Inhibitory Effects of 5-Fluorouracil on the Growth of 4-Hydroxytamoxifen-Resistant and Sensitive Breast Cancer Cells †
Abstract
:1. Introduction
2. Materials and Methods
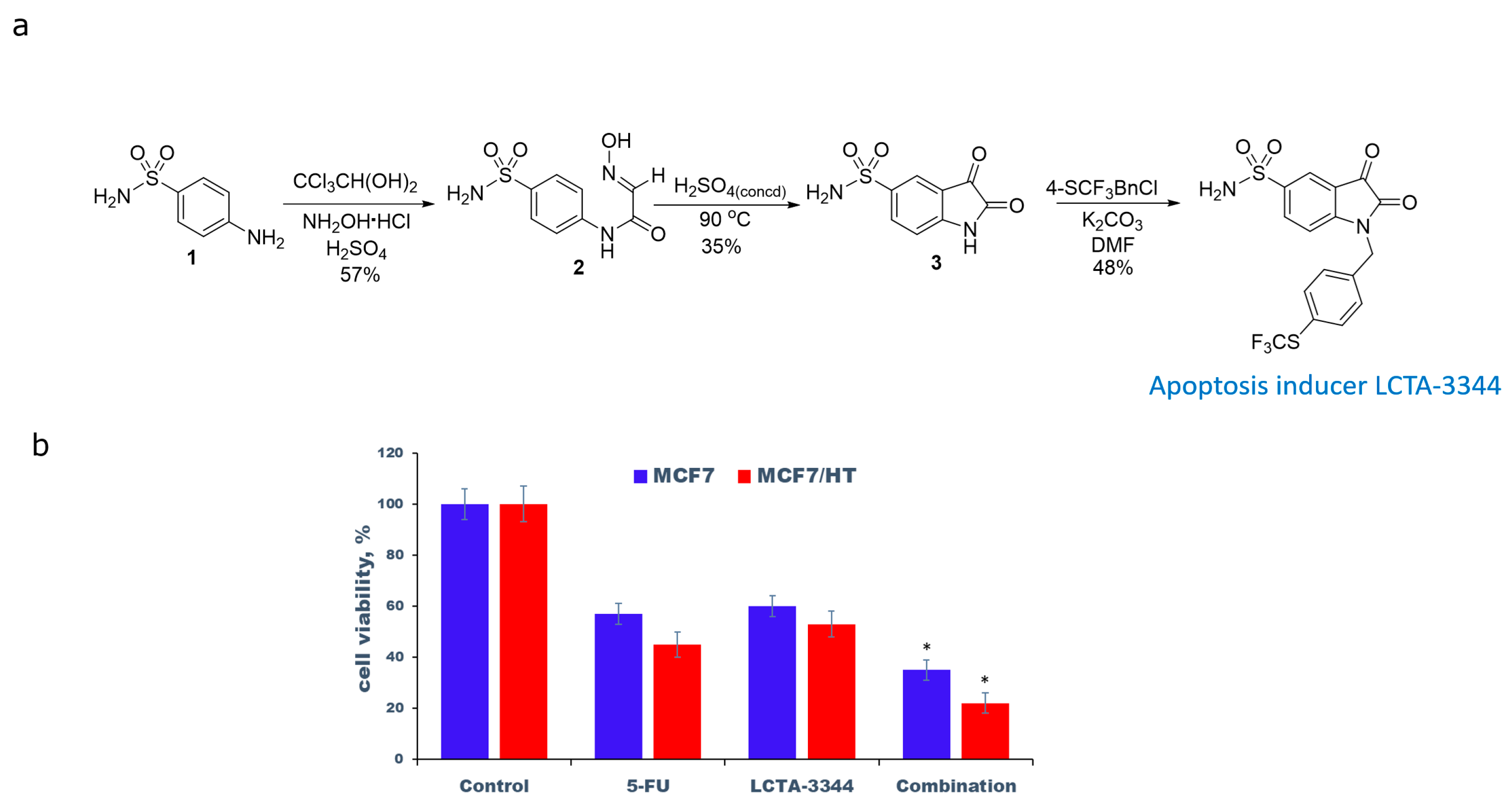
2.1. Synthesis, Reagents and Cell Lines
2.2. The Evaluation of the Antiproliferative Activity
2.3. Statistical Analysis
2.4. Immunoblotting
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Mojaddami, A.; Sakhteman, A.; Fereidoonnezhad, M.; Faghih, Z.; Najdian, A.; Khabnadideh, S.; Sadeghpour, H.; Rezaei, Z. Binding mode of triazole derivatives as aromatase inhibitors based on docking, protein ligand interaction fingerprinting, and molecular dynamics simulation studies. Res. Pharm. Sci. 2017, 12, 21–30. [Google Scholar] [PubMed]
- Kamruz, E.; Rostamizadeh, K.; Babazadeh, M. Synthesis and characterization of linear-dendritic of PEG and citric acid as a nanocarrier for tamoxifen. Res. Pharm. Sci. 2012, 7, S576. [Google Scholar]
- Jordan, V.C. 50th anniversary of the first clinical trial with ICI 46,474 (tamoxifen): Then what happened? Endocr.-Relat. Cancer 2021, 28, R11–R30. [Google Scholar] [CrossRef] [PubMed]
- Nabieva, N.; Fasching, P.A. Endocrine Treatment for Breast Cancer Patients Revisited-History, Standard of Care, and Possibilities of Improvement. Cancers 2021, 13, 5643. [Google Scholar] [CrossRef] [PubMed]
- Rutman, R.J.; Cantarow, A.; Paschkis, K.E. Studies in 2-acetylaminofluorene carcinogenesis. III. The utilization of uracil-2-C14 by preneoplastic rat liver and rat hepatoma. Cancer Res. 1954, 14, 119–123. [Google Scholar] [PubMed]
- Krymov, S.K.; Scherbakov, A.M.; Salnikova, D.I.; Sorokin, D.V.; Dezhenkova, L.G.; Ivanov, I.V.; Vullo, D.; De Luca, V.; Capasso, C.; Supuran, C.T.; et al. Synthesis, biological evaluation, and in silico studies of potential activators of apoptosis and carbonic anhydrase inhibitors on isatin-5-sulfonamide scaffold. Eur. J. Med. Chem. 2022, 228, 113997. [Google Scholar] [CrossRef] [PubMed]
- Iselt, M.; Holtei, W.; Hilgard, P. The tetrazolium dye assay for rapid in vitro assessment of cytotoxicity. Arzneimittelforschung 1989, 39, 747–749. [Google Scholar] [PubMed]
- Volkova, Y.A.; Antonov, Y.S.; Komkov, A.V.; Scherbakov, A.M.; Shashkov, A.S.; Menchikov, L.G.; Chernoburova, E.I.; Zavarzin, I.V. Access to steroidal pyridazines via modified thiohydrazides. RSC Adv. 2016, 6, 42863–42868. [Google Scholar] [CrossRef]
