Anticancer Activity of Cerium Oxide Nanoparticles Towards Human Lung Cancer Cells
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Experimental Section
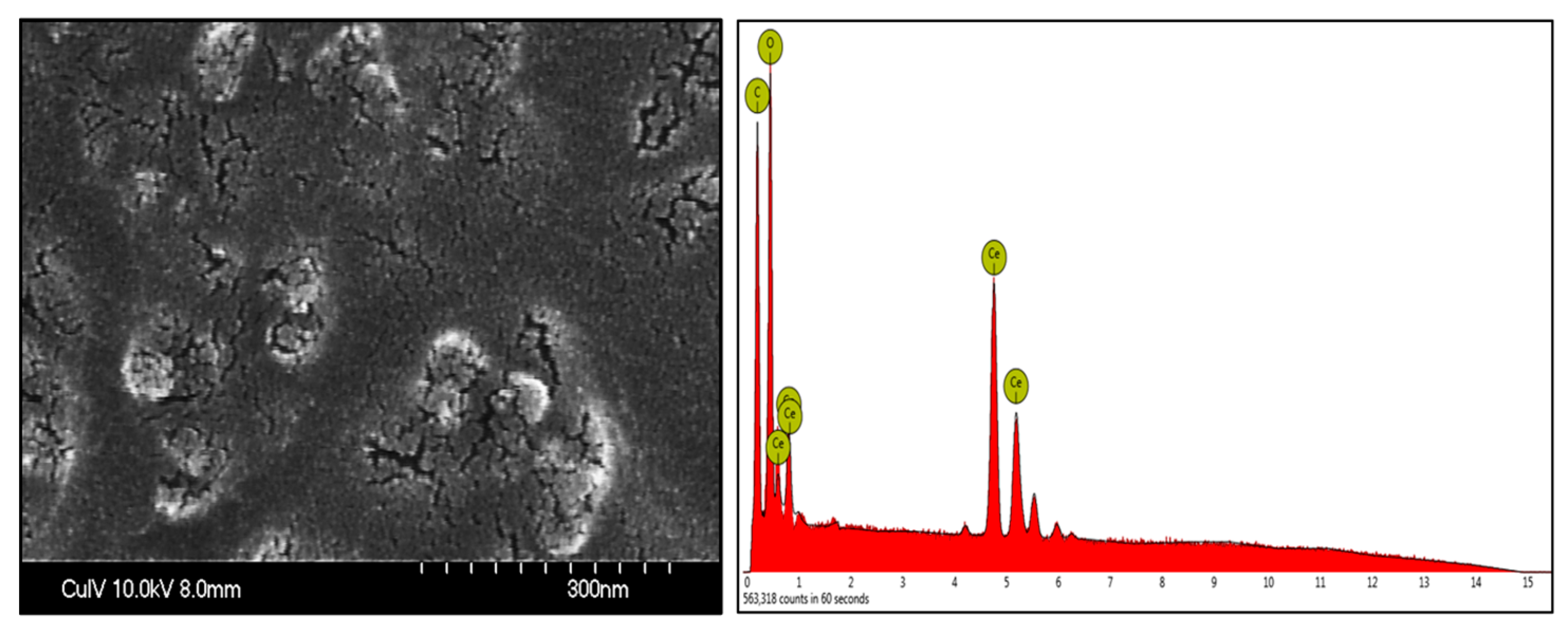
2.2.1. Field Emission Scanning Electron Microscopy (FESEM)
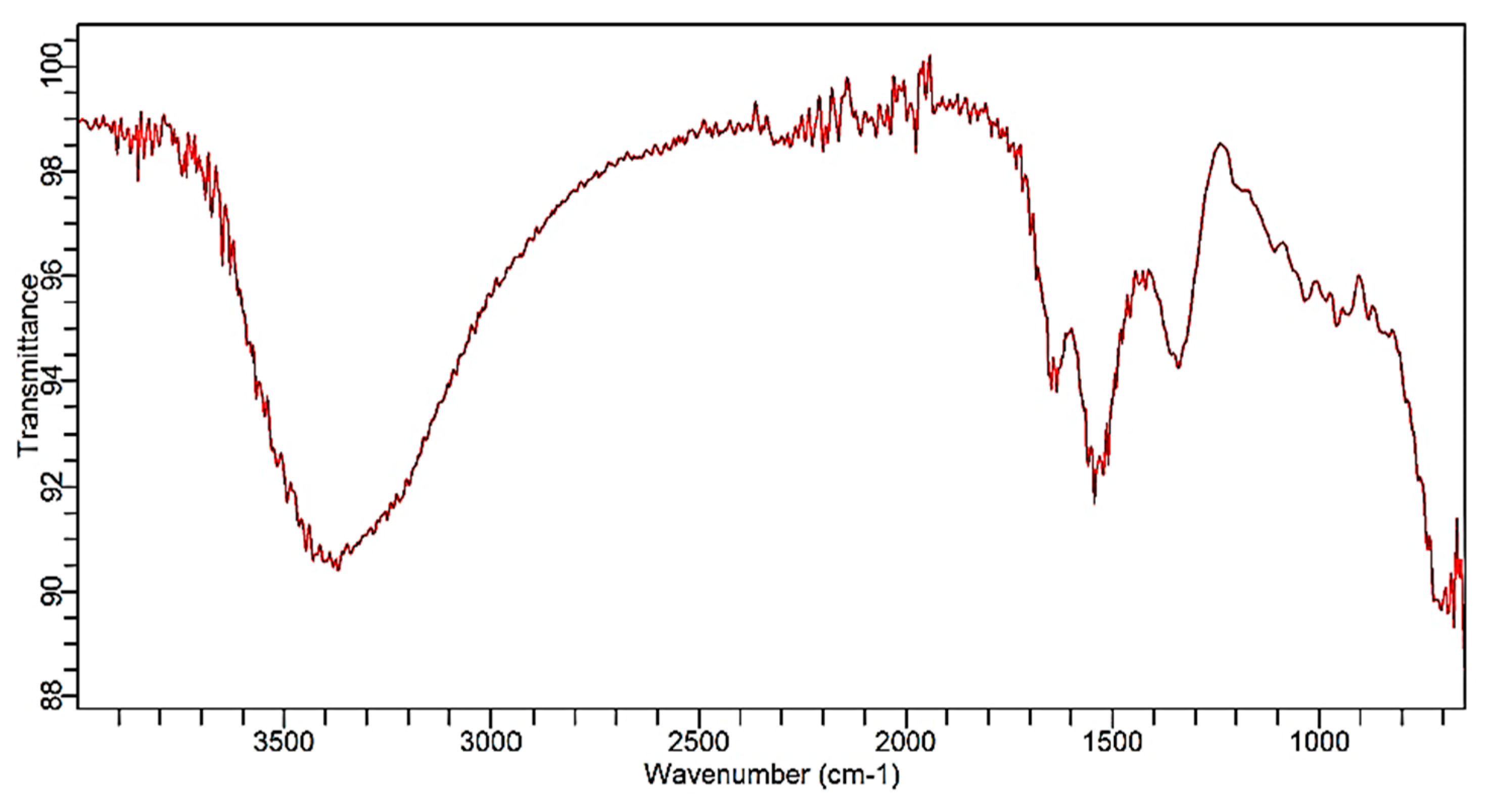
2.2.2. Fourier Transform Infrared Spectroscopy (FTIR)
2.2.3. Cell Culture
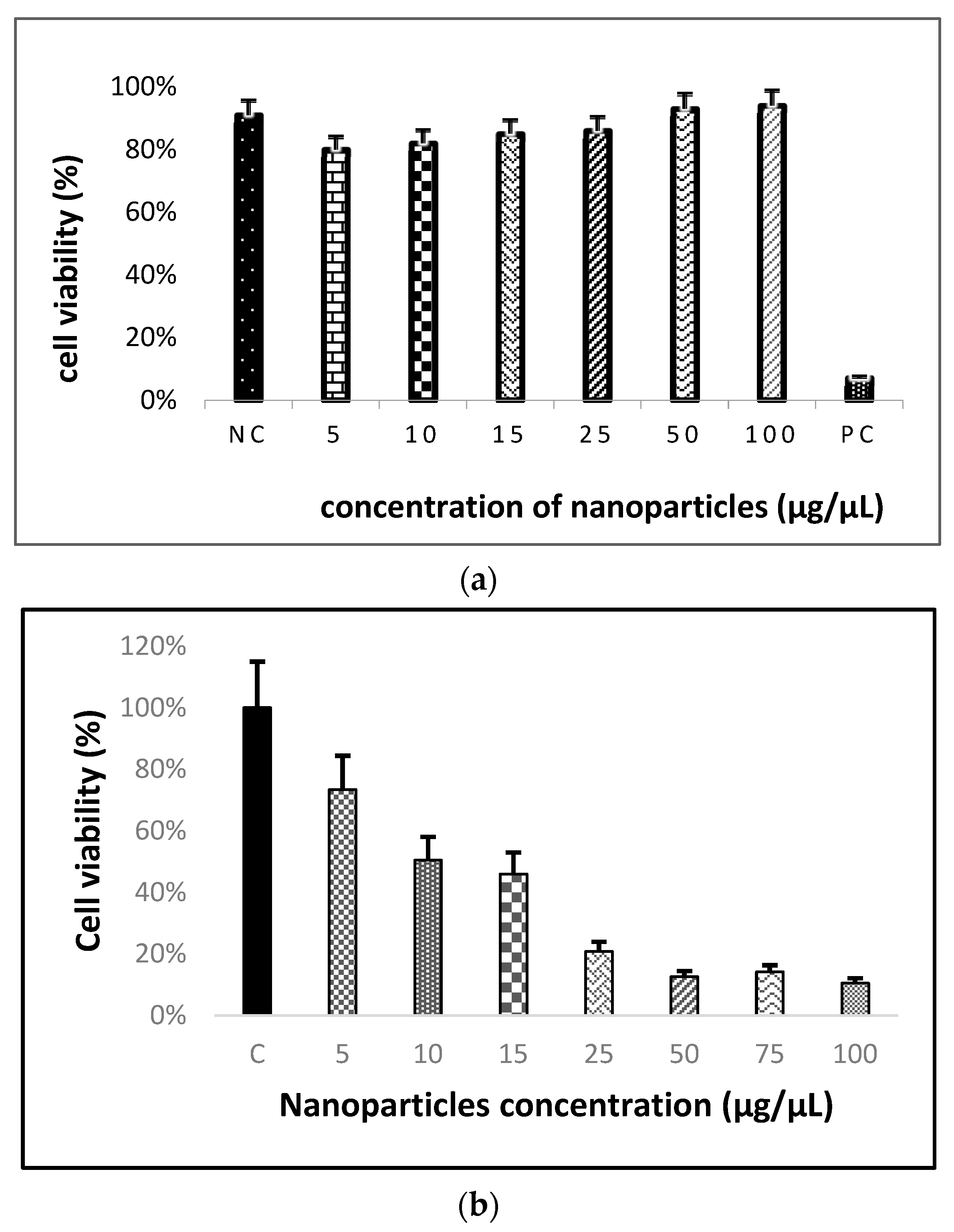
2.2.4. MTT Assay
2.2.5. Live/Dead Assay
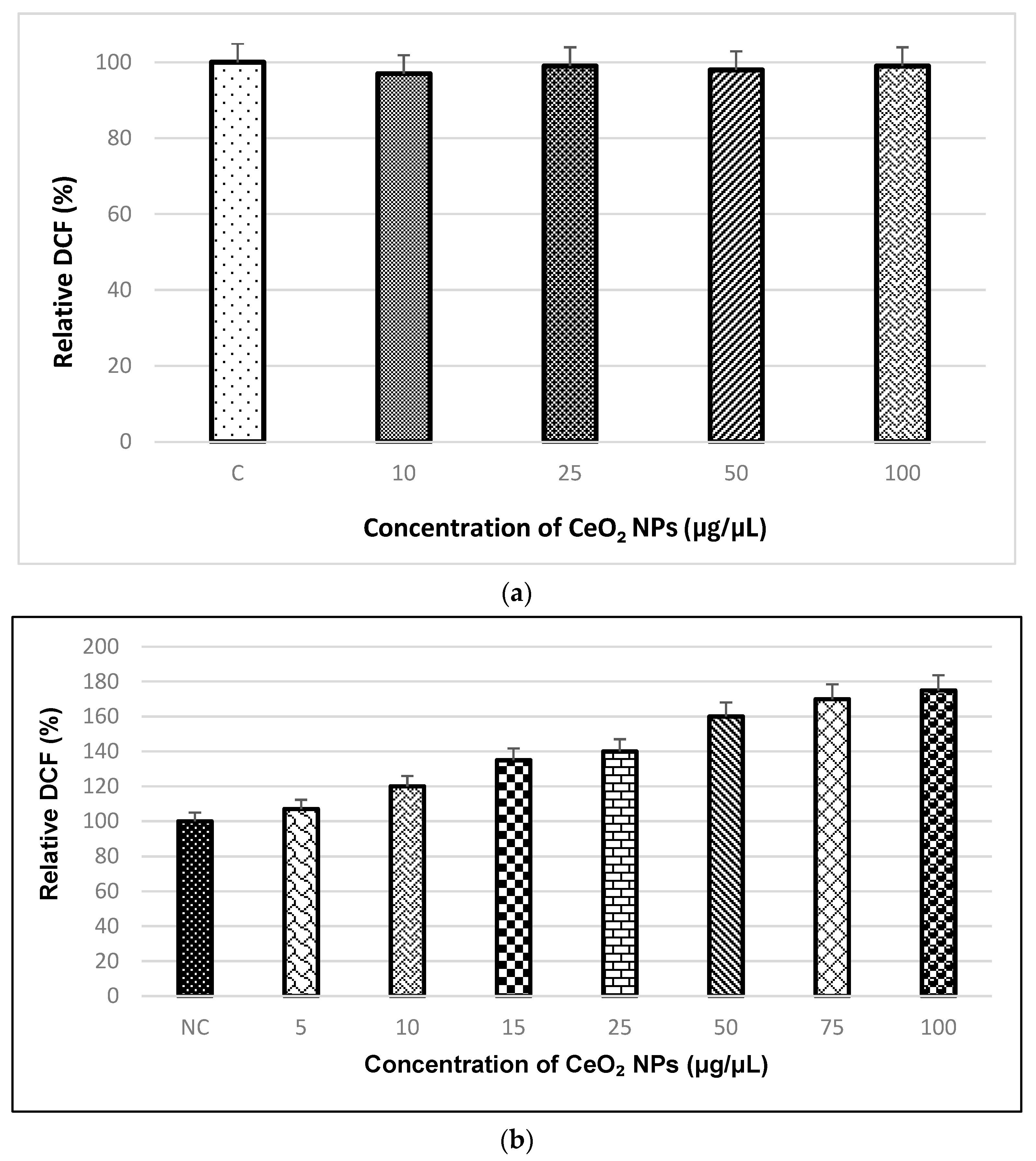
2.2.6. ROS Assay
2.2.7. Cell Proliferation Assay
3. Results
3.1. FESEM-EDS Analysis
3.2. FTIR Analysis
3.3. MTT Assay Analysis
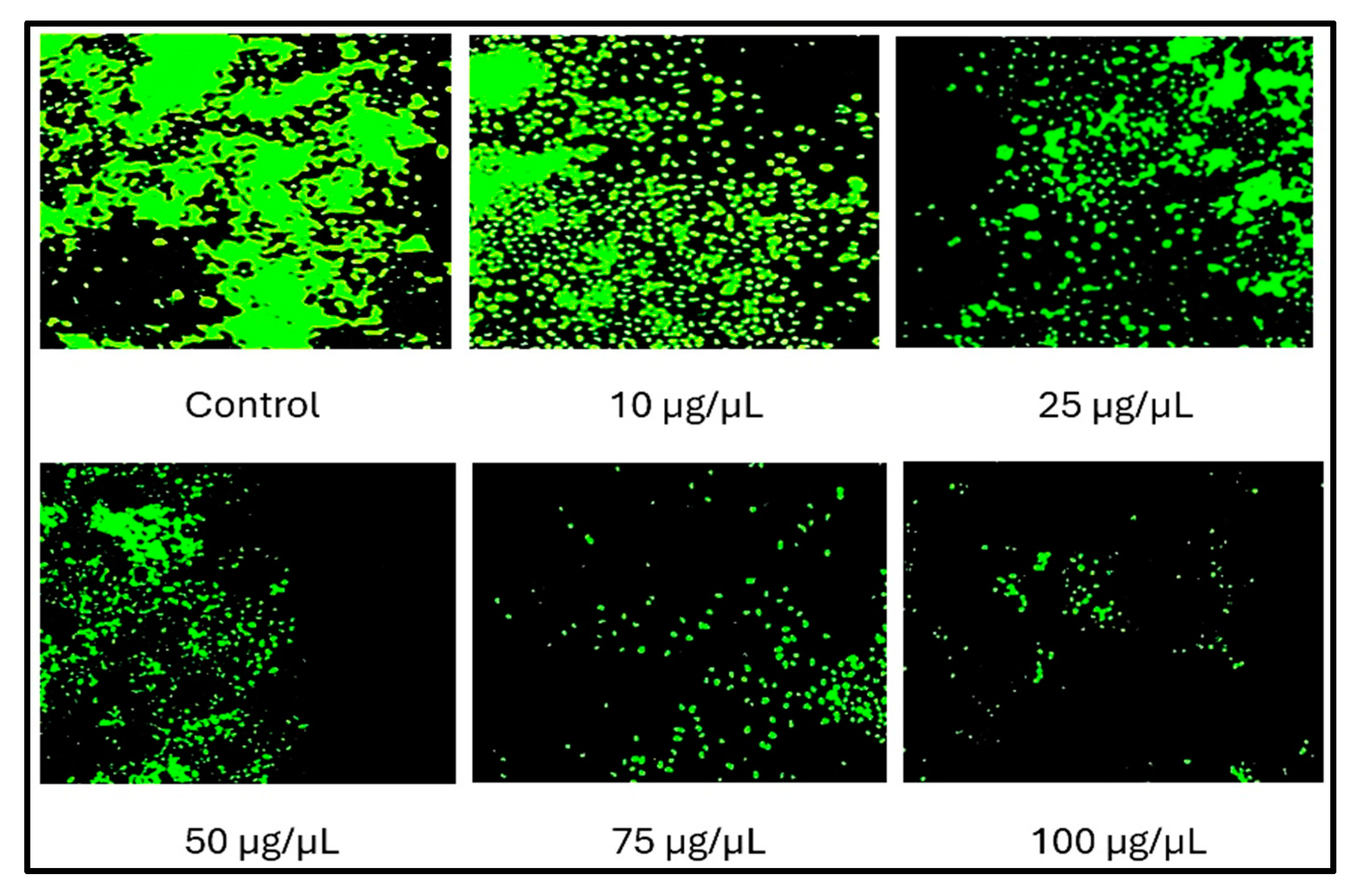
3.4. Live/Dead Assay Analysis
3.5. ROS Assay Analysis
3.6. Cell Proliferation Assay Analysis
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Hossain, N.; Mobarak, M.H.; Mimona, M.A.; Islam, M.A.; Hossain, A.; Zohura, F.T.; Chowdhury, M.A. Advances and significances of nanoparticles in semiconductor applications—A review. Results Eng. 2023, 19, 101347. [Google Scholar]
- Altammar, K.A. A review on nanoparticles: Characteristics, synthesis, applications, and challenges. Front. Microbiol. 2023, 14, 1155622. [Google Scholar]
