Longitudinal Changes in CD4+, CD8+ T Cell Phenotype and Activation Marker Expression Following Antiretroviral Therapy Initiation among Patients with Cryptococcal Meningitis
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Study Site Setting and Participants
2.3. Sample Size Estimation
2.4. PBMC Thawing and Stimulation
2.5. Cell Surface Staining
2.6. Data Analysis
2.7. Ethical Approval
3. Results
3.1. Participant Characteristics
3.2. Gating Strategy
3.3. CD4+ and CD8+ T Cell Counts Increase Four Weeks after ART Initiation
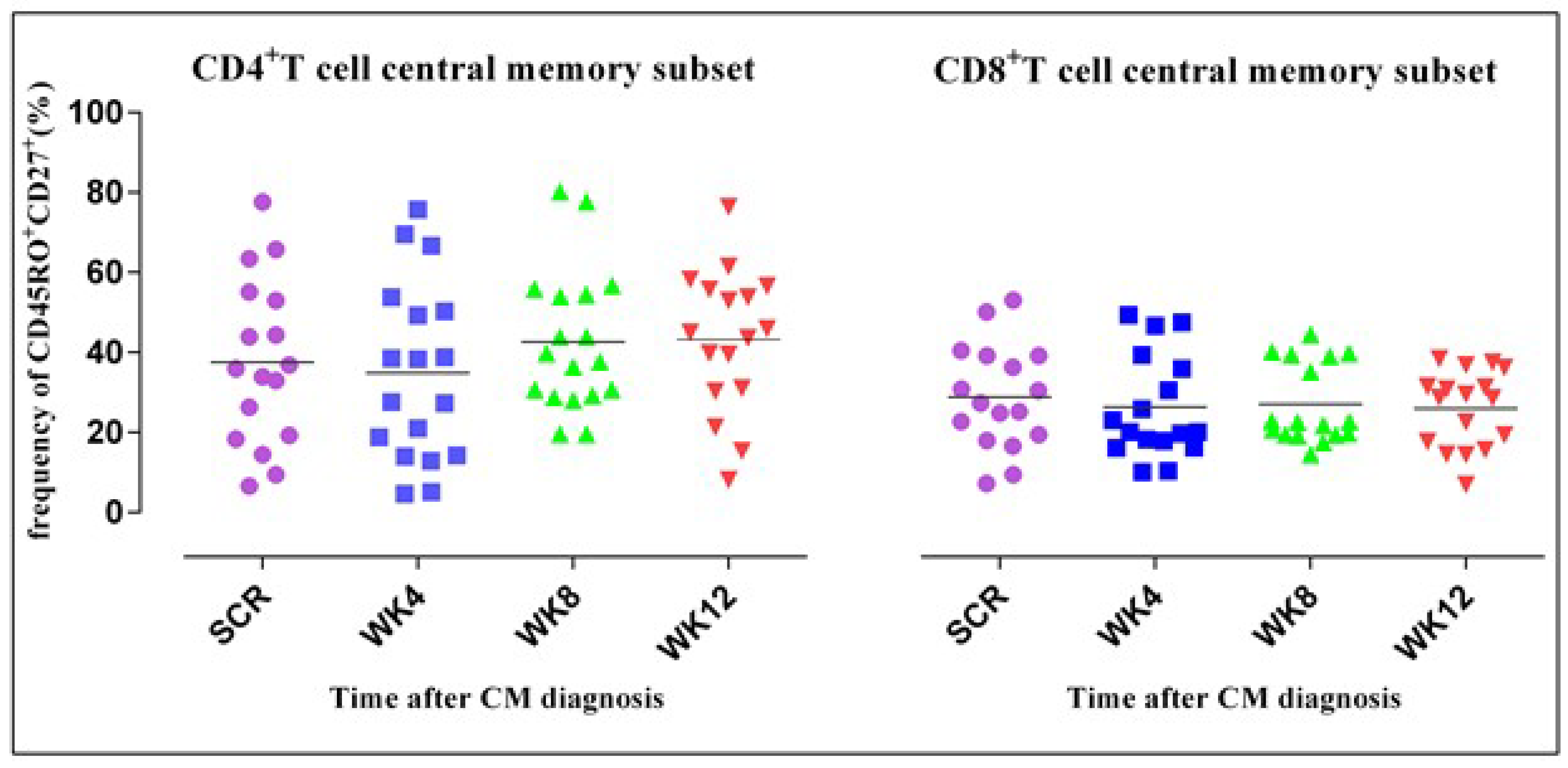
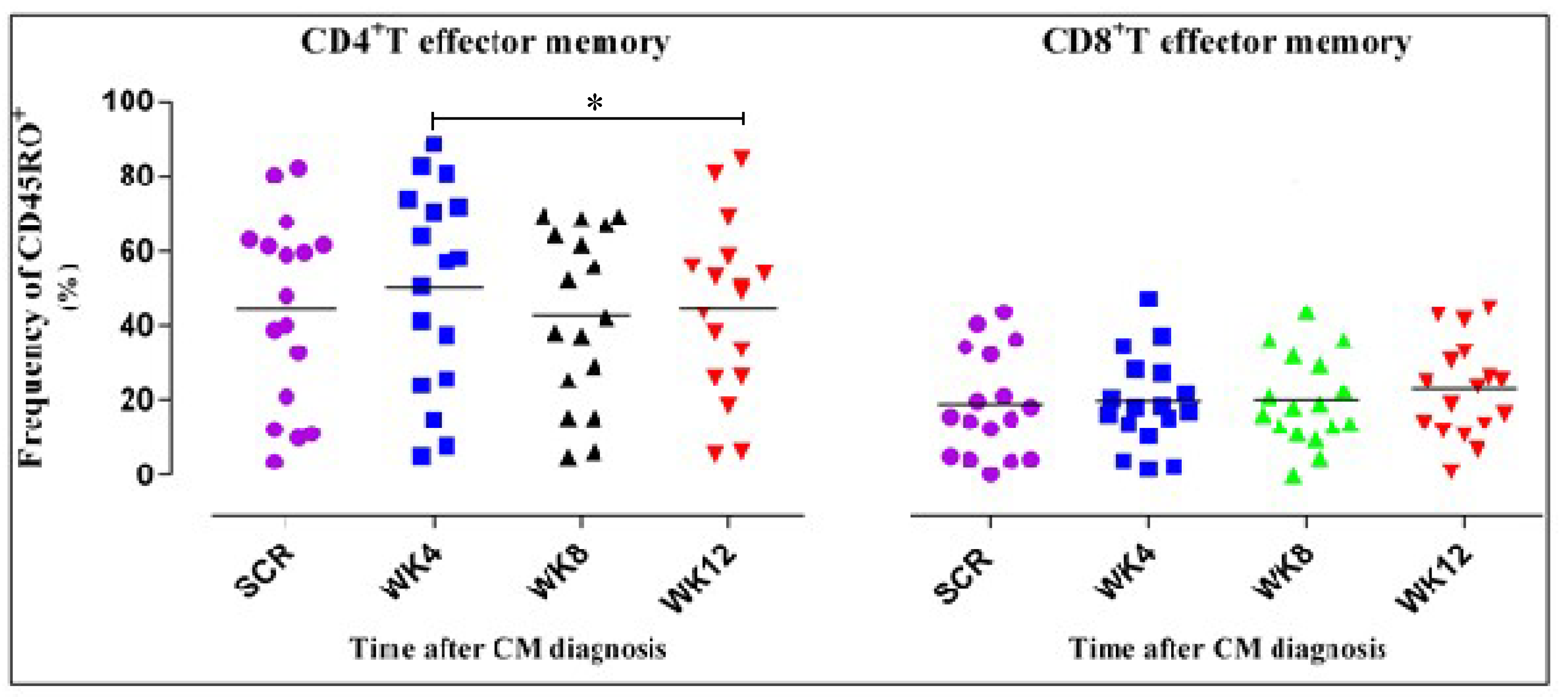
3.4. CD4+ T Cell Phenotypic Expression Profile
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Alemu, A.S.; Kempker, R.R.; Tenna, A.; Smitson, C.; Berhe, N.; Fekade, D.; Blumberg, H.M.; Aseffa, A. High prevalence of cryptococcal antigenemia among HIV-infected patients receiving antiretroviral therapy in Ethiopia. PLoS ONE 2013, 8, e58377. [Google Scholar] [CrossRef] [PubMed]
- Meya, D.B.; Okurut, S.; Zziwa, G.; Rolfes, M.A.; Kelsey, M.; Cose, S.; Joloba, M.; Naluyima, P.; Palmer, B.E.; Kambugu, A.; et al. Cellular immune activation in cerebrospinal fluid from Ugandans with cryptococcal meningitis and immune reconstitution inflammatory syndrome. J. Infect. Dis. 2015, 211, 1597–1606. [Google Scholar] [CrossRef] [PubMed]
- Durski, K.N.; Kuntz, K.M.; Yasukawa, K.; Virnig, B.A.; Meya, D.B.; Boulware, D.R. Cost-effective diagnostic checklists for meningitis in resource limited settings. J. Acquir. Immune Defic. Syndr. 2013, 63, e101–e108. [Google Scholar] [CrossRef] [PubMed]
- Warkentien, T.; Crum-cianflone, N.F. An update on cryptococcosis among HIV-infected persons. Int. J. STD AIDS 2010, 21, 679–684. [Google Scholar] [CrossRef] [PubMed]
