Toxicity and Teratogenic Potential of Piplartine from Piper tuberculatum Jacq. during Embryonic Development in Mice (Mus musculus)
Abstract
1. Introduction
2. Results
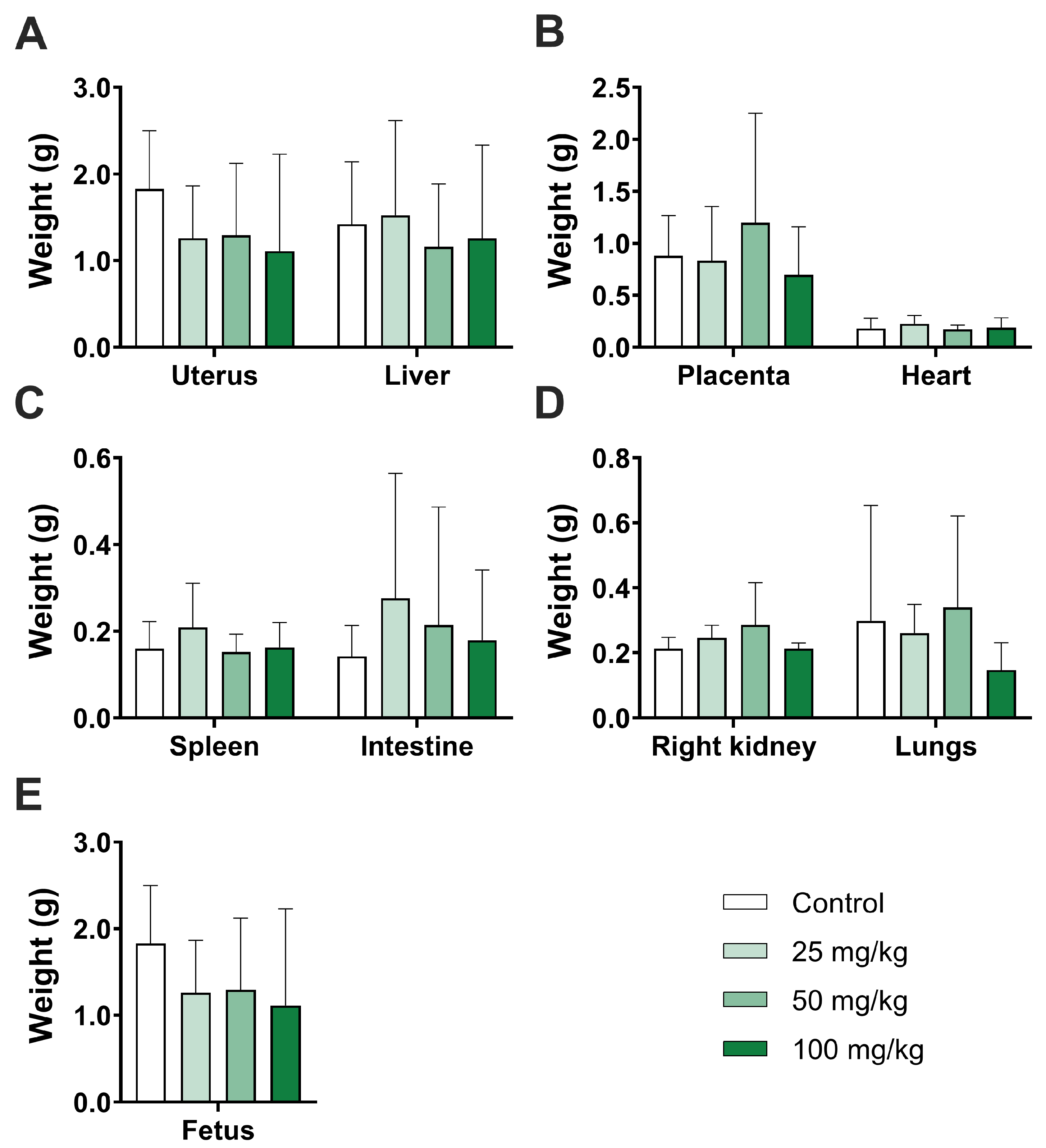
2.1. Maternal and Embryo–Fetal Toxicity
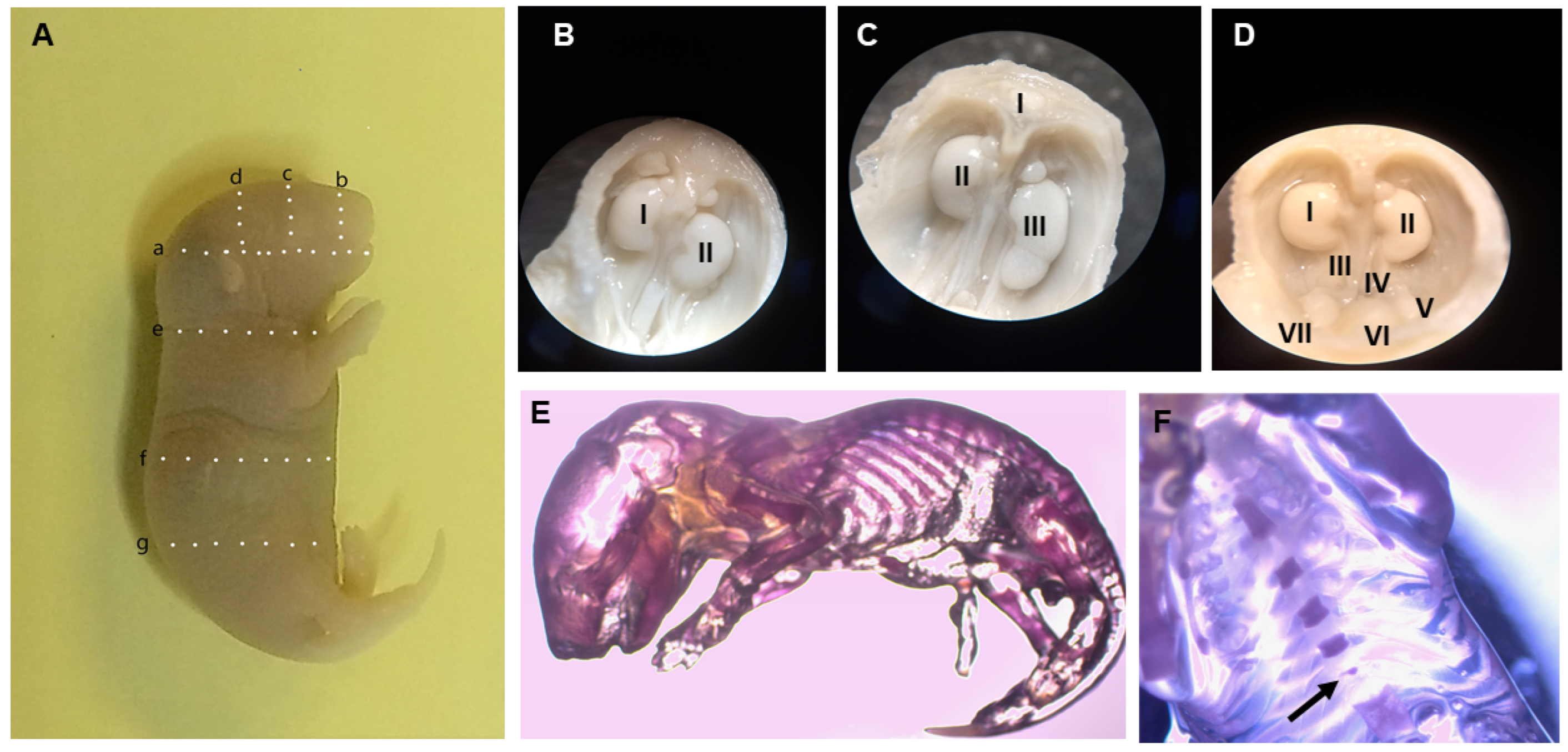
2.2. Fetal Visceral and Skeletal Anomalies
2.3. Hematological and Biochemical Analyses
2.4. DNA Fragmentation and Cell Cycle
3. Discussion
3.1. Maternal Toxicity
3.2. Embryo–Fetal Toxicity
3.3. Fetal Visceral Analysis
3.4. Fetal Skeletal Analysis
3.5. Hematological and Biochemical Analyses
3.6. DNA Fragmentation and Cell Cycle
3.7. Limitations
4. Materials and Methods
4.1. Piplartine Extraction and Isolation
4.2. Animals
4.3. Animal Mating
4.4. Maternal Toxicity
4.5. Laparotomy
4.6. Fetal Anomalies
4.7. Hematological and Biochemical Analysis
4.8. DNA Fragmentation and Cell Cycle
4.9. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Bezerra, D.P.; Pessoa, C.; De Moraes, M.O.; Saker-Neto, N.; Silveira, E.R.; Costa-Lotufo, L.V. Overview of the Therapeutic Potential of Piplartine (Piperlongumine). Eur. J. Pharm. Sci. 2013, 48, 453–463. [Google Scholar] [CrossRef]
- Huang, C.-H.; Wang, S.-C.; Chen, I.-C.; Chen, Y.-T.; Liu, P.-L.; Fang, S.-H.; Huang, S.-P.; Yeh, H.-C.; Liu, C.-C.; Lee, P.-Y.; et al. Protective Effect of Piplartine against LPS-Induced Sepsis through Attenuating the MAPKs/NF-κB Signaling Pathway and NLRP3 Inflammasome Activation. Pharmaceuticals 2021, 14, 588. [Google Scholar] [CrossRef]
- Shi, J.; Xia, Y.; Wang, H.; Yi, Z.; Zhang, R.; Zhang, X. Piperlongumine Is an NLRP3 Inhibitor With Anti-Inflammatory Activity. Front. Pharmacol. 2022, 12, 818326. [Google Scholar] [CrossRef]
- Meegan, M.J.; Nathwani, S.; Twamley, B.; Zisterer, D.M.; O’Boyle, N.M. Piperlongumine (Piplartine) and Analogues: Antiproliferative Microtubule-Destabilising Agents. Eur. J. Med. Chem. 2017, 125, 453–463. [Google Scholar] [CrossRef]
- Da Nóbrega, F.; Ozdemir, O.; Nascimento Sousa, S.; Barboza, J.; Turkez, H.; De Sousa, D. Piplartine Analogues and Cytotoxic Evaluation against Glioblastoma. Molecules 2018, 23, 1382. [Google Scholar] [CrossRef]
- Burci, L.M.; Pereira, I.T.; Da Silva, L.M.; Rodrigues, R.V.; Facundo, V.A.; Militão, J.S.L.T.; Santos, A.R.S.; Marques, M.C.A.; Baggio, C.H.; Werner, M.F.D.P. Antiulcer and Gastric Antisecretory Effects of Dichloromethane Fraction and Piplartine Obtained from Fruits of Piper tuberculatum Jacq. in Rats. J. Ethnopharmacol. 2013, 148, 165–174. [Google Scholar] [CrossRef]