- Vijayaram, S.; Razafindralambo, H.; Sun, Y.Z.; Vasantharaj, S.; Ghafarifarsani, H.; Hoseinifar, S.H.; Raeeszadeh, M. Applications of green synthesized metal nanoparticles—A review. Biol. Trace Elem. Res. 2024, 202, 360–386. [Google Scholar] [PubMed]
- Khan, S.; Hossain, M.K. Classification and properties of nanoparticles. In Nanoparticle-Based Polymer Composites; Woodhead Publishing: Sawston, UK, 2022; pp. 15–54. [Google Scholar]
- Gavas, S.; Quazi, S.; Karpiński, T.M. Nanoparticles for cancer therapy: Current progress and challenges. Nanoscale Res. Lett. 2021, 16, 173. [Google Scholar] [PubMed]
- Adan, A.; Kiraz, Y.; Baran, Y. Cell proliferation and cytotoxicity assays. Curr. Pharm. Biotechnol. 2016, 17, 1213–1221. [Google Scholar]
- Datta, A.; Mishra, S.; Manna, K.; Saha, K.D.; Mukherjee, S.; Roy, S. Pro-oxidant therapeutic activities of cerium oxide nanoparticles in colorectal carcinoma cells. ACS Omega 2020, 5, 9714–9723. [Google Scholar]
- Gao, Y.; Chen, K.; Ma, J.L.; Gao, F. Cerium oxide nanoparticles in cancer. OncoTargets Ther. 2014, 7, 835–840. [Google Scholar]
- Patil, N.; Bhaskar, R.; Vyavhare, V.; Dhadge, R.; Patil, Y. Overview on Methods of Synthesis of Nanoparticles. Int. J. Curr. Pharm. Res. 2021, 13, 11–16. [Google Scholar]
- Rajput, N. Methods of Preparation of Nanoparticles—A Review. Int. J. Adv. Eng. Technol. 2015, 7, 1806–1811. [Google Scholar]
