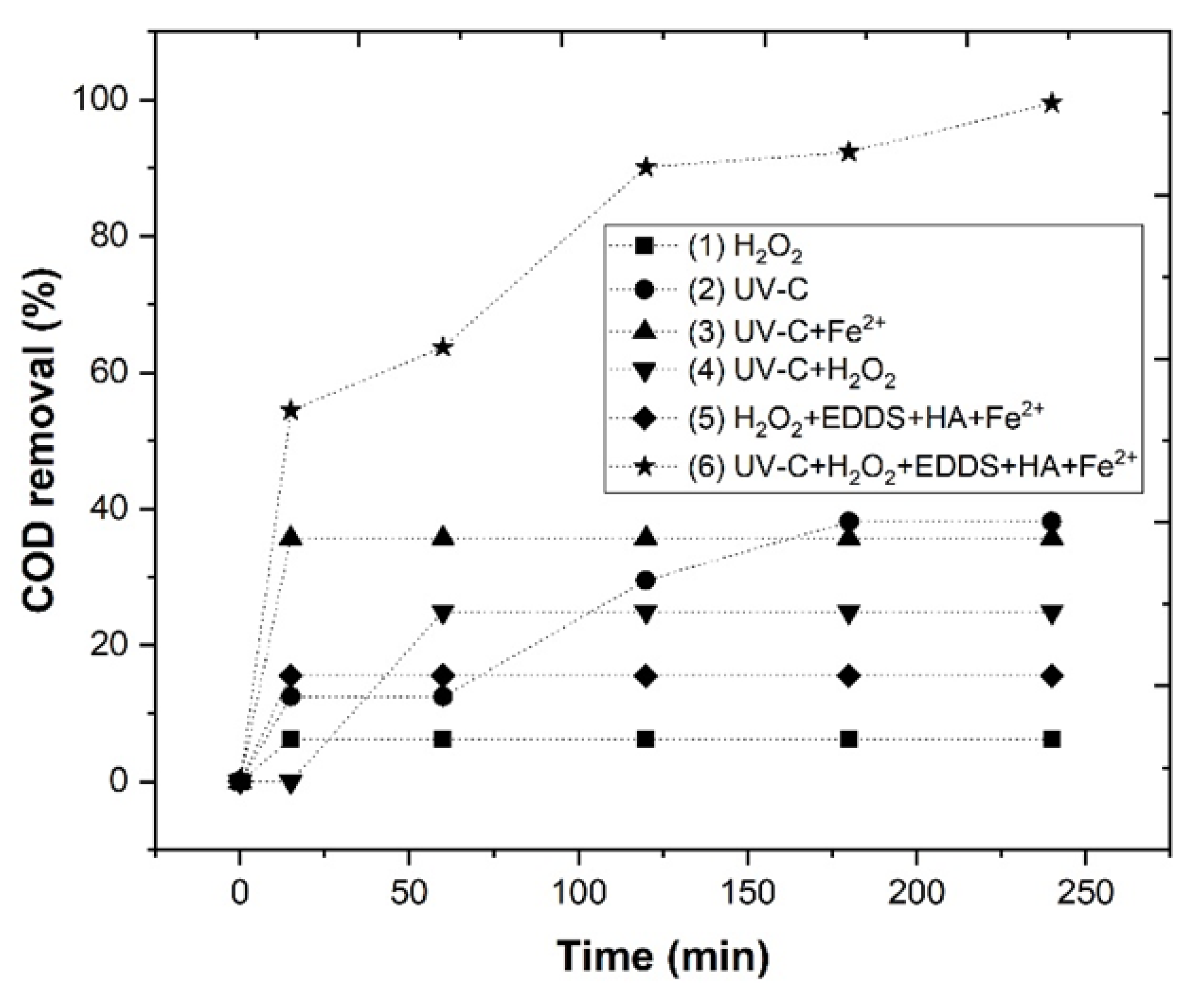
To evaluate the efficiency of the photo-Fenton system, the following experiments were carried out: (1) H2O2, (2) UV-C, (3) UV-C+Fe2+, (4) UV-C+H2O2, (5) H2O2+EDDS+HA+Fe2+, (6) UV-C+H2O2+EDDS+HA+Fe2+, reaching a COD removal of 6.2, 38.1, 35.7, 24.9, 15.5 and 99.5%, respectively (Figure 1). The high efficiency of the Fe2+-EDDS-photo-Fenton process can be attributed to the formation of more hydroxyl radicals () than the other processes. This means that the UV-C lamp shows a sensitive increase in COD removal in comparison to the EDDS-Fenton system.