- Zheng, C.F.; Ma, L.L.; Jones, G.J.; Gill, M.J.; Krensky, A.M.; Kubes, P.; Mody, C.H. Cytotoxic CD4+ T cells use granulysin to kill Cryptococcus neoformans, and activation of this pathway is defective in HIV patients. Blood 2007, 109, 2049–2057. [Google Scholar] [CrossRef] [PubMed]
- Retini, C.; Vecchiarelli, A.; Monari, C.; Bistoni, F.; Kozel, T.R. Encapsulation of Cryptococcus neoformans with glucuronoxylomannan inhibits the antigen-presenting capacity of monocytes. Infect. Immun. 1998, 66, 664–669. [Google Scholar] [PubMed]
- Jarvis, J.N.; Casazza, J.P.; Stone, H.H.; Meintjes, G.; Lawn, S.D.; Levitz, S.M.; Harrison, T.S.; Koup, R.A. The phenotype of the Cryptococcus-specific CD4+ memory T-cell response is associated with disease severity and outcome in HIV-associated cryptococcal meningitis. J. Infect. Dis. 2013, 207, 1817–1828. [Google Scholar] [CrossRef] [PubMed]
- Moir, S.; Chun, T.W.; Fauci, A.S. Pathogenic mechanisms of HIV disease. Annu. Rev. Pathol. 2011, 6, 223–248. [Google Scholar] [CrossRef]
- Tenforde, M.W.; Scriven, J.E.; Harrison, T.S.; Jarvis, J.N. Immune correlates of HIV-associated cryptococcal meningitis. PLoS Pathog. 2017, 13, e1006207. [Google Scholar] [CrossRef]
- De Rosa, S.C.; Herzenberg, L.A.; Herzenberg, L.A.; Roederer, M. 11-color, 13-parameter flow cytometry: Identification of human naive T cells by phenotype, function, and T-cell receptor diversity. Nat. Med. 2001, 7, 245–248. [Google Scholar] [CrossRef]
- Lacabaratz-Porret, C.; Urrutia, A.; Doisne, J.M.; Goujard, C.; Deveau, C.; Dalod, M.; Meyer, L.; Rouzioux, C.; Delfraissy, J.F.; Venet, A.; et al. Impact of antiretroviral therapy and changes in virus load on human immunodeficiency virus (HIV)–specific T cell responses in primary HIV infection. J. Infect. Dis. 2003, 187, 748–757. [Google Scholar] [CrossRef] [PubMed]
- Herout, S.; Mandorfer, M.; Breitenecker, F.; Reiberger, T.; Grabmeier-Pfistershammer, K.; Rieger, A.; Aichelburg, M.C. Impact of early initiation of antiretroviral therapy in patients with acute HIV infection in Vienna, Austria. PLoS ONE 2016, 11, e0152910. [Google Scholar] [CrossRef] [PubMed]
