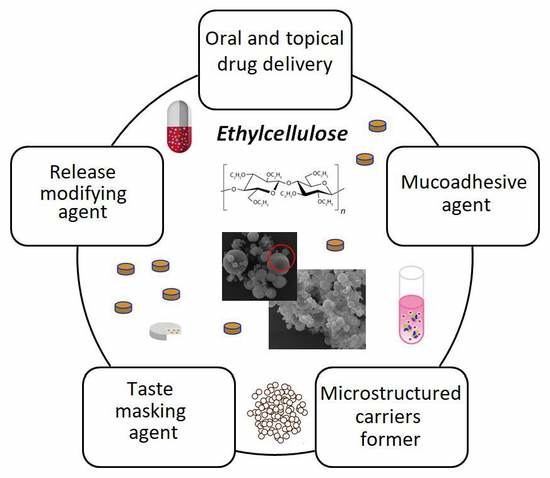
Ethylcellulose–A Pharmaceutical Excipient with Multidirectional Application in Drug Dosage Forms Development
Abstract
:1. Introduction
2. Physicochemical Characteristics of EC
3. Pharmaceutical Characteristics of EC
4. Applicability of EC in Pharmaceutical Formulations
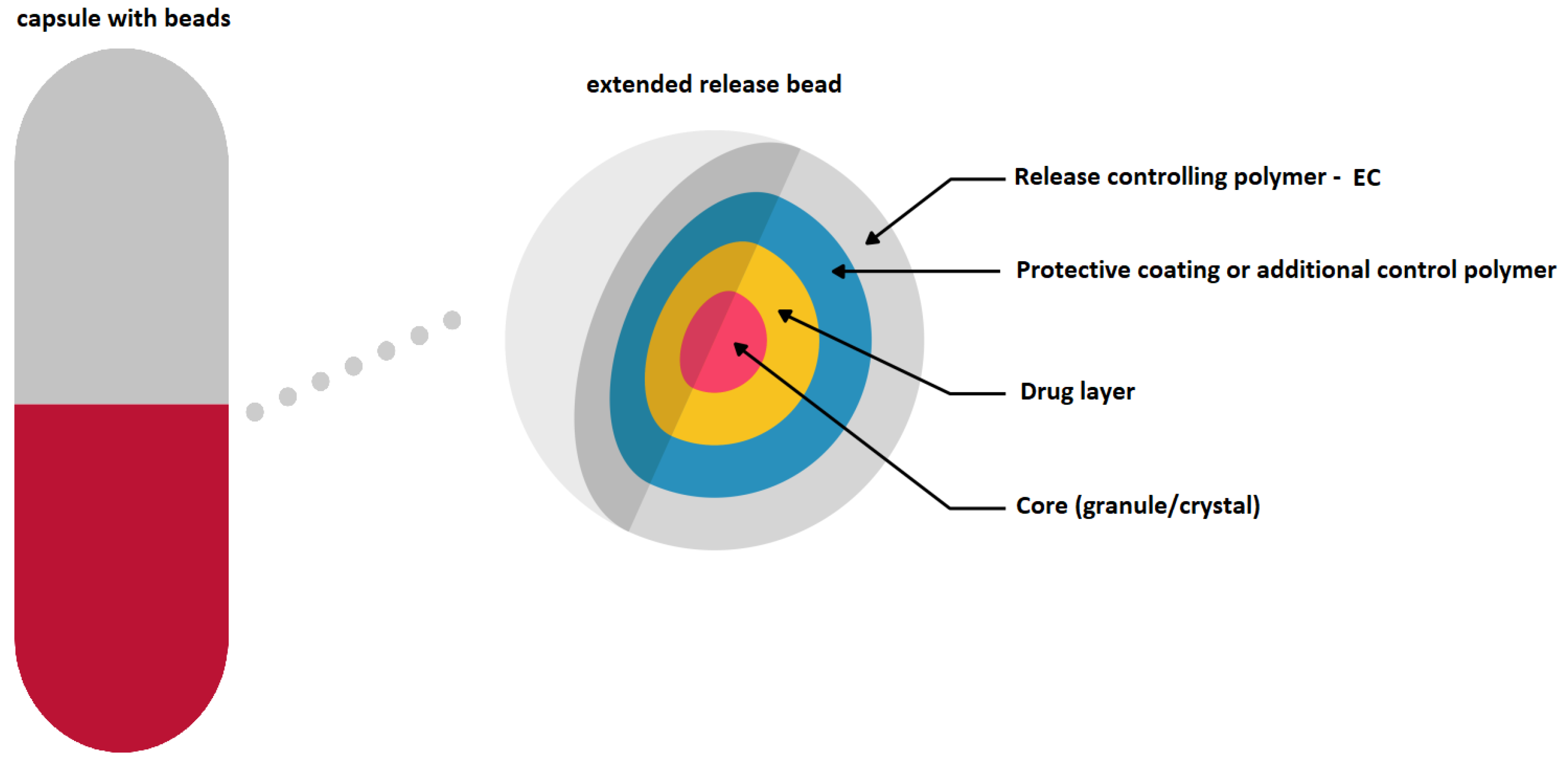
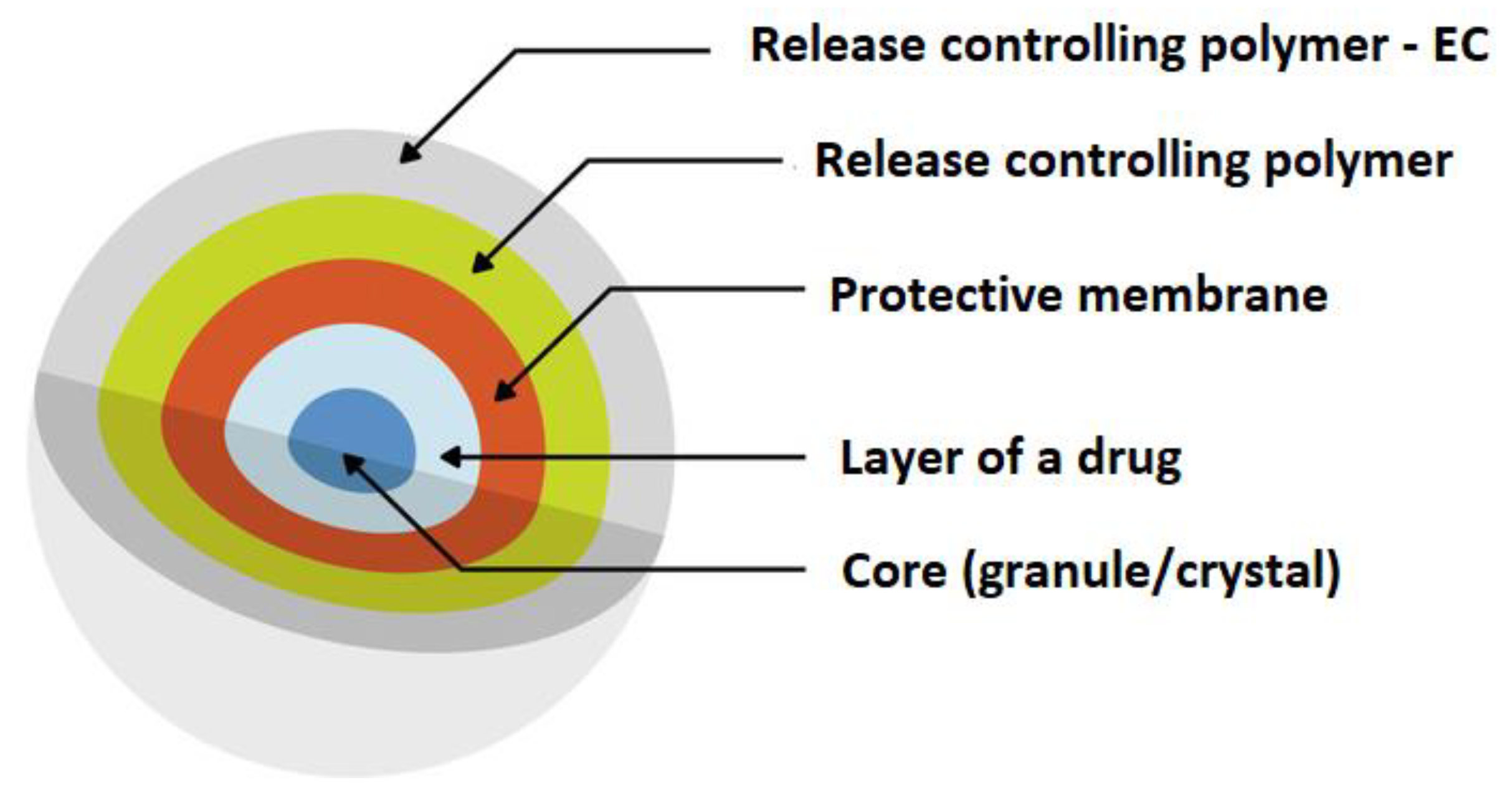
4.1. EC as An MR Coating Material in Oral Delivery Systems
4.2. EC as a Sustained Release Material in Topical Delivery Systems
4.3. EC-Based Solid Dispersions
4.4. EC-Based Micro- and Nanocarriers
4.5. EC in Mucoadhesive Delivery Systems
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Qiu, Y.; Lee, P.I. Rational design of oral modified-release drug delivery systems. In Developing Solid Oral Dosage Forms. Pharmaceutical Theory and Practice, 2nd ed.; Qui, Y., Zhang, G.G.Z., Mantri, R.V., Chen, Y., Yu, L., Eds.; Academic Press: Cambridge, MA, USA, 2017; pp. 1127–1160. [Google Scholar]
- FDA. Available online: https://www.fda.gov/downloads/Drugs/.../UCM291010.pdf (accessed on 20 July 2019).
- Rathbone, M.J.; Hadgraft, J.; Roberts, M.S.; Lane, M.E. Modified-Release Drug Delivery Technology, 2nd ed.; Informa Healthcare: London, UK, 2008; Volume 2. [Google Scholar]