- Piska, K.; Gunia-Krzyżak, A.; Koczurkiewicz, P.; Wójcik-Pszczoła, K.; Pękala, E. Piperlongumine (Piplartine) as a Lead Compound for Anticancer Agents—Synthesis and Properties of Analogues: A Mini-Review. Eur. J. Med. Chem. 2018, 156, 13–20. [Google Scholar] [CrossRef]
- Henrique, T.; Zanon, C.D.F.; Girol, A.P.; Stefanini, A.C.B.; Contessoto, N.S.D.A.; Da Silveira, N.J.F.; Bezerra, D.P.; Silveira, E.R.; Barbosa-Filho, J.M.; Cornélio, M.L.; et al. Biological and Physical Approaches on the Role of Piplartine (Piperlongumine) in Cancer. Sci. Rep. 2020, 10, 22283. [Google Scholar] [CrossRef]
- Parama, D.; Rana, V.; Girisa, S.; Verma, E.; Daimary, U.D.; Thakur, K.K.; Kumar, A.; Kunnumakkara, A.B. The Promising Potential of Piperlongumine as an Emerging Therapeutics for Cancer. Explor. Target. Anti-Tumor Ther. 2021, 2, 323. [Google Scholar] [CrossRef]
- Moraes, J.D.; Nascimento, C.; Lopes, P.O.M.V.; Nakano, E.; Yamaguchi, L.F.; Kato, M.J.; Kawano, T. Schistosoma mansoni: In Vitro Schistosomicidal Activity of Piplartine. Exp. Parasitol. 2011, 127, 357–364. [Google Scholar] [CrossRef]
- Mengarda, A.C.; Mendonça, P.S.; Morais, C.S.; Cogo, R.M.; Mazloum, S.F.; Salvadori, M.C.; Teixeira, F.S.; Morais, T.R.; Antar, G.M.; Lago, J.H.G.; et al. Antiparasitic Activity of Piplartine (Piperlongumine) in a Mouse Model of Schistosomiasis. Acta Trop. 2020, 205, 105350. [Google Scholar] [CrossRef]
- Araújo-Vilges, K.M.D.; Oliveira, S.V.D.; Couto, S.C.P.; Fokoue, H.H.; Romero, G.A.S.; Kato, M.J.; Romeiro, L.A.S.; Leite, J.R.S.A.; Kuckelhaus, S.A.S. Effect of Piplartine and Cinnamides on Leishmania amazonensis, Plasmodium falciparum and on Peritoneal Cells of Swiss Mice. Pharm. Biol. 2017, 55, 1601–1607. [Google Scholar] [CrossRef]
- Ticona, J.C.; Bilbao-Ramos, P.; Flores, N.; Dea-Ayuela, M.A.; Bolás-Fernández, F.; Jiménez, I.A.; Bazzocchi, I.L. (E)-Piplartine Isolated from Piper pseudoarboreum, a Lead Compound against Leishmaniasis. Foods 2020, 9, 1250. [Google Scholar] [CrossRef]
- Da Silva, M.A.; Fokoue, H.H.; Fialho, S.N.; Dos Santos, A.P.D.A.; Rossi, N.R.D.L.P.; Gouveia, A.D.J.; Ferreira, A.S.; Passarini, G.M.; Garay, A.F.G.; Alfonso, J.J.; et al. Antileishmanial Activity Evaluation of a Natural Amide and Its Synthetic Analogs against Leishmania (V.) braziliensis: An Integrated Approach in Vitro and in Silico. Parasitol. Res. 2021, 120, 2199–2218. [Google Scholar] [CrossRef]
- Mgbeahuruike, E.E.; Stålnacke, M.; Vuorela, H.; Holm, Y. Antimicrobial and Synergistic Effects of Commercial Piperine and Piperlongumine in Combination with Conventional Antimicrobials. Antibiotics 2019, 8, 55. [Google Scholar] [CrossRef]