- Holtick, U.; Frenzel, L.; Fröhlich, H.; Shimabukuro-Vornhagen, A.; Theurich, S.; Claasen, J.; Scheid, C.; von Bergwelt-Baildon, M.; Wendtner, C.; Chemnitz, J. Increased Functional T Cell. Defects In Patients With Low Cd4 Counts After Allogeneic Hematopoietic Stem Cell Transplantation. Bone Marrow Transplant. 2014, 49, S558–S559. [Google Scholar]
- Boulware, D.R.; Meya, D.B.; Muzoora, C.; Rolfes, M.A.; Huppler Hullsiek, K.; Musubire, A.; Taseera, K.; Nabeta, H.; Schutz, C.; Williams, D.; et al. Timing of antiretroviral therapy after diagnosis of cryptococcal meningitis. N. Engl. J. Med. 2014, 370, 2487–2498. [Google Scholar] [CrossRef] [PubMed]
- Meya, D.; Rajasingham, R.; Nalintya, E.; Tenforde, M.; Jarvis, J.N. Preventing Cryptococcosis—Shifting the Paradigm in the Era of Highly Active Antiretroviral Therapy. Curr. Trop. Med. Rep. 2015, 2, 81–89. [Google Scholar] [CrossRef] [PubMed]
- Meya, D.B.; Manabe, Y.C.; Castelnuovo, B.; Cook, B.A.; Elbireer, A.M.; Kambugu, A.; Kamya, M.R.; Bohjanen, P.R.; Boulware, D.R. Cost-effectiveness of serum cryptococcal antigen screening to prevent deaths among HIV-infected persons with a CD4+ cell count ≤ 100 cells/μL who start HIV therapy in resource-limited settings. Clin. Infect. Dis. 2010, 51, 448–455. [Google Scholar] [CrossRef] [PubMed]
- Conrad, J.A.; Ramalingam, R.K.; Duncan, C.B.; Smith, R.M.; Wei, J.; Barnett, L.; Simons, B.C.; Lorey, S.L.; Kalams, S.A. Antiretroviral Therapy Reduces the Magnitude and T Cell Receptor Repertoire Diversity of HIV-Specific T Cell Responses without Changing T Cell Clonotype Dominance. J. Virol. 2012, 86, 4213–4221. [Google Scholar] [CrossRef]
