1. Introduction
Synthetic colorants are widely applied chemicals in the textile sector. A diverse variety of these complex dyes is available on the international market with a range of properties and physiognomies. A significant amount of these toxic dyes is directly dumped every year into the aquatic bionetwork directly in the form of industrial effluents. The multifaceted aromatic chemistry of such complex dyes offers resistance to biodegradation, resulting in incomplete mineralization, thus metabolites of varying toxicity are produced [
1] which therefore increase the environmental health concerns. Presently, around 10,000 types and 700,000 tons of different dyes and pigments are manufactured worldwide annually with ever increasing trend [
2]. The reactive dyes class represents a major share of commercial grade synthetic colorants due to their excellent capability of forming covalent bonding between the reactive groups of the dyes and the functional groups of substrates like cellulose in the case of textile products [
3]. Dye-bath facilities lose a significant portion of these dyes due to hydrolysis reactions. The constituents of these organic chemicals are enormously noxious, consequentially leading towards skin and skin sensitivity, mutagenicity, carcinogenicity and eye irritation, etc. [
4,
5,
6]. Their threat level increases with their stable chemical nature due to aromatic rings and other intermediates produced on fragmentation. These health issues necessitate the treatment of such hazardous dyes in effluents prior to their dumping into the environment.
Currently, conventional technologies being adopted for the treatment of reactive dyes include photocatalytic degradation [
7], enzymatic action [
8], chemical coagulation/flocculation [
9], ion exchange [
10] and membrane separation [
11], etc. At an industrial scale these conventional treatment processes have certain limitations: e.g., phase, shape, band gap, light source, structure, reactor design and catalyst recovery [
12], the temperature specificity and alkaline conditions of laccase enzyme [
13], residual sludge generation during chemical coagulation [
14], slow pore diffusion, low accessible flow rates, high pressure drop and flow channeling in ion exchange [
15], and chemical incompatibility in membrane separation [
16], etc. Therefore, it is a pressing need for a treatment technology to solve the problem in a sustainable and cost-effective manner.
Adsorption appears to be an economical, effective and eco-friendly technology for the elimination of reactive dyes from effluents when compared with other methods due to its numerous advantages, which include low cost, ease of design at a large scale, facile manipulations, resistance to contaminants, higher efficiency rate, and an aptitude to cope with dilute as well as concentrated dye effluents [
17]. Generally, a wide range of adsorbents obtained from variety of sources such as clay [
18], nanoparticles [
19], nanotubes [
20], agricultural- and forest-derived wastes [
21,
22], activated carbon [
23], graphene composites [
24], zeolites [
25], chitosan [
26], and a number of polymeric adsorbents [
27] have been employed for the removal of reactive dyes. The high surface area, good adsorption capacity, mechanical and chemical stability of activated carbon (AC) ranks it as a promising substitute for existing dye removal techniques. The scalable production of AC can be ensured by using locally available waste biomass as raw precursor material, however the duration of treatment time, high temperature conditions and need for the continuous provision of a gaseous atmosphere could be possible reasons for the increased of these materials, compared to commercial AC. An efficient activated carbon that can be valorized at comparatively low cost, readily available locally and that can be regenerated easily are the most challenging characteristics demanded required by researchers.
In the current study, sawdust of widely available Melia azedarach was used as precursor raw material to produce AC on a laboratory scale using orthophosphoric acid as an activating agent. Melia azedarach is a locally grown woody plant mainly employed for obtaining hard lignocellulosic wood for making furniture and other construction industry-related items. The manufactured novel activated carbon was employed to treat an aqueous solution of reactive orange 16 (RO16) dye in batch reactors. The adsorbent material was characterized, and its surface charge and surface functional groups were determined. Surface area, pore volume and pore width diameter were also analyzed. Desorption and cost analyses of the studied treatment system were conducted to determine its suitability for field application.
3. Materials and Methods
3.1. Chemicals and Reagents
Orthophosphoric acid (H
3PO
4), nitric acid (HNO
3), sodium hydroxide (NaOH) and reactive orange 16 (RO16) dye were purchased from Sigma Aldrich (Darmstadt, Germany) and used without further purification. RO16 dye selected as a model pollutant has the key properties listed in
Table 6. A stock solution of RO16 dye was prepared by dissolving an appropriate amount of dye in 1000 mL of deionized water. Working as well as standard solutions for calibration curve generation were prepared by subsequent dilutions.
