Bone Tissue Engineering Scaffold Optimisation through Modification of Chitosan/Ceramic Composition
Abstract
1. Introduction
2. Materials and Methods
2.1. Synthesising Fluorapatite (FAp)
- An amount of 0.43 g of sodium fluoride (NaF) powder was added to 10 mL of 10 mol/L phosphoric acid and mixed using magnificent stirrer;
- Then, 5 g of hydroxyapatite was added to the solution and reacted for few minutes;
- The powder was mixed in mortar, producing FAp (Ca5(PO4)3F) through the following reaction.
2.2. Scaffold Preparation
2.3. Morphological SEM Study
2.4. Swelling Study
2.5. Characterisation of Scaffold (Fourier Transform Infrared)
2.6. Compression Test
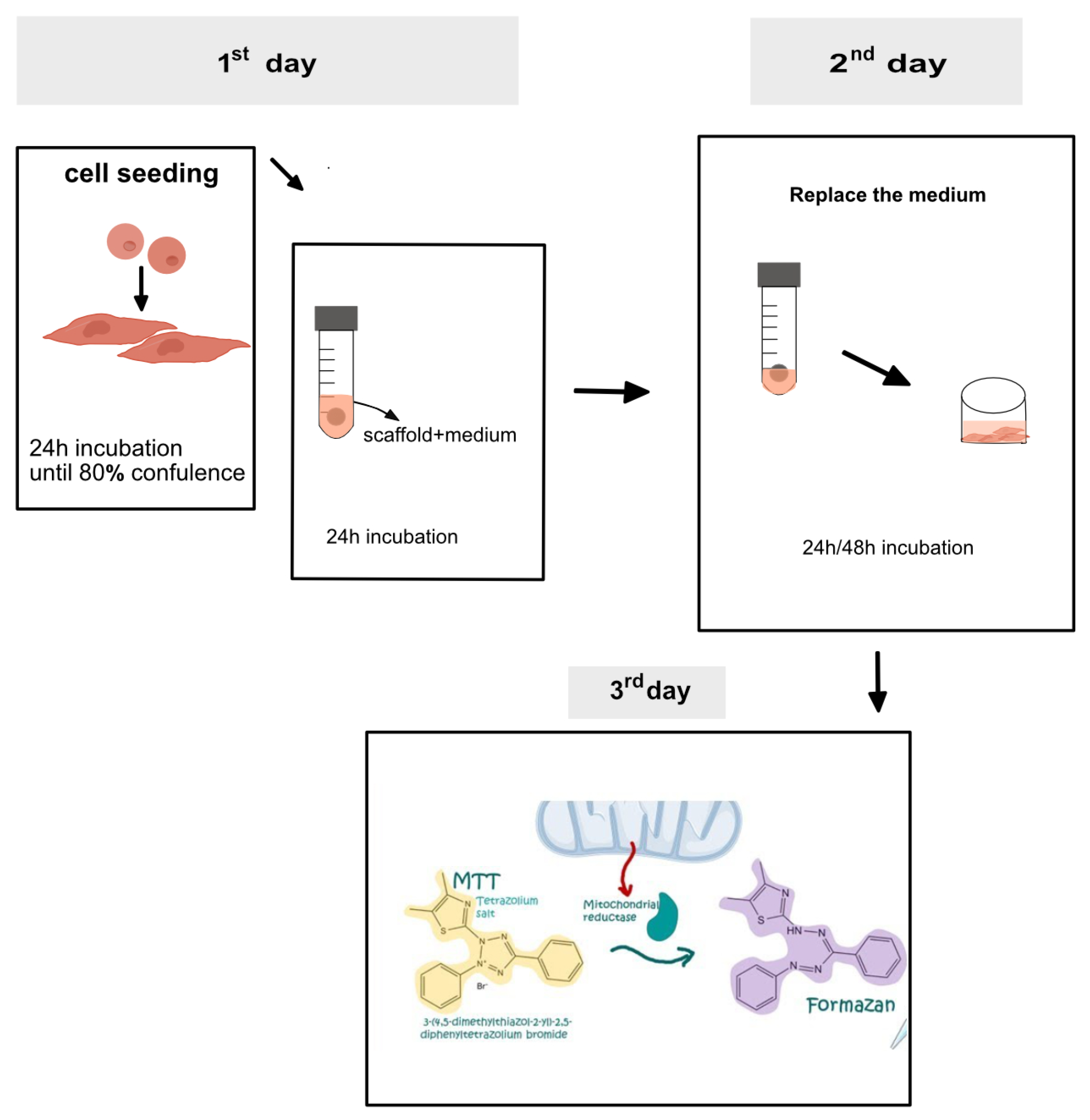
2.7. Cytotoxicity Evaluation
2.8. In Vitro Degradation Study
2.9. Statistical Analysis
3. Results and Discussion
3.1. SEM
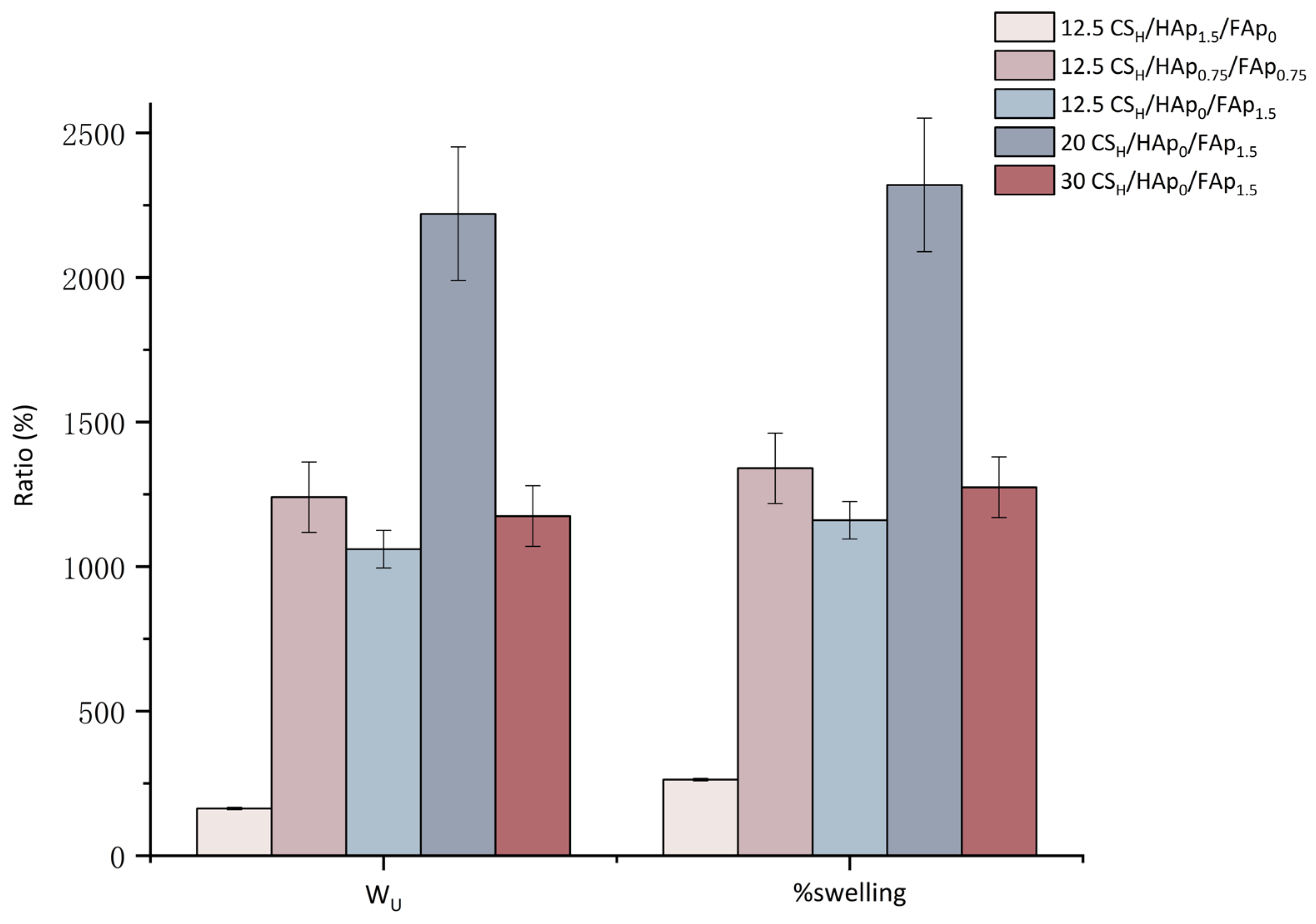
3.2. Swelling Study
3.3. FTIR
3.4. Compression Test
3.5. Cytotoxicity Evaluation
3.6. In Vitro Degradation Study
4. Conclusions
5. Future Work
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Pitacco, P.; Sadowska, J.M.; O’Brien, F.J.; Kelly, D.J. 3D Bioprinting of Cartilaginous Templates for Large Bone Defect Healing. Acta Biomater. 2023, 156, 61–74. [Google Scholar] [CrossRef]
- AO Research Institute Davos; Verrier, S.; Alini, M.; Alsberg, E.; Buchman, S.; Kelly, K.; Laschke, M.; Menger, M.; Murphy, W.; Stegemann, J.; et al. Tissue Engineering and Regenerative Approaches to Improving the Healing of Large Bone Defects. Eur. Cells Mater. 2016, 32, 87–110. [Google Scholar] [CrossRef]
- Dimitriou, R.; Mataliotakis, G.I.; Calori, G.M.; Giannoudis, P.V. The Role of Barrier Membranes for Guided Bone Regeneration and Restoration of Large Bone Defects: Current Experimental and Clinical Evidence. BMC Med. 2012, 10, 81. [Google Scholar] [CrossRef]
- Zhao, R.; Yang, R.; Cooper, P.R.; Khurshid, Z.; Shavandi, A.; Ratnayake, J. Bone Grafts and Substitutes in Dentistry: A Review of Current Trends and Developments. Molecules 2021, 26, 3007. [Google Scholar] [CrossRef]
- Mac, C.-H.; Chan, H.-Y.; Lin, Y.-H.; Sharma, A.K.; Song, H.-L.; Chan, Y.-S.; Lin, K.-J.; Lin, Y.-J.; Sung, H.-W. Engineering a Biomimetic Bone Scaffold That Can Regulate Redox Homeostasis and Promote Osteogenesis to Repair Large Bone Defects. Biomaterials 2022, 286, 121574. [Google Scholar] [CrossRef]
