1. Introduction
Polymerase chain reaction (PCR) is a technique that amplifies the target DNA. It is an important part of modern diagnostic medicine that can diagnose diseases with a small amount of DNA. Recently, it is possible to check the target DNA in real time using real-time PCR devices. As they are usually expensive, only large hospitals or laboratories can afford them. However, nowadays it is very important to quickly and accurately diagnose viruses for infectious diseases, requiring a PCR device capable of point-of-care test (POCT) [
1,
2]. For POCT devices, miniaturization and cost reduction are essential, which is what most PCR chips aim for [
1,
2,
3,
4].
In this paper, a low-cost and compact multiplexed real-time PCR system is presented. The proposed system utilizes a PCB-based PCR chip and a low-cost, high-performance, open-platform camera that is prevalent due to the development of smartphone cameras [
5]. The optics of the system are made very compact by adopting the lateral illumination method for fluorescence excitation, and they are able to multiplex up to four colors. As the PCB substrate of the PCR chip has a heater pattern and a thermistor attached, it was possible to easily implement thermal cycling without complicated structures. In this paper, the optical performance for single-dye fluorescence detection and the crosstalk between dyes was investigated with standard fluorescent materials. Additionally,
Chlamydia trachomatis DNA was employed for verification of the real-time quantification performance. The experimental results showed that the proposed system is highly feasible for a POC real-time PCR device.
2. Materials and Methods
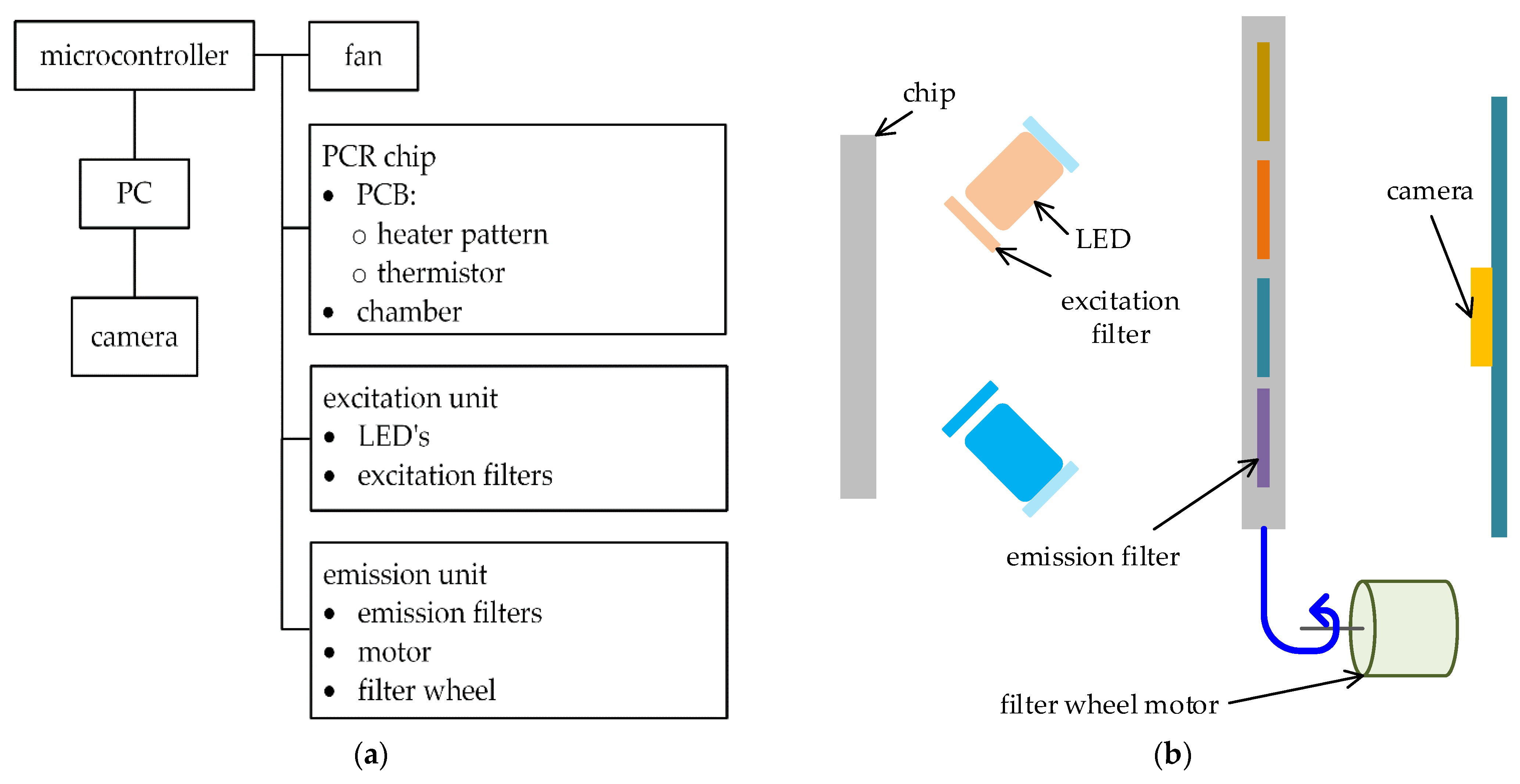
Figure 1 shows the functional block diagram (a) and the structure of the proposed system (b). The polycarbonate reaction chamber of the PCR chip was constructed on a PCB substrate with a heater pattern and a thermistor. The reaction chamber is structurally thin and wide, and its one side is optically transparent, providing the high-speed PCR thermal cycling and easy measurement of fluorescence brightness with a camera.
