Current Progress on Epidemiology, Diagnosis, and Treatment of Sporotrichosis and Their Future Trends
Abstract
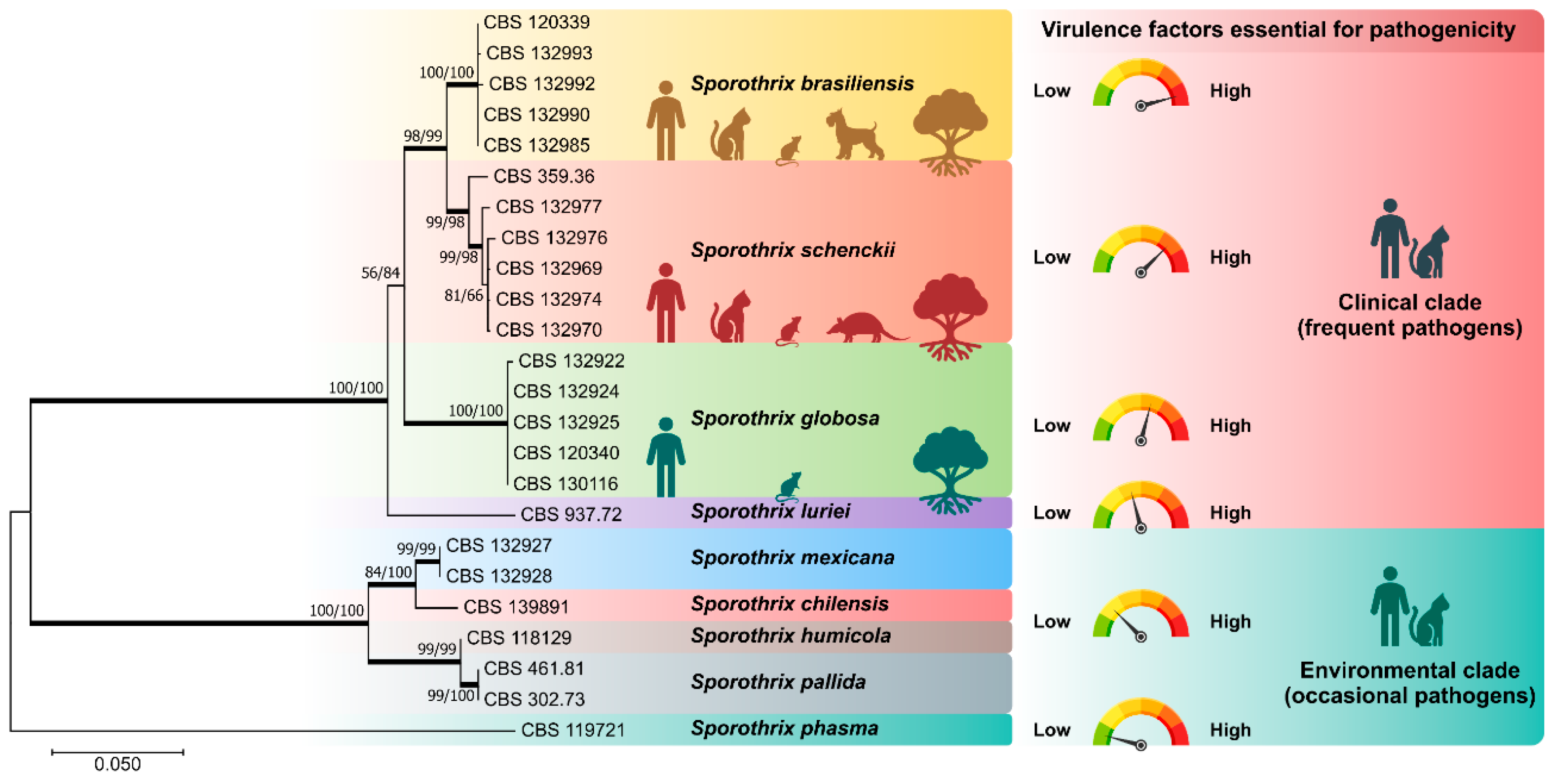
1. A Brief Introduction to the System Sporothrix-Sporotrichosis
2. Trends in the Epidemiology of Sporothrix Species
3. Trends in the Diagnosis of Sporotrichosis
3.1. Mycological Test
3.2. Direct Microscopic, Cytopathological, and Histopathological Examinations
3.3. Serology
3.4. Molecular Diagnosis
4. Trends in the Treatment of Sporotrichosis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Ramírez-Soto, M.; Aguilar-Ancori, E.; Tirado-Sánchez, A.; Bonifaz, A. Ecological determinants of sporotrichosis etiological agents. J. Fungi 2018, 4, 95. [Google Scholar] [CrossRef] [PubMed]
- Orofino-Costa, R.C.; Macedo, P.M.; Rodrigues, A.M.; Bernardes-Engemann, A.R. Sporotrichosis: An update on epidemiology, etiopathogenesis, laboratory and clinical therapeutics. An. Bras. De Dermatol. 2017, 92, 606–620. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues, A.M.; de Hoog, G.S.; de Camargo, Z.P. Sporothrix species causing outbreaks in animals and humans driven by animal-animal transmission. PLoS Pathog. 2016, 12, e1005638. [Google Scholar] [CrossRef]
- Rodrigues, A.M.; Bagagli, E.; de Camargo, Z.P.; Bosco, S.M.G. Sporothrix schenckii sensu stricto isolated from soil in an armadillo’s burrow. Mycopathologia 2014, 177, 199–206. [Google Scholar] [CrossRef] [PubMed]
- Gremião, I.D.; Miranda, L.H.; Reis, E.G.; Rodrigues, A.M.; Pereira, S.A. Zoonotic epidemic of sporotrichosis: Cat to human transmission. PLoS Pathog. 2017, 13, e1006077. [Google Scholar] [CrossRef]
- Bonifaz, A.; Vázquez-González, D. Diagnosis and treatment of lymphocutaneous sporotrichosis: What are the options? Curr. Fungal Infect. Rep. 2013, 7, 252–259. [Google Scholar] [CrossRef]
- Kauffman, C.A.; Bustamante, B.; Chapman, S.W.; Pappas, P.G. Clinical practice guidelines for the management of sporotrichosis: 2007 update by the Infectious Diseases Society of America. Clin. Infect. Dis. 2007, 45, 1255–1265. [Google Scholar] [CrossRef]
- Kauffman, C.A.; Hajjeh, R.; Chapman, S.W.; Group, M.S. Practice guidelines for the management of patients with sporotrichosis. Clin. Infect. Dis. 2000, 30, 684–687. [Google Scholar] [CrossRef]
- Aung, A.K.; Teh, B.M.; McGrath, C.; Thompson, P.J. Pulmonary sporotrichosis: Case series and systematic analysis of literature on clinico-radiological patterns and management outcomes. Med. Mycol. 2013, 51, 534–544. [Google Scholar] [CrossRef]
- Gremião, I.D.; Menezes, R.C.; Schubach, T.M.; Figueiredo, A.B.; Cavalcanti, M.C.; Pereira, S.A. Feline sporotrichosis: Epidemiological and clinical aspects. Med. Mycol. 2015, 53, 15–21. [Google Scholar] [CrossRef]