- Scherbakov, A.M.; Lobanova, Y.S.; Shatskaya, V.A.; Onopchenko, O.V.; Gershtein, E.S.; Krasil’nikov, M.A. Activation of mitogenic pathways and sensitization to estrogen-induced apoptosis: Two independent characteristics of tamoxifen-resistant breast cancer cells? Breast Cancer Res. Treat. 2006, 100, 1–11. [Google Scholar] [CrossRef]
- Mruk, D.D.; Cheng, C.Y. Enhanced chemiluminescence (ECL) for routine immunoblotting: An inexpensive alternative to commercially available kits. Spermatogenesis 2011, 1, 121–122. [Google Scholar] [CrossRef]
- Scherbakov, A.M.; Basharina, A.A.; Sorokin, D.V.; Mikhaevich, E.I.; Mizaeva, I.E.; Mikhaylova, A.L.; Bogush, T.A.; Krasil’nikov, M.A. Targeting hormone-resistant breast cancer cells with docetaxel: A look inside the resistance. Cancer Drug Resist. 2023, 6, 103–115. [Google Scholar] [CrossRef] [PubMed]
- Sarkaria, J.N.; Miller, E.M.; Parker, C.J.; Jordan, V.C.; Mulcahy, R.T. 4-Hydroxytamoxifen, an active metabolite of tamoxifen, does not alter the radiation sensitivity of MCF-7 breast carcinoma cells irradiated in vitro. Breast Cancer Res. Treat. 1994, 30, 159–165. [Google Scholar] [CrossRef] [PubMed]
- Custódio, J.B.; Almeida, L.M.; Madeira, V.M. The active metabolite hydroxytamoxifen of the anticancer drug tamoxifen induces structural changes in membranes. Biochim. Et Biophys. Acta 1993, 1153, 308–314. [Google Scholar] [CrossRef] [PubMed]
- Mohammed, H.; Russell, I.A.; Stark, R.; Rueda, O.M.; Hickey, T.E.; Tarulli, G.A.; Serandour, A.A.; Birrell, S.N.; Bruna, A.; Saadi, A.; et al. Progesterone receptor modulates ERα action in breast cancer. Nature 2015, 523, 313–317. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.J.; Gorski, J. Estrogen-induced transcription of the progesterone receptor gene does not parallel estrogen receptor occupancy. Proc. Natl. Acad. Sci. USA 1996, 93, 15180–15184. [Google Scholar] [CrossRef] [PubMed]
- Costa, R.L.B.; Han, H.S.; Gradishar, W.J. Targeting the PI3K/AKT/mTOR pathway in triple-negative breast cancer: A review. Breast Cancer Res. Treat. 2018, 169, 397–406. [Google Scholar] [CrossRef]
- Lu, Y.; Liu, B.; Liu, Y.; Yu, X.; Cheng, G. Dual effects of active ERK in cancer: A potential target for enhancing radiosensitivity. Oncol. Lett. 2020, 20, 993–1000. [Google Scholar] [CrossRef]
