1. Introduction
Clinical data indicate that, in humans, the excessive intake of saturated fatty acids increases the risk of developing chronic diseases, including cardiovascular disease (CVD) and type II diabetes, and may play a role in the onset of neurodegenerative disorders, such as Alzheimer’s disease [
1,
2,
3,
4]. Significant proportions of saturated fats in the human diet come from ruminant meat, milk, and milk-based products [
5]. However, foods originating from ruminants also provide high-quality proteins, bioactive lipids, and essential vitamins and minerals to the human diet [
6]. There is a direct correlation between healthy bioactive lipid composition in food products originating from ruminants and dietary fat composition [
7,
8,
9]. Food enrichment with long-chain omega-3 polyunsaturated fatty acids (LC-PUFAs) can serve as a strategy to prevent or reduce the economic and social burden of the aforementioned chronic diseases, while retaining the dietary benefits associated with macro- and micronutrients in ruminant meat and milk products and, importantly, without requiring changes to consumer eating habits [
8,
10]. Meta-analyses of clinical studies have demonstrated a strong relationship between the consumption of LC-PUFAs, specifically docosahexaenoic acid (DHA; 22:6n-3) and eicosapentaenoic acid (EPA; 20:5n-3), and the reduced incidence of CVD, hypertension, certain autoimmune and inflammatory diseases, dementia, cancer, and all-cause mortality [
11,
12,
13,
14,
15].
In dairy cows, fish meal (FM) and fish oils (FO) are the principal sources of dietary LC-PUFA EPA and DHA, neither of which are commonly found in dairy cow rations [
10]. However, ingredients of fish origin tend to affect the oxidative stability of milk, show substantial variability in composition and quality, and are susceptible to contamination with lipophilic organic chemicals present in marine ecosystems [
16]. These supplements can inhibit ruminal biohydrogenation of unsaturated fatty acids thus modifying the flow of fatty acids to the omasum, leading to substantive alterations in rumen microbiota and metabolism [
17,
18]. Moreover, the environmental sustainability of declining and fluctuating fish stocks needs to be considered when using FM and FO as sources of beneficial LC-PUFAs [
19]. There is an ongoing search for alternative PUFA ingredients, such as DHA-rich marine algae or thraustochytrids, as dietary supplements to increase PUFA availability in the food chain [
19,
20].
The thraustochytrid
Aurantiochytrium limacinum is a member of the heterotrophic protists, which are commonly (mis-)classified as microalgae, and also exhibit fungus-like characteristics [
21]. These organisms are the primary producers of DHA in the pelagic food chain and can be heterotrophically mass-cultured for industrial purposes using suitable simple carbon sources and without the requirement for sunlight [
22]. The synthesized DHA is accumulated as a constituent of triacylglycerols in intracellular lipid droplets within the unextracted biomass, offering protection against oxidation and thermal processing [
23]. Introducing DHA-rich microbial biomass to the diet of ruminants can potentially improve the nutritional value of milk and meat products [
8,
24]. However, this will only succeed if dietary inclusion does not suppress rumen fermentation and, thus, lower the productivity of dairy cows [
18,
25,
26]. It is also important for the introduced fatty acids to resist lipolytic degradation and/or biohydrogenation by rumen bacteria and bypass the rumen in an unsaturated form [
26,
27].
In the present study, we investigated the effect of an A. limacinum product (AURA) on rumen microbiota-mediated fermentation, which was measured via the production of gaseous and acidic metabolic end-products and the microbial biomass yield in an ex vivo rumen fermentation model. We also assessed the resistance of DHA, a fatty acid component of major interest in AURA products, to degradation and biohydrogenation by rumen microbes. Finally, we conducted a mechanistic seven-week pilot feeding trial with two rumen-cannulated dairy cows to verify the ex vivo findings and monitor diet-derived DHA deposition in milk. To our best knowledge, this is the first study to examine the effects of protist-based biomass on pivotal rumen fermentation parameters and the deposition of DHA in milk using a combination of ex vivo and in vivo approaches.