3.2. Preparation of Activated Carbon (AC)
Sawdust of Melia azedarach was acquired from a local market situated in Hazara (Abbottabad, Pakistan). The biomass was ground in a lab scale crusher to obtain uniform particle size (40–60 mesh size). Powdered material was washed multiple times with deionized water to remove any adhered impurities. The powdered biomass was then oven dried at 105 °C until no further weight loss was observed followed by packing in airtight containers for further use. The activated carbon preparation process was accomplished in two phases: (i) the first phase involved the mixing of H3PO4 (conc.) with raw powdered material having impregnation ratio of 2 in an orbital shaker at 30 °C while an agitation speed of 150 rpm was maintained for 2 h. The homogenized mixture was then exposed to modest heat stress (100 °C) inside ac muffle furnace under inert atmosphere of N2 for 12 h; (ii) in the second phase, the material was exposed to a high temperature (400 °C) for 30 min under a CO2 atmosphere. The obtained activated carbon was thoroughly washed with deionized water until neutralization of residual acid was achieved (pH > 6.5). Thereafter the produced carbonaceous material was oven dried at 105 °C until a constant weight was achieved and it was labelled as MA-AC400.
3.3. Characterization of MA-AC400
Infrared spectra of the MA-AC400 were obtained using a Fourier transform infrared spectrometer (Alpha Bruker, Karlsruhe, Germany). For FT-IR study, ground MA-AC400 was gently mixed with potassium bromide (KBr) to get a translucent pelletized sample and FTIR spectra runs were carried out in the 500 to 4000 cm−1 wavenumber range. The surface morphology and elemental composition of the MA-AC400 was examined using a scanning electron microscope (SEM) equipped with an energy dispersive X-ray spectroscopy (EDS) system using a JEOL JSM 5910 instrument (INCA 200, Oxford Instruments, High Wycombe, UK). Dynamic light scattering (DLS) measurements were performed at 25 °C in water on a Zetasizer Nano ZSP from Malvern Instruments Limited (Malvern, UK) using the 173° angle non-invasive backscatter mode and the M3-phase analysis light scattering mode, respectively. The instrument has a red 10.0 mW 633 nm He−Ne laser. The accuracy was better than ±2% on NIST-traceable latex standards. The multiple peak high-resolution fitting procedure was used to obtain the particle size distribution from the autocorrelation function. Intensity distribution curves for AC were considered for particle size distribution. Measurements were performed with AC at 0.1 mg mL−1 concentration. Fluorescence spectroscopy was performed at room temperature on a Fluoromax-4 spectrophotometer (Horiba, NY, USA). Measurements were taken by using the quartz cuvette with dimensions 10 × 10 mm, transparent from all sides. The slit width was fixed at 3 mm with a band pass of 12.75 nm. 0.1 mg mL−1 of as synthesized AC carbon was dispersed in distilled water to measure the fluorescence using a 50 Hz ultrasonicator (FSF-020S, Huanghua Faithful Instrument Co., LTD, Huanghua, Hebei Province, China) for 6 h.
3.3.1. pHPZC Determination and Boehm Titration
For pH at point of zero charge (pH
pzc) determination [
22], the surface charge distribution was analyzed by addition of 25 mg MA-AC400 adsorbent to 50 mL of 0.1 M NaCl solution and the pH was adjusted between 3–10 by addition of 0.1 M HCl and NaOH solutions. These mixtures were sealed and agitated at 150 rpm for 24 h under 30 °C. After separation, the final pH of each solution was measured quickly. pH
PZC corresponds to the net electrical neutrality of the adsorbent obtained from the intersection point by plotting
[
66].
3.3.2. Textural Properties of MA-AC400
The Boehm titration method [
67] was employed for the determination of various acidic/basic functional groups distributed onto the surface of MA-AC400 adsorbent. Briefly 1.0 g of MA-AC400 and 15 mL solution of NaHCO
3 (0.1 M), Na
2CO
3 (0.05 M) and NaOH (0.1 M for acidic groups) and 0.1 HCl for basic group/sites, respectively, were kept at room temperature for 2 days. Later, the aqueous solutions were back titrated with NaOH (0.1 M) and HCl (0.1 M) for basic and acid groups, respectively. The type and number of acidic groups were calculated by bearing in mind that NaOH neutralizes carboxylic acid, lactonic and phenolic groups while Na
2CO
3 neutralizes carboxylic acid and lactonic groups and that NaHCO
3 neutralizes only carboxylic acid groups. The quantity of functional groups containing oxygen, F
x, is calculated using Equations (1) and (2):
where F
x (mmol g
−1) V
bx, V
ex, M
t and DF are the total oxygen-containing functional groups, volume of titrant used to titrate the blank, volume of the titrant being used to titrate the extract, molarity of the titrant used and dilution factor, respectively.