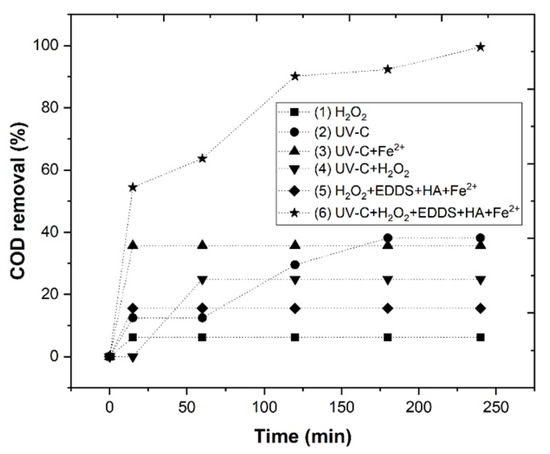
Figure 1.
Chemical treatment of WW under operational conditions, as follows: (Fe2+) = 5 mM, (H2O2) = 175 mM, (EDDS) = 1 mM, (HA) = 1 mM, pH 3.0, agitation 350 rpm, temperature 298 K, UV lamp = TNN 15/32 Heraeus, reaction time 240 min.
3.2. Optimization of Photo-Fenton Process
In the photo-Fenton process, the Fe2+ reacts with H2O2 to produce hydroxyl radicals (), as observed in Equation (3) [9]. In this study, the oxidant H2O2 concentration was initially optimized. The results show a COD removal of 82.5% with the application of 175 mM H2O2. The application of a H2O2 concentration above the excess of H2O2 consumed the radicals, as observed in Equation (4) [10]:
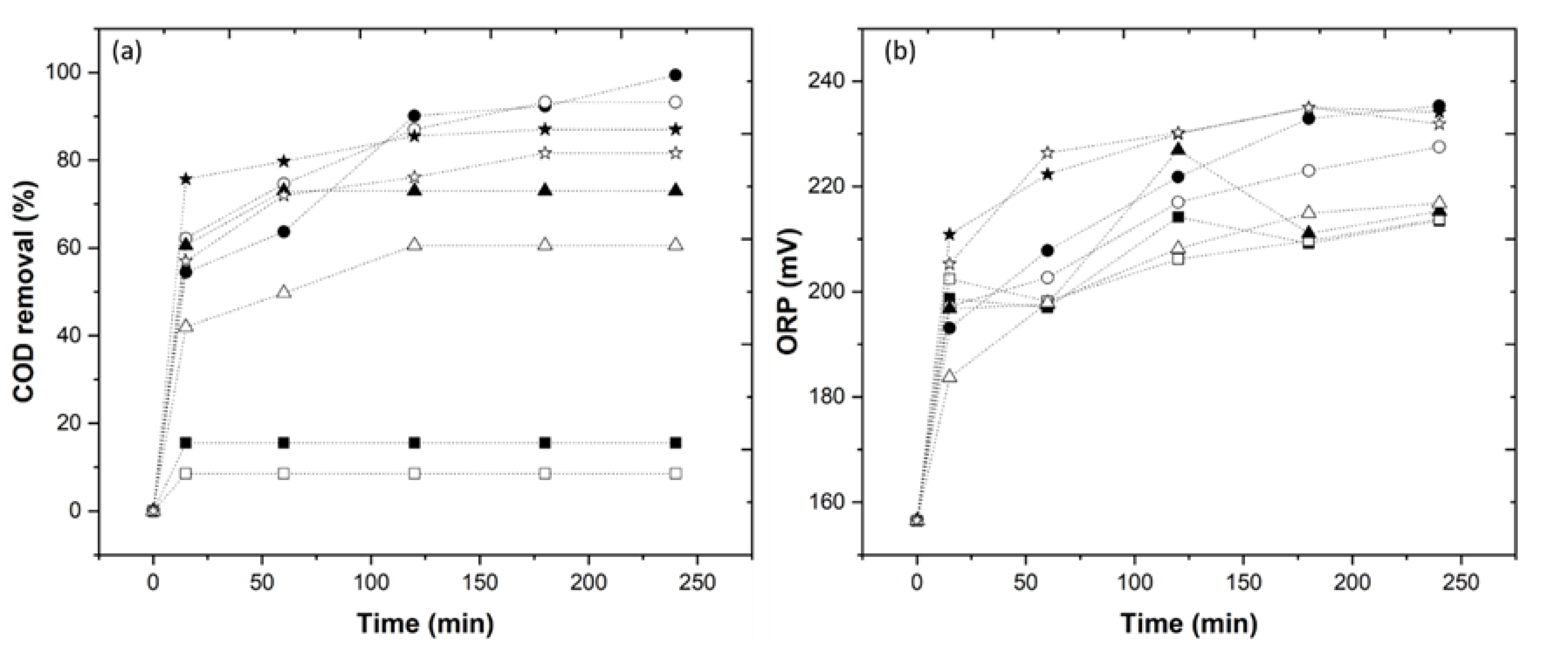
The catalyst Fe2+ concentration varies from 1.0 to 10 mM, and the results show a high COD removal with the application of 5.0 mM Fe2+. The catalyst type also varies, and the results show a higher efficiency with the application of the Fe2+ catalyst, similar to the study by Rodríguez-Chueca et al. [11]. The complexing agents, EDDS (1.0–10 mM) and HA (1.0–10 mM), were tested, and the results show a higher COD removal with the application of a molar ratio 1/5/1 in the EDDS-Fe2+/HA system (data not shown). Finally, the pH varied (3.0 and 6.0), and with application of pH 6.0, there was a COD removal of 93.2%, as observed in Figure 2a (UV-C) > 81.6% (solar radiation) > 60.6% (UV-A) > 8.5% (dark Fenton). As shown in Figure 2b, the ORP values at pH 6.0 employing dark Fenton, UV-C, UV-A and solar radiation (213.8, 227.5, 216.8 and 231.9 mV, respectively) were similar to the ORP values at pH 3.0 (213.5, 235.3, 215.3 and 234.1 mV, respectively).