2. Materials and Methods
2.1. Basal Diet for Ex Vivo Fermentation
The basal diet used as substrate in the ex vivo fermentation studies was composed of fresh grass silage (energy 10.8 MJ/kg, crude protein 16%) and commercial dairy cow concentrate (Lypsykrossi, Suomen Rehu Ltd., Hyvinkää, Finland; energy 12.3 MJ/kg, crude protein 19%), dosed 60:40 on a dry matter (DM) basis. The grass silage used was a combination of timothy (Phleum pratense) and meadow fescue (Festuca pratensis), cut into 3–8 mm pieces. A subsample of chopped silage was taken for DM determination, but fresh silage was used for the actual fermentation studies since drying is likely to affect fermentation kinetics. Commercial concentrate pellets based on oats, barley, wheat bran, rapeseed meal, sugar beet pulp, vitamins, and minerals were milled to ensure uniform particle size. The total amount of basal feed material was 0.25 g DM per vessel. Each feed component, including the AURA supplement, was separately weighed into individual fermentation vessels to ensure similar ratios.
2.2. Rumen Ex Vivo Protocol
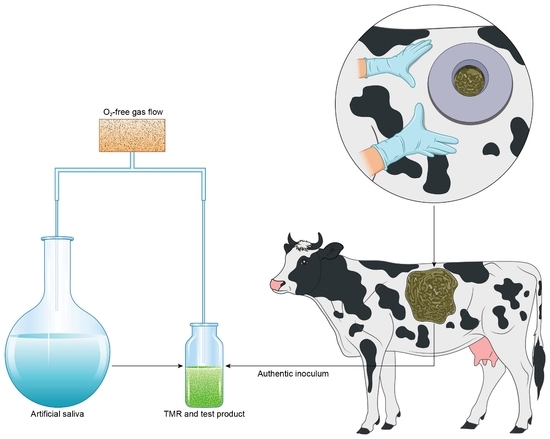
The two rumen-cannulated dairy cows used as donors of rumen fluid inoculum (one animal per experiment) were lactating at the time of sampling and were fed 8 kg DM per day of a commercial compound feed (Opti-Maituri 26, Lantmännen Feed Oy, Turku, Finland; energy 12.8 MJ/kg, crude protein 26%) and approximately 40 kg of grass silage (~13 kg DM with energy 10.8 MJ/kg and crude protein 16%). Rumen fluid was collected from the middle part of the rumen before the first daily feeding using a vacuum pump. The rumen fluid was immediately sealed in an airtight thermos container, transported to the laboratory, and used as inoculum within one hour of collection. Prior to inoculation, large solid particles were removed from the fresh rumen fluid by was straining through a 3 mm steel mesh under protective anoxic gas flow.
Glass serum bottles (20 mL) were used as the fermentation vessels. The vessels, containing the feed components and test products, were flushed with CO
2 gas that had been passed through a hot copper catalyst, to scavenge any residual O
2, and sealed with thick butyl rubber stoppers. Fermentation was initiated by introducing temperature-adjusted (+38 °C) anaerobic artificial saliva, including phosphate and bicarbonate, as buffering agents [
28], and fresh rumen fluid (10% inoculum) into the vessels to a total volume of 10 mL under protective O
2-free CO
2 gas flow. Finally, the vessels were sealed with butyl rubber stoppers and aluminum seals and incubated at +38 °C in a gyratory shaker at 100 rpm. The exact time of inoculation for each vessel was recorded and considered when sampling and stopping fermentation. Inoculation of the fermentation vessels was conducted in a random order to avoid any bias resulting from time of inoculation or freshness of the inoculum.
Gas production was measured at chosen timepoints by manually puncturing the rubber stopper with a needle that was connected to a high-precision glass syringe with a sensitive ground plunger and recording the volume of gas released from the fermentation vessels. In both ex vivo experiments, five replicate vessels of each dietary treatment were withdrawn from the study at each time point for various analyses. Thus, none of the test vessels was sampled during fermentation to avoid potential effects on fermentation kinetics. The total duration of the individual ex vivo experiments, and their intermediate sampling points, are described below.