- Namakka, M.; Rahman, M.R.; Said, K.A.M.B.; Mannan, M.A.; Patwary, A.M. A Review of Nanoparticle Synthesis Methods, Classifications, Applications, and Characterization. Environ. Nanotechnol. Monit. Manag. 2023, 20, 100900. [Google Scholar]
- Goornavar, V.; Biradar, S.; Ezeagwu, C.; Ezeagwu, D.; Hall, J.C.; Ramesh, G.T. Toxicity of raw and purified single-walled carbon nanotubes in rat’s lung epithelial and cervical cancer cells. J. Nanosci. Nanotechnol. 2015, 15, 2105–2114. [Google Scholar] [PubMed]
- Rane, A.V.; Kanny, K.; Abitha, V.K.; Thomas, S. Methods for Synthesis of Nanoparticles and Fabrication of Nanocomposites. In Synthesis of Inorganic Nanomaterials; Woodhead Publishing: Sawston, UK, 2018; pp. 121–139. [Google Scholar]
- Wason, M.S.; Zhao, J. Cerium oxide nanoparticles: Potential applications for cancer and other diseases. Am. J. Transl. Res. 2013, 5, 126–131. [Google Scholar] [PubMed]
- Nelson, B.C.; Johnson, M.E.; Walker, M.L.; Riley, K.R.; Sims, C.M. Antioxidant cerium oxide nanoparticles in biology and medicine. Antioxidants 2016, 5, 15. [Google Scholar] [CrossRef]
- Charbgoo, F.; Ramezani, M.; Darroudi, M. Bio-sensing applications of cerium oxide nanoparticles: Advantages and disadvantages. Biosens. Bioelectron. 2017, 96, 33–43. [Google Scholar]
