Biophysics and Quantum Limitation of Photoreceptive Processes
Definition
:1. Introduction
2. Basic Concepts
3. Nonimaging Photoreceptive Process
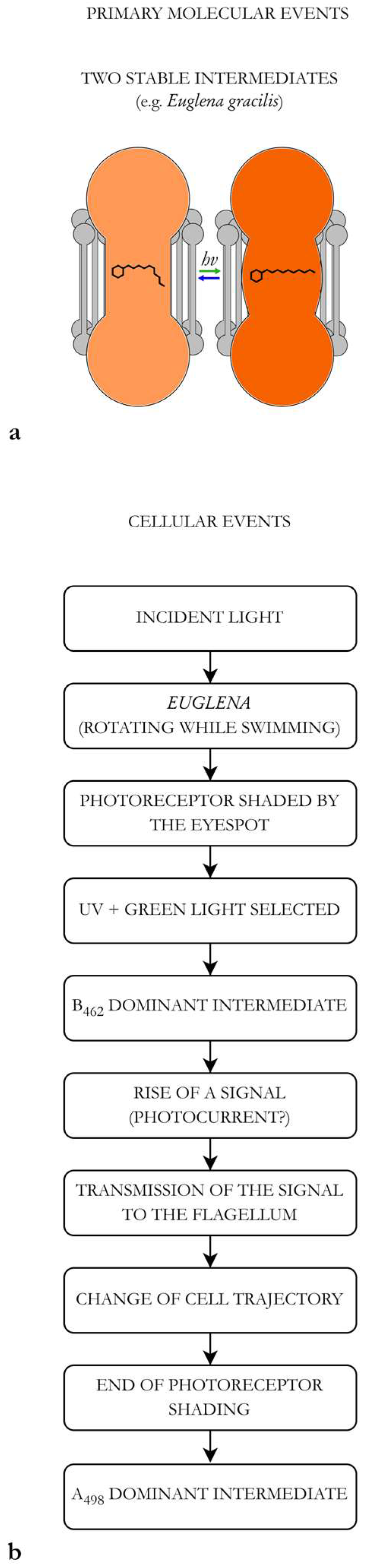
3.1. Structural Characteristics of a Photoreceptive Apparatus (e.g., Euglena gracilis)
3.2. Biophysics and Quantum Limitation of the Photoreceptive Process
3.3. Hypothesis on the Euglena Photoreceptive Mechanism
4. Imaging Visual Process
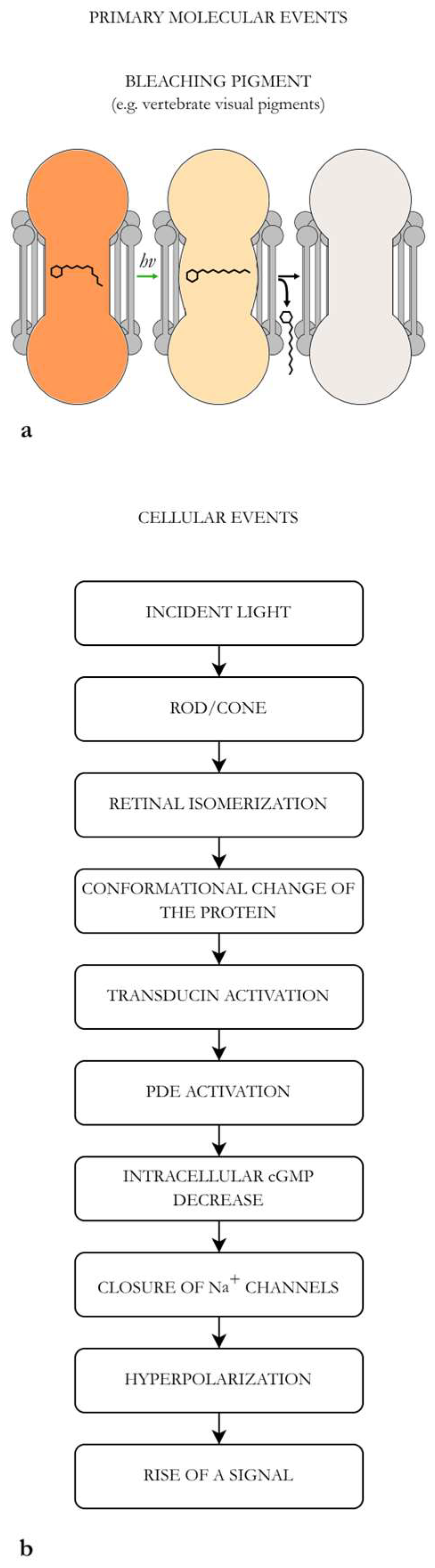
4.1. Structural Characteristics of a Visual Apparatus (e.g., the Human Eye)
4.2. Biophysics and Quantum Limitation of the Retinal Visual Process
4.2.1. The Finite Number of Incoming Photons and Their Random Distribution in Time and Space
4.2.2. False Alarm and Quantum Efficiency
Author Contributions
Funding
Conflicts of Interest
References
- Rose, A. Vision: Human and Electronic; Springer Science & Business Media: New York, NY, USA, 1973. [Google Scholar]
- Oesterhelt, D.; Stoeckenius, W. Rhodopsin-like protein from the purple membrane of Halobacterium halobium. Nat. New Biol. 1971, 233, 149–152. [Google Scholar] [CrossRef] [PubMed]
- Rosati, G.; Verni, F.; Barsanti, L.; Passarelli, V.; Gualtieri, P. Ultrastructure of the apical zone of Euglena gracilis: Photoreceptors and motor apparatus. Electron Microsc. Rev. 1991, 4, 319–342. [Google Scholar] [CrossRef] [PubMed]