The textural properties of MA-AC400 were evaluated through frequently adopted nitrogen adsorption-desorption isotherm analysis at 77.30 K using a GEMINI VII 2390 surface area analyzer (Micromeritics Instrument Corp, Norcross, GA, USA). The Brunauer–Emmett–Teller (BET) method was adopted to calculate the specific surface areas (S
BET). Additionally, the micropore surface area (S
micro) and micropore volume (V
micro) were calculated by De Boer’s t-plot method. The external surface area (S
external) was calculated as the difference between SBET and S
micro. The total pore volume (V
total) was estimated in terms of the amount of nitrogen gas adsorption at a relative pressure (P/P
0) of 0.98 by using the Horvath–Kawazoe method [
68]. The micropore area and volume were calculated using the t-plot method [
69].
3.4. Batch Reactor Studies
Batch adsorption investigations were conducted in aqueous phase mixtures of MA-AC400 and RO16 dye and all adsorption studies were performed at 30 ± 2 °C. A series of 50 mL Erlenmeyer flasks containing 20 mL of RO16 solution having variable initial concentrations of 20 to 100 mg L
−1 and MA-AC400 dosage of 0.40 to 2 g L
−1 were employed at a chosen pH of 2 to 10 and pH were adjusted by adding 0.1 M HCl or 0.1 M NaOH solutions. These flasks were shaken using shaker equipped with digital fuzzy control system (Wise Cube, Seoul, Korea) at 150 rpm for a specific period of contact time (10–600 min) in order to achieve equilibration time. The filtrates were then analyzed using UV-Visible double beam spectrophotometer (T80
+ PG instruments, Leicestershire, UK) at λ
max = 492 nm under calibrated conditions. The adsorption capacities of MA-AC400 at time t (q
t, mg g
−1) and at equilibrium time (q
e, mg g
−1) and percentage RO16 dye removal of MA-AC400 were calculated by employing the following Equations (3)–(5):
where V (L), m (g), C
0, C
t, and C
f are the volume of solution, mass of adsorbent, initial, at any time “t” and final RO16 dye concentration in aqueous solution (mg L
−1), respectively.
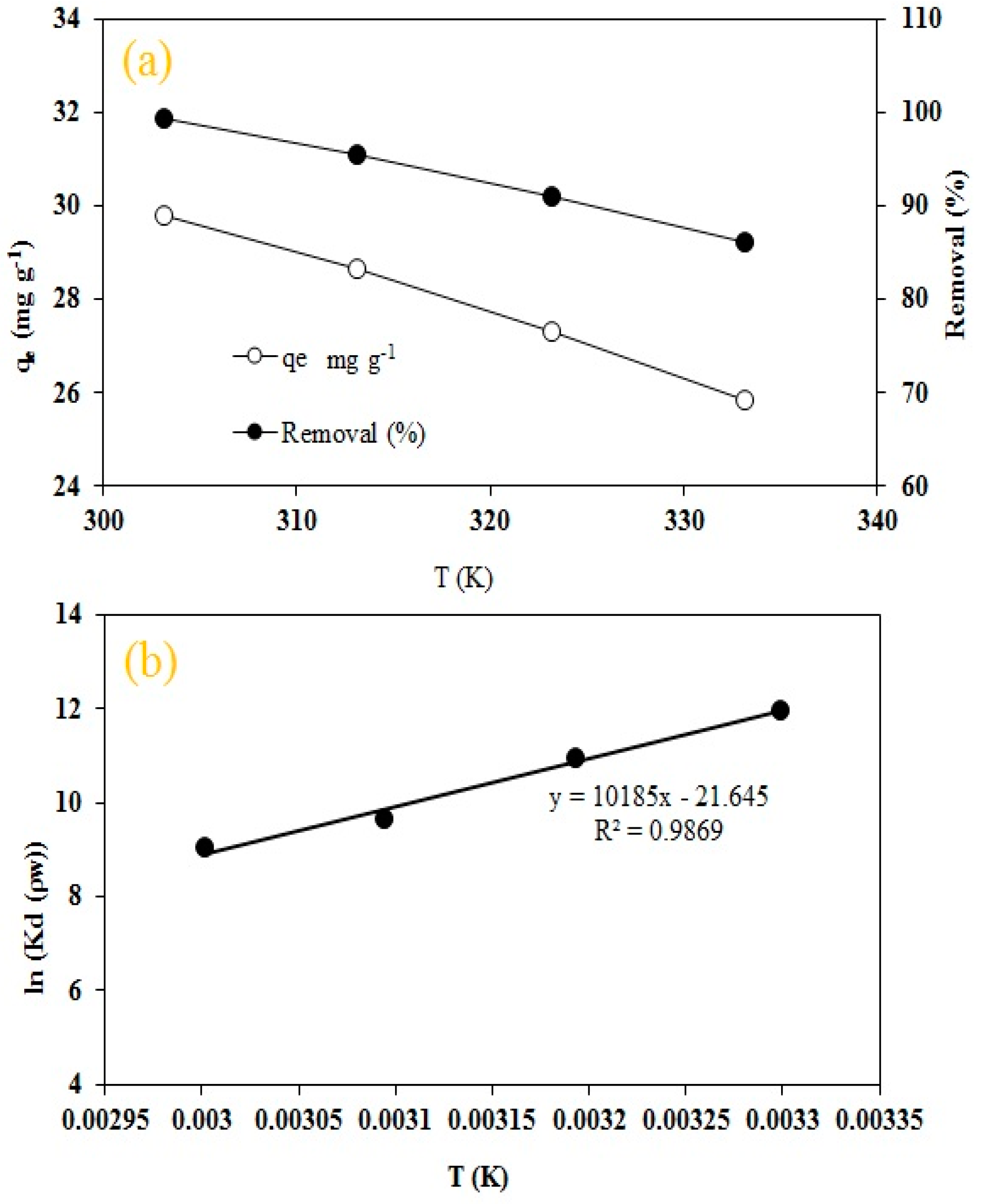
3.5. Thermodynamic Studies