- Rowe, R.C.; Sheskey, P.J.; Quinn, M.E. Handbook of Pharmaceutical Excipients, 6th ed.; Pharmaceutical Press: London, UK; Chicago, IL, USA; Washington, DC, USA, 2009; pp. 262–267. [Google Scholar]
- Ethyl Cellulose. Available online: https://pubchem.ncbi.nlm.nih.gov/compound/24832091#section= Probable-Routes-of-Human-Exposure (accessed on 21 July 2019).
- The European Pharmacopoeia, 9th ed.; Council of Europe: Strasburg, France, 2016.
- The United States Pharmacopeia and National Formulary (USP41-NF 36); Pharmacopeia Convention: Rockville, MD, USA, 2018; Volume 2.
- FDA Inactive Ingredients Database. Available online: https://search.fda.gov/search?utf8=%E2%9C%93&affiliate=fda1&query=ethylcellulose&commit=Search (accessed on 20 July 2019).
- Canadian List of Acceptable Non-Medicinal Ingredients. Available online: https://www.canada.ca/en/health-canada/services/drugs-health-products/natural-non-prescription/applications-submissions/product-licensing/compendium-monographs.html (accessed on 20 July 2019).
- The Pharmacopoeia of the People’s Republic of China 2015 Edition; The Stationery Office: London, UK, 2015.
- The Japanese Pharmacopoeia, Seventeenth Edition (JP17); The Ministry of Health, Labour and Welfare: Tokyo, Japan, 2016.
- Ethocel, Colorcon. Available online: https://www.pharma.dupont.com/pharmaceutical-products/ethocel. html (accessed on 20 July 2019).