- Walkey, C.; Das, S.; Seal, S.; Erlichman, J.; Heckman, K.; Ghibelli, L.; Traversa, E.; McGinnis, J.F.; Self, W.T. Catalytic properties and biomedical applications of cerium oxide nanoparticles. Environ. Sci. Nano 2015, 2, 33–53. [Google Scholar] [PubMed]
- Huston, M.; DeBella, M.; DiBella, M.; Gupta, A. Green synthesis of nanomaterials. Nanomaterials 2021, 11, 2130. [Google Scholar]
- Zholobak, N.M.; Shcherbakov, A.B.; Ivanova, O.S.; Reukov, V.; Baranchikov, A.E.; Ivanov, V.K. Nanoceria-curcumin conjugate: Synthesis and selective cytotoxicity against cancer cells under oxidative stress conditions. J. Photochem. Photobiol. B Biol. 2020, 209, 111921. [Google Scholar]
- Hosseinzadeh, R.; Khorsandi, K.; Esfahani, H.S.; Habibi, M.; Hosseinzadeh, G. Preparation of cerium-curcumin and cerium-quercetin complexes and their LEDs irradiation assisted anticancer effects on MDA-MB-231 and A375 cancer cell lines. Photodiagnosis Photodyn. Ther. 2021, 34, 102326. [Google Scholar]
- Javid, H.; Hashemy, S.I.; Heidari, M.F.; Esparham, A.; Gorgani-Firuzjaee, S. The anticancer role of cerium oxide nanoparticles by inducing antioxidant activity in esophageal cancer and cancer stem-like ESCC spheres. BioMed Res. Int. 2022, 2022, 3268197. [Google Scholar]