- Bicanic, T.; Meintjes, G.; Wood, R.; Hayes, M.; Rebe, K.; Bekker, L.G.; Harrison, T. Fungal burden, early fungicidal activity, and outcome in cryptococcal meningitis in antiretroviral-naive or antiretroviral-experienced patients treated with amphotericin B or fluconazole. Clin. Infect. Dis. 2007, 45, 76–80. [Google Scholar] [CrossRef]
- Velu, V.; Shetty, R.D.; Larsson, M.; Shankar, E.M. Role of PD-1 co-inhibitory pathway in HIV infection and potential therapeutic options. Retrovirology 2015, 12, 14. [Google Scholar] [CrossRef]
- Catalfamo, M.; Wilhelm, C.; Tcheung, L.; Proschan, M.; Friesen, T.; Park, J.H.; Adelsberger, J.; Baseler, M.; Maldarelli, F.; Davey, R.; et al. CD4 and CD8 T cell immune activation during chronic HIV infection: Roles of homeostasis, HIV, type I IFN, and IL-7. J. Immunol. 2011, 186, 2106–2116. [Google Scholar] [CrossRef]
- Rallón, N.; Sempere-Ortells, J.M.; Soriano, V.; Benito, J.M. Central memory CD4 T cells are associated with incomplete restoration of the CD4 T cell pool after treatment-induced long-term undetectable HIV viraemia. J. Antimicrob. Chemother. 2013, 68, 2616–2625. [Google Scholar] [CrossRef] [PubMed]
- Jarvis, J.N.; Harrison, T.S. HIV-associated cryptococcal meningitis. AIDS 2007, 21, 2119–2129. [Google Scholar] [CrossRef] [PubMed]
- Akakpo, K.P.; Quayson, S.E.; Lartey, M. Disseminated cryptococcosis in a patient with HIV/AIDS at a teaching hospital in Ghana. SAGE Open Med. Case Rep. 2015, 3, 2050313X14565421. [Google Scholar] [CrossRef] [PubMed]
- Voelz, K.; May, R.C. Cryptococcal interactions with the host immune system. Eukaryot. Cell 2010, 9, 835–846. [Google Scholar] [CrossRef] [PubMed]
- Guidelines for the Use of Antiretroviral Agents in HIV-1-Infected Adults and Adolescents. 2016. Available online: http://aidsinfo.nih.gov/guidelines (accessed on 10 June 2016).
- World Health Organisation STEPS Sample Size Calculator and Sampling Spreadsheet. Available online: http://www.who.int/chp/steps/resources/sampling/en/ (accessed on 10 January 2017).