- Pereira, S.A.; Gremião, I.D.F.; Menezes, R.C. Sporotrichosis in Animals: Zoonotic Transmission. In Sporotrichosis: New Developments and Future Prospects; Zeppone Carlos, I., Ed.; Springer International Publishing: Cham, Switzerland, 2015; pp. 83–102. [Google Scholar] [CrossRef]
- Pereira, S.A.; Gremião, I.D.; Kitada, A.A.; Boechat, J.S.; Viana, P.G.; Schubach, T.M. The epidemiological scenario of feline sporotrichosis in Rio de Janeiro, State of Rio de Janeiro, Brazil. Rev. Da Soc. Bras. De Med. Trop. 2014, 47, 392–393. [Google Scholar] [CrossRef] [PubMed]
- Schubach, A.; Schubach, T.M.; Barros, M.B.; Wanke, B. Cat-transmitted sporotrichosis, Rio de Janeiro, Brazil. Emerg. Infect. Dis. 2005, 11, 1952–1954. [Google Scholar] [CrossRef] [PubMed]
- Gremião, I.D.F.; Martins da Silva da Rocha, E.; Montenegro, H.; Carneiro, A.J.B.; Xavier, M.O.; de Farias, M.R.; Monti, F.; Mansho, W.; de Macedo Assunção Pereira, R.H.; Pereira, S.A.; et al. Guideline for the management of feline sporotrichosis caused by Sporothrix brasiliensis and literature revision. Braz. J. Microbiol. Publ. Braz. Soc. Microbiol. 2021, 52, 107–124. [Google Scholar] [CrossRef]
- Rodrigues, A.M.; Della Terra, P.P.; Gremiao, I.D.; Pereira, S.A.; Orofino-Costa, R.; de Camargo, Z.P. The threat of emerging and re-emerging pathogenic Sporothrix species. Mycopathologia 2020, 185, 813–842. [Google Scholar] [CrossRef] [PubMed]
- Schenck, B.R. On refractory subcutaneous abscesses caused by a fungus possibly related to the Sporotricha. Bull. Johns Hopkins Hosp. 1898, 9, 286–290. [Google Scholar]
- Hektoen, L.; Perkins, C.F. Refractory subcutaneous abscesses caused by Sporothrix schenckii: A new pathogenic fungus. J. Exp. Med. 1900, 5, 77–89. [Google Scholar] [CrossRef] [PubMed]
- Marimon, R.; Gené, J.; Cano, J.; Trilles, L.; Dos Santos Lazéra, M.; Guarro, J. Molecular phylogeny of Sporothrix schenckii. J. Clin. Microbiol. 2006, 44, 3251–3256. [Google Scholar] [CrossRef]
- de Beer, Z.W.; Duong, T.A.; Wingfield, M.J. The divorce of Sporothrix and Ophiostoma: Solution to a problematic relationship. Stud. Mycol. 2016, 83, 165–191. [Google Scholar] [CrossRef]
- Rodrigues, A.M.; Cruz Choappa, R.; Fernandes, G.F.; De Hoog, G.S.; Camargo, Z.P. Sporothrix chilensis sp. nov. (Ascomycota: Ophiostomatales), a soil-borne agent of human sporotrichosis with mild-pathogenic potential to mammals. Fungal Biol. 2016, 120, 246–264. [Google Scholar] [CrossRef]