- Falacho, R.I.; Palma, P.J.; Marques, J.A.; Figueiredo, M.H.; Caramelo, F.; Dias, I.; Viegas, C.; Guerra, F. Collagenated Porcine Heterologous Bone Grafts: Histomorphometric Evaluation of Bone Formation Using Different Physical Forms in a Rabbit Cancellous Bone Model. Molecules 2021, 26, 1339. [Google Scholar] [CrossRef]
- Sharmin, F.; O’Sullivan, M.; Malinowski, S.; Lieberman, J.R.; Khan, Y. Large Scale Segmental Bone Defect Healing through the Combined Delivery of VEGF and BMP-2 from Biofunctionalized Cortical Allografts. J. Biomed. Mater. Res. B Appl. Biomater. 2019, 107, 1002–1010. [Google Scholar] [CrossRef] [PubMed]
- Kattimani, V.S.; Kondaka, S.; Lingamaneni, K.P. Hydroxyapatite—Past, Present, and Future in Bone Regeneration. Bone Tissue Regen. Insights 2016, 7, BTRI-S36138. [Google Scholar] [CrossRef]
- Oryan, A.; Alidadi, S.; Moshiri, A.; Maffulli, N. Bone Regenerative Medicine: Classic Options, Novel Strategies, and Future Directions. J. Orthop. Surg. 2014, 9, 18. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez, A.; Nowzari, H. The Long-Term Risks and Complications of Bovine-Derived Xenografts: A Case Series. J. Indian Soc. Periodontol. 2019, 23, 487–492. [Google Scholar] [CrossRef] [PubMed]
- Ji, C.; Bi, L.; Li, J.; Fan, J. Salvianolic Acid B-Loaded Chitosan/Hydroxyapatite Scaffolds Promotes the Repair of Segmental Bone Defect by Angiogenesis and Osteogenesis. Int. J. Nanomed. 2019, 14, 8271–8284. [Google Scholar] [CrossRef] [PubMed]
- Polo-Corrales, L.; Latorre-Esteves, M.; Ramirez-Vick, J.E. Scaffold Design for Bone Regeneration. J. Nanosci. Nanotechnol. 2014, 14, 15–56. [Google Scholar] [CrossRef]
- Gou, L.; Cheng, S.Q.; Ran, J.G.; Su, B.H. Characterization of Porous Calcium Phosphate Bioceramics Modified by Porosifier. Key Eng. Mater. 2007, 336–338, 1618–1621. [Google Scholar] [CrossRef]
- Zhu, T.; Cui, Y.; Zhang, M.; Zhao, D.; Liu, G.; Ding, J. Engineered Three-Dimensional Scaffolds for Enhanced Bone Regeneration in Osteonecrosis. Bioact. Mater. 2020, 5, 584–601. [Google Scholar] [CrossRef] [PubMed]
- Omar, A.M.; Hassan, M.H.; Daskalakis, E.; Ates, G.; Bright, C.J.; Xu, Z.; Powell, E.J.; Mirihanage, W.; Bartolo, P.J.D.S. Geometry-Based Computational Fluid Dynamic Model for Predicting the Biological Behavior of Bone Tissue Engineering Scaffolds. J. Funct. Biomater. 2022, 13, 104. [Google Scholar] [CrossRef]
