1. Introduction
Taeniid tapeworms represent an important group of parasites that threaten the health of both livestock and humans. The impact of taeniids in both veterinary and public health is primarily confined to the metacestode stage of the parasite in the intermediate host [
1]. One important taeniid species is
T. multiceps, a cestode of dogs and wild canids, which is the cause of a fatal disease called cerebral coenurosis in small ruminants [
2,
3,
4,
5]. The parasite has zoonotic potential, with occasional cases of coenurosis in humans reported in different parts of the world including Italy, Israel, North America, Uganda, and Egypt [
6,
7,
8]. Human teniid infection, such as cerebral coenurosis (
T. multiceps), occurs with the ingestion of parasite eggs from contaminated environment, food, or water [
1]. However, human infections are not involved in the complete life cycle of these parasites making human a dead-end host. Small ruminants become infected with
T. multiceps following ingestion of eggs from contaminated pastures and water. Following ingestion (in both human and livestock hosts), the hatched oncosphere penetrates the intestinal wall and enters the blood stream and finally makes its way to the brain and spinal cord [
3,
9,
10]. Metacestode larvae have a high affinity to cerebrospinal fluid (CSF), which stimulates differentiation and development into cysts [
11].
Transmission to dogs and other canids, the definitive hosts, occurs when metacestodes in the brain or spinal cord of coenurosis-affected goats or sheep are eaten and develop into mature
T. multiceps worms in the intestines [
2]. The adult worms shed proglottids into the host’s faeces with each mature proglottid containing up to 37,000 eggs, allowing canid hosts to amplify infection within an ecosystem [
3,
12,
13]. In dogs,
T. multiceps infection is typically subclinical with few health impacts; however, under heavy infestation dogs can demonstrate non-specific gastrointestinal syndrome such as abdominal pain, diarrhea, and constipation [
14]. Although there is limited clinical impact in dogs, treatment of
T. multiceps and related cestodes (such as
Echinococcus granulosus) in dogs is recommended to safeguard ruminant hosts [
14,
15]. Deworming treatment of dogs using praziquantel and other preventive strategies such as avoidance of feeding raw offal from animal carcasses to dogs, can be highly effective in the control of these taeniids [
5,
16].
The parasite is distributed worldwide and is well-recognised as a cause of small ruminant disease [
2]. However, there is evidence to suggest that coenurosis due to
T. multiceps has become a growing threat to the health of small ruminants in eastern and other parts of Africa [
17,
18,
19,
20,
21,
22,
23,
24]. However, despite the availability of preventive measures, very little attention is being given to address the disease. In Tanzania, cerebral coenurosis appears to be emerging as a major health problem in small ruminants in pastoral communities [
20,
24,
25,
26]. In a recent small-scale study in northern Tanzania [
24], over 80% of cases of neurological syndrome in sheep and goats were caused by the
T. multiceps metacestode.
Coenurus cerebralis and coenurosis was identified as the cause of very high mortality in small ruminants [
24]. In another study, farmers ranked neurological syndromes, locally known as
Ormilo, as the most important health problem in sheep and goats in pastoral communities of northern Tanzania [
26]. Although canids are known to be the definitive host of
T. multiceps, little is known about the prevalence and determinants of taeniid infection in dogs in these areas. The aim of this study was to: (a) determine the prevalence of coenurosis in small ruminants and taeniid infection in dogs across a range of agro-ecological systems in northern Tanzania: (b) identify risk factors for coenurosis in small ruminants and taeniid infection in dogs.
3. Discussion
The current study assessed the prevalence of owner-reported neurological syndrome, known locally as
Ormilo, and associated risk factors in small ruminants in agro-pastoral and pastoral livestock systems in northern Tanzania. Furthermore, the study investigated the determinants of taeniid prevalence in dogs and the farmers’ knowledge and practices in relation to coenurosis prevention established. The study confirmed coenurosis in a high proportion (69%) of animals slaughtered at home due to suspected
Ormilo, somewhat lower than previous studies (82%) [
24] but consistent with the interpretation that a large proportion of neurological cases in small ruminants in these communities is caused by
T. multiceps. This interpretation is supported by the risk factor analysis showing that households practicing home (backyard) slaughter of small ruminants were thirteen times more likely to report cases of neurological disease in sheep and goats than those that did not. Since
T. multiceps is known to be transmitted by consumption of cysts by definitive hosts (canids), our conclusion that the neurological condition,
Ormilo, is caused primarily by
T. multiceps is also strengthened by the observation of a higher prevalence of the neurological condition in pastoralist villages where risk behaviours such as feeding brains of small ruminants to dogs as described by Hughes et al., 2018 are reported more often.