- Mgbeahuruike, E.; Fyhrquist, P.; Vuorela, H.; Julkunen-Tiitto, R.; Holm, Y. Alkaloid-Rich Crude Extracts, Fractions and Piperamide Alkaloids of Piper Guineense Possess Promising Antibacterial Effects. Antibiotics 2018, 7, 98. [Google Scholar] [CrossRef]
- Kumar, J.U.; Shankaraiah, G.; Kumar, R.S.C.; Pitke, V.V.; Rao, G.T.; Poornima, B.; Babu, K.S.; Sreedhar, A.S. Synthesis, Anticancer, and Antibacterial Activities of Piplartine Derivatives on Cell Cycle Regulation and Growth Inhibition. J. Asian Nat. Prod. Res. 2013, 15, 658–669. [Google Scholar] [CrossRef]
- Petrovska, B. Historical Review of Medicinal Plants′ Usage. Pharmacogn. Rev. 2012, 6, 1–5. [Google Scholar] [CrossRef]
- Valli, M.; Russo, H.M.; Bolzani, V.S. The Potential Contribution of the Natural Products from Brazilian Biodiversity to Bioeconomy. An. Acad. Bras. Ciênc. 2018, 90, 763–778. [Google Scholar] [CrossRef]
- Campelo, Y.; Ombredane, A.; Vasconcelos, A.; Albuquerque, L.; Moreira, D.; Plácido, A.; Rocha, J.; Hilarion Fokoue, H.; Yamaguchi, L.; Mafud, A.; et al. Structure–Activity Relationship of Piplartine and Synthetic Analogues against Schistosoma mansoni and Cytotoxicity to Mammalian Cells. Int. J. Mol. Sci. 2018, 19, 1802. [Google Scholar] [CrossRef]
- Lad, N.P.; Kulkarni, S.; Sharma, R.; Mascarenhas, M.; Kulkarni, M.R.; Pandit, S.S. Piperlongumine Derived Cyclic Sulfonamides (Sultams): Synthesis and in Vitro Exploration for Therapeutic Potential against HeLa Cancer Cell Lines. Eur. J. Med. Chem. 2017, 126, 870–878. [Google Scholar] [CrossRef]
- Bezerra, D.P.; Moura, D.J.; Rosa, R.M.; De Vasconcellos, M.C.; E Silva, A.C.R.; De Moraes, M.O.; Silveira, E.R.; Lima, M.A.S.; Henriques, J.A.P.; Costa-Lotufo, L.V.; et al. Evaluation of the Genotoxicity of Piplartine, an Alkamide of Piper tuberculatum, in Yeast and Mammalian V79 Cells. Mutat. Res. /Genet. Toxicol. Environ. Mutagen. 2008, 652, 164–174. [Google Scholar] [CrossRef]
- Santoro, A.; Chianese, R.; Troisi, J.; Richards, S.; Nori, S.L.; Fasano, S.; Guida, M.; Plunk, E.; Viggiano, A.; Pierantoni, R.; et al. Neuro-Toxic and Reproductive Effects of BPA. Curr. Neuropharmacol. 2019, 17, 1109–1132. [Google Scholar] [CrossRef]
- Phipps, K.R.; Danielewska-Nikiel, B.; Mushonganono, J.; Baldwin, N. Reproductive and Developmental Toxicity Screening Study of an Acetone Extract of Rosemary. Regul. Toxicol. Pharmacol. 2021, 120, 104840. [Google Scholar] [CrossRef]