- Streeck, H.; Jessen, H.; Alter, G.; Teigen, N.; Waring, M.T.; Jessen, A.; Stahmer, I.; van Lunzen, J.; Lichterfeld, M.; Gao, X.; et al. Immunological and virological impact of highly active antiretroviral therapy initiated during acute HIV-1 infection. J. Infect. Dis. 2006, 194, 734–739. [Google Scholar] [CrossRef]
- Wilson, E.M.; Sereti, I. Immune restoration after antiretroviral therapy: The pitfalls of hasty or incomplete repairs. Immunol. Rev. 2013, 254, 343–354. [Google Scholar] [CrossRef] [PubMed]
- Sallusto, F.; Geginat, J.; Lanzavecchia, A. Central memory and effector memory T cell subsets: Function, generation, and maintenance. Annu. Rev. Immunol. 2004, 22, 745–763. [Google Scholar] [CrossRef]
- Lanzavecchia, A.; Sallusto, F. Understanding the generation and function of memory T cell subsets. Curr. Opin. Immunol. 2005, 17, 326–332. [Google Scholar] [CrossRef]
- Von Wyl, V.; Gianella, S.; Fischer, M.; Niederoest, B.; Kuster, H.; Battegay, M.; Bernasconi, E.; Cavassini, M.; Rauch, A.; Hirschel, B.; et al. Early antiretroviral therapy during primary HIV-1 infection results in a transient reduction of the viral setpoint upon treatment interruption. PLoS ONE 2011, 6, e27463. [Google Scholar] [CrossRef]
- Serrano-Villar, S.; Gutierrez, C.; Vallejo, A.; Hernandez-Novoa, B.; Diaz, L.; Fernandez, M.A.; Madrid, N.; Dronda, F.; Zamora, J.; Munoz-Fernandez, M.A.; et al. The CD4/CD8 ratio in HIV-infected subjects is independently associated with T-cell activation despite long-term viral suppression. J. Infect. 2013, 66, 57–66. [Google Scholar] [CrossRef]
- Gengis, E.H.; Deeks, S.G. CD4+ T cell recovery with antiretroviral therapy: More than the sum of the parts. Clin. Infect. Dis. 2009, 48, 362–364. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Jenkins, M.K.; Moon, J.J. The role of naive T cell precursor frequency and recruitment in dictating immune response magnitude. J. Immunol. 2012, 188, 4135–4140. [Google Scholar] [CrossRef] [PubMed]
- Butler, E.K.; Boulware, D.R.; Bohjanen, P.R.; Meya, D.B. Long Term 5-Year Survival of Persons with Cryptococcal Meningitis or Asymptomatic Subclinical Antigenemia in Uganda. PLoS ONE 2012, 7, e51291. [Google Scholar] [CrossRef] [PubMed]
- Robbins, G.K.; Spritzler, J.G.; Chan, E.S.; Asmuth, D.M.; Gandhi, R.T.; Rodriguez, B.A.; Skowron, G.; Skolnik, P.R.; Shafer, R.W.; Pollard, R.B.; et al. Incomplete reconstitution of T cell subsets on combination antiretroviral therapy in the AIDS Clinical Trials Group protocol 384. Clin. Infect. Dis. 2009, 48, 350–361. [Google Scholar] [CrossRef] [PubMed]
