High Expression Levels of the Genes cyclin-A2 and glucocorticoid receptor Are Associated with High-Quality Embryos in Gilthead Sea Bream (Sparus aurata L.)
Abstract
:1. Introduction
2. Materials and Methods
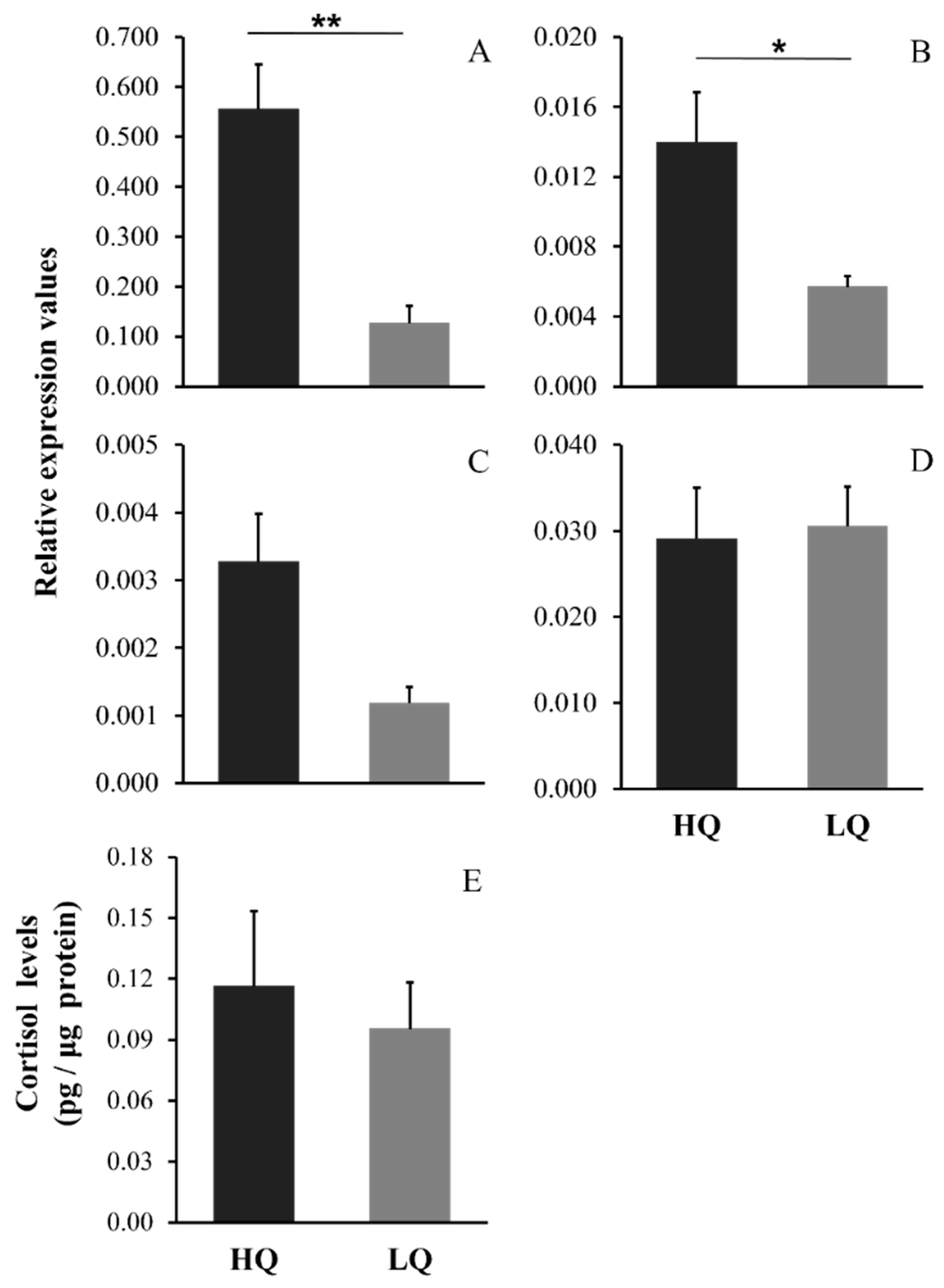
3. Results
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Bromage, N.; Jones, J.; Randall, C.; Thrush, M.; Davies, B.; Springate, J.; Duston, J.; Barker, G. Broodstock management, fecundity, egg quality and the timing of egg production in the rainbow trout (Oncorhynchus mykiss). Aquaculture 1992, 100, 141–166. [Google Scholar] [CrossRef]
- Kjørsvik, E.; Hoehne-Reitan, K.; Reitan, K. Egg and larval quality criteria as predictive measures for juvenile production in turbot (Scophthalmus maximus L.). Aquaculture 2003, 227, 9–20. [Google Scholar] [CrossRef]
- Lahnsteiner, F.; Patarnello, P. The shape of the lipid vesicle is a potential marker for egg quality determination in the gilthead seabream, Sparus aurata, and in the sharpsnout seabream, Diplodus puntazzo. Aquaculture 2005, 246, 423–435. [Google Scholar] [CrossRef]
- Kamler, E. Parent–egg–progeny relationships in teleost fishes: An energetics perspective. Rev. Fish Biol. Fish. 2005, 15, 399–421. [Google Scholar] [CrossRef]
- Carnevali, O.; Mosconi, G.; Cardinali, M.; Meiri, I.; Polzonetti-Magni, A. Molecular components related to egg viability in the gilthead sea bream, Sparus aurata. Mol. Reprod. Dev. 2001, 58, 330–335. [Google Scholar] [CrossRef]
- Carrillo, M.; Bromage, N.; Zanuy, S.; Serrano, R.; Prat, F. The effect of modifications in photoperiod on spawning time, ovarian development and egg quality in the sea bass (Dicentrarchus labrax L.). Aquaculture 1989, 81, 351–365. [Google Scholar] [CrossRef]
- Kjørsvik, E.; Mangor-Jensen, A.; Holmefjord, I. Egg Quality in Fishes: Advance Marine Biology; Elsevier: Amsterdam, The Netherlands, 1990; pp. 71–113. [Google Scholar]
- Mansour, N.; Lahnsteiner, F.; Patzner, R.A. Distribution of lipid droplets is an indicator for egg quality in brown trout, Salmo trutta fario. Aquaculture 2007, 273, 744–747. [Google Scholar] [CrossRef]
- Çoban, D.; Kamacı, H.O.; Suzer, C.; Yıldırım, Ş.; Arda, G.; Korkut, A.Y.; Saka, Ş.; Fırat, K. Effect of some morphometric characteristics on egg quality in common Dentex, Dentex dentex (Linnaeus, 1758). Turk. J. Fish. Aquat. Sci. 2011, 11, 425–431. [Google Scholar] [CrossRef]
- Lahnsteiner, F.; Giménez, G.; Alicia, E. Egg quality determination based on the shape of the lipid vesicle in common dentex Dentex dentex. Aquacult. Res. 2008, 39, 144–149. [Google Scholar] [CrossRef]