- Jiang, H.; Zuo, J.; Li, B.; Chen, R.; Luo, K.; Xiang, X.; Lu, S.; Huang, C.; Liu, L.; Tang, J.; et al. Drug-induced oxidative stress in cancer treatments: Angel or devil? Redox Biol. 2023, 63, 102754. [Google Scholar]
- Lin, W.; Huang, Y.W.; Zhou, X.D.; Ma, Y. Toxicity of cerium oxide nanoparticles in human lung cancer cells. Int. J. Toxicol. 2006, 25, 451–457. [Google Scholar] [PubMed]
- Kumar, P.; Nagarajan, A.; Uchil, P.D. Analysis of cell viability by the MTT assay. Cold Spring Harb. Protoc. 2018, 2018, pdb-prot095505. [Google Scholar]
- Bahuguna, A.; Khan, I.; Bajpai, V.K.; Kang, S.C. MTT assay to evaluate the cytotoxic potential of a drug. Bangladesh J. Pharmacol. 2017, 12, 115–118. [Google Scholar]
- Johnson, S.; Nguyen, V.; Coder, D. Assessment of cell viability. Curr. Protoc. Cytom. 2013, 64, 9.2.1–9.2.26. [Google Scholar]
- Zhou, S.; Cui, Z.; Urban, J. Dead Cell Counts During Serum Cultivation Are Underestimated by the Fluorescent Live/Dead Assay; WILEY-VCH Verlag: Weinheim, Germany, 2011. [Google Scholar]
- Najahi-Missaoui, W.; Arnold, R.D.; Cummings, B.S. Safe nanoparticles: Are we there yet? Int. J. Mol. Sci. 2020, 22, 385. [Google Scholar] [CrossRef]