- Dall’Oca, C.; Maluta, T.; Micheloni, G.M.; Cengarle, M.; Morbioli, G.; Bernardi, P.; Sbarbati, A.; Degl’Innocenti, D.; Lavini, F.; Magnan, B. The Biocompatibility of Bone Cements: Progress in Methodological Approach. Eur. J. Histochem. 2017, 61, 2673. [Google Scholar] [CrossRef]
- Yeng, C.M.; Salmah, H.; Sam, S.T. Corn Cob Filled Chitosan Biocomposite Films. Adv. Mater. Res. 2013, 747, 649–652. [Google Scholar] [CrossRef]
- Satitsri, S. Chitin and Chitosan Derivatives as Biomaterial Resources for Biological and Biomedical Applications. Mol. Pharm. 2020, 25, 5961. [Google Scholar] [CrossRef]
- Nady, N.; Kandil, S.H. Novel Blend for Producing Porous Chitosan-Based Films Suitable for Biomedical Applications. Membranes 2018, 8, 2. [Google Scholar] [CrossRef]
- Chen, Y.; Liu, X.; Liu, R.; Gong, Y.; Wang, M.; Huang, Q.; Feng, Q.; Yu, B. Zero-Order Controlled Release of BMP2-Derived Peptide P24 from the Chitosan Scaffold by Chemical Grafting Modification Technique for Promotion of Osteogenesis in Vitro and Enhancement of Bone Repair in Vivo. Theranostics 2017, 7, 1072–1087. [Google Scholar] [CrossRef]
- Borkowski, L.; Przekora, A.; Belcarz, A.; Palka, K.; Jozefaciuk, G.; Lübek, T.; Jojczuk, M.; Nogalski, A.; Ginalska, G. Fluorapatite Ceramics for Bone Tissue Regeneration: Synthesis, Characterization and Assessment of Biomedical Potential. Mater. Sci. Eng. C 2020, 116, 111211. [Google Scholar] [CrossRef]
- Lin, C.-C.; Lin, S.-C.; Chiang, C.-C.; Chang, M.-C.; Lee, O.K.-S. Reconstruction of Bone Defect Combined with Massive Loss of Periosteum Using Injectable Human Mesenchymal Stem Cells in Biocompatible Ceramic Scaffolds in a Porcine Animal Model. Stem Cells Int. 2019, 2019, 6832952. [Google Scholar] [CrossRef] [PubMed]
- Wei, S.; Ma, J.; Xu, L.; Gu, X.; Ma, X. Biodegradable Materials for Bone Defect Repair. Mil. Med. Res. 2020, 7, 54. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Chen, G.; Huang, C.; Tu, H.; Zou, J. Bone Marrow Stem Cells-Derived Extracellular Matrix Is a Promising Material. Oncotarget 2017, 8, 98336–98347. [Google Scholar] [CrossRef]
- Fernandez de Grado, G.; Keller, L.; Idoux-Gillet, Y.; Wagner, Q.; Musset, A.M.; Benkirane-Jessel, N.; Bornert, F.; Offner, D. Bone Substitutes: A Review of Their Characteristics, Clinical Use, and Perspectives for Large Bone Defects Management. J. Tissue Eng. 2018, 9, 2041731418776819. [Google Scholar] [CrossRef] [PubMed]