- Sil, A.; Andrianopoulos, A. Thermally dimorphic human fungal pathogens—Polyphyletic pathogens with a convergent pathogenicity trait. Cold Spring Harb. Perspect. Med. 2014, 5, a019794. [Google Scholar] [CrossRef]
- Fernandes, G.F.; dos Santos, P.O.; Amaral, C.C.; Sasaki, A.A.; Godoy-Martinez, P.; Camargo, Z.P.d. Characteristics of 151 Brazilian Sporothrix schenckii isolates from 5 different geographic regions of Brazil: A forgotten and re-emergent pathogen. Open Mycol. J. 2009, 3, 48–58. [Google Scholar] [CrossRef]
- Fernandes, G.F.; dos Santos, P.O.; Rodrigues, A.M.; Sasaki, A.A.; Burger, E.; de Camargo, Z.P. Characterization of virulence profile, protein secretion and immunogenicity of different Sporothrix schenckii sensu stricto isolates compared with S. globosa and S. brasiliensis species. Virulence 2013, 4, 241–249. [Google Scholar] [CrossRef] [PubMed]
- Della Terra, P.P.; Rodrigues, A.M.; Fernandes, G.F.; Nishikaku, A.S.; Burger, E.; de Camargo, Z.P. Exploring virulence and immunogenicity in the emerging pathogen Sporothrix brasiliensis. PLoS Negl. Trop. Dis. 2017, 11, e0005903. [Google Scholar] [CrossRef]
- Almeida-Paes, R.; de Oliveira, L.C.; Oliveira, M.M.E.; Gutierrez-Galhardo, M.C.; Nosanchuk, J.D.; Zancopé Oliveira, R.M. Phenotypic characteristics associated with virulence of clinical isolates from the Sporothrix complex. BioMed Res. Int. 2015, 2015, 1–10. [Google Scholar] [CrossRef]
- Arrillaga-Moncrieff, I.; Capilla, J.; Mayayo, E.; Marimon, R.; Mariné, M.; Gené, J.; Cano, J.; Guarro, J. Different virulence levels of the species of Sporothrix in a murine model. Clin. Microbiol. Infect. 2009, 15, 651–655. [Google Scholar] [CrossRef] [PubMed]
- de Macedo, P.M.; Sztajnbok, D.C.; Camargo, Z.P.; Rodrigues, A.M.; Lopes-Bezerra, L.M.; Bernardes-Engemann, A.R.; Orofino-Costa, R. Dacryocystitis due to Sporothrix brasiliensis: A case report of a successful clinical and serological outcome with low-dose potassium iodide treatment and oculoplastic surgery. Br. J. Dermatol. 2015, 172, 1116–1119. [Google Scholar] [CrossRef]
- Silva-Vergara, M.L.; de Camargo, Z.P.; Silva, P.F.; Abdalla, M.R.; Sgarbieri, R.N.; Rodrigues, A.M.; dos Santos, K.C.; Barata, C.H.; Ferreira-Paim, K. Disseminated Sporothrix brasiliensis infection with endocardial and ocular involvement in an HIV-infected patient. Am. J. Trop. Med. Hyg. 2012, 86, 477–480. [Google Scholar] [CrossRef]
- Almeida-Paes, R.; de Oliveira, M.M.; Freitas, D.F.; do Valle, A.C.; Zancope-Oliveira, R.M.; Gutierrez-Galhardo, M.C. Sporotrichosis in Rio de Janeiro, Brazil: Sporothrix brasiliensis is associated with atypical clinical presentations. PLoS Negl. Trop. Dis. 2014, 8, e3094. [Google Scholar] [CrossRef] [PubMed]