- Brydson, J.A. Plastics Materials, 6th ed.; Butterworth-Heinemann: Oxford, UK, 1995. [Google Scholar]
- Mehta, R.; Teckoe, J.; Schoener, C.; Workentine, S.; Ferrizzi, D.; Rajabi-Siahoboomi, A. Investigation into effect of ethylcellulose viscosity variation on the drug release of metoprolol tartrate and acetaminophen extended release multiparticulates—Part I. AAPS PharmSciTech 2016, 17, 1366–1375. [Google Scholar] [CrossRef]
- Lokhande, A.B.; Ravindra, S.M.; Jitendra, D.K.; Naik, B. Influence of different viscosity grade ethylcellulose polymers on encapsulation and in vitro release study of drug loaded nanoparticles metformin. J. Pharm. Res. 2013, 5, 414–420. [Google Scholar]
- Trofimiuk, M.; Wasilewska, K.; Winnicka, K. How to modify drug release in paediatric dosage forms? Novel technologies and modern approaches with regard to children’s population. Int. J. Mol. Sci. 2019, 20, 3200. [Google Scholar] [CrossRef]
- Hamedelniel, E.I.; Bajdik, J.; Pintye-Hódi, K. Optimization of preparation of matrix pellets containing ethylcellulose. Chem. Eng. Process. 2010, 49, 120–124. [Google Scholar] [CrossRef]
- Mallipeddi, R.; Saripella, K.K.; Neau, S.H. Use of fine particle ethylcellulose as the diluent in the production of pellets by extrusion-spheronization. SPJ. 2014, 22, 360–371. [Google Scholar] [CrossRef]
- Grund, J.; Koerber, M.; Walther, M.; Bodmeier, R. The effect of polymer properties on direct compression and drug release from water-insoluble controlled release matrix tablets. Int. J. Pharm. 2014, 469, 94–101. [Google Scholar] [CrossRef]
- Cifuentes, C.; Aguilar-de-Leyva, A.; Rajabi-Siahboomi, A.R.; Caraballo, I. Critical points in ethylcellulose matrices: Influence of the polymer, drug and filler properties. Acta Pharm. 2013, 63, 115–129. [Google Scholar] [CrossRef] [Green Version]
- Bruschi, M. Strategies to Modify Drug Release from Pharmaceutics Systems, 1st ed.; Woodhead Publishing: Cambridge, UK, 2015; pp. 87–119. [Google Scholar]
- Mehta, R.Y.; Missaghi, S.; Tiwari, S.B.; Rajabi-Siahboomi, A.R. Application of ethylcellulose coating to hydrophilic matrices: A strategy to modulate drug release profile and reduce drug release variability. AAPS Pharm. Sci. Tech. 2014, 15, 1049–1059. [Google Scholar] [CrossRef]
- Pawar, A.; Deepak, V.B.; Vineeta, V.K.; Vilasrao, J.K. Advances in pharmaceutical coatings. Int. J. Chem. Tech. Res. 2010, 1, 733–737. [Google Scholar]
- Aqualon, Ashland. Available online: https://www.ashland.com/industries/pharmaceutical/oral-soliddose/aqualon-ethylcellulose (accessed on 20 July 2019).
- Surelease, Colorcon. Available online: https://www.colorcon.com/products-formulation/all-products/ film-coatings/sustained-release/surelease (accessed on 20 July 2019).
- Aquacoat, ECD. Available online: http://www.fmcbiopolymer.com/Pharmaceutical/Products/Aquacoat. aspx (accessed on 20 July 2019).
- Aquarius, Ashland. Available online: https://www.ashland.com/file_source/Ashland/links/PTR-112_Aquarius_Control_ECD_reconstitution_instructions.pdf (accessed on 20 July 2019).
- AshaKote, Ashaland. Available online: http://www.ashacel.com/AshaKote.PDF (accessed on 20 July 2019).
- Shin, T.H.; Ho, M.J.; Kim, S.R.; Im, S.H.; Kim, C.H.; Lee, S.; Kang, M.J.; Choi, Y.W. Formulation and in vivo pharmacokinetic evaluation of ethyl cellulose-coated sustained release multiple-unit system of tacrolimus. Int. J. Biol. Macromol. 2018, 1, 544–550. [Google Scholar] [CrossRef]
- Full Prescribing Information Advagraf®. Available online: https://www.ema.europa.eu/en/documents/product-information/advagraf-epar-product-information_en.pdf (accessed on 20 July 2019).
- Shah, N.; Sharma, O.P.; Mehta, T.; Amin, A. Design of experiment approach for formulating multi-unit colon-targeted drug delivery system: In vitro and in vivo studies. Drug Dev. Ind. Pharm. 2016, 42, 825–835. [Google Scholar] [CrossRef]
- Kim, M.S.; Yeom, D.W.; Kim, S.R.; Yoon, H.Y.; Kim, C.H.; Son, H.Y.; Kim, J.H.; Lee, S.; Choi, Y.W. Development of a chitosan based double layer-coated tablet as a platform for colon-specific drug delivery. Drug Des. Devel. Ther. 2017, 11, 45–57. [Google Scholar] [CrossRef]
- Wan, H.; Park, K. Oral Controlled Release Formulation Design and Delivery. Theory to Practice; Wiley: Hoboken, NJ, USA, 2010. [Google Scholar]
- Full Prescribing Information Micro-K™. Available online: https://www.rxlist.com/micro-k-drug.htm (accessed on 20 July 2019).
- Full Prescribing Information Theo-24®. Available online: https://www.drugs.com/dosage/theo-24.html (accessed on 20 July 2019).
- Mohamed, F.A.; Roberts, M.; Seton, L.; Ford, J.L.; Levina, M.; Rajabi-Siahboomi, A.R. Film-coated matrix mini-tablets for the extended release of a water-soluble drug. Drug Dev. Ind. Pharm. 2015, 41, 623–630. [Google Scholar] [CrossRef]
- Full Prescribing Information Metadate CD®. Available online: https://www.accessdata.fda.gov/drugsatfda_docs/label/2009/021259s021lbl.pdf (accessed on 20 July 2019).
- Full Prescribing Information Lamictal® XR. Available online: https://www.fda.gov/downloads/drugs/developmentapprovalprocess/developmentresources/ucm215664.pdf (accessed on 20 July 2019).
- Venkata, P.D.B. Spheroidal oral drug absorption system (SODAS). J. Glob. Pharma Technol. 2011, 3, 1–5. [Google Scholar]
- Elan Drug Technologies. Spheroidal Drug Absorption System (SODAS®). Available online: http://www.elandrugtechnologies.com/oral_controlled_release/sodas (accessed on 20 July 2019).
- Full Prescribing Information Adzenys XR-ODT®. Available online: https://www.accessdata.fda.gov/drugsatfda_docs/label/2017/204326s002lbl.pdf (accessed on 20 July 2019).
- Full Prescribing Information Cotempla XR-ODT®. Available online: https://www.accessdata.fda.gov/drugsatfda_docs/label/2017/205489s000lbl.pdf (accessed on 20 July 2019).
- Full Prescribing Information Inderal® LA. Available online: https://www.accessdata.fda.gov/drugsatfda_docs/label/2011/016418s080,016762s017,017683s008lbl.pdf (accessed on 20 July 2019).
- Full Prescribing Information Dilacor-XR®. Available online: https://www.drugs.com/pro/dilacor-xr.htmlDilcaor (accessed on 20 July 2019).