- Cummings, A.M. Toxicological Mechanisms of Implantation Failure. Toxicol. Sci. 1990, 15, 571–579. [Google Scholar] [CrossRef]
- Taylor, P. Practical Teratology; Academic Press: London, UK, 1986; ISBN 978-0-12-683860-2. [Google Scholar]
- Fofaria, N.M.; Qhattal, H.S.S.; Liu, X.; Srivastava, S.K. Nanoemulsion Formulations for Anti-Cancer Agent Piplartine—Characterization, Toxicological, Pharmacokinetics and Efficacy Studies. Int. J. Pharm. 2016, 498, 12–22. [Google Scholar] [CrossRef]
- Bezerra, D.P.; Castro, F.O.D.; Alves, A.P.N.N.; Pessoa, C.; Moraes, M.O.D.; Silveira, E.R.; Lima, M.A.S.; Elmiro, F.J.M.; Alencar, N.M.N.D.; Mesquita, R.O.; et al. In Vitro and in Vivo Antitumor Effect of 5-FU Combined with Piplartine and Piperine. J. Appl. Toxicol. 2008, 28, 156–163. [Google Scholar] [CrossRef]
- Branzei, D.; Foiani, M. Regulation of DNA Repair throughout the Cell Cycle. Nat. Rev. Mol. Cell Biol. 2008, 9, 297–308. [Google Scholar] [CrossRef]
- Wendler, A.; Wehling, M. The Translatability of Animal Models for Clinical Development: Biomarkers and Disease Models. Curr. Opin. Pharmacol. 2010, 10, 601–606. [Google Scholar] [CrossRef]
- Leenaars, C.H.C.; Kouwenaar, C.; Stafleu, F.R.; Bleich, A.; Ritskes-Hoitinga, M.; De Vries, R.B.M.; Meijboom, F.L.B. Animal to Human Translation: A Systematic Scoping Review of Reported Concordance Rates. J. Transl. Med. 2019, 17, 223. [Google Scholar] [CrossRef]
- Mukherjee, P.; Roy, S.; Ghosh, D.; Nandi, S.K. Role of Animal Models in Biomedical Research: A Review. Lab. Anim. Res. 2022, 38, 18. [Google Scholar] [CrossRef]
- Von Keutz, E. Specific Toxicity Tests for Neurotoxicity, Immunotoxicity, Allergy, Irritation, Reprotoxicity, and Carcinogenicity. In Regulatory Toxicology; Reichl, F.-X., Schwenk, M., Eds.; Springer International Publishing: Cham, Switzerland, 2021; pp. 129–138. ISBN 978-3-030-57498-7. [Google Scholar]
- Dasharathy, S.; Arjunan, S.; Maliyur Basavaraju, A.; Murugasen, V.; Ramachandran, S.; Keshav, R.; Murugan, R. Mutagenic, Carcinogenic, and Teratogenic Effect of Heavy Metals. Evid. -Based Complement. Altern. Med. 2022, 2022, 8011953. [Google Scholar] [CrossRef]
- Li, G.; Li, D.; Rao, H.; Liu, X. Potential Neurotoxicity, Immunotoxicity, and Carcinogenicity Induced by Metribuzin and Tebuconazole Exposure in Earthworms (Eisenia fetida) Revealed by Transcriptome Analysis. Sci. Total Environ. 2022, 807, 150760. [Google Scholar] [CrossRef] [PubMed]