- Hansen, Ø.J.; Puvanendran, V. Fertilization success and blastomere morphology as predictors of egg and juvenile quality for domesticated Atlantic cod, Gadus morhua, broodstock. Aquacult. Res. 2010, 41, 1791–1798. [Google Scholar] [CrossRef]
- Kohn, Y.Y.; Symonds, J.E. Evaluation of egg quality parameters as predictors of hatching success and early larval survival in hapuku (Polyprion oxygeneios). Aquaculture 2012, 342, 42–47. [Google Scholar] [CrossRef]
- Rideout, R.; Trippel, E.; Litvak, M. Predicting haddock embryo viability based on early cleavage patterns. Aquaculture 2004, 230, 215–228. [Google Scholar] [CrossRef]
- Aegerter, S.; Jalabert, B.; Bobe, J. Large scale real-time PCR analysis of mRNA abundance in rainbow trout eggs in relationship with egg quality and post-ovulatory ageing. Mol. Reprod. Dev. 2005, 72, 377–385. [Google Scholar] [CrossRef] [PubMed]
- Bonnet, E.; Fostier, A.; Bobe, J. Microarray-based analysis of fish egg quality after natural or controlled ovulation. BMC Genom. 2007, 8, 55. [Google Scholar] [CrossRef] [PubMed]
- Carnevali, O.; Centonze, F.; Brooks, S.; Marota, I.; Sumpter, J.P. Molecular Cloning and Expression of Ovarian Cathepsin D in Seabream, Sparus aurata. Biol. Reprod. 1999, 61, 785–791. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ma, H.; Martin, K.; Dixon, D.; Hernandez, A.G.; Weber, G.M. Transcriptome analysis of egg viability in rainbow trout, Oncorhynchus mykiss. BMC Genom. 2019, 20, 319. [Google Scholar] [CrossRef] [Green Version]
- Mommens, M.; Fernandes, J.M.; Tollefsen, K.E.; Johnston, I.A.; Babiak, I. Profiling of the embryonic Atlantic halibut (Hippoglossus hippoglossus L.) transcriptome reveals maternal transcripts as potential markers of embryo quality. BMC Genom. 2014, 15, 829. [Google Scholar] [CrossRef] [Green Version]
- Bobe, J. Egg quality in fish: Present and future challenges. Anim. Front. 2015, 5, 66–72. [Google Scholar] [CrossRef] [Green Version]
- Darias, M.; Lan Chow Wing, O.; Cahu, C.; Zambonino-Infante, J.-L.; Mazurais, D. Double staining protocol for developing European sea bass (Dicentrarchus labrax) larva. J. Appl. Ichthyol. 2010, 26, 280–285. [Google Scholar] [CrossRef] [Green Version]
- Darias, M.J.; Mazurais, D.; Koumoundouros, G.; Le Gall, M.M.; Huelvan, C.; Desbruyeres, E.; Quazuguel, P.; Cahu, C.L.; Zambonino-Infante, J.L. Imbalanced dietary ascorbic acid alters molecular pathways involved in skeletogenesis of developing European sea bass (Dicentrarchus labrax). Comp. Biochem. Physiol. Part A Mol. Integr. Physiol. 2011, 159, 46–55. [Google Scholar] [CrossRef]
- Marques, C.; Fernández, I.; Rosa, J.; Viegas, M.; Cancela, M.L.; Laizé, V. Spatiotemporal expression and retinoic acid regulation of bone morphogenetic proteins 2, 4 and 16 in Senegalese sole. J. Appl. Ichthyol. 2014, 30, 713–720. [Google Scholar] [CrossRef]