2.3. Ex Vivo Rumen Experiment I
This ex vivo experiment examined changes in the overall abundance and stability of LC-PUFAs in the rumen environment that resulted from the
A. limacinum (AURA) biomass test product (Alltech Inc., Nicholasville, KY, USA). The effect of the spray-dried AURA product on rumen fermentation was also assessed. Corresponding analyses were also performed on the ethyl acetate (EtOAc)-soluble fraction of the AURA product, which was obtained as follows: (1) 9.0 g of the AURA product was suspended in H
2O, (2) the pH was adjusted to 3 with HCl, and water was added to a total added volume of 21 mL, (3) 27 mL of EtOAc was added and the mixture was shaken at room temperature overnight, (4) the extract was centrifuged at 3000×
g for 10 min, (5) the organic phase was withdrawn and its volume was adjusted to 25 mL with fresh EtOAc, and (6) long-chain fatty acid (LCFA) composition was analyzed as described below. The volume of EtOAc extract used in the fermentation study was adjusted so that the LCFA content corresponded to that in the intact AURA product. The extract was introduced to empty vessels, and the solvent was evaporated to dryness before adding other components. For comparison, an FO product commonly used as a feed ingredient was tested in the experiment. The AURA and FO product doses are listed in
Table 1. For all test products, the total LCFA additions were the same for doses 1 through 4. The sampling time points in fermentation were 0 and 8 h.
2.4. Ex Vivo Rumen Experiment II
In this experiment, we measured the dose–response relationship of the AURA test product with rumen fermentation and the resistance of DHA to bacterial attack by rumen microbes by using a wider AURA product concentration range than that used in experiment I. The exact concentrations of AURA used are listed in
Table 1. In experiment II, the sampling time points were 0, 4, and 8 h of fermentation.
2.5. In Vivo Experiment with Rumen-Cannulated Cows—Animals, Trial Schedule, and Dietary Treatments
Two rumen-cannulated lactating dairy cows were assigned to the in vivo study. The animals were kept in the research facility of Alimetrics Ltd. in Southern Finland, in accordance with EU Directive 2010/63/EU, and cannulation was approved by the Animal Experiment Board in Finland. The animals were kept in a herd of approximately 30 dairy cows in a loose-stall system and used in a seven-week trial comprising three feeding phases: (1) a two-week pre-trial control diet, (2) a four-week AURA diet, and (3) a one-week washout phase. The diet used for both the control and the washout phase was that used routinely on the farm: 7–8 kg of oat-based compound feed and grass silage fed ad libitum (~8 kg DM). During the compound feeding period, the experimental animals were tied within individual stalls. Since the normal diet of the experimental cows did not change, no adaptation period was required prior to the start of the trial. During the AURA diet period, the spray-dried product (140 g/head/day) was manually mixed into the compound feed every day and served in four portions. Grass silage was always freely available to the animals, except when the compound feed was served.
2.6. Sampling of Rumen Fluid and Milk
A vacuum pump was used to collect rumen fluid samples (each 100 mL) for short-chain fatty acid (SCFA) and microbial analyses through the cannula of both animals in the morning of four consecutive days during the final week of each dietary phase (control, AURA, washout) of the trial. The samples were drawn from the mid-ventral region of the rumen immediately prior to the first feeding of each day and then immediately frozen on dry ice. To analyze the LCFA content in milk, a 100 mL sample was drawn during both morning and evening milking on two consecutive days during each week of the three dietary phases and then frozen on dry ice. Prior to LCFA analysis in the laboratory, the samples from each day (morning and evening sample) were thawed and pooled at a ratio corresponding to the respective volumes of milk obtained in the morning and evening milking.
2.7. Analysis of Short-Chain Fatty Acids in Rumen Fluid
Volatile fatty acids (VFA) and lactic acid, referred to hereafter as SCFA, were analyzed as free acids in the rumen fluid samples from both ex vivo and in vivo experiments using pivalic acid (Sigma-Aldrich, St. Louis, MO, USA) as an internal standard [
29]. For this, 400 μL of rumen fluid and 2.4 mL of 1.0 mM pivalic acid solution were mixed, shaken vigorously for 5 min, and centrifuged at 3000×
g for 10 min. Then, 800 μL of the supernatant was mixed with 400 μL of saturated oxalic acid solution, incubated at 4 °C for 60 min, and centrifuged at 18,000×
g for 10 min. The supernatant was analyzed by gas chromatography (Agilent Technologies, Santa Clara, CA, USA) using a glass column packed with 80/120 Carbopack B-DA/4% Carbowax stationary phase, helium as the carrier gas, and a flame ionization detector. The quantified acids were acetic, propionic, butyric, valeric, isobutyric, 2-methylbutyric, isovaleric, and lactic acid.