- Abdul Halim, N.A.; Hussein, M.Z.; Kandar, M.K. Nanomaterials-Upconverted Hydroxyapatite for Bone Tissue Engineering and a Platform for Drug Delivery. Int. J. Nanomed. 2021, 16, 6477–6496. [Google Scholar] [CrossRef] [PubMed]
- Bhadang, K.A.; Gross, K.A. Influence of Fluorapatite on the Properties of Thermally Sprayed Hydroxyapatite Coatings. Biomaterials 2004, 25, 4935–4945. [Google Scholar] [CrossRef]
- Kokubo, T.; Takadama, H. How Useful Is SBF in Predicting in Vivo Bone Bioactivity? Biomaterials 2006, 27, 2907–2915. [Google Scholar] [CrossRef]
- Killion, J.; Geever, L.; Devine, D.; Kennedy, J.; Higginbotham, C. Development of Synthetic Alternatives For Bone Tissue Engineering. Aust. J. Basic Appl. Sci. 2013, 7, 150–156. [Google Scholar]
- Zhang, J.; Nie, J.; Zhang, Q.; Li, Y.; Wang, Z.; Hu, Q. Preparation and Characterization of Bionic Bone Structure Chitosan/Hydroxyapatite Scaffold for Bone Tissue Engineering. J. Biomater. Sci. Polym. Ed. 2014, 25, 61–74. [Google Scholar] [CrossRef] [PubMed]
- Brun, V.; Guillaume, C.; Mechiche Alami, S.; Josse, J.; Jing, J.; Draux, F.; Bouthors, S.; Laurent-Maquin, D.; Gangloff, S.C.; Kerdjoudj, H.; et al. Chitosan/Hydroxyapatite Hybrid Scaffold for Bone Tissue Engineering. Biomed. Mater. Eng. 2014, 24, 63–73. [Google Scholar] [CrossRef]
- Jeong, J.; Kim, J.H.; Shim, J.H.; Hwang, N.S.; Heo, C.Y. Bioactive Calcium Phosphate Materials and Applications in Bone Regeneration. Biomater. Res. 2019, 23, 4. [Google Scholar] [CrossRef]
- Nordquist, W.D.; Okudera, H.; Kitamura, Y.; Kimoto, K.; Okudera, T.; Krutchkoff, D.J. Part II: Crystalline Fluorapatite-Coated Hydroxyapatite Implant Material: A Dog Study With Histologic Comparison of Osteogenesis Seen With FA-Coated HA Grafting Material Versus HA Controls: Potential Bacteriostatic Effect of Fluoridated HA. J. Oral Implantol. 2011, 37, 35–42. [Google Scholar] [CrossRef] [PubMed]
- Jia, F.; Zhou, L.; Li, S.; Lin, X.; Wen, B.; Lai, C.; Ding, X. Phosphoric Acid and Sodium Fluoride: A Novel Etching Combination on Titanium. Biomed. Mater. 2014, 9, 035004. [Google Scholar] [CrossRef] [PubMed]
- Zaichick, V.; Tzaphlidou, M. Determination of Calcium, Phosphorus, and the Calcium/Phosphorus Ratio in Cortical Bone from the Human Femoral Neck by Neutron Activation Analysis. Appl. Radiat. Isot. 2002, 56, 781–786. [Google Scholar] [CrossRef]