- Eakin, R.M. Evolution of photoreceptors. Cold Spring Harb. Symp. Quant. Biol. 1965, 30, 363–370. [Google Scholar] [CrossRef] [PubMed]
- Stirbet, A.; Lazár, D.; Guo, Y.; Govindjee, G. Photosynthesis: Basics, history and modelling. Ann. Bot. 2019, 126, 511–537. [Google Scholar] [CrossRef]
- Stryer, L. Biochemistry, 3rd ed.; W.H. Freeman: New York, NY, USA, 1988. [Google Scholar]
- Gualtieri, P. A biological point of view on photoreception (no-imaging vision) in algae. J. Photochem. Photobiol. B Biol. 1993, 18, 95–97. [Google Scholar] [CrossRef]
- Holmes, R.M.; Victora, M.; Wang, R.F.; Kwiat, P.G. Testing the limits of human vision with quantum states of light: Past, present, and future experiments. In Advanced Photon Counting Techniques XII, 1065903, Proceedings of SPIE—The International Society for Optical Engineering, Orlando, FL, USA, 15–19 April 2018; Campbell, J.C., Itzler, M.A., Eds.; SPIE: Bellingham, WA, USA, 2018; Volume 10659. [Google Scholar]
- Horrigan, D.M.; Makino, C.L. How Do We See? An Introduction to the Biophysics of Visual Transduction. Tutorial Posted on the Biophisical Society Web Page. 2023. Available online: https://www.biophysics.org/ (accessed on 17 November 2023).