- Nepomuceno Araujo, M.; Nihei, C.H.; Rodrigues, A.M.; Higashino, H.; Ponzio, V.; Campos Pignatari, A.C.; Barcellos, M.A.; Braga, O.; Duayer, I.F. Case report: Invasive sinusitis due to Sporothrix brasiliensis in a renal transplant recipient. Am. J. Trop. Med. Hyg. 2021, 105, 1218–1221. [Google Scholar] [CrossRef]
- do Monte Alves, M.; Pipolo Milan, E.; da Silva-Rocha, W.P.; Soares de Sena da Costa, A.; Araujo Maciel, B.; Cavalcante Vale, P.H.; de Albuquerque, P.R.; Lopes Lima, S.; Salles de Azevedo Melo, A.; Messias Rodrigues, A.; et al. Fatal pulmonary sporotrichosis caused by Sporothrix brasiliensis in Northeast Brazil. PLoS Negl. Trop. Dis. 2020, 14, e0008141. [Google Scholar] [CrossRef]
- Barros, M.B.; de Almeida Paes, R.; Schubach, A.O. Sporothrix schenckii and sporotrichosis. Clin. Microbiol. Rev. 2011, 24, 633–654. [Google Scholar] [CrossRef]
- Dangerfield, L.F.; Gear, J. Sporotrichosis among miners on the Witwatersrand gold mines. S.A. Med. J. 1941, 15, 128–131. [Google Scholar]
- Pijper, A.; Pullinger, B.D. An outbreak of sporotrichosis among South African native miners. Lancet 1927, 210, 914–916. [Google Scholar] [CrossRef]
- Brown, R.; Bowen, J.W.; Weintroub, D.; Cluver, E.H.; Buchanan, G.; Simson, F.W.; Lurie, H.I.; Du Toit, C.J.; Golberg, L.; Goedvolk, C.J.; et al. Sporotrichosis infection on mines of the Witwatersrand. In Proceedings of the Transvaal Mine Medical Officer’s Association, Cape Town, South Africa, 1 January 1947; pp. 1–67. [Google Scholar]
- Govender, N.P.; Maphanga, T.G.; Zulu, T.G.; Patel, J.; Walaza, S.; Jacobs, C.; Ebonwu, J.I.; Ntuli, S.; Naicker, S.D.; Thomas, J. An outbreak of lymphocutaneous sporotrichosis among mine-workers in South Africa. PLoS Negl. Trop. Dis. 2015, 9, e0004096. [Google Scholar] [CrossRef]
- Vismer, H.F.; Hull, P.R. Prevalence, epidemiology and geographical distribution of Sporothrix schenckii infections in Gauteng, South Africa. Mycopathologia 1997, 137, 137–143. [Google Scholar] [CrossRef] [PubMed]
- Nenoff, P.; Reinel, D.; Kruger, C.; Grob, H.; Mugisha, P.; Suss, A.; Mayser, P. Tropical and travel-related dermatomycoses: Part 2: Cutaneous infections due to yeasts, moulds, and dimorphic fungi. Der Hautarzt Z. Fur. Dermatol. Venerol. Und Verwandte Geb. 2015, 66, 522–532. [Google Scholar] [CrossRef]
- Callens, S.F.; Kitetele, F.; Lukun, P.; Lelo, P.; Van Rie, A.; Behets, F.; Colebunders, R. Pulmonary Sporothrix schenckii infection in a HIV positive child. J. Trop. Pediatrics 2006, 52, 144–146. [Google Scholar] [CrossRef] [PubMed]
- Ponnighaus, M.; Grosser, S.; Baum, H.P.; Mischke, D.; Kowalzick, L. Sporotrichosis as the cause of a leg ulcer. Der Hautarzt; Z. Fur Dermatol. Venerol. Und Verwandte Geb. 2003, 54, 64–66. [Google Scholar] [CrossRef]