- Azaman, F.A.; Zhou, K.; Blanes-Martínez, M.D.M.; Fournet, M.B.; Devine, D.M. Bioresorbable Chitosan-Based Bone Regeneration Scaffold Using Various Bioceramics and the Alteration of Photoinitiator Concentration in an Extended UV Photocrosslinking Reaction. Gels 2022, 8, 696. [Google Scholar] [CrossRef]
- Nga, N.K.; Thanh Tam, L.T.; Ha, N.T.; Hung Viet, P.; Huy, T.Q. Enhanced Biomineralization and Protein Adsorption Capacity of 3D Chitosan/Hydroxyapatite Biomimetic Scaffolds Applied for Bone-Tissue Engineering. RSC Adv. 2020, 10, 43045–43057. [Google Scholar] [CrossRef]
- Killion, J.A.; Geever, L.M.; Devine, D.M.; Higginbotham, C.L. Fabrication and in Vitro Biological Evaluation of Photopolymerisable Hydroxyapatite Hydrogel Composites for Bone Regeneration. J. Biomater. Appl. 2014, 28, 1274–1283. [Google Scholar] [CrossRef] [PubMed]
- Hong, M.-H.; Lee, J.H.; Jung, H.S.; Shin, H.; Shin, H. Biomineralization of Bone Tissue: Calcium Phosphate-Based Inorganics in Collagen Fibrillar Organic Matrices. Biomater. Res. 2022, 26, 42. [Google Scholar] [CrossRef]
- Wu, X.; Walsh, K.; Hoff, B.L.; Camci-Unal, G. Mineralization of Biomaterials for Bone Tissue Engineering. Bioengineering 2020, 7, 132. [Google Scholar] [CrossRef]
- Liu, D.; Liu, Z.; Zou, J.; Li, L.; Sui, X.; Wang, B.; Yang, N.; Wang, B. Synthesis and Characterization of a Hydroxyapatite-Sodium Alginate-Chitosan Scaffold for Bone Regeneration. Front. Mater. 2021, 8, 648980. [Google Scholar] [CrossRef]
- Borkowski, L.; Przekora, A.; Belcarz, A.; Palka, K.; Jojczuk, M.; Lukasiewicz, P.; Nogalski, A.; Ginalska, G. Highly Porous Fluorapatite/Β-1,3-glucan Composite for Bone Tissue Regeneration: Characterization and in Vitro Assessment of Biomedical Potential. Int. J. Mol. Sci. 2021, 22, 10414. [Google Scholar] [CrossRef]
- Xue, X.; Hu, Y.; Wang, S.; Chen, X.; Jiang, Y.; Su, J. Fabrication of Physical and Chemical Crosslinked Hydrogels for Bone Tissue Engineering. Bioact. Mater. 2022, 12, 327–339. [Google Scholar] [CrossRef]
- Surowiec, R.K.; Allen, M.R.; Wallace, J.M. Bone Hydration: How We Can Evaluate It, What Can It Tell Us, and Is It an Effective Therapeutic Target? Bone Rep. 2022, 16, 101161. [Google Scholar] [CrossRef] [PubMed]