The study adds to earlier studies [
20,
24,
25,
26], confirming that cerebral coenurosis is a major disease problem in pastoral farming systems in Tanzania, but additionally demonstrates a high disease prevalence (7.3%) in agro-pastoral flocks. The impact on productivity, food security and rural livelihoods in Tanzania is likely to be profound, given that these sectors comprise a very large proportion (35%) of livestock-owning households in the country, and provide more than 50% of milk and 90% of meat consumed in Tanzania [
27].
Backyard slaughter practices can be linked to poor management of offal including brain material from infected small ruminants, which are often either fed to dogs or thrown away. Community social events and celebrations such as circumcision festivals and traditional wedding ceremonies, traditional medication retreat for Maasai (orpul) often involve massive, unsupervised backyard slaughter. Similarly, backyard slaughter has been reported commonly during festivals in Egypt [
28]. Unsupervised backyard slaughter might be due to a shortage of human capacity to carry out extension services and meat inspection and is likely to be common in most rural settings in Tanzania [
29]. Furthermore, Amer et al., 2017 stated that backyard slaughter was characterised by limited infrastructure for meat inspection leading to unsupervised disposal of brains rendering them accessible to dogs. Factors associated with backyard slaughter are likely be an important reason for the high prevalence of cerebral coenurosis in goats and sheep reported in many African countries [
18,
20,
24,
30].
Using faecal egg counts, we demonstrated a high prevalence of taeniid eggs in dog faeces in the study area. Morphologically, it is not possible to differentiate these eggs to species level using microscopic examination, so these results are not a definitive diagnosis of
T. multiceps infection in these hosts. Despite this limitation, these results provide new insights into the epidemiology of dog-associated parasite infections in Tanzania. There is little information available on taeniid infections in local dogs, with previous work limited to a single study in Ngorongoro Conservation Area [
31]. Our results demonstrate that taeniid infections are widespread in our study area and consistent with questionnaire results reporting a lack of worming practices in local households. Although the current study did not type taeniid eggs to the species level, the high prevalence of coenurosis provides circumstantial evidence that
T. multiceps is likely to be present, potentially alongside other taeniids of zoonotic or animal health importance such as
Taenia hydatigena, Taenia ovis,
Echinococcus granulosus. Similarly, this study revealed a higher prevalence of taeniid infection in pastoral as compared with agropastoral systems, consistent with the higher prevalence of the coenurosis-related neurological syndrome reported in small ruminants in these pastoral livestock keeping communities.
Despite a high reported prevalence of coenurosis in small ruminants, this study demonstrated a lack of awareness among livestock keepers around the value of deworming dogs. For those that reportedly dewormed their dogs, only very few reported using an appropriate deworming drug for taeniids such as praziquantel. The common deworming agents used were those that treat round worm infections in ruminants (levamisole and albendazole), none of which were formulated to treat tapeworm infection in dogs. In Europe, deworming has been effective in controlling taeniids in dogs [
32]. However, this approach is likely to be challenging in Tanzania, although cost was not commonly given as a reason for not deworming dogs, there may be little awareness of the costs involved in sustaining a high-frequency (approximately every 2 months) dosing regimen needed to control taeniid infections. Opportunities may arise for embedding control of Taenia with other dog-related interventions for public health, such as rabies vaccinations as proposed in a case study on the control of echinococcus in Morocco [
33].
Despite uncertainties around the feasibility of de-worming dogs as a method of control, this study clearly highlights that feeding small ruminant brains to dogs poses a significant risk for taeniid infections. This provides an opportunity for potential low-cost behavioural interventions for disease control and prevention. These might include, for example, the cessation of feeding brains of slaughtered small ruminants to dogs and/or throwing the brains away in places where they may be scavenged by dogs from other households or by wild carnivores. Instead, brains should be burnt. Although these interventions are not costly, there may be constraints in relation to the lack of availability of other food for dogs, particularly at times when household food (which comprises a major part of dogs’ diets) is in short supply. Nonetheless, dissemination of information about the life cycle of T. multiceps and the value of behavioural changes for prevention of coenurosis should be considered a high priority for animal health professionals working in communities suffering from a high disease burden.