- Findlay, G. Sporotrichosis research in the Transvaal-how it began 60 years ago. S. Afr. Med. J. Suid-Afrik. Tydskr. Vir Geneeskd. 1985, 68, 117–118. [Google Scholar]
- Dalis, J.S.; Kazeem, H.M.; Kwaga, J.K.; Kwanashie, C.N. Severe generalized skin lesions due to mixed infection with Sporothrix schenkii and Dermatophilus congolensis in a bull from Jos, Nigeria. Vet. Microbiol. 2014, 172, 475–478. [Google Scholar] [CrossRef]
- Rasamoelina, T.; Maubon, D.; Raharolahy, O.; Razanakoto, H.; Rakotozandrindrainya, N.; Rakotomalala, F.A.; Bailly, S.; Sendrasoa, F.; Ranaivo, I.; Andrianarison, M. Sporotrichosis in the Highlands of Madagascar, 2013–2017. Emerg. Infect. Dis. 2019, 25, 1893–1902. [Google Scholar] [CrossRef] [PubMed]
- Ross, M.D.; Gelfand, M. Deep fungal infections in Rhodesia—A 10-year survey of histological material. Part I Cent. Afr. J. Med. 1978, 24, 208–212. [Google Scholar] [PubMed]
- Jacyk, W.K.; Lawande, R.V.; Tulpule, S.S. Deep mycoses in West Africa: A report of 13 cases and review of the Nigerian literature. J. Natl. Med. Assoc. 1981, 73, 251–256. [Google Scholar] [PubMed]
- Gumaa, S.A. Sporotrichosis in Sudan. Trans. R. Soc. Trop. Med. Hyg. 1978, 72, 637–640. [Google Scholar] [CrossRef]
- Chakrabarti, A.; Bonifaz, A.; Gutierrez-Galhardo, M.C.; Mochizuki, T.; Li, S. Global epidemiology of sporotrichosis. Med. Mycol. 2015, 53, 3–14. [Google Scholar] [CrossRef]
- Sharma, N.L.; Mehta, K.I.; Mahajan, V.K.; Kanga, A.K.; Sharma, V.C.; Tegta, G.R. Cutaneous sporotrichosis of face: Polymorphism and reactivation after intralesional triamcinolone. Indian J. Dermatol. Venereol. Leprol. 2007, 73, 188–190. [Google Scholar] [CrossRef]
- Mehta, K.I.S.; Sharma, N.L.; Kanga, A.K.; Mahajan, V.K.; Ranjan, N. Isolation of Sporothrix schenckii from the environmental sources of cutaneous sporotrichosis patients in Himachal Pradesh, India: Results of a pilot study. Mycoses 2007, 50, 496–501. [Google Scholar] [CrossRef]
- Verma, S.; Verma, G.K.; Singh, G.; Kanga, A.; Shanker, V.; Singh, D.; Gupta, P.; Mokta, K.; Sharma, V. Sporotrichosis in Sub-Himalayan India. PLoS Negl. Trop. Dis. 2012, 6, e1673. [Google Scholar] [CrossRef]
- Rudramurthy, S.M.; Shankarnarayan, S.A.; Hemashetter, B.M.; Verma, S.; Chauhan, S.; Nath, R.; Savio, J.; Capoor, M.; Kaur, H.; Ghosh, A.K.; et al. Phenotypic and molecular characterisation of Sporothrix globosa of diverse origin from India. Braz. J. Microbiol. Publ. Braz. Soc. Microbiol. 2021, 52, 91–100. [Google Scholar] [CrossRef]