- Nishimoto, S.K.; Waite, J.H.; Nishimoto, M.; Kriwacki, R.W. Structure, activity, and distribution of fish osteocalcin. J. Biol. Chem. 2003, 278, 11843–11848. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Alsop, D.; Vijayan, M.M. Development of the corticosteroid stress axis and receptor expression in zebrafish. Am. J. Physiol.-Regul. Integr. Comp. Physiol. 2008, 294, 711–719. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Feist, G.; Schreck, C.B.; Fitzpatrick, M.S.; Redding, J.M. Sex steroid profiles of coho salmon (Oncorhynchus kisutch) during early development and sexual differentiation. Gen. Comp. Endocrinol. 1990, 80, 299–313. [Google Scholar] [CrossRef]
- Barry, T.P.; Malison, J.A.; Held, J.A.; Parrish, J.J. Ontogeny of the cortisol stress response in larval rainbow trout. Gen. Comp. Endocrinol. 1995, 97, 57–65. [Google Scholar] [CrossRef]
- Sampath-Kumar, R.; Lee, S.; Tan, C.; Munro, A.; Lam, T. Biosynthesis in vivo and excretion of cortisol by fish larvae. J. Exp. Zool. 1997, 277, 337–344. [Google Scholar] [CrossRef]
- Campbell, P.; Pottinger, T.; Sumpter, J. Preliminary evidence that chronic confinement stress reduces the quality of gametes produced by brown and rainbow trout. Aquaculture 1994, 120, 151–169. [Google Scholar] [CrossRef]
- Eriksen, M.; Espmark, Å.; Braastad, B.; Salte, R.; Bakken, M. Long-term effects of maternal cortisol exposure and mild hyperthermia during embryogeny on survival, growth and morphological anomalies in farmed Atlantic salmon Salmo salar offspring. J. Fish Biol. 2007, 70, 462–473. [Google Scholar] [CrossRef]
- Nesan, D.; Vijayan, M.M. Embryo exposure to elevated cortisol level leads to cardiac performance dysfunction in zebrafish. Mol. Cell. Endocrinol. 2012, 363, 85–91. [Google Scholar] [CrossRef]
- Skaalsvik, T.H.; Bolla, S.L.; Thornqvist, P.-O.; Babiak, I. Quantitative characteristics of Atlantic halibut (Hippoglossus hippoglossus L.) egg quality throughout the reproductive season. Theriogenology 2015, 83, 38–47. [Google Scholar] [CrossRef]
- Faustino, M.; Power, D. Osteologic development of the viscerocranial skeleton in sea bream: Alternative ossification strategies in teleost fish. J. Fish Biol. 2001, 58, 537–572. [Google Scholar] [CrossRef]
- Koumoundouros, G.; Oran, G.; Divanach, P.; Stefanakis, S.; Kentouri, M. The opercular complex deformity in intensive gilthead sea bream (Sparus aurata L.) larviculture. Moment of apparition and description. Aquaculture 1997, 156, 165–177. [Google Scholar] [CrossRef]