2.8. Extraction of Microbial DNA from Rumen Fluid Samples
Rumen fluid subsamples from ex vivo experiment II and the in vivo trial were collected for analysis of the abundance of different microbial species, groups, or genera via real-time quantitative PCR (qPCR). Microbial DNA was extracted from rumen fluid using the following protocol. First, 0.4 mL of each rumen fluid sample was suspended in 1.6 mL of 50 mM phosphate-buffered saline with EDTA, and the suspension was shaken vigorously for 2 min. Next, the microcentrifuge tubes were centrifuged at 18,000× g for 10 min to pellet the microbial cells, after which the pellet was resuspended in 600 µL of phosphate lysis buffer with 100 mM Tris and 50 mM EDTA (pH 8.0). The suspension was transferred to a screw-cap microcentrifuge tube containing 20 µL of proteinase K (20 mg/mL; Roche Diagnostics GmbH, Mannheim, Germany) and 0.4 g of sterile glass beads (Scientific Industries Inc., Bohemia, NY, USA), and incubated at 65 °C for 60 min with shaking for 30 s (1400 rpm) at 10 min intervals. The microbial cells were disrupted by two 1 min rounds of bead beating (FastPrep-24™, MP Biomedicals, Irvine, CA, USA) at 6.5 m/s, after which genomic DNA was purified from the homogenates through phenol–chloroform–isoamyl alcohol (24:1) extraction with centrifugation at 10,000× g for 10 min, followed by chloroform–isoamyl alcohol purification with centrifugation at 10,000× g for 10 min. The DNA was precipitated by the addition of 0.6 volumes of 100% isopropanol and pelleted by centrifugation at 18,000× g for 10 min. Finally, the DNA pellet was washed twice with 1 mL of ice-cold 70% ethanol, dried, and resuspended in 100 µL of Tris-EDTA buffer with 10 mM Tris and 1.0 mM EDTA (pH 8.0) (AppliChem, Darmstadt, Germany).
2.9. qPCR Analysis of Rumen Microorganisms
qPCR analyses were performed using an ABI Prism Sequence Detection System 7500 instrument (Thermo Fisher Scientific Inc., Waltham, MA, USA). Amplifications were conducted in 15 µL volumes with SYBR Select Master Mix (Thermo Fisher Scientific Inc., Waltham, MA, USA), 0.25 µM of both primers, and 5 µL of 1:100 diluted template DNA or deionized sterile water as a no-template control. The rRNA gene-targeted primer sequences and PCR product sizes used for enumeration of the target microorganisms are listed in
Table 2. The thermal cycling conditions involved one cycle of preheating at 50 °C for 2 min and an initial denaturation at 95 °C for 10 min, followed by 40 cycles of denaturation at 95 °C for 15 s and annealing and extension at the primer-specific annealing temperatures for 60 s. To verify the specificity of amplifications based on the melting temperature of PCR products, a melt curve analysis was carried out in conjunction with each qPCR run by slowly decreasing the temperature from 95 to 60 °C, with fluorescence determination at 0.3 °C intervals.
Tenfold serial dilutions ranging from 1 × 108 to 1 × 102 synthetic small-subunit rRNA gene copies of target microorganism (gBlocks® Gene Fragments, IDT, Coralville, IA, USA) were included in each 96-well plate. The fractional cycle number at which the fluorescence passed the threshold (set at 0.3 fluorescence units) was determined for the unknowns and compared with the standard curves. Taking into account the original volume of the starting material, elution volume, and PCR template dilution, the numbers of small-subunit rRNA gene targets were determined per mL of rumen fluid. For data analysis, the rRNA gene copies/mL values were log10-transformed to achieve a normal distribution.