- Sullivan, L.B.; Chandel, N.S. Mitochondrial reactive oxygen species and cancer. Cancer Metab. 2014, 2, 17. [Google Scholar]
- Lewinski, N.; Colvin, V.; Drezek, R. Cytotoxicity of nanoparticles. Small 2008, 4, 26–49. [Google Scholar] [PubMed]
- Riss, T.; Niles, A.; Moravec, R.; Karassina, N.; Vidugiriene, J. Cytotoxicity assays: In vitro methods to measure dead cells. In Assay Guidance Manual; NIH: Bethesda, MD, USA, 2019. [Google Scholar]
- Wiesenthal, A.; Hunter, L.; Wang, S.; Wickliffe, J.; Wilkerson, M. Nanoparticles: Small and mighty. Int. J. Dermatol. 2011, 50, 247–254. [Google Scholar]
- Kalaycıoğlu, Z.; Geçim, B.; Erim, F.B. Green synthesis of cerium oxide nanoparticles from turmeric and kinds of honey: Characterisations, antioxidant and photocatalytic dye degradation activities. Adv. Nat. Sci. Nanosci. Nanotechnol. 2022, 13, 015016. [Google Scholar]
- Andrabi, S.M.; Majumder, S.; Gupta, K.C.; Kumar, A. Dextran based amphiphilic nano-hybrid hydrogel system incorporated with curcumin and cerium oxide nanoparticles for wound healing. Colloids Surf. B Biointerfaces 2020, 195, 111263. [Google Scholar]
- Aseyd Nezhad, S.; Es-haghi, A.; Tabrizi, M.H. Green synthesis of cerium oxide nanoparticle using Origanum majorana L. leaf extract, its characterization and biological activities. Appl. Organomet. Chem. 2020, 34, e5314. [Google Scholar]