- Fukushiro, R. Epidemiology and ecology of sporotrichosis in Japan. Zent. Bakteriol. Mikrobiol. Hyg. A 1984, 257, 228–233. [Google Scholar] [CrossRef]
- Ishizaki, H.; Kawasaki, M. Molecular epidemiology of Sporothrix schenckii. Nihon Ishinkin Gakkai Zasshi 2000, 41, 245–249. [Google Scholar] [CrossRef] [PubMed]
- Takeda, Y.; Kawasaki, M.; Ishizaki, H. Phylogeny and molecular epidemiology of Sporothrix schenckii in Japan. Mycopathologia 1991, 116, 9–14. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, K.; Kawasaki, M.; Ishizaki, H. Analysis of restriction profiles of mitochondrial DNA from Sporothrix schenckii and related fungi. Mycopathologia 1988, 103, 147–151. [Google Scholar] [CrossRef] [PubMed]
- Ishizaki, H.; Kawasaki, M.; Aoki, M.; Wu, S.; Lin, J.; Kim, J.A.; Won, Y.H.; Calvo, C.R. Mitochondrial DNA analysis of Sporothrix schenckii from China, Korea and Spain. Nihon Ishinkin Gakkai Zasshi 2004, 45, 23–25. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Ishizaki, H.; Kawasaki, M.; Aoki, M.; Vismer, H.; Muir, D. Mitochondrial DNA analysis of Sporothrix schenckii in South Africa and Australia. Med. Mycol. 2000, 38, 433–436. [Google Scholar] [CrossRef]
- Ishizaki, H.; Kawasaki, M.; Aoki, M.; Matsumoto, T.; Padhye, A.A.; Mendoza, M.; Negroni, R. Mitochondrial DNA analysis of Sporothrix schenckii in North and South America. Mycopathologia 1998, 142, 115–118. [Google Scholar] [CrossRef]
- Mochizuki, H.; Anzawa, K.; Mochizuki, T. Genotyping of intraspecies polymorphisms of Sporothrix globosa using partial sequence of mitochondrial DNA. J. Derm. 2022, 49, 263–271. [Google Scholar] [CrossRef]
- Kawasaki, M.; Anzawa, K.; Mochizuki, T.; Ishizaki, H. New strain typing method with Sporothrix schenckii using mitochondrial DNA and polymerase chain reaction restriction fragment length polymorphism (PCR–RFLP) technique. J. Derm. 2012, 39, 362–365. [Google Scholar] [CrossRef] [PubMed]
- Moussa, T.A.; Kadasa, N.M.; Al Zahrani, H.S.; Ahmed, S.A.; Feng, P.; Gerrits van den Ende, A.H.; Zhang, Y.; Kano, R.; Li, F.; Li, S.; et al. Origin and distribution of Sporothrix globosa causing sapronoses in Asia. J. Med. Microbiol. 2017, 66, 560–569. [Google Scholar] [CrossRef]
- Liu, T.-T.; Zhang, K.; Zhou, X. Molecular identification of Sporothrix clinical isolates in China. J. Zhejiang Univ. Sci. B 2014, 15, 100–108. [Google Scholar] [CrossRef]
- Yu, X.; Wan, Z.; Zhang, Z.; Li, F.; Li, R.; Liu, X. Phenotypic and molecular identification of Sporothrix isolates of clinical origin in Northeast China. Mycopathologia 2013, 176, 67–74. [Google Scholar] [CrossRef] [PubMed]