2.10. LCFA Analysis of Rumen Fluid and Milk Samples
DHA and other LCFAs were analyzed in the test products (FO, AURA, and the EtOAc extract of AURA), the ex vivo rumen fluid simulation samples, and the milk samples from the in vivo feeding trial. The samples were prepared according to a previously described procedure [
35] and analyzed on an Agilent 7890 gas chromatograph equipped with a 5975C Mass Selective Detector (Agilent Technologies, Santa Clara, CA, USA).
2.11. Statistical Analysis
2.11.1. Ex Vivo Results
To enable statistical comparisons between the different test product doses and time points, Tukey’s honestly significant difference (HSD) post hoc test was performed following an analysis of variance (ANOVA) of the ex vivo simulation data with the type I error rate set to α = 0.05. The statistical significance of the change in the residual concentration of each LCFA characteristic within each test product during 8 h of fermentation in ex vivo experiment I was determined through an independent sample t-test, which was performed separately for each test product and dose. This approach was used because the objective was to discover the potential concentration change in each individual acid during fermentation, rather than an overall change in acid composition.
2.11.2. In Vivo Results
The mechanistic feeding trial involved two cows, which were in synchrony for two weeks on the control diet, then for four weeks on the AURA diet and, finally, for one week on the control diet (washout). When we statistically analyzed the effects of the AURA test product on the rumen microbial parameters, we compared data from samples collected during the final week of each feeding period (control, AURA, washout); in practice, we compared the means of data generated from samples collected on four consecutive days in weeks two (control diet), six (AURA diet), and seven (washout).
Since there were animal-specific differences in the abundances of rumen microbes and SCFAs, the microbial densities and SCFA concentrations in the experimental animals were normalized such that the mean of each parameter in all analyzed samples was the same for both animals. This allowed the Tukey’s HSD post hoc test to indicate true shifts within each period of measurement rather than merely reflecting differences between the animals in their baseline levels of rumen microorganisms or SCFA concentrations.
All statistical tests on both ex vivo and in vivo data were conducted using the IBM SPSS Statistics Subscription (Build 1.0.0.1327) software package (Chicago, IL, USA).
4. Discussion
Previous studies have examined the effects of LC-PUFAs on rumen fermentation and production parameters [
8,
36,
37]. Pure acids have been tested in some studies, whereas in others, complex acid sources composed of mixtures of acids have been used [
8,
38,
39,
40,
41,
42]. The results of these studies have been inconsistent, partly because of the different experimental models used and partly because of the different test product compositions and doses applied. It is indisputable that the replacement of saturated fats in animal products with LC-PUFAs would be beneficial for the health of human end-consumers. However, in commercial practice, this concept will only be viable if there is no negative effect on productivity and if the LC-PUFA supplement is reasonably priced. Practical application will also be nonviable if strong inhibition of total rumen fermentation activity (VFA production) occurs, or if microbial fermentation shifts in an unfavorable direction and can no longer support body weight gain or the production of high volumes of milk of the correct composition. In this study, we conducted a detailed investigation of one potential protist source of healthy fatty acids at realistic doses. The use of high product doses would not be meaningful because excess LC-PUFA is very well known to inhibit bacterial fermentation and because the benefits gained in product quality could not compensate for the higher price of high doses.
There are many possible variations of the laboratory rumen fermentation experiment. Laboratory arrangements are always models and therefore intended to mimic conditions and the fermentation activity in the rumen with sufficient accuracy for considering the questions at hand. In the model used in this study, special attention was paid to proper physicochemical conditions, the careful handling of inoculum, and precise execution of the fermentation, sampling, and analysis of the vessels. The five fermentation vessels with independently introduced feed components, test products, and inoculum were considered as replicates in the statistical evaluation. The experimental arrangement allows accurately determining the response of the specific rumen microbiota introduced in the vessels to the test products used. The question remains whether the rumen content used has a microbiota composition that adequately represents the microbiota of the whole herd of the farm, all farms in a specific country, or all countries with different feeding regimens. Although the magnitude of test product effects could vary, there are no reports suggesting that the existence and direction of fermentation effects would be different in individual animals on a given diet. In the two ex vivo experiments, we used rumen fluid inoculum from two different dairy cows with identical results. It remains to be shown whether similar results are obtained, for example, with American beef cattle fed with a high-grain diet.