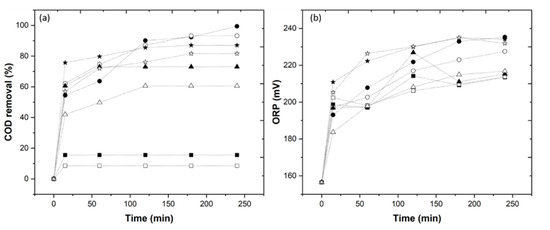
Figure 2.
Evolution of the (a) COD removal, (b) ORP value through the photo-Fenton experiments, under the influence of different UV sources at pH 3 and 6. Dark Fenton/pH 3 (  ), dark Fenton/pH 6 (
), dark Fenton/pH 6 (  ), UV-C/pH 3 (
), UV-C/pH 3 (  ), UV-C/pH 6 (
), UV-C/pH 6 (  ), UV-A/pH 3 (
), UV-A/pH 3 (  ), UV-A/pH 6 (
), UV-A/pH 6 (  ), solar radiation/pH 3 (
), solar radiation/pH 3 (  ), solar radiation/pH 6 (
), solar radiation/pH 6 (  ).
).
 ), dark Fenton/pH 6 (
), dark Fenton/pH 6 (  ), UV-C/pH 3 (
), UV-C/pH 3 (  ), UV-C/pH 6 (
), UV-C/pH 6 (  ), UV-A/pH 3 (
), UV-A/pH 3 (  ), UV-A/pH 6 (
), UV-A/pH 6 (  ), solar radiation/pH 3 (
), solar radiation/pH 3 (  ), solar radiation/pH 6 (
), solar radiation/pH 6 (  ).
).
4. Conclusions
In this work, the photo-Fenton process was optimized by the addition of EDDS and HA to decrease the precipitation of iron and increase the regeneration of Fe3+ to Fe2+. In addition, three kind of radiations were tested (UV-C, UV-A and solar), and it is concluded that:
- The application of 175 mM H2O2 achieves a high COD removal (82.5%);
- The generation of radicals is greatly promoted by the addition of hydroxylamine, and the molar ratio of EDDS-Fe/HA system (1/5/1) achieves a higher COD removal (99.4%);
- With the application of the Fe2+-EDDS/HA system, it is concluded that the photo-Fenton process at pH 6.0 achieves similar COD reductions regarding pH 3.0.
- Solar radiation achieves a similar COD removal than UV-A radiation and can be a viable and cheap alternative.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/ECP2022-12652/s1.