- Nadeem, M.; Khan, R.; Afridi, K.; Nadhman, A.; Ullah, S.; Faisal, S.; Mabood, Z.U.; Hano, C.; Abbasi, B.H. Green synthesis of cerium oxide nanoparticles (CeO2 NPs) and their antimicrobial applications: A review. Int. J. Nanomed. 2020, 15, 5951–5961. [Google Scholar]
- Gunasekaran, N.K.; Tumkur, P.P.; Bayon, N.N.; Prabhakaran, K.; Arasho, W.D.; Hall, J.C.; Ramesh, G.T. Cerium Oxide Nanoparticles: Synthesis and Characterization Using Curcuma longa (Turmeric Rhizome). Energy Environ. Focus 2023, 7, 83–89. [Google Scholar]
- Dutta, A. Fourier transform infrared spectroscopy. In Spectroscopic Methods for Nanomaterials Characterization; Elsevier: Amsterdam, The Netherlands, 2017; pp. 73–93. [Google Scholar]
- Jaggi, N.; Vij, D.R. Fourier transform infrared spectroscopy. In Handbook of Applied Solid State Spectroscopy; Springer: Boston, MA, USA, 2006; pp. 411–450. [Google Scholar]
- Ismail, A.A.; van de Voort, F.R.; Sedman, J. Fourier transform infrared spectroscopy: Principles and applications. In Techniques and Instrumentation in Analytical Chemistry; Elsevier: Amsterdam, The Netherlands, 1997; Volume 18, pp. 93–139. [Google Scholar]