- Bandyopadhayaya, S.; Akimov, M.G.; Verma, R.; Sharma, A.; Sharma, D.; Kundu, G.C.; Gretskaya, N.M.; Bezuglov, V.V.; Mandal, C.C. N-arachidonoyl dopamine inhibits epithelial-mesenchymal transition of breast cancer cells through ERK signaling and decreasing the cellular cholesterol. J. Biochem. Mol. Toxicol. 2021, 35, e22693. [Google Scholar] [CrossRef]
- Testa, J.R.; Tsichlis, P.N. AKT signaling in normal and malignant cells. Oncogene 2005, 24, 7391–7393. [Google Scholar] [CrossRef]
- Kong, D.; Yamori, T. Advances in development of phosphatidylinositol 3-kinase inhibitors. Curr. Med. Chem. 2009, 16, 2839–2854. [Google Scholar] [CrossRef]
- Liao, Y.; Hung, M.C. Physiological regulation of Akt activity and stability. Am. J. Transl. Res. 2010, 2, 19–42. [Google Scholar] [PubMed]

| IC50 of HT, µM | IC50 of 5-FU, µM | |
|---|---|---|
| MCF7 | 5.2 ± 0.6 | 6.3 ± 0.7 |
| MCF7/HT | 9.8 ± 1.0 * | 2.1 ± 0.3 * |
| Resistance index # | 1.9 | 0.3 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sorokin, D.V.; Krymov, S.K.; Cherednichenko, M.N.; Mikhaylova, A.L.; Salnikova, D.I.; Shchekotikhin, A.E.; Scherbakov, A.M. Inhibitory Effects of 5-Fluorouracil on the Growth of 4-Hydroxytamoxifen-Resistant and Sensitive Breast Cancer Cells. Eng. Proc. 2023, 56, 192. https://doi.org/10.3390/ASEC2023-16332
Sorokin DV, Krymov SK, Cherednichenko MN, Mikhaylova AL, Salnikova DI, Shchekotikhin AE, Scherbakov AM. Inhibitory Effects of 5-Fluorouracil on the Growth of 4-Hydroxytamoxifen-Resistant and Sensitive Breast Cancer Cells. Engineering Proceedings. 2023; 56(1):192. https://doi.org/10.3390/ASEC2023-16332
Chicago/Turabian StyleSorokin, Danila Vladimirovich, Stepan K. Krymov, Margo N. Cherednichenko, Alexandra L. Mikhaylova, Diana I. Salnikova, Andrey E. Shchekotikhin, and Alexander M. Scherbakov. 2023. "Inhibitory Effects of 5-Fluorouracil on the Growth of 4-Hydroxytamoxifen-Resistant and Sensitive Breast Cancer Cells" Engineering Proceedings 56, no. 1: 192. https://doi.org/10.3390/ASEC2023-16332
APA StyleSorokin, D. V., Krymov, S. K., Cherednichenko, M. N., Mikhaylova, A. L., Salnikova, D. I., Shchekotikhin, A. E., & Scherbakov, A. M. (2023). Inhibitory Effects of 5-Fluorouracil on the Growth of 4-Hydroxytamoxifen-Resistant and Sensitive Breast Cancer Cells. Engineering Proceedings, 56(1), 192. https://doi.org/10.3390/ASEC2023-16332