- Full Prescribing Information Cardizem® CD. Available online: https://www.accessdata.fda.gov/drugsatfda_docs/label/2010/020062s040lbl.pdf (accessed on 20 July 2019).
- Amelian, A.; Winnicka, K. Polymers in pharmaceutical taste masking applications. Polimery 2017, 6, 417–496. [Google Scholar] [CrossRef]
- Faisal, W.; Farag, F.; Abdellatif, A.A.H.; Abbas, A. Taste masking approaches for medicines. Curr. Drug Deliv. 2018, 15, 167–185. [Google Scholar] [CrossRef]
- Taki, M.; Tagami, T.; Ozeki, T. Preparation of polymer-blended quinine nanocomposite particles by spray drying and assessment of their instrumental bitterness-masking effect using a taste sensor. Drug. Dev. Ind. Pharm. 2017, 43, 715–722. [Google Scholar] [CrossRef]
- Wasilewska, K.; Szekalska, M.; Ciosek-Skibinska, P.; Lenik, J.; Jacyna, J.; Markuszewski, M.; Winnicka, K. Ethylcellulose in organic solution or aqueous dispersion form in designing taste-masked microparticles by the spray drying technique with a model bitter drug: Rupatadine fumarate. Polymers 2019, 11, 522. [Google Scholar] [CrossRef]
- Ito, D.; Tackoe, J.; Rajabi-Siahboomo, A. Investigation of Taste Masking Performance of an Aqueous Ethylcellulose Dispersion (Surelease®) on Acetaminophen Granules. Poster (AAPS 2013). Available online: https://www.colorcon.com/products-formulation/all-products/download/311/552/34?method=view (accessed on 20 July 2019).
- Pimparade, M.B.; Morott, J.T.; Park, J.B.; Kulkarni, V.I.; Majumdar, S.; Murthy, S.N.; Lian, Z.; Pinto, E.; Bi, V.; Durig, T.; et al. Development of taste masked caffeine citrate formulations utilizing hot melt extrusion technology and in vitro-in vivo evaluations. Int. J. Pharm. 2015, 20, 167–176. [Google Scholar] [CrossRef]
- Rao, M.R.; Bhingole, R.C. Nanosponge-based pediatric-controlled release dry suspension of gabapentin for reconstitution. Drug. Dev. Ind. Pharm. 2015, 41, 2029–2036. [Google Scholar] [CrossRef]
- Full Prescribing Information Aciphex® Sprinkle™. Available online: https://www.accessdata.fda.gov/drugsatfda_docs/label/2014/020973s035204736s005lbl.pdf (accessed on 20 July 2019).
- Full Prescribing Information Dilatrate-SR. Available online: https://www.drugs.com/pro/dilatrate-sr.html (accessed on 20 July 2019).
- Full Prescribing Information Durlaza. Available online: https://www.drugs.com/pro/durlaza.html (accessed on 20 July 2019).
- Full Prescribing Information Enjuvia. Available online: https://www.drugs.com/pro/enjuvia.html (accessed on 20 July 2019).
- Full Prescribing Information Entocort EC. Available online: https://www.drugs.com/pro/entocort-ec.html (accessed on 20 July 2019).
- Full Prescribing Information Levbid®. Available online: https://www.rxlist.com/levbid-extended-release-drug.htm#indications (accessed on 20 July 2019).
- Orfiril Long Minitablets Information. Available online: https://ec.europa.eu/health/documents/community-register/2018/20180531140837/anx_140837_en.pdf (accessed on 20 July 2019).
- Full Prescribing Information Palladone. Available online: https://www.drugs.com/pro/palladone.html (accessed on 20 July 2019).
- Full Prescribing Information Pentasa. Available online: https://www.accessdata.fda.gov/drugsatfda_docs/label/2018/020049s031lbl.pdf (accessed on 20 July 2019).
- Full Prescribing Information Qsymia. Available online: https://www.drugs.com/qsymia.html (accessed on 20 July 2019).
- Full Prescribing Information Tegretol® -XR. Available online: https://www.accessdata.fda.gov/drugsatfda_docs/label/2009/016608s101,018281s048lbl.pdf (accessed on 20 July 2019).
- Full Prescribing Information Trokendi XR. Available online: https://www.drugs.com/pro/trokendi-xr.html (accessed on 20 July 2019).
- Kathe, K.; Kathpalia, H. Film forming systems for topical and transdermal drug delivery. Asian J. Pharm. Sci. 2017, 12, 487–497. [Google Scholar] [CrossRef]
- Joshi, M.; Sharma, V.; Pathak, K. Matrix based system of isotretinoin as nail lacquer to enhance transungal delivery across human nail plate. Int. J. Pharm. 2015, 478, 268–277. [Google Scholar] [CrossRef] [PubMed]
- Vipin, K.V.; Sarath, C.C.; Ann, R.A.; Premaletha, K.; Muriakose, M.R. Formulation and evaluation of an antifungual nail lacquer for onychomycosis. Br. Biomed. Bull. 2014, 2, 242–248. [Google Scholar]
- Šveikauskaitė, I.; Briedis, V. Effect of film-forming polymers on release of naftifine hydrochloride from nail lacquers. Int. J. Polym. Sci. 2017, 1, 7. [Google Scholar] [CrossRef]
- Baranowski, P.; Karolewicz, B.; Gajda, M.; Pluta, J. Ophthalmic drug dosage forms: Characterisation and research methods. Sci. World J. 2014, 2014, 1–14. [Google Scholar] [CrossRef]
- Paradkar, M.; Thakkar, V.; Soni, T.; Gandhi, T.; Gohel, M. Formulation and evaluation of clotrimazole transdermal spray. Drug Dev. Ind. Pharm. 2015, 41, 1718–1725. [Google Scholar] [CrossRef]
- Goudanavar, P.; Ambhore, N.; Hiremath, D.; Udupi, R. Comparative evaluation of polymer combination in the design of brimonidine tartrate ocular inserts. Indian Drugs 2012, 49, 30–35. [Google Scholar]
- Mundada, A.S.; Shrikhande, B.K. Design and evaluation of soluble ocular drug insert for controlled release of ciprofloxacin hydrochloride. Drug Dev. Ind. Pharm. 2006, 32, 443–448. [Google Scholar] [CrossRef]
- Khan, S.; Ali, A.; Singhavi, D.; Yeole, P. Preparation of ophthalmic insert of acyclovir using ethylcellulose rate-controlling membrane. J. Appl. Ther. Res. 2008, 6, 19–24. [Google Scholar]