- Lv, S.; Hu, X.; Liu, Z.; Lin, Y.; Wu, H.; Li, F. Clinical epidemiology of sporotrichosis in Jilin province, China (1990-2019): A series of 4969 cases. Infect. Drug Resist. 2022, 15, 1753–1765. [Google Scholar] [CrossRef]
- Song, Y.; Li, S.S.; Zhong, S.X.; Liu, Y.Y.; Yao, L.; Huo, S.S. Report of 457 sporotrichosis cases from Jilin province, northeast China, a serious endemic region. J. Eur. Acad. Derm. Venereol. 2013, 27, 313–318. [Google Scholar] [CrossRef] [PubMed]
- Yao, L.; Song, Y.; Cui, Y.; Zhou, J.F.; Zhong, S.X.; Zhao, D.Y.; Li, S.S. Pediatric sporotrichosis in Jilin province of China (2010-2016): A retrospective study of 704 cases. J. Pediatric Infect. Dis. Soc. 2019, 9, 342–348. [Google Scholar] [CrossRef]
- Yao, L.; Song, Y.; Zhou, J.F.; Cui, Y.; Li, S.S. Epidemiological and clinical comparisons of pediatric and adult sporotrichosis in Jilin Province, China. Mycoses 2020, 63, 308–313. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Hagen, F.; Stielow, B.; Rodrigues, A.M.; Samerpitak, K.; Zhou, X.; Feng, P.; Yang, L.; Chen, M.; Deng, S.; et al. Phylogeography and evolutionary patterns in Sporothrix spanning more than 14,000 human and animal case reports. Persoonia 2015, 35, 1–20. [Google Scholar] [CrossRef]
- Zamri-Saad, M.; Salmiyah, T.S.; Jasni, S.; Cheng, B.Y.; Basri, K. Feline sporotrichosis: An increasingly important zoonotic disease in Malaysia. Vet. Rec. 1990, 127, 480. [Google Scholar]
- Tang, M.M.; Tang, J.J.; Gill, P.; Chang, C.C.; Baba, R. Cutaneous sporotrichosis: A six-year review of 19 cases in a tertiary referral center in Malaysia. Int. J. Derm. 2012, 51, 702–708. [Google Scholar] [CrossRef]
- Kano, R.; Okubo, M.; Siew, H.H.; Kamata, H.; Hasegawa, A. Molecular typing of Sporothrix schenckii isolates from cats in Malaysia. Mycoses 2015, 58, 220–224. [Google Scholar] [CrossRef]
- Beurmann, L.; Ramond, L. Abcès sous-cutanés multiples d’origine mycosique. Ann. Derm. Syph. 1903, 4, 678–685. [Google Scholar]
- Matruchot, L. Sur un nouveau groupe de champignons pathogènes, agents des sporotrichoses. Comptes Rendus De L’Académie De Sci. 1910, 150, 543–545. [Google Scholar]
- Beurmann, L.; Gougerot, H. Les Sporotrichose; Librairie Felix Alcan: Paris, France, 1912. [Google Scholar]
- Romeo, O.; Scordino, F.; Criseo, G. New insight into molecular phylogeny and epidemiology of Sporothrix schenckii species complex based on calmodulin-encoding gene analysis of Italian isolates. Mycopathologia 2011, 172, 179–186. [Google Scholar] [CrossRef] [PubMed]
- Criseo, G.; Romeo, O. Ribosomal DNA sequencing and phylogenetic analysis of environmental Sporothrix schenckii strains: Comparison with clinical isolates. Mycopathologia 2010, 169, 351–358. [Google Scholar] [CrossRef] [PubMed]