- Polo, A.; Yufera, M.; Pascual, E. Effects of temperature on egg and larval development of Sparus aurata L. Aquaculture 1991, 92, 367–375. [Google Scholar] [CrossRef]
- Divanach, P. Contribution a la Connaissance de la Biologie et de l’ Elevange de 6 Sparides Mediterranees: Sparus aurata, Diplodus sargus, Diplodus vulgaris, Diplodus annularis, Lithignathus mormyrous, Puntazzi pintazzo (Poissons telesteens); Universite des Sciences et Techniques du Languedoc, Station de Biologie: Saint-Hippolyte, QC, Canada, 1985. [Google Scholar]
- Koumoundouros, G. Morpho-anatomical abnormalities in Mediterranean marine aquaculture. Recent Adv. Aquac. Res. 2010, 66, 125–148. [Google Scholar]
- Schmittgen, T.D.; Livak, K.J. Analyzing real-time PCR data by the comparative CT method. Nat. Protoc. 2008, 3, 1101–1108. [Google Scholar] [CrossRef]
- De Jesus, E.G.T.; Hirano, T. Changes in whole body concentrations of cortisol, thyroid hormones, and sex steroids during early development of the chum salmon Oncorhynchus keta. Gen. Comp. Endocrinol. 1992, 85, 55–61. [Google Scholar] [CrossRef]
- Bradford, M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef]
- Sokal, R.R.; Rohlf, F.J. Biometry: The Principles and Practice of Statistics in Biological Research, 2nd ed.; W. H. Freeman and Company: San Francisco, CA, USA, 1981; p. 859. [Google Scholar]
- Fernández, C.G.; Roufidou, C.; Antonopoulou, E.; Sarropoulou, E. Expression of developmental-stage-specific genes in the gilthead sea bream Sparus aurata L. Mar. Biotechnol. 2013, 15, 313–320. [Google Scholar] [CrossRef]
- Mommens, M.; Fernandes, J.M.; Bizuayehu, T.T.; Bolla, S.L.; Johnston, I.A.; Babiak, I. Maternal gene expression in Atlantic halibut (Hippoglossus hippoglossus L.) and its relation to egg quality. BMC Res. Notes 2010, 3, 138. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Weber, G.M.; Birkett, J.; Martin, K.; Dixon, D.; Gao, G.; Leeds, T.D.; Vallejo, R.L.; Ma, H. Comparisons among rainbow trout, Oncorhynchus mykiss, populations of maternal transcript profile associated with egg viability. BMC Genom. 2021, 22, 448. [Google Scholar] [CrossRef] [PubMed]
- Nesan, D.; Kamkar, M.; Burrows, J.; Scott, I.C.; Marsden, M.; Vijayan, M.M. Glucocorticoid receptor signaling is essential for mesoderm formation and muscle development in zebrafish. Endocrinology 2012, 153, 1288–1300. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pikulkaew, S.; Benato, F.; Celeghin, A.; Zucal, C.; Skobo, T.; Colombo, L.; Valle, L.D. The knockdown of maternal glucocorticoid receptor mRNA alters embryo development in zebrafish. Dev. Dyn. 2011, 240, 874–889. [Google Scholar] [CrossRef] [PubMed]