- Arshavsky, V.Y.; Burns, M.E. Current understanding of signal amplification in phototransduction. Cell. Logist. 2014, 4, e29390. [Google Scholar] [CrossRef]
- Mayr, E. Toward a New Philosophy of Biology: Observations of an Evolutionist; Belknap Press of Harvard University Press: Cambridge, MA, USA, 1988. [Google Scholar]
- Dawkins, R. Climbing Mount Improbable; Penguin Books Limited: London, UK, 2006. [Google Scholar]
- Uhl, R.; Hegemann, P. Probing visual transduction in a plant cell: Optical recording of rhodopsin-induced structural changes from Chlamydomonas reinhardtii. Biophys. J. 1990, 58, 1295–1302. [Google Scholar] [CrossRef]
- Andruniów, T.; Ferré, N.; Olivucci, M. Structure, initial excited-state relaxation, and energy storage of rhodopsin resolved at the multiconfigurational perturbation theory level. Proc. Natl. Acad. Sci. USA 2004, 101, 17908–17913. [Google Scholar] [CrossRef]
- Barsanti, L.; Evangelista, V.; Passarelli, V.; Frassanito, A.; Gualtieri, P. Fundamental questions and concepts about photoreception and the case of Euglena gracilis. Integr. Biol. Quant. Biosci. Nano Macro 2011, 4, 22–36. [Google Scholar] [CrossRef]
- Barsanti, L.; Coltelli, P.; Evangelista, V.; Passarelli, V.; Frassanito, A.M.; Vesentini, N.; Gualtieri, P. Low-resolution characterization of the 3D structure of the Euglena gracilis photoreceptor. Biochem. Biophys. Res. Commun. 2008, 375, 471–476. [Google Scholar] [CrossRef]
- Gehring, W.J. New perspectives on eye development and the evolution of eyes and photoreceptors. J. Hered. 2005, 96, 171–184. [Google Scholar] [CrossRef] [PubMed]
- Gunkel, M.; Schöneberg, J.; Alkhaldi, W.; Irsen, S.; Noé, F.; Kaupp, U.B.; Al-Amoudi, A. Higher-order architecture of rhodopsin in intact photoreceptors and its implication for phototransduction kinetics. Structure 2015, 23, 628–638. [Google Scholar] [CrossRef] [PubMed]
- Goldberg, A.F.X.; Moritz, O.L.; Williams, D.S. Molecular basis for photoreceptor outer segment architecture. Prog. Retin. Eye Res. 2016, 55, 52–81. [Google Scholar] [CrossRef] [PubMed]
- McFadden, J.; Al-Khalili, J. Life on the Edge: The Coming of Age of Quantum Biology; Crown Publishers: New York, NY, USA, 2014. [Google Scholar]
- Saen-Oon, S.; Lucas, M.F.; Guallar, V. Electron transfer in proteins: Theory, applications and future perspectives. Phys. Chem. Chem. Phys. 2013, 15, 15271–15285. [Google Scholar] [CrossRef] [PubMed]
- Trixler, F. Quantum tunnelling to the origin and evolution of life. Curr. Org. Chem. 2013, 17, 1758–1770. [Google Scholar] [CrossRef] [PubMed]
- Xin, H.; Sim, W.J.; Namgung, B.; Choi, Y.; Li, B.; Lee, L.P. Quantum biological tunnel junction for electron transfer imaging in live cells. Nat. Commun. 2019, 10, 3245. [Google Scholar] [CrossRef] [PubMed]
- Barsanti, L.; Passarelli, V.; Walne, P.L.; Gualtieri, P. In vivo photocycle of the Euglena gracilis photoreceptor. Biophys. J. 1997, 72, 545–553. [Google Scholar] [CrossRef]
- Evangelista, V.; Passarelli, V.; Barsanti, L.; Gualtieri, P. Fluorescence Behavior of Euglena Photoreceptor. Photochem. Photobiol. 2003, 78, 93–97. [Google Scholar] [CrossRef]
- Barsanti, L.; Coltelli, P.; Evangelista, V.; Passarelli, V.; Frassanito, A.M.; Vesentini, N.; Santoro, F.; Gualtieri, P. In Vivo Absorption Spectra of the Two Stable States of the Euglena Photoreceptor Photocycle. Photochem. Photobiol. 2009, 85, 304–312. [Google Scholar] [CrossRef]
- Dusenbery, W.H. Sensory Ecology: How Organisms Acquire and Respond to Information; Freeman: New York, NY, USA, 1992. [Google Scholar]