- Kala, S.; Gurudiwn, P.; Juyal, D. Formulation and evaluation of besifloxacin loaded in situ gel for ophthalmic delivery. UKJPB. 2018, 6, 36–40. [Google Scholar] [CrossRef]
- Mortazavi, S.A.; Jaffariazar, Z.; Damercheli, E. Formulation and in-vitro evaluation of ocular ciprofloxacin-containing minitablets prepared with different combinations of carbopol 974P and various cellulose derivatives. IJPR. 2010, 9, 107–114. [Google Scholar] [PubMed]
- John, L.; Kumar, A.; Samuel, S. Formulation and evaluation of amlodipine transdermal patches using ethyl cellulose. Int. Res. J. Pharm. 2013, 4, 84–88. [Google Scholar] [CrossRef]
- Idrees, A.; Rahman, N.U.; Javaid, Z.; Kashif, M.; Aslam, I.; Abbas, K.; Hussain, T. In vitro evaluation of transdermal patches of flurbiprofen with ethyl cellulose. Acta Pol. Pharm. 2014, 71, 287–295. [Google Scholar] [PubMed]
- Cherukuri, S.; Ba, U.R. Formulation and evaluation of transdermal drug delivery of topiramate. Int. J. Pharm. Investig. 2017, 7, 10–17. [Google Scholar] [PubMed] [Green Version]
- Akhlaq, M.; Arshad, M.S.; Mudassir, A.M.; Hussain, A.; Kucuk, I.; Haj-Ahmad, R.; Rasekh, M.; Ahmad, Z. Formulation and evaluation of anti-rheumatic dexibuprofen transdermal patches: A quality-by-design approach. J. Drug. Targ. 2016, 24, 603–612. [Google Scholar] [CrossRef]
- Gupta, V.; Singh, S.; Srivarstava, M.; Ahmad, H.; Pachauri, S.D.; Khandelwal, K.; Dwivedi, P.; Dwivedi, A.K. Effect of polydimethylsiloxane and ethylcellulose on in vitro permeation of centchroman from its transdermal patches. Drug Deliv. 2016, 23, 113–122. [Google Scholar] [CrossRef]
- Huang, Y.; Dai, W.G. Fundamental aspects of solid dispersion technology for poorly soluble drugs. Acta Pharm. Sin. B 2014, 4, 18–25. [Google Scholar] [CrossRef]
- Tsunashima, D.; Yamashita, K.; Ogawara, K.; Sako, K.; Higaki, K. Preparation of extended release solid dispersion formulations of tacrolimus using ethylcellulose and hydroxypropylmethylcellulose by solvent evaporation metod. J. Pharm. Pharmacol. 2016, 68, 316–323. [Google Scholar] [CrossRef]
- Sadeghi, F.; Garekani, H.A.; Sadeghi, R. Comparison of ethylcellulose matrix characteristics prepared by solid dispersion technique or physical mixing. Daru J. Fac. Pharm. 2012, 11, 27–34. [Google Scholar]
- Ohara, T.; Kitamura, S.; Kitagawa, T.; Terada, K. Dissolution mechanism of poorly water-soluble drug from extended release solid dispersion system with ethylcellulose and hydroxypropylmethylcellulose. Int. J. Pharm. 2005, 302, 95–102. [Google Scholar] [CrossRef]
- Samba, M.; Kumawat, M.; Sambamoorthly, U. Formulation of sustain release solid dispersion of verapamil hydrochloride using ethyl cellulose and Eudragit RSPO. Int. J. Pharm. Pharm. Sci. 2011, 3, 116–122. [Google Scholar]
- Singh, M.N.; Hemant, K.S.Y.; Ram, M.; Shivakumar, H.G. Microencapsulation: A promising technique for controlled drug delivery. Res. Pharm. Sci. 2010, 5, 65–77. [Google Scholar] [PubMed]
- Chandrasekaran, P.; Kandasamy, R. Solid oral flexible formulations for pediatric and geriatric patients: Age-appropriate formulation platforms. Indian J. Pharm. Sci. 2018, 80, 14–25. [Google Scholar] [CrossRef]
- Murtaza, G. Ethylcellulose microparticles: A review. Acta Pol. Pharm. 2012, 69, 11–22. [Google Scholar]
- Pan-In, P.; Banlunara, W.; Chaichanawongsaroj, N.; Wanichwecharungruang, S. Ethyl cellulose nanoparticles: Clarithomycin encapsulation and eradication of H. pylori. Carbohydr. Polym. 2014, 109, 22–27. [Google Scholar] [CrossRef]
- Djerafi, R.; Swanepoel, A.; Crampon, C.; Kalombo, L.; Labuschagne, P.; Badens, E.; Masmoudi, Y. Supercritical antisolvent co-precipitation of rifampicin and ethyl cellulose. Eur. J. Pharm. Sci. 2017, 102, 161–171. [Google Scholar] [CrossRef]
- Villa Nova, M.; Gonçalves Mde, C.; Nogueira, A.C.; Herculano Lda, S.; Medina, A.N.; Bazotte, R.B.; Bruschi, M.L. Formulation and characterization of ethylcellulose microparticles containing L-alanyl-L-glutamine. Drug Dev. Ind. Pharm. 2014, 40, 1308–1317. [Google Scholar] [CrossRef]
- Shankar, N.B.; Kumar, R.P.; Kumar, N.U.; Brata, B.B. Development and characterization of bioadhesive gel of microencapsulated metronidazole for vaginal use. Iran J. Pharm. Res. 2010, 9, 209–219. [Google Scholar]
- Balzus, B.; Sahle, F.F.; Hönzke, S.; Gerecke, C.; Schumacher, F.; Hedtrich, S.; Kleuser, B.; Bodmeier, R. Formulation and ex vivo evaluation of polymeric nanoparticles for controlled delivery of corticosteroids to the skin and the corneal epithelium. Eur. J. Pharm. Biopharm. 2017, 115, 122–130. [Google Scholar] [CrossRef]
- El-Habashy, S.E.; Allam, A.N.; El-Kamel, A.H. Ethyl cellulose nanoparticles as a platform to decrease ulcerogenic potential of piroxicam: Formulation and in vitro/in vivo evaluation. Int. J. Nanomed. 2016, 11, 2369–2380. [Google Scholar]
- Begum, N.; Sailaja, A.K. Formulation and evaluation of Cox-2 inhibitor (etoricoxib) loaded ethyl cellulose nanoparticles for topical drug delivery. Nano. Biomed. Eng. 2018, 10, 1–9. [Google Scholar]
- Chassot, J.M.; Ribas, D.; Silveira, E.F.; Grünspan, L.D.; Pires, C.C.; Farago, P.V.; Braganhol, E.; Tasso, L.; Cruz, L. Beclomethasone dipropionate-loaded polymeric nanocapsules: Development, in vitro cytotoxicity, and in vivo evaluation of acute lung injury. J. Nanosci. Nanotechnol. 2015, 15, 855–864. [Google Scholar] [CrossRef] [PubMed]
- Tussionex® Drug Information: Description, User Reviews, Drug Side Effects, Interactions—Prescribing Information. Available online: https://www.rxlist.com/tussionex-drug.htm (accessed on 20 July 2019).