- Bucy, R.P.; Hockett, R.D.; Derdeyn, C.A.; Saag, M.S.; Squires, K.; Sillers, M.; Mitsuyasu, R.T.; Kilby, J.M. Initial increase in blood CD4+ lymphocytes after HIV antiretroviral therapy reflects redistribution from lymphoid tissues. J. Clin. Investig. 1999, 103, 1391–1398. [Google Scholar] [CrossRef]
- Smith, C.J.; Sabin, C.A.; Youle, M.S.; Kinloch-de Loes, S.; Lampe, F.C.; Madge, S.; Cropley, I.; Johnson, M.A.; Phillips, A.N. Factors influencing increases in CD4 cell counts of HIV-positive persons receiving long-term highly active antiretroviral therapy. J. Infect. Dis. 2004, 190, 1860–1868. [Google Scholar] [CrossRef] [PubMed]
- Kambugu, A.; Meya, D.B.; Rhein, J.; O’brien, M.; Janoff, E.N.; Ronald, A.R.; Kamya, M.R.; Mayanja-Kizza, H.; Sande, M.A.; Bohjanen, P.R.; et al. Outcomes of cryptococcal meningitis in Uganda before and after the availability of highly active antiretroviral therapy. Clin. Infect. Dis. 2008, 46, 1694–1701. [Google Scholar] [CrossRef]
- Jarvis, J.N.; Bicanic, T.; Loyse, A.; Namarika, D.; Jackson, A.; Nussbaum, J.C.; Longley, N.; Muzoora, C.; Phulusa, J.; Taseera, K.; et al. Determinants of mortality in a combined cohort of 501 patients with HIV-associated cryptococcal meningitis: Implications for improving outcomes. Clin. Infect. Dis. 2014, 58, 736–745. [Google Scholar] [CrossRef]
- Wang, W.; Carm, A.R. Clinical manifestations of AIDS with cryptococcal meningitis. Chin. Med. J. 2001, 114, 841–843. [Google Scholar]
- Moore, R.D.; Keruly, J.C. CD4+ cell count 6 years after commencement of highly active antiretroviral therapy in persons with sustained virologic suppression. Clin. Infect. Dis. 2007, 44, 441–446. [Google Scholar] [CrossRef]
- Goldman, D.L.; Lee, S.C.; Mednick, A.J.; Montella, L.; Casadevall, A. Persistent Cryptococcus neoformansPulmonary Infection in the Rat Is Associated with Intracellular Parasitism, Decreased Inducible Nitric Oxide Synthase Expression, and Altered Antibody Responsiveness to Cryptococcal Polysaccharide. Infect. Immun. 2000, 68, 832–838. [Google Scholar] [CrossRef] [PubMed]
- Chéret, A.; Bacchus-Souffan, C.; Avettand-Fenoël, V.; Mélard, A.; Nembot, G.; Blanc, C.; Samri, A.; Sáez-Cirión, A.; Hocqueloux, L.; Lascoux-Combe, C.; et al. Combined ART started during acute HIV infection protects central memory CD4+ T cells and can induce remission. J. Antimicrob. Chemother. 2015, 70, 2108–2120. [Google Scholar]
- Kalia, V.; Sarkar, S.; Ahmed, R. CD8 T-cell memory differentiation during acute and chronic viral infections. In Memory T Cells; Springer: New York, NY, USA, 2010; pp. 79–95. [Google Scholar]