- Pikulkaew, S.; De Nadai, A.; Belvedere, P.; Colombo, L.; Dalla Valle, L. Expression analysis of steroid hormone receptor mRNAs during zebrafish embryogenesis. Gen. Comp. Endocrinol. 2010, 165, 215–220. [Google Scholar] [CrossRef] [PubMed]
- Boglione, C.; Gisbert, E.; Gavaia, P.; Witten, P.E.; Moren, M.; Fontagné, S.; Koumoundouros, G. Skeletal anomalies in reared E uropean fish larvae and juveniles. Part 2: Main typologies, occurrences and causative factors. Rev. Aquac. 2013, 5, 121–167. [Google Scholar] [CrossRef] [Green Version]
- Loizides, M.; Georgiou, A.; Somarakis, S.; Witten, P.; Koumoundouros, G. A new type of lordosis and vertebral body compression in Gilthead sea bream, Sparus aurata L.: Aetiology, anatomy and consequences for survival. J. Fish Dis. 2014, 37, 949–957. [Google Scholar] [CrossRef] [Green Version]
- Faustino, M.; Power, D. Development of osteological structures in the sea bream: Vertebral column and caudal fin complex. J. Fish Biol. 1998, 52, 11–22. [Google Scholar] [CrossRef]
- Walker, M.; Kimmel, C. A two-color acid-free cartilage and bone stain for zebrafish larvae. Biotech. Histochem. 2007, 82, 23–28. [Google Scholar] [CrossRef]
| Tank | n-Fert | Abn | Normal | Group | Mean n-Fert | Mean Abn | Mean Normal |
|---|---|---|---|---|---|---|---|
| T1 | 3 | 4 | 93 | HQ | 1.3 ± 1.4 a | 2.8 ± 1.0 a | 95.8 ± 2.3 a |
| T2 | 3 | 4 | 93 | ||||
| T3 | 1 | 2 | 97 | ||||
| T4 | 1 | 3 | 96 | ||||
| T5 | 0 | 2 | 98 | ||||
| T6 | 0 | 2 | 98 | ||||
| T7 | 3 | 13 | 84 | LQ | 3.5 ± 1.4 b | 12.3 ± 1.6 b | 84.2 ± 0.8 b |
| T8 | 6 | 10 | 84 | ||||
| T9 | 4 | 11 | 85 | ||||
| T10 | 3 | 12 | 85 | ||||
| T11 | 3 | 14 | 83 | ||||
| T12 | 2 | 14 | 84 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Georgiou, A.N.; Georga, I.; Stamopoulou, A.; Tzokas, K.; Koumoundouros, G.; Flytzanis, C.N. High Expression Levels of the Genes cyclin-A2 and glucocorticoid receptor Are Associated with High-Quality Embryos in Gilthead Sea Bream (Sparus aurata L.). Aquac. J. 2022, 2, 51-58. https://doi.org/10.3390/aquacj2020005
Georgiou AN, Georga I, Stamopoulou A, Tzokas K, Koumoundouros G, Flytzanis CN. High Expression Levels of the Genes cyclin-A2 and glucocorticoid receptor Are Associated with High-Quality Embryos in Gilthead Sea Bream (Sparus aurata L.). Aquaculture Journal. 2022; 2(2):51-58. https://doi.org/10.3390/aquacj2020005
Chicago/Turabian StyleGeorgiou, Anastasia N., Ioanna Georga, Andriana Stamopoulou, Kostas Tzokas, George Koumoundouros, and Constantin N. Flytzanis. 2022. "High Expression Levels of the Genes cyclin-A2 and glucocorticoid receptor Are Associated with High-Quality Embryos in Gilthead Sea Bream (Sparus aurata L.)" Aquaculture Journal 2, no. 2: 51-58. https://doi.org/10.3390/aquacj2020005