5. Data Management and Analysis
5.1. Data Analysis Plan
Data from both surveys were entered into Microsoft Excel® (Microsoft Corporation, Washington, WA, USA) before analysis using R statistical environment version 4.0.3 R Core Team (2021). Descriptive statistics on village data were summarised as: percentage of responses on risk determinants of coenurosis in small ruminants and various management options for dogs across livestock systems in rural northern Tanzania. A chi-square analysis was carried out to examine differences in household risk practices in each management option for dogs and small ruminants in different agro-ecological settings. In each chi-square analysis, a p-value of ≤0.05 was considered statistically significant. Determinants of risk factors for coenurosis in small ruminants and taeniid infection in dogs were identified through univariate and multivariate mixed logistic regressions.
5.2. Prevalence
In small ruminants, the farmer-reported twelve-month period prevalence for coenurosis was calculated as the number of cases of suspected coenurosis reported in the village flock over the last twelve months divided by the total number of small ruminants owned by the farmer over the past twelve months. In addition, postmortem examination surveys were conducted from identified slaughter points available within the village. Therefore, prevalence of coenurosis from postmortem surveys were determined as the total number of heads in which coenurus cysts were recovered divided by the total number of all heads inspected for coenurus cysts. However, home-slaughtered small ruminants inspected for coenurosis were not considered during estimation of the prevalence due to bias since were known to be sick and salvaged due to their health status.
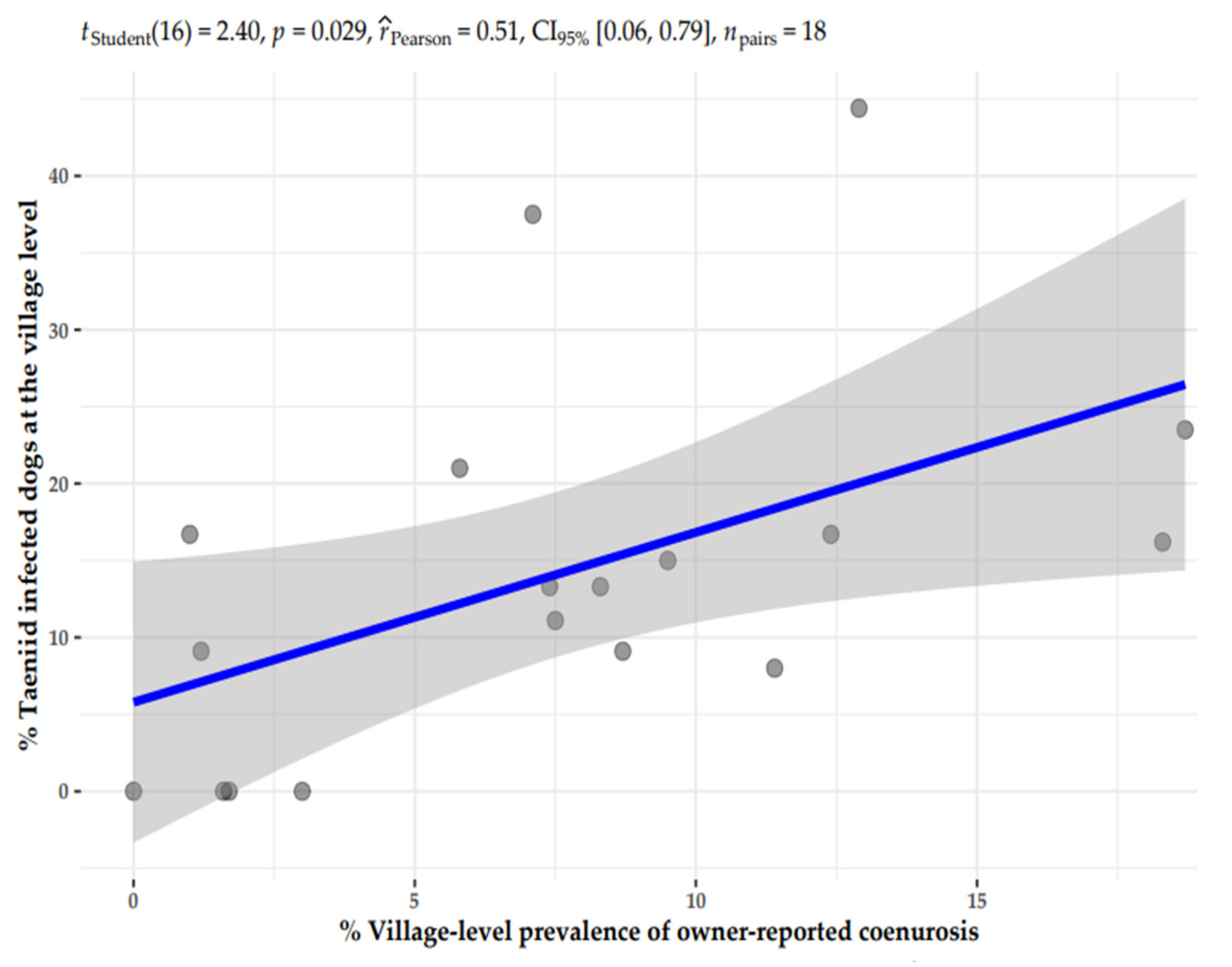
The prevalence of taeniid infections was estimated as the proportion of taeniid positive dogs out of the total number of dogs tested in each agro-ecological setting. Finally, the preliminary analysis performed a Pearson’s correlation analysis between taeniid infection in dogs and neurological syndrome prevalence at the village flock to establish a relationship between the two aspects involved in the T. multiceps cycles.
5.3. Logistic Regression Analysis for Risk Determinants for T. multiceps
A mixed effects logistic regression analysis was employed to identify and investigate potential determinants of coenurosis infections in small ruminants and taeniid infection in dogs.
- (a)
T. multiceps coenurosis infection in small ruminants