- Tuntland, T.; Ethell, B.; Kosaka, T.; Blasco, F.; Zang, R.X.; Jain, M.; Gould, T.; Hoffmaster, K. Implementation of Pharmacokinetic and Pharmacodynamic Strategies in Early Research Phases of Drug Discovery and Development at Novartis Institute of Biomedical Research. Front. Pharmacol. 2014, 5, 174. [Google Scholar] [CrossRef]
- Palmer, M.E.; Andrews, L.J.; Abbey, T.C.; Dahlquist, A.E.; Wenzler, E. The Importance of Pharmacokinetics and Pharmacodynamics in Antimicrobial Drug Development and Their Influence on the Success of Agents Developed to Combat Resistant Gram Negative Pathogens: A Review. Front. Pharmacol. 2022, 13, 888079. [Google Scholar] [CrossRef]
- Ball, K.; Bruin, G.; Escandón, E.; Funk, C.; Pereira, J.N.S.; Yang, T.-Y.; Yu, H. Characterizing the Pharmacokinetics and Biodistribution of Therapeutic Proteins: An Industry White Paper. Drug Metab. Dispos. 2022, 50, 858–866. [Google Scholar] [CrossRef] [PubMed]
- Stielow, M.; Witczyńska, A.; Kubryń, N.; Fijałkowski, Ł.; Nowaczyk, J.; Nowaczyk, A. The Bioavailability of Drugs—The Current State of Knowledge. Molecules 2023, 28, 8038. [Google Scholar] [CrossRef] [PubMed]
- Melo, A.; Alves, M. Piper (Piperaceae) in Eastern Northeast Brazil. Phytotaxa 2021, 505, 1–38. [Google Scholar] [CrossRef]
- Kopf, R.; Lorenz, D.; Salewski, E. Der Einfluß von Thalidomid auf die Fertilität von Ratten im Generationsversuch über zwei Generationen. Naunyn-Schmiedebergs Arch. 1964, 247, 121–135. [Google Scholar] [CrossRef]
- Staples, R.E.; Schnell, V.L. Refinements in Rapid Clearing Technic in the KOH-Alizarin Red S Method for Fetal Bone. Stain Technol. 1964, 39, 61–63. [Google Scholar]
- Wilson, J.G. Methods for Administering Agents and Detecting Malformations in Experimental Animals. In Teratology: Principals and Techniques; Wilson, J.G., Warkany, J., Eds.; University of Chicago Press: Chicago, IL, USA, 1965; pp. 262–277. [Google Scholar]
- Barrow, M.V.; Taylor, W.J. A Rapid Method for Detecting Malformations in Rat Fetuses. J. Morphol. 1969, 127, 291–305. [Google Scholar] [CrossRef] [PubMed]
- Riond, B.; Weissenbacher, S.; Hofmann-Lehmann, R.; Lutz, H. Performance Evaluation of the S Ysmex Poc H-100i V D Iff Hematology Analyzer for Analysis of Canine, Feline, Equine, and Bovine Blood. Vet. Clin. Pathol. 2011, 40, 484–495. [Google Scholar] [CrossRef] [PubMed]
- Yeung, A.W.K.; Souto, E.B.; Durazzo, A.; Lucarini, M.; Novellino, E.; Tewari, D.; Wang, D.; Atanasov, A.G.; Santini, A. Big Impact of Nanoparticles: Analysis of the Most Cited Nanopharmaceuticals and Nanonutraceuticals Research. Curr. Res. Biotechnol. 2020, 2, 53–63. [Google Scholar] [CrossRef]