- Kateriya, S.; Nagel, G.; Bamberg, E.; Hegemann, P. “Vision” in single-celled algae. Physiology 2004, 19, 133–137. [Google Scholar] [CrossRef]
- Böhm, M.; Kreimer, G. Orient in the World with a Single Eye: The Green Algal Eyespot and Phototaxis. In Progress in Botany; Cánovas, F.M., Lüttge, U., Risueño, M.C., Pretzsch, H., Eds.; Springer: Cham, Switzerland, 2020; Volume 82, pp. 259–304. [Google Scholar]
- Ooka, H.; Ishii, T.; Hashimoto, K.; Nakamura, R. Light-induced cell aggregation of Euglena gracilis towards economically feasible biofuel production. RSC Adv. 2014, 4, 20693–20698. [Google Scholar] [CrossRef]
- Mercatelli, R.; Quercioli, F.; Barsanti, L.; Evangelista, V.; Coltelli, P.; Passarelli, V.; Frassanito, A.M.; Gualtieri, P. Intramolecular photo-switching and intermolecular energy transfer as primary photo-events in photoreceptive processes: The case of Euglena gracilis. Biochem. Biophys. Res. Commun. 2009, 385, 176–180. [Google Scholar] [CrossRef] [PubMed]
- Hong, F.T. An electrochemical approach to the design of membrane-based molecular optoelectronic devices. In Molecular Electronics: Biosensors and Biocomputers; Springer Science and Business Media: New York, NY, USA, 1989; pp. 105–114. [Google Scholar]
- Hameroff, S.R. Ultimate Computing. Biomolecular Consciousness and Nanotechnology, 1st ed.; Elsevier Science Pub. Co. Inc.: New York, NY, USA, 1987. [Google Scholar]
- Del Giudice, E.; Doglia, S.; Milani, M. Self-focusing and Ponderomotive Forces of Coherent Electric Waves: A Mechanism for Cytoskeleton Formation and Dynamics. In Proceedings of the Coherent Excitations in Biological Systems, Bad Neuenahr, Germany, 29 November–1 December 1982; Springer: Berlin/Heidelberg, Germany, 1983; pp. 123–127. [Google Scholar]
- Insinna, E.M.; Zaborski, P.; Tuszynski, J. Electrodynamics of microtubular motors: The building blocks of a new model. Biosystems 1996, 39, 187–226. [Google Scholar] [CrossRef] [PubMed]
- Artal, P. The Eye as an Optical Instrument. In Optics in Our Time; Al-Amri, M., El-Gomati, M., Zubairy, M., Eds.; Springer: Cham, Switzerland, 2016. [Google Scholar]
- Kaplan, H.J. Anatomy and function of the eye. Chem. Immunol. Allergy 2007, 92, 4–10. [Google Scholar]
- Bloomer, C.M. Principles of Visual Perception; Herbert Press: London, UK, 1990. [Google Scholar]
- Ripps, H.; Weale, R.A. Color Vision. Annu. Rev. Phycol. 1969, 20, 193–216. [Google Scholar] [CrossRef]
- Boynton, R.M. Human Color Vision; Holt, Rinehart and Winston: New York, NY, USA, 1979. [Google Scholar]
- Begbie, G.H. Seeing and the Eye, 1st ed.; Natural History Press: New York, NY, USA, 1969. [Google Scholar]
- Enoch, J.M.; Tobey, F.L. Vertebrate Photoreceptor Optics; Springer Series in Optical Sciences; Springer: Berlin/Heidelberg, Germany, 1981. [Google Scholar]
- Rose, A. Quantum effects in human vision. Adv. Biol. Med. Phys. 1957, 5, 211–242. [Google Scholar]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Barsanti, L.; Gualtieri, P. Biophysics and Quantum Limitation of Photoreceptive Processes. Encyclopedia 2024, 4, 26-35. https://doi.org/10.3390/encyclopedia4010003
Barsanti L, Gualtieri P. Biophysics and Quantum Limitation of Photoreceptive Processes. Encyclopedia. 2024; 4(1):26-35. https://doi.org/10.3390/encyclopedia4010003
Chicago/Turabian StyleBarsanti, Laura, and Paolo Gualtieri. 2024. "Biophysics and Quantum Limitation of Photoreceptive Processes" Encyclopedia 4, no. 1: 26-35. https://doi.org/10.3390/encyclopedia4010003
APA StyleBarsanti, L., & Gualtieri, P. (2024). Biophysics and Quantum Limitation of Photoreceptive Processes. Encyclopedia, 4(1), 26-35. https://doi.org/10.3390/encyclopedia4010003