First, univariate regression analysis was performed for each predictor variable for coenurosis infection in small ruminants, The outcome variable was the “number of neurological cases” in the last twelve months and predictor variables were “backyard slaughter”, “livestock system”, “introduced animals”, “water source (dry season and wet season)”, “transhumance practices” and “communal grazing”. Second, variables that were significant at a level of p ≤ 0.25 in univariate analysis were included in the final multivariable model. The final model was arrived at by backward stepwise approach where non-significant predictors were iteratively removed while those with p < 0.05 retained and the Akaike information criterion (AIC) was used parsimoniously to select the best model. In multivariable analysis, mixed effect logistic regression was employed with households and villages included as random effects. Additionally, interactions and confounding effects between independent variables in the multivariable analysis were tested by adjusting for significant variables included in the model using Likelihood Ratio Tests (LRTs).
- (b)
Taeniid infection in dogs
To establish the key predictors for prevalence of taeniid infections in dogs, a univariate regression analysis of each predictor variable for taeniid infection in dogs was performed first. The outcome variable was taeniid positivity in a dog (yes/no) determined as either presence of eggs or recovery of segments of mature taeniid in the coproscopic examination. The predictor variables were: age, sex (male or female), livestock system (pastoral or agro-pastoral), and behavioural characteristics, including whether the dog was used for herding (yes or no), moved with the herders during grazing, and had been dewormed in the past twelve months (yes or no), and feeding practices, including whether brains were fed to dogs (yes or no), or the type of feeding practices (offal, porridge, leftovers, scavenging). The age as the only continuous variable for dogs, for the purpose of analysis the age groups (adult and juveniles) were categorized further by grouping as: mature juveniles (0.5 ≥ years < 1), early adulthood (1 ≥years < 3), middle aged (3 ≥ years < 6), later adulthood (6 ≥ years < 9), and seniors (9 ≥ years) as described by Wallis et al. [
40]. Second, variables that were significant at a level of
p ≤ 0.25 in univariate analysis were included in the full multivariable models for taeniid infection in dogs. The final models were built by backwards elimination, dropping one variable at time from the full model based on (AIC). Likelihood ratio tests (LRTs) were used to check the significance of each variable in the final model. A
p-value ≤ 0.05 of the LRT was considered significant and the variables were retained in the model. Interaction and confounding effects were tested using LRT and the significance of
p-values on the presence or absence of the variable with respect to others.