In the studies described here, the concentration of the AURA test product ranged from 0.08 to 2.5 mg/mL, supplying 0.04–1.3 mg/mL of total fatty acids or 0.013–0.40 mg/mL of DHA. The dose of AURA applied in the feeding experiment with dairy cows was 140 g/head/day, which was administered in four servings during the day. Assuming a rumen volume of 60 L [
43], the introduced dose corresponds to 0.93 mg/mL, which is within the concentration range tested in the ex vivo models. Two independent fermentation experiments confirmed that none of the AURA doses tested affected rumen fermentation in any way, with no changes detected in either gas or SCFA production nor in total eubacteria abundance. A recent study investigating the potential antimicrobial effects of AURA on nine pure cultures of intestinal bacteria related to human health showed that the product did not inhibit bacterial growth at 10 mg/mL [
44], which is four times the highest concentration used in this study. On the other hand, studies of the pure cultures of rumen bacteria involved in the biohydrogenation of LC-PUFAs (
Butyrivibrio fibrisolvens and
B. proteoclasticus) have shown that >30 µg/mL of various pure unsaturated fatty acids can inhibit growth [
45,
46]. In vitro studies using complex rumen microbiota as an inoculum and ruminant feed as a substrate have not observed inhibition of rumen fermentation in cows. For example, a batch fermentation study found that neither pure EPA nor DHA at 0.2 mg/mL (2% of feed DM) had any significant effect on microbial gas or VFA production, or the on the levels of feed DM [
47]. A study examining sunflower, fish, and algae oils in a RUSITEC continuous fermentation system showed that all oil combinations at 2–3% of feed DM suppressed methane production but increased total VFA and propionate [
48].
Previous tests on various LC-PUFAs have produced inconsistent results; therefore, the benefits of feed supplements containing healthy unsaturated fatty acids have been questioned. There are several reasons for these apparent contradictions in the published literature. One is that the effective concentration of LC-PUFAs to which the bacteria are exposed in the test system may not be the same as the introduced dose for a given volume of culture or rumen contents. It is worth noting that in studies with pure bacterial cultures and liquid growth medium, the lipophilic compounds tend to be enriched in biological membranes and, consequently, the density of bacteria in solution can play a significant role. A dense microbial system tolerates higher product concentrations because the effective product dose per bacterium is lower. This implies that the microbial community in the true rumen would tolerate higher LC-PUFA concentrations than that in any laboratory fermentation model. Another important element is the presence of a solid feed matrix, which can be expected to protect bacteria by binding some potentially inhibitory compounds in a reversible fashion, as observed in other biological systems [
49]. Typically, a solid matrix is absent in studies with pure bacterial cultures, while it is commonly included when complex microbial communities are used as the inoculum in batch or continuous fermentation models. When a solid matrix is present, it is worth considering whether the concentration of the test product should be calculated against the feed DM content or the culture volume. It is difficult to judge which is the correct approach, but it is important to acknowledge that this may have a major impact on the tolerance of rumen bacteria to LC-PUFA compounds. In the ex vivo arrangement used in the experiments described in this paper, the highest concentration of AURA product was 2.5 mg/mL when calculated against the liquid volume (corresponding to 1.3 mg/mL of total fatty acids or 0.4 mg/mL of DHA), but it comprised 9.1% when calculated as a percentage of feed DM. When the effect of a product on microbial fermentation is sought, it is necessary to consider the effective concentration of the product that the bacteria will encounter. The effective concentration is heavily influenced by the water solubility of the product and the presence of a solid matrix to which part of or the entire product may potentially attach.