- Kasashima, Y.; Uchida, S.; Yoshihara, K.; Yasuji, T.; Sako, K.; Namiki, N. Oral sustained-release suspension based on a lauryl sulfate salt/complex. Int. J. Pharm. 2016, 30, 677–683. [Google Scholar] [CrossRef] [PubMed]
- Singh, I.; Rehni, A.K.; Kalra, R.; Joshi, G.; Kumar, M.; Aboul-Enein, H.Y. Ion exchange resins: Drug delivery and therapeutic applications. FABAD J. Pharm. Sci. 2007, 32, 91–100. [Google Scholar]
- Maulvi, F.A.; Soni, T.G.; Shah, D.O. Extended release of timolol from ethyl cellulose microparticles laden hydrogel contact lenses. Open Pharm. Sci. 2015, 2, 1–12. [Google Scholar] [CrossRef]
- Maulvi, F.A.; Lakdawala, D.H.; Shaikh, A.A.; Desai, A.R.; Choksi, H.H.; Vaidya, R.J.; Ranch, K.M.; Koli, A.R.; Vyas, B.A.; Shah, D.O. In vitro and in vivo evaluation of novel implantation technology in hydrogel contact lenses for controlled drug delivery. J. Control Release 2016, 226, 47–56. [Google Scholar] [CrossRef]
- Arici, M.; Topbas, O.; Karavana, S.Y.; Ertan, G.; Sariisik, M.; Ozturk, C. Preparation of naproxen-ethyl cellulose microparticles by spray drying technique and their application to textile materials. J. Microencapsul. 2014, 31, 654–666. [Google Scholar] [CrossRef]
- Patel, S.; Patel, H.; Seth, A. Microsponge drug delivery system: An overview. JGPT. 2010, 2, 1–9. [Google Scholar]
- Aloorkar, N.; Kulkarni, A.; Ingale, D.; Patil, R. Microsponges as innovative drug delivery systems. Int. J. Pharm. Sci. Nanotechnol. 2012, 5, 1597–1606. [Google Scholar]
- Kappor, D.; Patel, M.; Vyas, R.; Lad, C.; Tyagi, B. A review on microsponge drug delivery system. JDDT. 2014, 4, 29–35. [Google Scholar] [CrossRef]
- Bothiraja, C.; Gholap, A.D.; Shaikh, K.S.; Pawar, A.P. Investigation of ethyl cellulose microsponge gel for topical delivery of eberconazole nitrate for fungal therapy. Ther. Deliv. 2014, 5, 781–794. [Google Scholar] [CrossRef] [PubMed]
- Jelvehgari, M.; Siahi-Shadbad, M.R.; Azarmi, S.; Martin, G.P.; Nokhodchi, A. The microsponge delivery system of benzoyl peroxide: Preparation, characterization and release studies. Int. J. Pharm. 2006, 308, 124–132. [Google Scholar] [CrossRef] [PubMed]
- Yadav, E.; Rao, R.; Kumar, S.; Mahant, S.; Vohra, P. Microsponge based gel of tea tree oil for dermatological microbial infections. Nat. Prod. J. 2018, 8, 1. [Google Scholar] [CrossRef]
- Pawar, A.P.; Gholap, A.P.; Kuchekar, A.B.; Bothiraja, C.; Mali, A.J. Formulation and evaluation of optimized oxybenzone microsponge gel for topical delivery. J. Drug. Deliv. 2015, 1–9. [Google Scholar] [CrossRef]
- Zaman, M.; Qureshi, S.; Sultana, K.; Hanif, M.; Mahmood, A.; Shaheryar, Z.A.; Gulzar, F.; Barkat, K.; Abdel-Daim, M.M. Application of quasi-emulsification and modified double emulsification techniques for formulation of tacrolimus microsponges. Int. J. Nanomed. 2018, 13, 4537–4548. [Google Scholar] [CrossRef]
- Obiedallah, M.M.; Abdel-Mageed, A.M.; Elfaham, T.H. Ocular administration of acetazolamide microsponges in situ gel formulations. SPJ 2018, 6, 909–920. [Google Scholar] [CrossRef]
- Bharathala, S.; Sharma, P. Biomedical applications of nanoparticles. Recent Trends and Future Perspectives. In Nanotechnology in Modern Animal Biotechnology; Maurya, P.K., Singh, S., Eds.; Springer Singapore: Singapore, 2019; pp. 113–132. [Google Scholar]
- Liu, M.; Zhang, Y.; Sun, S.; Khan, A.R.; Ji, J.; Yang, M.; Zhai, G. Recent advances in electrospun for drug delivery purpose. J. Drug Target. 2019, 27, 270–282. [Google Scholar] [CrossRef]
- Goyal, R.; Macri, L.K.; Kaplan, H.M.; Kohn, J. Nanoparticles and nanofibers for topical drug delivery. J. Control Release 2016, 28, 77–92. [Google Scholar] [CrossRef]
- Li, H.; Liu, K.; Williams, G.R.; Wu, J.; Wu, J.; Wang, H.; Niu, S.; Zhu, L.M. Dual temperature and pH responsive nanofiber formulations prepared by electrospinning. Colloids Surf. B 2018, 171, 142–149. [Google Scholar] [CrossRef]
- You, D.G.; Li, X.Y.; Wang, X.; Yang, J.H.; Bligh, S.A.; Williams, G.R. Nanofibers fabricated using triaxial electrospinning as zero order drug delivery systems. ACS Appl. Mater. Interfaces 2015, 7, 18891–18897. [Google Scholar] [CrossRef]
- Illangakoon, U.E.; Yu, D.G.; Ahmad, B.S.; Chatterton, N.P.; Williams, D.R. 5-Fluorouracil loaded Eudragit fibers prepared by electrospinning. Int. J. Pharm. 2015, 495, 895–902. [Google Scholar] [CrossRef] [PubMed]
- Lu, H.; Wang, Q.; Li, G.; Qiu, Y.; Wei, Q. Electrospun water-stable zein/ethyl cellulose composite nanofiber and its drug release properties. Mater. Sci. Eng. C Matter. Biol. Appl. 2017, 74, 86–93. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Deng, L.; Zhang, C.; Feng, F.; Zhang, H. Tunable physical properties of ethylcellulose/gelatin composite nanofibers by electrospinning. J. Agric. Food Chem. 2018, 28, 1907–1915. [Google Scholar] [CrossRef] [PubMed]
- Bagul, U.; Gujar, K.; Dhat, S.; Aphale, S.; Bhavsar, M. In vitro study of mucoadhesive strength of polymers for mucoadhesive drug delivery systems. Int. J. Curr. Pharm. Res. 2009, 1, 42–46. [Google Scholar]