- Giacone, D.V.; Dartora, V.F.M.C.; De Matos, J.K.R.; Passos, J.S.; Miranda, D.A.G.; De Oliveira, E.A.; Silveira, E.R.; Costa-Lotufo, L.V.; Maria-Engler, S.S.; Lopes, L.B. Effect of Nanoemulsion Modification with Chitosan and Sodium Alginate on the Topical Delivery and Efficacy of the Cytotoxic Agent Piplartine in 2D and 3D Skin Cancer Models. Int. J. Biol. Macromol. 2020, 165, 1055–1065. [Google Scholar] [CrossRef] [PubMed]

| Variable | Group | p-Value | |||
|---|---|---|---|---|---|
| Control | 25 mg/kg | 50 mg/kg | 100 mg/kg | ||
| Organ weight | |||||
| Uterus | 1.8 ± 0.6 | 1.2 ± 0.6 | 1.2 ± 0.8 | 1.1 ± 1.1 | 0.8872 |
| (n = 8) | (n = 6) | (n = 7) | (n = 3) | ||
| Heart | 0.1 ± 0.9 | 0.2 ± 0.1 | 0.1 ± 0.0 | 0.1 ± 0.1 | 0.9983 |
| (n = 8) | (n = 8) | (n = 8) | (n = 7) | ||
| Liver | 1.4 ± 0.7 | 1.5 ± 1.0 | 1.1 ± 0.7 | 1.2 ± 1.0 | 0.9864 |
| (n = 8) | (n = 8) | (n = 8) | (n = 7) | ||
| Spleen | 0.1 ± 0.1 | 0.2 ± 0.1 | 0.1 ± 0.0 | 0.1 ± 0.0 | 0.8695 |
| (n = 8) | (n = 7) | (n = 8) | (n = 7) | ||
| Intestine | 0.1 ± 0.1 | 0.2 ± 0.2 | 0.2 ± 0.2 | 0.1 ± 0.1 | 0.9473 |
| (n = 8) | (n = 7) | (n = 8) | (n = 6) | ||
| Right kidney | 0.2 ± 0.0 | 0.2 ± 0.0 | 0.2 ± 0.1 | 0.2 ± 0.0 | 0.4092 |
| (n = 7) | (n = 7) | (n = 8) | (n = 7) | ||
| Lungs | 0.2 ± 0.3 | 0.2 ± 0.0 | 0.3 ± 0.2 | 0.1 ± 0.1 | 0.9346 |
| (n = 9) | (n = 7) | (n = 8) | (n = 7) | ||
| Maternal weight (g) | |||||
| Initial | 31.0 ± 5.8 | 31.7 ± 4.4 | 30.3 ± 2.2 | 32.0 ± 2.2 | 0.9931 |
| (n = 10) | (n = 10) | (n = 9) | (n = 7) | ||
| Final | 45.6 ± 8.8 | 44.4 ± 8.3 | 46.6 ± 5.2 | 46.8 ± 8.9 | 0.9965 |
| (n = 10) | (n = 9) | (n = 9) | (n = 7) | ||
| Maternal weight gain (g) | |||||
| 6th day–Day 0 | 31.1 ± 5.9 | 31.0 ± 4.1 | 30.4 ± 2.1 | 31.8 ± 2.2 | 0.9970 |
| 9th day–6th day | 31.9 ± 6.6 | 32.7 ± 3.6 | 31.7 ± 1.7 | 32.5 ± 3.3 | 0.9983 |
| 12th day–9th day | 34.8 ± 6.4 | 35.4 ± 5.7 | 34.9 ± 2.5 | 35.3 ± 2.5 | 0.9997 |
| 15th day–12th day | 39.1 ± 6.9 | 41.3 ± 7.1 | 39.1 ± 4.0 | 41.0 ± 3.5 | 0.9889 |
| 18th day–15th day | 45.6 ± 9.9 | 45.4 ± 10.3 | 46.3 ± 5.4 | 48.5 ± 6.2 | 0.9946 |
| Δ weight | 11.9 ± 6.85 | 17.7 ± 8.01 | 13.7 ± 5.28 | 12.2 ± 7.78 | 0.9296 |
| Variable | Group | p-Value | |||
|---|---|---|---|---|---|
| Control | 25 mg/kg | 50 mg/kg | 100 mg/kg | ||
| Implantations | 16.7 ± 4.7 | 18.3 ± 7.5 | 20.2 ± 5.9 | 18.8 ± 9.7 | 0.9296 |
| (n = 10) | (n = 10) | (n = 9) | (n = 7) | ||
| Resorptions | 8.4 ± 3.7 | 10.7 ± 4.6 | 12.3 ± 3.5 | 10.6 ± 4.1 | 0.9108 |
| (n = 10) | (n = 9) | (n = 9) | (n = 6) | ||