Previous in vivo feeding studies have mainly concentrated on fatty acid biohydrogenation, potential shifts in rumen microbial community structure, and the physiology of the animal, but not on parameters connected to overall fermentation activity. Algal biomass at 1% and FO at 2–3% of feed DM have been found to inhibit biohydrogenation and affect the abundance of bacteria responsible for biohydrogenation [
39,
50]. Furthermore, DHA-rich algal biomass provided at 100 g/head/day has been found to improve reproductive performance in dairy cows, an additional health benefit of feeding with fatty acids [
51]. In the in vivo experiment in the present study, the residual concentration of total SCFAs was slightly reduced during the four weeks of the AURA diet. However, all the available facts suggest that this reduction was not due to the suppression of rumen fermentation but is most likely attributable to the improved efficiency of VFA uptake through the rumen epithelium when LC-PUFAs are present in the diet. This proposal is supported by the following facts based on our study: (1) the calculated LC-PUFA concentration in the rumen was less than half the actual concentration, showing that there was no inhibition in the ex vivo experiments; (2) the actual LC-PUFA concentration in the rumen is likely to be lower than that calculated, since the acids are continuously absorbed; (3) the density of bacteria and the feed DM content were significantly higher in the rumen than in the ex vivo experiments, thus protecting the bacteria in the rumen environment from potentially inhibitory LC-PUFAs; and (4) the total density of bacteria in the rumen was not reduced upon exposure to the AURA product.
Multiple studies have shown that LC-PUFA-containing products alter the composition of rumen microbiota, since different microorganisms react differently to the presence of PUFAs in the growth habitat [
39,
48,
50,
52]. Some microbial changes were observed in the in vivo experiment in this study. The most significant finding was related to bacteria involved in lactic acid metabolism pathways. At the end of the four-week AURA diet period, growth of a major lactic acid-producing genus,
Lactobacillus spp., was significantly suppressed, as was that of representatives of the genus
Megasphaera, which is known to be involved in rumen lactic acid utilization [
53,
54]. Moreover, the residual concentration of lactic acid in rumen fluid was significantly decreased. This may indicate that the risk of lactic acidosis is reduced when the AURA product is used in dairy cow diets, but further studies are needed to reveal the detailed mode of action. Interestingly, the density of methanogens was also lower at the end of the AURA diet period. There were similar findings in a previous study when various oils rich in unsaturated fatty acids were introduced into the rumen system [
48,
55,
56,
57]. In the present study, microbial alterations occurred when AURA was added to the diet, but these began to revert after the AURA product was removed from the diet. This indicates that the observed changes were not a result of temporal effects related to the season or phase of lactation but were truly associated with the introduction and withdrawal of the AURA test product in the diet.
Although understanding the effects of an LC-PUFA dietary supplement on rumen microbial activity is essential for its commercial adoption, it is equally important to demonstrate that the essential omega-3 fatty acids survive the complex rumen ecosystem and are deposited in the milk and meat. In our ex vivo experiments that included the AURA product, the disappearance of the C22 fatty acid DHA was concentration-dependent; the higher the concentration of introduced AURA, the smaller the proportion of DHA that disappeared during fermentation. At levels corresponding to the dose recommended for commercial production animals, approximately 10% of the DHA was degraded or transformed by rumen microorganisms. For EPA, a characteristic C20 fatty acid in FO, the reduction during fermentation was nearly 20%. One type of transformation is biohydrogenation catalyzed by anaerobic rumen bacteria that use unsaturated fatty acids as ultimate electron acceptors in their electron transport chains. It is worth noting that LCFAs do not disappear solely due to biohydrogenation but are also used as building blocks in bacterial anabolism. In this study, almost 10% of the fully saturated palmitic acid, which cannot be used as an electron acceptor, disappeared during fermentation. Ruminal polyunsaturated C18 fatty acids have been widely studied, and the active role of the rumen bacteria, especially that of
Butyrivibrio species, in the partial or complete ruminal biohydrogenation of C18 has been confirmed [
39,
48,
50,
58]. It appears that the complete reduction of the carbon double bonds is a synergistic process that involves a consortium of bacteria, each with a specific role. Consequently, differences in these species’ susceptibility to LC-PUFAs may lead to the accumulation of intermediates at certain acid concentrations. In general, the fatty acids with the longest carbon skeletons (EPA and DHA) are more resistant to biohydrogenation than shorter-chain PUFAs, such as linoleic and α-linolenic acid [
45,
58]. DHA and EPA have also been reported to inhibit the hydrogenation of unsaturated C18 acids [
47]. Based on these findings, it is unlikely that the DHA in AURA or the EPA in FO would be degraded in the rumen to an extent that would impair the deposition of in-feed DHA or EPA in edible products.