- Descours, B.; Avettand-Fenoel, V.; Blanc, C.; Samri, A.; Mélard, A.; Supervie, V.; Theodorou, I.; Carcelain, G.; Rouzioux, C.; Autran, B.; et al. Immune responses driven by protective human leukocyte antigen alleles from long-term nonprogressors are associated with low HIV reservoir in central memory CD4 T cells. Clin. Infect. Dis. 2012, 54, 1495–1503. [Google Scholar] [CrossRef] [PubMed]
- Lefrançois, L. Development, trafficking, and function of memory T-cell subsets. Immunol. Rev. 2006, 211, 93–103. [Google Scholar] [CrossRef] [PubMed]
- Lakkis, F.G.; Sayegh, M.H. Memory T cells: A hurdle to immunologic tolerance. J. Am. Soc. Nephrol. 2003, 14, 2402–2410. [Google Scholar] [CrossRef] [PubMed]
- Sabiiti, W.; May, R.C. Mechanisms of infection by the human fungal pathogen Cryptococcus neoformans. Future Microbiol. 2012, 7, 1297–1313. [Google Scholar] [CrossRef] [PubMed]
- Charlier, C.; Nielsen, K.; Daou, S.; Brigitte, M.; Chretien, F.; Dromer, F. Evidence of a role for monocytes in dissemination and brain invasion by Cryptococcus neoformans. Infect. Immun. 2009, 77, 120–127. [Google Scholar] [CrossRef] [PubMed]
- Williamson, P.R. Advancing Translational Immunology in HIV-Associated Cryptococcal Meningitis. J. Infect. Dis. 2013, 207, 1793–1795. [Google Scholar] [CrossRef] [PubMed]
- Boulware, D.R.; Meya, D.B.; Bergemann, T.L.; Wiesner, D.L.; Rhein, J.; Musubire, A.; Lee, S.J.; Kambugu, A.; Janoff, E.N.; Bohjanen, P.R. Clinical features and serum biomarkers in HIV immune reconstitution inflammatory syndrome after cryptococcal meningitis: A prospective cohort study. PLoS Med. 2010, 7, e1000384. [Google Scholar] [CrossRef]
- Hunt, P.W.; Cao, H.L.; Muzoora, C.; Ssewanyana, I.; Bennett, J.; Emenyonu, N.; Kembabazi, A.; Neilands, T.B.; Bangsberg, D.R.; Deeks, S.G.; et al. Impact of CD8+ T cell activation on CD4+ T cell recovery and mortality in HIV-infected Ugandans initiating antiretroviral therapy. AIDS 2011, 25, 2123–2131. [Google Scholar] [CrossRef]
- Jankovic, D.; Feng, C.G. CD4+ T cell differentiation in infection: Amendments to the Th1/Th2 axiom. Front. Immunol. 2015, 6, 198. [Google Scholar] [CrossRef] [PubMed]
- Fauci, A.S. Pathogenesis of HIV disease: Opportunities for new prevention interventions. Clin. Infect. Dis. 2007, 45, S206–S212. [Google Scholar] [CrossRef] [PubMed]
- Jain, N.; Guerrero, A.; Fries, B.C. Phenotypic switching and its implications for the pathogenesis of Cryptococcus neoformans. FEMS Yeast Res. 2006, 6, 480–488. [Google Scholar] [CrossRef] [PubMed]
- Hoffmann, M.; Pantazis, N.; Martin, G.E.; Hickling, S.; Hurst, J.; Meyerowitz, J.; Willberg, C.B.; Robinson, N.; Brown, H.; Fisher, M.; et al. Exhaustion of Activated CD8 T Cells Predicts Disease Progression in Primary HIV-1 Infection. PLoS Pathog. 2016, 12, e1005661. [Google Scholar] [CrossRef] [PubMed]