- Zamborini, F.P.; Bao, L.; Dasari, R. Nanoparticles in measurement science. Anal. Chem. 2012, 84, 541–576. [Google Scholar] [PubMed]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gunasekaran, N.K.; Nazario Bayon, N.; Tumkur, P.P.; Prabhakaran, K.; Hall, J.C.; Ramesh, G.T. Anticancer Activity of Cerium Oxide Nanoparticles Towards Human Lung Cancer Cells. Nanomanufacturing 2025, 5, 6. https://doi.org/10.3390/nanomanufacturing5020006
Gunasekaran NK, Nazario Bayon N, Tumkur PP, Prabhakaran K, Hall JC, Ramesh GT. Anticancer Activity of Cerium Oxide Nanoparticles Towards Human Lung Cancer Cells. Nanomanufacturing. 2025; 5(2):6. https://doi.org/10.3390/nanomanufacturing5020006
Chicago/Turabian StyleGunasekaran, Nithin Krisshna, Nicole Nazario Bayon, Prathima Prabhu Tumkur, Krishnan Prabhakaran, Joseph C. Hall, and Govindarajan T. Ramesh. 2025. "Anticancer Activity of Cerium Oxide Nanoparticles Towards Human Lung Cancer Cells" Nanomanufacturing 5, no. 2: 6. https://doi.org/10.3390/nanomanufacturing5020006
APA StyleGunasekaran, N. K., Nazario Bayon, N., Tumkur, P. P., Prabhakaran, K., Hall, J. C., & Ramesh, G. T. (2025). Anticancer Activity of Cerium Oxide Nanoparticles Towards Human Lung Cancer Cells. Nanomanufacturing, 5(2), 6. https://doi.org/10.3390/nanomanufacturing5020006