Thermodynamic parameters were calculated to evaluate thermodynamically the nature of adsorption in the RO16 and MA-AC400 system. The thermodynamic equilibrium constant was calculated using Equation (6). To make it dimensionless, this equilibrium constant was multiplied by solution (water) density (ρw ≈ 1000 g L
−1) [
22,
70,
71]. Changes in enthalpy ΔH (kJ mol
−1) and entropy ΔS (J mol
−1 K
−1) were obtained via intercept and slope of Van’t Hoff plot lnKd(ρw) against 1/T (Equation (7)) and finally the Gibb’s free energy ΔG (kJ mol
−1) was determined using Equation (8):
3.6. Isotherm Studies
Adsorption isotherm models are mathematical tools extensively employed to designate the type of molecule coverage, adsorbent surface homogeneity/heterogeneity, binding energies, physical/chemical nature of interaction and adsorption heat of molecules. The equilibrium data of RO16 dye on MA-AC400 was evaluated using linearized forms of the widely applied Langmuir and Freundlich models and the results of parameters and constants were calculated. The Langmuir isotherm [
72] assumes a monolayer coverage of adsorbate molecules onto the surface of an adsorbent predominantly via chemical interaction. The Freundlich isotherm [
73] is generally applied to heterogeneous surfaces assuming a multilayer adsorption of adsorbate molecules onto the surface of adsorbent and binding sites are not equally distributed. The linearized forms of the Langmuir and Freundlich isotherm models are given by Equations (7) and (8), respectively:
The linear plot of 1/q
e vs. 1/C
e yields Langmuir parameters of calculated q
e cal (mg g
−1) and KL (L mg
−1). KL measures the adsorption affinity of adsorbate (RO16 dye molecules) to the adsorption sites of adsorbent, i.e., MA-AC400 (in current study). Freundlich isotherm can be described as follows:
where KF is the Freundlich constant which is related to the affinity between interacting species and 1/n measures the nature of the adsorption for which the heterogeneity increases as the value gets nearer to zero [
74]. KF and 1/n can be determined from intercept and slope of plot log (q
e) versus log (C
e) respectively.