- Cockerham, L.R.; Jain, V.; Sinclair, E.; Glidden, D.V.; Hartogenesis, W.; Hatano, H.; Hunt, P.W.; Martin, J.N.; Pilcher, C.D.; Sekaly, R.; et al. Programmed death-1 expression on CD4+ and CD8+ T cells in treated and untreated HIV disease. AIDS 2014, 28, 1749. [Google Scholar] [CrossRef]
- Mora, D.J.; Fortunato, L.R.; Andrade-Silva, L.E.; Ferreira-Paim, K.; Rocha, I.H.; Vasconcelos, R.R.; Silva-Teixeira, D.N.; Nascentes, G.A.; Silva-Vergara, M.L. Cytokine Profiles at Admission Can Be Related to Outcome in AIDS Patients with Cryptococcal Meningitis. PLoS ONE 2015, 10, e0120297. [Google Scholar] [CrossRef]
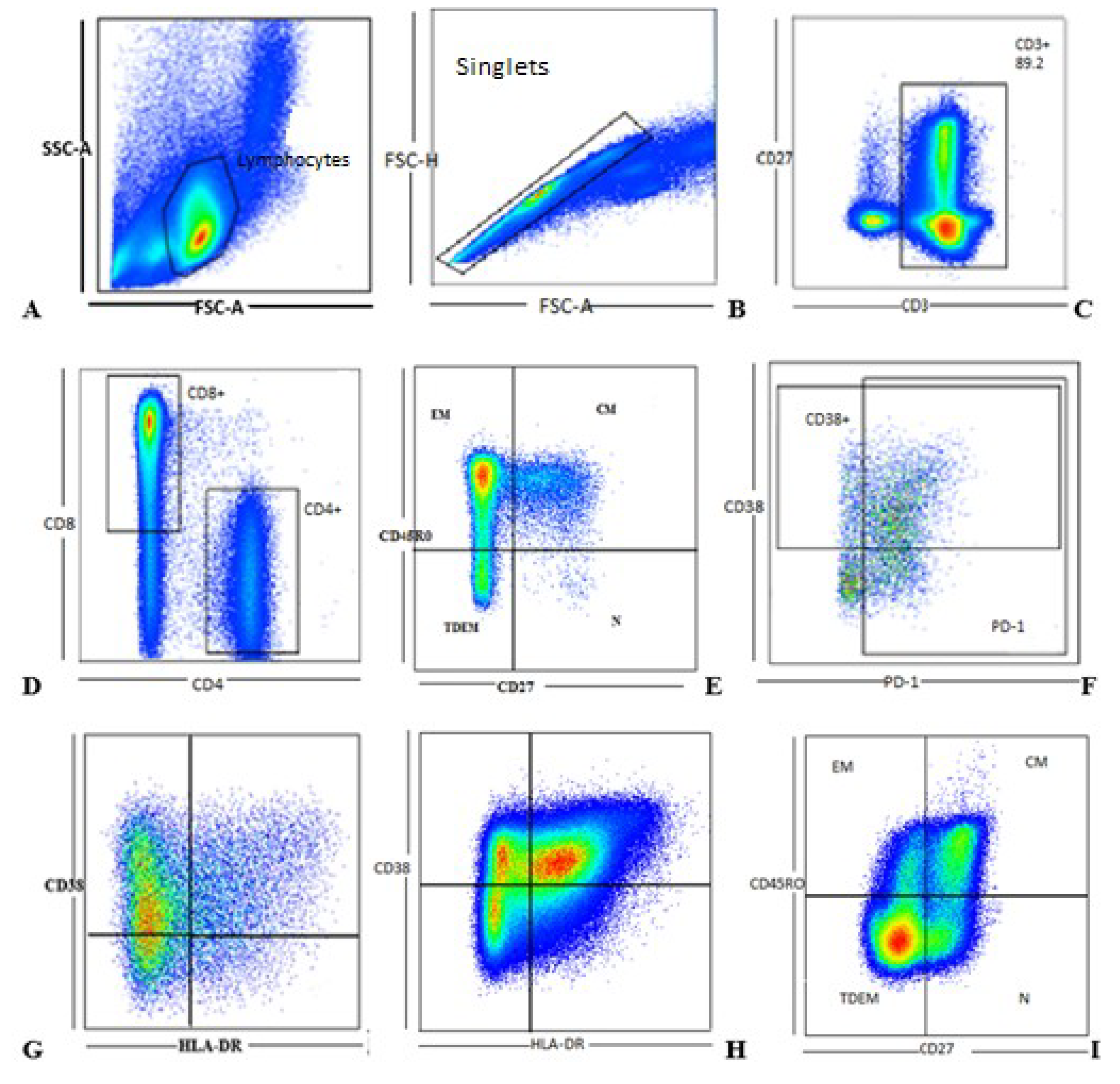



| Cell Marker | Phenotype Function |
|---|---|
| CD3 | T cell lineage marker |
| CD4 | CD4+ T lineage |
| CD8 | CD8+ T lineage |
| CD38 | Immune activation |
| CD45R0 | T cell memory |
| CD27 | T cell memory |
| PD-1 | Immune exhaustion |
| HLA-DR | Immune activation |
| Mean Age, Years | 37 |
| Female, n (%) | 7 (35%) |
| Male, n (%) | 13 (65%) |
| Baseline CD4 T cell count, mean (SD) | 34 (±32) cells/mm3 |
| Viral load (log10), Mean (SD) | 6.33 ± 0.66 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bayiyana, A.; Okurut, S.; Nabatanzi, R.; Zziwa, G.; Boulware, D.R.; Lutwama, F.; Meya, D. Longitudinal Changes in CD4+, CD8+ T Cell Phenotype and Activation Marker Expression Following Antiretroviral Therapy Initiation among Patients with Cryptococcal Meningitis. J. Fungi 2019, 5, 63. https://doi.org/10.3390/jof5030063
Bayiyana A, Okurut S, Nabatanzi R, Zziwa G, Boulware DR, Lutwama F, Meya D. Longitudinal Changes in CD4+, CD8+ T Cell Phenotype and Activation Marker Expression Following Antiretroviral Therapy Initiation among Patients with Cryptococcal Meningitis. Journal of Fungi. 2019; 5(3):63. https://doi.org/10.3390/jof5030063
Chicago/Turabian StyleBayiyana, Alice, Samuel Okurut, Rose Nabatanzi, Godfrey Zziwa, David R. Boulware, Fredrick Lutwama, and David Meya. 2019. "Longitudinal Changes in CD4+, CD8+ T Cell Phenotype and Activation Marker Expression Following Antiretroviral Therapy Initiation among Patients with Cryptococcal Meningitis" Journal of Fungi 5, no. 3: 63. https://doi.org/10.3390/jof5030063
APA StyleBayiyana, A., Okurut, S., Nabatanzi, R., Zziwa, G., Boulware, D. R., Lutwama, F., & Meya, D. (2019). Longitudinal Changes in CD4+, CD8+ T Cell Phenotype and Activation Marker Expression Following Antiretroviral Therapy Initiation among Patients with Cryptococcal Meningitis. Journal of Fungi, 5(3), 63. https://doi.org/10.3390/jof5030063