- Abruzzo, A.; Nicoletta, F.P.; Dalena, F.; Cerchiara, T.; Luppi, B.; Bigucci, F. Bilayered buccal films as child-appropriate dosage form for systemic administration of propranolol. Int. J. Pharm. 2017, 5, 257–265. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Liu, Y.; Li, W.; Gao, P.; Xiang, D.; Ren, X.; Liu, D. Mucoadhesive buccal film containing ornidazole and dexamethasone for oral ulcers: In vitro and in vivo studies. Pharm. Dev. Technol. 2019, 24, 118–126. [Google Scholar] [CrossRef] [PubMed]
- Laffleur, F.; Krouská, J.; Tkacz, J.; Pekař, M.; Aghai, F.; Netsomboon, K. Buccal adhesive films with moisturizer-the next level for dry mouth syndrome? Int. J. Pharm. 2018, 550, 309–315. [Google Scholar] [CrossRef] [PubMed]
- Ammar, H.O.; Ghorab, M.M.; Mahmoud, A.A.; Shahin, H.I. Design and in vitro/in vivo evaluation of ultra-thin mucoadhesive buccal film containing fluticasone propionate. AAPS PharmSciTech. 2017, 18, 93–103. [Google Scholar] [CrossRef]
- Mukherjee, D.; Bharath, S. Design and characterization of double layered mucoadhesive system containing bisphosphonate derivative. ISRN Pharm. 2013, 19, 604690. [Google Scholar] [CrossRef]
- Roh, J.; Han, M.; Kim, K.N.; Kim, K.M. The in vitro and in vivo effects of a fast-dissolving mucoadhesive bi-layered strip as topical anesthetics. Dent. Mater. J. 2016, 35, 601–605. [Google Scholar] [CrossRef] [Green Version]
- Phaechamud, T.; Mahadlek, J. Solvent exchange-induced in situ forming gel comprising ethyl cellulose-antimicrobial drugs. Int. J. Pharm. 2015, 494, 381–392. [Google Scholar] [CrossRef] [PubMed]
- Sallam, A.S.; Hamudi, F.F.; Khalil, E.A. Effect of ethylcellulose and propylene glycol on the controlled-release performance of glyceryl monooleate–mertronidazole periodontal gel. Pharm. Dev. Technol. 2015, 20, 159–168. [Google Scholar] [CrossRef] [PubMed]
- Pathak, K.; Sharma, V.; Akhtar, N.; Rastogi, P. Localization of fluconazole in oral cavity by preferential coating of buccoadhesive tablet for treatment of oral thrush. Int. J. Pharm. Invest. 2016, 6, 106–115. [Google Scholar] [CrossRef] [PubMed] [Green Version]







| Organic EC products |
| Ethocel™ |
| Available in many varieties that differ in grade and viscosity (e.g., Ethocel™ Standard Grade 4 Premium, 7 Premium, 10 Premium, 20 Premium, 45 Premium, 100 Premium). Ethocel™ are white to light-tan granular powders in physical appearance with bulk density and specific gravity of about 0.4 g/cm3 and 1.12–1.15 g/cm3 respectively and glass transition temperatures range between 129 and 133 °C. They dissolve in a wide range of solvents such as aliphatic alcohols, chlorinated solvents, and natural oils. They are practically insoluble in glycerin, propylene glycol, and water. Films made from Ethocel™ are tough, with high tensile strength and high flexibility even at low temperatures. They can be combined with water soluble polymers such as methylcellulose and hypromellose (HPMC) in aqueous coating liquids. They are characterized by thermoplastic nature and ability to soften at 135–160 °C which makes them versatile in pharmaceutical hot melt extrusion processes. They ensure desired drug release properties and improved bioavailability of especially poorly soluble drugs [12]. |
| Aqualon™ ethylcellulose |
| During preparation, the substitution of ethoxyl groups is controlled to obtain commercially useful range of 48–52% ethoxyl (or 2.3–2.6 ethoxyl groups out of a theoretical maximum of 3.0) per anhydroglucose unit. Over this ethoxyl range, Aqualon™ ethylcellulose is classified into three ethoxyl types: N (low substitution), T (mid substitution), and X (high substitution). The improved compressible grade (Aqualon™ T10) was developed with optimized compactability (high ethoxyl content and low viscosity) and good powder flow. The grades of Aqualon™ ethylcellulose are compliant with the monograph requirements of the United States Pharmacopoeia (USP) and the European Pharmacopoeia (Ph. Eur.) [24]. |
| Aqueous dispersion of ethylcellulose |
| Surelease® |
| Surelease® is a family of fully formulated, aqueous dispersion products which constitute plasticized aqueous dispersions of EC with 25% (w/w) solid content available in four types: E-7-19029, E-7-19030, E7-19040, E-7-19050. They are plasticized (depending on type) with dibutyl sebacate (3.5%-E-7-19029, E-7-19030) and oleic acid (1.9%-E7-19040). Surelease® is produced in the following steps: EC is blended with plasticizer, then extruded and melted. The molten plasticized EC is then directly emulsified in ammoniated water in a high-shear mixing device under pressure. Ammonium oleate is formed in situ in order to stabilize and form the dispersion of plasticized EC particles. Then, purified water is added to achieve the final solids content. Applications of Surelease® include beads and particles coating, matrix granulation (the dispersion can be used as a binder in wet granulation for the production of free-flowing granules, which can subsequently be compressed into tablets), taste-masking coating, or nutritional enteric coating [25]. |
| Aquacoat® ECD |