- Cui, W.; Song, X.; Chen, J.; Chen, Y.; Li, Y.; Zhao, C. Adsorption Behaviors of Different Water Structures on the Fluorapatite (001) Surface: A DFT Study. Front. Mater. 2020, 7, 47. [Google Scholar] [CrossRef]
- Ali, H.U.; Iqbal, D.N.; Iqbal, M.; Ezzine, S.; Arshad, A.; Zeeshan, R.; Chaudhry, A.A.; Alshawwa, S.Z.; Nazir, A.; Khan, A.F. HPMC Crosslinked Chitosan/Hydroxyapatite Scaffolds Containing Lemongrass Oil for Potential Bone Tissue Engineering Applications. Arab. J. Chem. 2022, 15, 103850. [Google Scholar] [CrossRef]
- Kazimierczak, P.; Wessely-Szponder, J.; Palka, K.; Barylyak, A.; Zinchenko, V.; Przekora, A. Hydroxyapatite or Fluorapatite—Which Bioceramic Is Better as a Base for the Production of Bone Scaffold?—A Comprehensive Comparative Study. Int. J. Mol. Sci. 2023, 24, 5576. [Google Scholar] [CrossRef]
- Shi, C.; Hou, X.; Zhao, D.; Wang, H.; Guo, R.; Zhou, Y. Preparation of the Bioglass/Chitosan-Alginate Composite Scaffolds with High Bioactivity and Mechanical Properties as Bone Graft Materials. J. Mech. Behav. Biomed. Mater. 2022, 126, 105062. [Google Scholar] [CrossRef]
- Ashok, M.; Meenakshi Sundaram, N.; Narayana Kalkura, S. Crystallization of Hydroxyapatite at Physiological Temperature. Mater. Lett. 2003, 57, 2066–2070. [Google Scholar] [CrossRef]
- Llanos, J.H.R.; Vercik, L.C.D.O.; Vercik, A. Physical Properties of Chitosan Films Obtained after Neutralization of Polycation by Slow Drip Method. J. Biomater. Nanobiotechnol. 2015, 6, 276–291. [Google Scholar] [CrossRef]
- Liuyun, J.; Yubao, L.; Chengdong, X. Preparation and Biological Properties of a Novel Composite Scaffold of Nano-Hydroxyapatite/Chitosan/Carboxymethyl Cellulose for Bone Tissue Engineering. J. Biomed. Sci. 2009, 16, 65. [Google Scholar] [CrossRef]
- Devine, D.M.; Higginbotham, C.L. The Synthesis of a Physically Crosslinked NVP Based Hydrogel. Polymer 2003, 44, 7851–7860. [Google Scholar] [CrossRef]
- Taktak, R.; Elghazel, A.; Bouaziz, J.; Charfi, S.; Keskes, H. Tricalcium Phosphate-Fluorapatite as Bone Tissue Engineering: Evaluation of Bioactivity and Biocompatibility. Mater. Sci. Eng. C 2018, 86, 121–128. [Google Scholar] [CrossRef] [PubMed]
- Venkatesan, J.; Pallela, R.; Bhatnagar, I.; Kim, S.K. Chitosan-Amylopectin/Hydroxyapatite and Chitosan-Chondroitin Sulphate/Hydroxyapatite Composite Scaffolds for Bone Tissue Engineering. Int. J. Biol. Macromol. 2012, 51, 1033–1042. [Google Scholar] [CrossRef] [PubMed]