| Placental weight | 0.8 ± 0.3 | 0.8 ± 0.5 | 1.1 ± 1.0 | 0.6 ± 0.4 | 0.9710 |
| (n = 8) | (n = 8) | (n = 7) | (n = 3) | ||
| Number of fetuses | 7.8 ± 2.5 | 8.6 ± 4.3 | 8.1 ± 3.3 | 9.7 ± 5.3 | 0.9883 |
| (n = 10) | (n = 10) | (n = 9) | (n = 7) | ||
| Fetus weight | 1.6 ± 0.8 | 1.3 ± 0.2 | 1.3 ± 0.1 | 1.5 ± 0.2 | 0.9694 |
| (n = 10) | (n = 9) | (n = 7) | (n = 4) | ||
| Visceral Anomaly | Present | Group | |||
|---|---|---|---|---|---|
| Control | 25 mg/kg | 50 mg/kg | 100 mg/kg | ||
| Palate | Yes | 1 | 3 | 0 | 0 |
| No | 55 | 56 | 45 | 61 | |
| Heart ventricle | Yes | 1 | 3 | 0 | 1 |
| No | 55 | 56 | 44 | 60 | |
| Heart atrium | Yes | 0 | 5 | 0 | 2 |
| No | 56 | 54 | 44 | 59 | |
| Intestinal loop | Yes | 0 | 1 | 0 | 0 |
| No | 57 | 58 | 44 | 61 | |
| Kidney | Yes | 0 | 2 | 0 | 1 |
| No | 57 | 57 | 44 | 60 | |
| Adrenal | Yes | 0 | 0 | 0 | 2 |
| No | 57 | 59 | 44 | 59 | |
| Thymus | Yes | 0 | 0 | 2 | 0 |
| No | 56 | 59 | 42 | 61 | |
| Lung | Yes | 0 | 0 | 1 | 0 |
| No | 56 | 59 | 43 | 61 | |
| Liver | Yes | 0 | 0 | 1 | 0 |
| No | 56 | 59 | 44 | 61 | |
| Skeletal Anomaly | Present | Dose | |||
|---|---|---|---|---|---|
| Control | 25 mg/kg | 50 mg/kg | 100 mg/kg | ||
| Sternebrae | Yes | 0 | 9 | 6 | 4 |
| No | 32 | 18 | 12 | 13 | |
| Hematological Analysis | Control | 25 mg/kg | 50 mg/kg | 100 mg/kg | p-Value |
|---|---|---|---|---|---|
| RBC (×106/µL) | (n = 10) 8.75 ± 1.16 | (n = 10) 9.40 ± 1.19 | (n = 9) 8.79 ± 0.49 | (n = 7) 9.28 ± 1.60 | 0.5649 |
| HGB | 13.16 ± 1.71 | 13.86 ± 1.79 | 13.29 ± 0.90 | 13.80 ± 2.30 | 0.334 |
| (g/dL) | |||||
| HCT | 34.32 ± 4.39 | 36.14 ± 4.49 | 34.73 ± 2.28 | 36.47 ± 6.25 | 0.446 |
| (%) | |||||
| MCV | 39.27 ± 1.49 | 38.48 ± 1.69 | 39.50 ± 1.39 | 39.31 ± 1.03 | 0.3526 |
| (fL) | |||||
| MCH | 15.07 ± 0.71 | 14.78 ± 0.95 | 15.13 ± 0.73 | 14.91 ± 0.52 | 0.7469 |
| (pg) | |||||
| MCHC | 38.36 ± 1.07 | 38.34 ± 1.31 | 38.28 ± 1.58 | 37.92 ± 1.16 | 0.8644 |
| (g/dL) | |||||
| RDW-CV | 18.24 ± 1.06 | 19.79 ± 2.14 | 18.22 ± 1.85 | 18.14 ± 1.09 | 0.1523 |
| (%) | |||||
| WBC | 3.13 ± 1.78 | 3.32 ± 1.68 | 4.50 ± 4.95 | 3.01 ± 1.00 | 0.9791 |
| (×103/µL) | |||||
| W-SCR | 61.79 ± 14.29 | 64.05 ± 9.69 | 68.24 ± 2.42 | 66.89 ± 8.88 | 0.4596 |
| (%) | |||||
| W-MCR | 36.59 ± 14.34 | 34.60 ± 8.79 | 29.84 ± 1.45 | 32.32 ± 9.01 | 0.3237 |
| (%) | |||||
| W-LCR | 1.62 ± 1.37 | 1.35 ± 1.71 | 2.11 ± 1.82 | 0.79 ± 0.73 | 0.2281 |
| (fL) | |||||
| PLT | 1162.27 ± 443.11 | 1373.23 ± 542.46 | 1179.44 ± 491.07 | 1208.44 ± 454.24 | 0.7446 |
| (×103/µL) |
| Biochemical Marker | Control | 25 mg/kg | 50 mg/kg | 100 mg/kg | p-Value |