| It contains primarily EC with a surfactant and a stabilizer from the emulsion stage (sodium lauryl sulfate (SLS) and cetyl alcohol (CA)). Depending on type it consists of EC (24.5–29.5%), CA (1.7–3.3%), and SLS (0.9–1.7%). Traces of dimethylopolysiloxane to enhance foaming during distillation may also be present. EC is dissolved in a water-immiscible organic solvent with CA addition as a dispersion stabilizer. The solution is then emulsified into an aqueous SLS solution. The resulting crude emulsion is homogenized to yield a submicron emulsion which is then distilled to remove the organic solvent and water to yield a solid dispersion. EC is present in the dispersion as spherical particles in the size range of 0.1–0.3 μm. It exists as a milky white liquid with the characteristic odor of EC. Product does not contain plasticizer. Recommended plasticizers include dibutyl sebacate, acetylated monoglycerides, triacetin or glyceryl triacetate, acetyltriethyl citrate, and triethyl citrate. It is used for the aqueous film coating of solid dosage forms to extend drug release, taste mask, or to protect against moisture [26]. |
|
Product (Manufacturer) | Drug | Dosage Form | EC Role | Reference |
|---|---|---|---|---|
|
Aciphex® Sprinkle™ (Eisai Management Co., Ltd.) | rabeprazole | granules in capsule | coating, delayed release | [53] |
|
Adzenys XR-ODT™ (Neos Therapeutics) | amphetamine | extended release orally disintegrating tablet with beads | coating, extended release | [41] |
| Advagraf XL™ | tacrolimus | capsules | extended release | [30] |
|
Cotempla XR-ODT® (Neos Therapeutics) | methylphenidate | extended release orally disintegrating tablet with beads | coating, extended release | [42] |
|
Cardizem CD® (Abbot Laboratories) | diltiazem | tablets in capsule | coating, extended release | [45] |
|
Dilacor XR® (Actavis Pharma, Inc.) | diltiazem | tablets in capsule | extended release | [44] |
|
Dilatrate-SR
(Epic Pharma, LLC) | diltiazem | beads in capsule | extended release | [54] |
|
Durlaza
(New Haven Pharmaceuticals, Inc.) | acetylsalicylic acid | capsules | extended release | [55] |
|
Enjuvia (Teva Pharmaceuticals USA, Inc.) | estrogens | coated tablets | extended release | [56] |
|
Entocort EC (Astra Zeneca) | budesonide | coated granules in capsules | sustained release | [57] |
|
Inderal® LA
(Wyeth Pharmaceuticals, Inc.) | propranolol | granules in capsules | coating, extended release | [43] |
|
Levbid® (Mylan Pharmaceuticals Inc.) | hyoscyamine | tablets | extended release | [58] |
|
Metadate CD® (UCB Manufacturing, Inc.) | methylphenidate | granules in capsule | coating, extended release | [37] |
|
Micro-K® (KV Pharmaceutical) | potassium | microcapsules in capsules | coating, extended release | [34] |
|
Lamictal® XR
(GSK) | lamotrigine | tablets | coating, extended release | [38] |
|
Orfiril Long
(Desitin Arzneimittel GmbH) | natrii valproas | minitablets | extended release | [59] |
|
Palladone
(PF Laboratories Inc.) | hydromorphone | capsules | extended release | [60] |
|
Pentasa® (Ferring GmbH) | mesalamine | granules | prolonged release | [61] |
|
Qsymia® (Vivus, Inc.) | phentermine, topiramate | capsules | extended release | [62] |
|
Tegretol® XL
(Novartis) | carbamazepine | tablets | matrix, extended release | [63] |
|
Theo-24® (UCB Pharma, Inc.) | theophylline | beads in capsule | coating, extended release | [35] |
|
Trokendi XR™ (Catalent Pharma Solutions) | topiramate | capsules | extended release | [64] |
| Drug Dosage Form | Active Substance | Used Excipients | EC Role | Reference |
|---|---|---|---|---|
| Ocular formulations | ||||
| ocular inserts | brimonidine | sodium alginate, EC | hydrophobic, sustained release coating | [71] |
| ocular inserts | ciprofloxacin | gelatin, EC | rate-controlling, increasing residence time membrane | [72] |
| ocular insert | acyclovir | sodium alginate, EC | rate-controlling membrane | [73] |
| in situ ophthalmic hydrogel | besifloxacin | sodium alginate, xanthan gum, EC | increase the pre-corneal residence time | [74] |
| minitablet | ciprofloxacin | HPMC, sodium carboxymethyl cellulose, EC, hydroxyethyl cellulose, carbopol | sustained release | [75] |
| Transdermal formulations | ||||
| transdermal patch | amlodipine | EC | sustained release | [76] |
| transdermal patch | flurbiprofen | EC, propylene glycol, dibutyl phthalate | constant drug release | [77] |
| transdermal delivery system | topiramate | EC, povidone, Eudragit L 100, carbopol | extended release | [78] |
| transdermal patch | dexibuprofen | EC, povidone, di-N-butyl phthalate | matrix formation | [79] |
| transdermal patch | centchroman | EC, polydimethylsiloxane, propylene glycol, Di-n-butyl-phthalate | Film-forming polymer | [80] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wasilewska, K.; Winnicka, K. Ethylcellulose–A Pharmaceutical Excipient with Multidirectional Application in Drug Dosage Forms Development. Materials 2019, 12, 3386. https://doi.org/10.3390/ma12203386
Wasilewska K, Winnicka K. Ethylcellulose–A Pharmaceutical Excipient with Multidirectional Application in Drug Dosage Forms Development. Materials. 2019; 12(20):3386. https://doi.org/10.3390/ma12203386
Chicago/Turabian StyleWasilewska, Katarzyna, and Katarzyna Winnicka. 2019. "Ethylcellulose–A Pharmaceutical Excipient with Multidirectional Application in Drug Dosage Forms Development" Materials 12, no. 20: 3386. https://doi.org/10.3390/ma12203386
APA StyleWasilewska, K., & Winnicka, K. (2019). Ethylcellulose–A Pharmaceutical Excipient with Multidirectional Application in Drug Dosage Forms Development. Materials, 12(20), 3386. https://doi.org/10.3390/ma12203386