- Ehterami, A.; Kazemi, M.; Nazari, B.; Saraeian, P.; Azami, M. Fabrication and Characterization of Highly Porous Barium Titanate Based Scaffold Coated by Gel/HA Nanocomposite with High Piezoelectric Coefficient for Bone Tissue Engineering Applications. J. Mech. Behav. Biomed. Mater. 2018, 79, 195–202. [Google Scholar] [CrossRef]
- Woodard, J.R.; Hilldore, A.J.; Lan, S.K.; Park, C.J.; Morgan, A.W.; Eurell, J.A.C.; Clark, S.G.; Wheeler, M.B.; Jamison, R.D.; Wagoner Johnson, A.J. The Mechanical Properties and Osteoconductivity of Hydroxyapatite Bone Scaffolds with Multi-Scale Porosity. Biomaterials 2007, 28, 45–54. [Google Scholar] [CrossRef] [PubMed]
- Devine, D.M.; Hoctor, E.; Hayes, J.S.; Sheehan, E.; Evans, C.H. Extended Release of Proteins Following Encapsulation in Hydroxyapatite/Chitosan Composite Scaffolds for Bone Tissue Engineering Applications. Mater. Sci. Eng. C 2018, 84, 281–289. [Google Scholar] [CrossRef]
- Elghazel, A.; Taktak, R.; Bouaziz, J.; Charfi, S.; Keskes, H. TCP-Fluorapatite Composite Scaffolds: Mechanical Characterization and In Vitro/In Vivo Testing. In Scaffolds in Tissue Engineering—Materials, Technologies and Clinical Applications; Baino, F., Ed.; InTech: London, UK, 2017. [Google Scholar] [CrossRef]
- Killion, J.A.; Geever, L.M.; Devine, D.M.; Farrell, H.; Higginbotham, C.L. Compressive Strength and Bioactivity Properties of Photopolymerizable Hybrid Composite Hydrogels for Bone Tissue Engineering. Int. J. Polym. Mater. Polym. Biomater. 2013, 63, 641–650. [Google Scholar] [CrossRef]
- Roohani-Esfahani, S.I.; Newman, P.; Zreiqat, H. Design and Fabrication of 3D Printed Scaffolds with a Mechanical Strength Comparable to Cortical Bone to Repair Large Bone Defects. Sci. Rep. 2016, 6, srep19468. [Google Scholar] [CrossRef]
- Johnston, N.R.; Strobel, S.A. Principles of Fluoride Toxicity and the Cellular Response: A Review. Arch. Toxicol. 2020, 94, 1051–1069. [Google Scholar] [CrossRef]
- Wang, M.O.; Etheridge, J.M.; Thompson, J.A.; Vorwald, C.E.; Dean, D.; Fisher, J.P. Evaluation of the In Vitro Cytotoxicity of Cross-Linked Biomaterials. Biomacromolecules 2013, 14, 1321–1329. [Google Scholar] [CrossRef] [PubMed]