Closeness between predicted adsorption by both linear models and experimental adsorption of MA-AC400 for RO16 was determined through normalized standard deviation, which is expressed as:
Error Function Analysis for Linear Isotherms
Linear regression is one of the distinct and widely adopted statistical tools employed for connecting and validating experimental and modeled data in adsorption studies. These error functions are widely employed to cross-examine the findings of mathematical, empirical and semi-empirical models with those of experimental findings in order to maintain consistency and credibility of scientific data. Numerous research studies have revealed that the error range of experimental data is generally altered throughout the conversion of adsorption isotherms into forms after linearization [
75]. In our current work, five different error functions including coefficient of determination (R
2) [
22], chi-square error (χ
2) [
76], sum of absolute error (SAE) [
77], Marquardt’s percent standard deviation (MPSD) [
78] and HYBRID functional error (HYBRID) [
79] were calculated and the goodness of fit for Langmuir and Freundlich was suggested. All error functions are represented by Equations (12)–(16):
3.7. Kinetic Modelling
Adsorption kinetic studies are crucial in the optimization of adsorption process conditions for contaminant adsorption. The adsorption kinetics of RO16 dye were investigated using three widely employed models, namely the pseudo-first order (PFO), pseudo-second order (PSO) and intraparticle diffusion model (IPD), which can be represented in Equations (17)–(19). Adsorption kinetics were studied at 20, 40, 60, 80 and 100 mg L
−1 of initial dye concentrations over a time duration of 180 min:
Here K1, K2 and Kpi represent PFO, PSO and IPD constants. Ci determines the boundary layer thickness.
3.8. Desorption Study
To investigate the reusability of spent MA-AC400, a desorption study was performed. In brief, RO16-loaded MA-AC400 adsorbent was oven dried and added into a 50 mL flask. Eluents, namely 0.5, 1.0 and 1.5 M strength solution of 30 mL of each sodium hydroxide (NaOH) and potassium hydroxide (KOH), respectively, were added into the flasks. These mixtures were agitated at 150 rpm for 8 h at 30 °C in shaking incubator (Wise Cube). Afterwards, the adsorbent was removed via vacuum filtration and the quantity of RO16 dye desorbed into the solution was analyzed by spectrophotometry. The desorption efficiency (%) of MA-AC400 was calculated using Equation (20) [
36]:
4. Conclusions
In this study, Melia azedarach was used as the precursor biomass for the production of H3PO4- activated MA-AC400 adsorbent at a comparatively low temperature exposure of 400 °C and contact time of 30 min, biomass and acid ratio 2, compared to the published reports. MA-AC400 was better characterized by SEM, EDS, FTIR, DLS and fluorescence measurements. The fluorescent MA-AC400 is characterized as fine particles with a majority of particles in the range of 0.12 to 0.30 µm and emission fluorescence at 497 nm. The textural properties of MA-AC400 correspond to a mesoporous AC with a BET surface area of 293.13 m2 g−1 and a Langmuir surface area of 440.83 m2 g−1, pore volume of 0.149 cm3 g−1, and average pore width (4V/A by BET) of 20.3 Å. Moreover Boehm titration results confirm the acidic character of MA-AC400 with total acidic functional groups (3.5 mmol g−1) and total basic groups of 0.45 mmol g−1. The number of phenolic groups was found to be higher, i.e., 1.7 mmol g−1, followed by carboxylic acid (0.30 mmol g−1) and lactonic (1.5 mmol g−1) groups. The chemical interactions between RO16 and MA-AC400 were evidenced from EDS and FTIR analyses. The experimental adsorption capacity of MA-AC400 for RO16 was observed to be 58.54 mg g−1 with the adsorbent dosage of 0.4 g L−1 and solution pH of 6.2. The RO16 adsorption over MA-AC400 follows PSO and intraparticle diffusion kinetics at lower and higher initial concentrations of dye, respectively. Equilibrium data reveal a better fit to the Langmuir isotherm rather than the Freundlich isotherm with higher R2 and maximum Langmuir adsorption capacity predicted as 65.79 mg per gram of MA-AC400. Error analysis confirmed the Langmuir chemisorption for RO16 with higher coefficient of determination and lower values of chi-square error (χ2), sum of absolute error (SAE), Marquardt’s percent standard deviation (MPSD) and HYBRID functional error (HYBRID). MA-AC400 adsorbent could be used under suitable conditions to remove anionic RO16 dye and considered to be effectively applied in pilot scale projects. Economic cost of MA-AC400 adsorbent was calculated to be 1388.33 PKR ($8.36 US/kg). It is recommended to explore the potential of this MA-AC400 for the scavenging of toxic metal ions and other emerging contaminants present in industrial wastewater. The testing of this adsorbent in real wastewater treatment technology can further unveil its capabilities and shortcomings. It is further recommended to develop commercial production facility for the development of MA-AC400.