|---|---|---|---|---|---|
| (n = 10) | (n = 10) | (n = 9) | (n = 7) | ||
| ALT | 50.82 ± 26.17 | 34.00 ± 10.39 | 41.50 ± 23.55 | 33.43 ± 13.26 | 0.2699 |
| (U/L) | |||||
| AST | 91.00 ± 41.15 | 61.56 ± 18.63 | 74.14 ± 22.42 | 73.29 ± 28.82 | 0.3415 |
| (U/L) | |||||
| Albumin | 1.14 ± 0.44 | 1.44 ± 0.43 | 1.60 ± 0.43 | 1.38 ± 0.46 | 0.1513 |
| (g/dL) | |||||
| Creatinine | 0.44 ± 0.13 | 0.42 ± 0.08 | 0.37 ± 0.06 | 0.39 ± 0.08 | 0.3976 |
| (mg/dL) | |||||
| Urea | 48.27 ± 10.64 | 54.64 ± 20.08 | 54.67 ± 5.77 | 43.86 ± 9.55 | 0.0864 |
| (mg/dL) | |||||
| LDH | 893.38 ± 521.70 | 698.70 ± 260.61 | 933.38 ± 413.48 | 724.33 ± 214.89 | 0.4954 |
| (mg/dL) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Silva, G.R.; Miranda, L.T.G.; da Silva, S.A.d.C.A.; Andrade, L.R.d.; Souza, N.C.d.; Sá, B.S.; Santana, E.R.d.; Vasconcelos, A.G.; Moreira, D.C.; Pic-Taylor, A.; et al. Toxicity and Teratogenic Potential of Piplartine from Piper tuberculatum Jacq. during Embryonic Development in Mice (Mus musculus). Drugs Drug Candidates 2024, 3, 353-367. https://doi.org/10.3390/ddc3020021
Silva GR, Miranda LTG, da Silva SAdCA, Andrade LRd, Souza NCd, Sá BS, Santana ERd, Vasconcelos AG, Moreira DC, Pic-Taylor A, et al. Toxicity and Teratogenic Potential of Piplartine from Piper tuberculatum Jacq. during Embryonic Development in Mice (Mus musculus). Drugs and Drug Candidates. 2024; 3(2):353-367. https://doi.org/10.3390/ddc3020021
Chicago/Turabian StyleSilva, Giulliano Rezende, Lívia Thaís Gontijo Miranda, Shirley Aline da Costa Arteaga da Silva, Laise Rodrigues de Andrade, Natanael Carvalho de Souza, Bruno Silva Sá, Elivaldo Ribeiro de Santana, Andreanne Gomes Vasconcelos, Daniel Carneiro Moreira, Aline Pic-Taylor, and et al. 2024. "Toxicity and Teratogenic Potential of Piplartine from Piper tuberculatum Jacq. during Embryonic Development in Mice (Mus musculus)" Drugs and Drug Candidates 3, no. 2: 353-367. https://doi.org/10.3390/ddc3020021
APA StyleSilva, G. R., Miranda, L. T. G., da Silva, S. A. d. C. A., Andrade, L. R. d., Souza, N. C. d., Sá, B. S., Santana, E. R. d., Vasconcelos, A. G., Moreira, D. C., Pic-Taylor, A., Durazzo, A., Lucarini, M., Yamaguchi, L. F., Kato, M. J., Damazo, A. S., Arcanjo, D. D. R., Leite, J. R. d. S. d. A., & Baroneza, J. E. (2024). Toxicity and Teratogenic Potential of Piplartine from Piper tuberculatum Jacq. during Embryonic Development in Mice (Mus musculus). Drugs and Drug Candidates, 3(2), 353-367. https://doi.org/10.3390/ddc3020021