- Ghiasi, M.S.; Chen, J.E.; Rodriguez, E.K.; Vaziri, A.; Nazarian, A. Computational Modeling of Human Bone Fracture Healing Affected by Different Conditions of Initial Healing Stage. BMC Musculoskelet. Disord. 2019, 20, 562. [Google Scholar] [CrossRef] [PubMed]
- Seyedmajidi, S.; Seyedmajidi, M. Fluorapatite: A Review of Synthesis, Properties and Medical Applications vs Hydroxyapatite. Iran. J. Mater. Sci. Eng. 2022, 19, 1–20. [Google Scholar] [CrossRef]







| Scaffold ID | HAp/(%wt) | FAp/(%wt) | Volume of AA/(mL) |
|---|---|---|---|
| 12.5 CSH/HAp1.5/FAp0 | 1.5 | 0 | 12.5 |
| 12.5 CSH/HAp0.7.5/FAp0.75 | 0 | 1.5 | 12.5 |
| 12.5 CSH/HAp0/FAp1.5 | 0.75 | 0.75 | 12.5 |
| 20 CSH/HAp1.5/FAp0 | 0 | 1.5 | 20 |
| 20 CSH/HAp0.7.5/FAp0.75 | 0.75 | 0.75 | 20 |
| 20 CSH/HAp0/FAp1.5 | 0 | 1.5 | 30 |
| 30 CSH/HAp1.5/FAp0 | 1.5 | 0 | 30 |
| 30 CSH/HAp0.7.5/FAp0.75 | 0.75 | 0.75 | 30 |
| 30 CSH/HAp0/FAp1.5 | 0 | 1.5 | 30 |
| Atomic Percent (%) | 12.5 CSH/HAp1.5/FAp0 | 12.5 CSH/HAp0.75/FAp0.75 | 12.5 CSH/HAp0/FAp1.5 | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Ca | P | Ca/P ratio | Ca | P | Ca/P Ratio | Ca | P | Ca/P Ratio | |
| W0 | 9.821 | 3.39 | 2.89 | 9.9 | 4.99 | 1.98 | 5.82 | 6.18 | 0.94 |
| W2 | 24.56 | 10.29 | 2.38 | 15.79 | 7.49 | 2.1 | 8.82 | 3.54 | 2.94 |
| W8 | 29.53 | 13.25 | 2.22 | 30.88 | 13.15 | 2.34 | 9.87 | 3.6 | 2.7 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhou, K.; Azaman, F.A.; Cao, Z.; Brennan Fournet, M.; Devine, D.M. Bone Tissue Engineering Scaffold Optimisation through Modification of Chitosan/Ceramic Composition. Macromol 2023, 3, 326-342. https://doi.org/10.3390/macromol3020021
Zhou K, Azaman FA, Cao Z, Brennan Fournet M, Devine DM. Bone Tissue Engineering Scaffold Optimisation through Modification of Chitosan/Ceramic Composition. Macromol. 2023; 3(2):326-342. https://doi.org/10.3390/macromol3020021
Chicago/Turabian StyleZhou, Keran, Farah Alwani Azaman, Zhi Cao, Margaret Brennan Fournet, and Declan M. Devine. 2023. "Bone Tissue Engineering Scaffold Optimisation through Modification of Chitosan/Ceramic Composition" Macromol 3, no. 2: 326-342. https://doi.org/10.3390/macromol3020021
APA StyleZhou, K., Azaman, F. A., Cao, Z., Brennan Fournet, M., & Devine, D. M. (2023). Bone Tissue Engineering Scaffold Optimisation through Modification of Chitosan/Ceramic Composition. Macromol, 3(2), 326-342. https://doi.org/10.3390/macromol3020021