Author Contributions
Conceptualization, N.J., C.A., A.R.T. and L.M.; methodology, N.J., C.A., A.R.T. and L.M.; software, N.J.; validation, N.J., A.R.T. and L.M.; formal analysis, N.J.; investigation, N.J.; resources, N.J., C.A., M.S.L. and J.A.P.; data curation, N.J.; writing—original draft preparation, N.J.; writing—review and editing, N.J., M.S.L. and J.A.P.; visualization, N.J., M.S.L. and J.A.P.; supervision, M.S.L. and J.A.P.; project administration, J.A.P.; funding acquisition, J.A.P. All authors have read and agreed to the published version of the manuscript.
Funding
The authors are grateful for the financial support of the Project AgriFood XXI, operation nº NORTE-01-0145-FEDER-000041, and to the Fundação para a Ciência e a Tecnologia (FCT) for the financial support provided to CQVR through UIDB/00616/2020. Ana R. Teixeira also thanks the FCT for the financial support provided through the doctoral scholarship UI/BD/150847/2020.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Amaral-Silva, N.; Martins, R.C.; Paiva, C.; Castro-Silva, S.; Quinta-Ferreira, R.M. A New Winery Wastewater Treatment Approach during Vintage Periods Integrating Ferric Coagulation, Fenton Reaction and Activated Sludge. J. Environ. Chem. Eng. 2016, 4, 2207–2215. [Google Scholar] [CrossRef]
- Ferreira, R.; Gomes, J.; Martins, R.C.; Costa, R.; Quinta-Ferreira, R.M. Winery Wastewater Treatment by Integrating Fenton’s Process with Biofiltration by Corbicula Fluminea. J. Chem. Technol. Biotechnol. 2018, 93, 333–339. [Google Scholar] [CrossRef]
- Zhang, Y.; Klamerth, N.; Messele, S.A.; Chelme-Ayala, P.; El-Din, M.G. Kinetics Study on the Degradation of a Model Naphthenic Acid by Ethylenediamine-N, N’-Disuccinic Acid-Modified Fenton Process. J. Hazard. Mater. 2016, 318, 371–378. [Google Scholar] [CrossRef] [PubMed]
- Dowd, K.O.; Pillai, S.C. Photo-Fenton Disinfection at near Neutral PH: Process, Parameter Optimization and Recent Advances. J. Environ. Chem. Eng. 2020, 8, 104063. [Google Scholar] [CrossRef]
- Ahile, U.J.; Wuana, R.A.; Itodo, A.U.; Sha, R.; Dantas, R.F. A Review on the Use of Chelating Agents as an Alternative to Promote Photo-Fenton at Neutral PH: Current Trends, Knowledge Gap and Future Studies. Sci. Total Environ. 2020, 710, 134872. [Google Scholar] [CrossRef] [PubMed]
- Lucas, M.S.; Mosteo, R.; Maldonado, M.I.; Malato, S.; Peres, J.A. Solar Photochemical Treatment of Winery Wastewater in a CPC Reactor. J. Agric. Food Chem. 2009, 57, 11242–11248. [Google Scholar] [CrossRef] [PubMed]
- Jorge, N.; Teixeira, A.R.; Matos, C.C.; Lucas, M.S.; Peres, J.A. Combination of Coagulation–Flocculation–Decantation and Ozonation Processes for Winery Wastewater Treatment. Int. J. Environ. Res. Public Health 2021, 18, 8882. [Google Scholar] [CrossRef] [PubMed]
- Jorge, N.; Teixeira, A.R.; Guimarães, V.; Lucas, M.S.; Peres, J.A. Treatment of Winery Wastewater with a Combination of Adsorption and Thermocatalytic Processes. Processes 2022, 10, 75. [Google Scholar] [CrossRef]
- Tan, W.; Ai, J.; Fan, Y.; Liu, X.; Xu, Y.; Zhang, H.; Huang, Y.H. Enhanced Mineralization of Reactive Black 5 by Waste Iron Oxide via Photo-Fenton Process. Res. Chem. Intermed. 2020, 46, 4423–4431. [Google Scholar] [CrossRef]
- Baba, Y.; Yatagai, T.; Harada, T.; Kawase, Y. Hydroxyl Radical Generation in the Photo-Fenton Process: Effects of Carboxylic Acids on Iron Redox Cycling. Chem. Eng. J. 2015, 277, 229–241. [Google Scholar] [CrossRef]
- Rodríguez-Chueca, J.; Amor, C.; Silva, T.; Dionysiou, D.D.; Li, G.; Lucas, M.S.; Peres, J.A. Treatment of Winery Wastewater by Sulphate Radicals: HSO5-/Transition Metal/UV-A LEDs. Chem. Eng. J. 2017, 310, 473–483. [Google Scholar] [CrossRef] [Green Version]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).