A pilot study conducted with two rumen-cannulated dairy cows confirmed that the introduction of the AURA product into the diet led to a clear change in the fatty acid profile in milk. The DHA concentration in milk increased over 25-fold during the four-week AURA diet period and then decreased by 30–40% within one week of washout. Interestingly, DHA was deposited in fat tissues during the four weeks of the AURA diet and then slowly released into milk during the subsequent week on the control (washout) diet. The effects of feeding with
A. limacinum biomass on the milk production and composition have been previously reported [
9,
24,
51]. Similar increases in DHA deposition in milk were found in these studies, but the total milk fat depression results were inconsistent. On the other hand, the total milk yield and yields of protein and lactose were significantly increased when the DHA-rich AURA product was added to the diet.
To summarize, this study shows that the addition of AURA, a product comprising DHA-rich microbial biomass, to the diet of dairy cows had no negative effects on rumen fermentation intensity and showed some positive effects on rumen microbial composition. DHA, the most important omega-3 fatty acid in AURA, was demonstrated to be stable and only underwent modest degradation in the rumen system, resulting in the successful deposition of DHA in the milk of cows that had been fed a diet supplemented with AURA.
 AURA;
AURA;  AURA extract;
AURA extract;  FO.
FO.
 AURA;
AURA;  AURA extract;
AURA extract;  FO.
FO.
 acid concentration at 0 h;
acid concentration at 0 h;  acid concentration at 8 h. AURA concentration in fermentation medium: Dose 1 (0.08 mg/mL), Dose 2 (0.15 mg/mL), Dose 3 (0.30 mg/mL) and Dose 4 (0.60 mg/mL). FO doses in fermentation medium: Dose 1 (0.05 mg/mL), Dose 2 (0.11 mg/mL), Dose 3 (0.22 mg/mL) and Dose 4 (0.44 mg/mL).
acid concentration at 8 h. AURA concentration in fermentation medium: Dose 1 (0.08 mg/mL), Dose 2 (0.15 mg/mL), Dose 3 (0.30 mg/mL) and Dose 4 (0.60 mg/mL). FO doses in fermentation medium: Dose 1 (0.05 mg/mL), Dose 2 (0.11 mg/mL), Dose 3 (0.22 mg/mL) and Dose 4 (0.44 mg/mL).
 acid concentration at 0 h;
acid concentration at 0 h;  acid concentration at 8 h. AURA concentration in fermentation medium: Dose 1 (0.08 mg/mL), Dose 2 (0.15 mg/mL), Dose 3 (0.30 mg/mL) and Dose 4 (0.60 mg/mL). FO doses in fermentation medium: Dose 1 (0.05 mg/mL), Dose 2 (0.11 mg/mL), Dose 3 (0.22 mg/mL) and Dose 4 (0.44 mg/mL).
acid concentration at 8 h. AURA concentration in fermentation medium: Dose 1 (0.08 mg/mL), Dose 2 (0.15 mg/mL), Dose 3 (0.30 mg/mL) and Dose 4 (0.60 mg/mL). FO doses in fermentation medium: Dose 1 (0.05 mg/mL), Dose 2 (0.11 mg/mL), Dose 3 (0.22 mg/mL) and Dose 4 (0.44 mg/mL).

 AURA 0.25 mg/mL;
AURA 0.25 mg/mL;  AURA 0.63 mg/mL;
AURA 0.63 mg/mL;  AURA 1.50 mg/mL;
AURA 1.50 mg/mL;  AURA 2.50 mg/mL.
AURA 2.50 mg/mL.
 AURA 0.25 mg/mL;
AURA 0.25 mg/mL;  AURA 0.63 mg/mL;
AURA 0.63 mg/mL;  AURA 1.50 mg/mL;
AURA 1.50 mg/mL;  AURA 2.50 mg/mL.
AURA 2.50 mg/mL.