The PCR chip is placed vertically to the camera direction (
Figure 1b). The color bands of the excitation and emission filters were selected to effectively detect HEX, ROX, CY5, and FAM dyes. The excitation light for each color dye consists of an LED and an excitation filter illuminated at an angle of 45 degree right next to the transparent side of the chamber (
Figure 1b). Four pairs of LEDs and excitation filters were placed at 90-degree intervals on the parallel plane to the transparent chamber side. An emission filter is selected by a filter wheel driven by a linear servo motor. Fluorescence passing through the corresponding emission filter was detected by the camera.
Two separate experiments were performed to verify the fluorescence detection performance: single dye fluorescence experiment and fluorescence crosstalk experiment. The brightness of fluorescence of the plateau phase of PCR was emulated by 2 pico-mole/36 µL dye solution for each fluorescence. In the single dye experiment, two fluorescence images obtained by injecting the emulation dye solution and double-distilled water (DDW) into the chip, respectively, were compared. To investigate crosstalk between dyes, the image of the chip with the emulation solution containing all dyes and that with the solution without only the dye to be measured were compared.
The quantitative analysis performance was also investigated with DNA amplification experiments. The reagents used in the experiment were composed of a 36 µL solution with 6 copies of Chlamydia trachomatis DNA, Master mix, Primer mix, and DDW. The PCR thermal cycling conditions were 50 °C for 2 min, 95 °C for 10 min, followed by 40 cycles of 95 °C for 15 s and 60 °C for 1 min. The chip image was captured in the last 2 s of the 60 °C annealing step of each cycle to measure the fluorescence brightness. The region of interest (ROI) for determining the brightness of an image was set to the smallest rectangular area covering the chamber through image processing.
3. Results and Discussions
Figure 2 shows the ROI images of each experiment result. The left of the figure shows the result images for the single dye experiment. Each column indicates the dye to be measured. The “DDW” labeled row shows the image taken by injected DDW and the “Target” row shows the images when the corresponding emulation solutions were injected. The right picture of
Figure 2 shows the crosstalk experiment result images. In this picture, the row labeled “ALL” shows the images of the chip with all the dye, and the row labeled “Target” shows the images excluding the corresponding dye. From the resulting figure, it can be seen that the proposed system performs well in fluorescence detection without crosstalk.
Table 1 summarizes the average brightness of each ROI image in
Figure 2, the difference in brightness according to the presence or absence of each fluorescent dye, and the relative difference divided by the small brightness. For all fluorescent dyes, regardless of the presence or absence of other dyes, the relative gaps were 9.5 or more, and the relative gain difference between the two experiments was within ±2. These results proved that fluorescence can be stably detected without interference.
Figure 3 shows the result of amplification experiment by labeling Chlamydia trachomatis DNA with FAM dye.
Figure 3a shows the ROI images taken in each cycle, and
Figure 3b shows the average ROI brightness for each cycle (blue curve). The red curve is the fluorescence brightness measured each cycle by the existing photodiode-based system for reference. The cycle threshold, that is, the quantitative result of the real-time PCR system, differs only less than 1 cycle for both, showing that there is no difference in performance between the systems.
This paper presents the use of a miniaturized, low-cost, real-time PCR system using PCR chip, lateral illumination excitation, and open platform camera. The results of single dye, dye crosstalk, and actual DNA amplification experiments showed that the proposed system is sufficiently feasible as a POC multiplexed real-time PCR device.