- Criseo, G.; Malara, G.; Romeo, O.; Puglisi Guerra, A. Lymphocutaneous sporotrichosis in an immunocompetent patient: A case report from extreme southern Italy. Mycopathologia 2008, 166, 159–162. [Google Scholar] [CrossRef]
- Marimon, R.; Cano, J.; Gené, J.; Sutton, D.A.; Kawasaki, M.; Guarro, J. Sporothrix brasiliensis, S. globosa, and S. mexicana, three new Sporothrix species of clinical interest. J. Clin. Microbiol. 2007, 45, 3198–3206. [Google Scholar] [CrossRef]
- Madrid, H.; Cano, J.; Gene, J.; Bonifaz, A.; Toriello, C.; Guarro, J. Sporothrix globosa, a pathogenic fungus with widespread geographical distribution. Rev. Iberoam. De Micol. 2009, 26, 218–222. [Google Scholar] [CrossRef]
- Rachman, R.; Ligaj, M.; Chinthapalli, S.; Serafino Wani, R. Zoonotic acquisition of cutaneous Sporothrix brasiliensis infection in the UK. BMJ Case Rep. 2022, 15, e248418. [Google Scholar] [CrossRef]
- McGuinness, S.L.; Boyd, R.; Kidd, S.; McLeod, C.; Krause, V.L.; Ralph, A.P. Epidemiological investigation of an outbreak of cutaneous sporotrichosis, Northern Territory, Australia. BMC Infect. Dis. 2016, 16, 16. [Google Scholar] [CrossRef]
- New, D.; Beukers, A.G.; Kidd, S.E.; Merritt, A.J.; Weeks, K.; van Hal, S.J.; Arthur, I. Identification of multiple species and subpopulations among Australian clinical Sporothrix isolates using whole genome sequencing. Med. Mycol. 2019, 57, 905–908. [Google Scholar] [CrossRef]
- Schauer, A.; Campbell, A.J.; Arthur, I.; Blyth, C.C.; Bowen, A.C. Spotting sporotrichosis skin infection: The first Australian paediatric case series. J. Paediatr. Child Health 2020, 56, 408–410. [Google Scholar] [CrossRef]
- Mackay, B.; Menrath, V.; Ridley, M.; Kelly, W. Sporotrichosis in a cat. Aust. Vet Pr. 1986, 16, 3–5. [Google Scholar]
- Thomson, J.; Trott, D.J.; Malik, R.; Galgut, B.; McAllister, M.M.; Nimmo, J.; Renton, D.; Kidd, S.E. An atypical cause of sporotrichosis in a cat. Med. Mycol. Case Rep. 2019, 23, 72–76. [Google Scholar] [CrossRef] [PubMed]
- Dixon, D.M.; Salkin, I.F.; Duncan, R.A.; Hurd, N.J.; Haines, J.H.; Kemna, M.E.; Coles, F.B. Isolation and characterization of Sporothrix schenckii from clinical and environmental sources associated with the largest U.S. epidemic of sporotrichosis. J. Clin. Microbiol. 1991, 29, 1106–1113. [Google Scholar] [CrossRef] [PubMed]
- Coles, F.B.; Schuchat, A.; Hibbs, J.R.; Kondracki, S.F.; Salkin, I.F.; Dixon, D.M.; Chang, H.G.; Duncan, R.A.; Hurd, N.J.; Morse, D.L. A multistate outbreak of sporotrichosis associated with Sphagnum moss. Am. J. Epidemiol. 1992, 136, 475–487. [Google Scholar] [CrossRef] [PubMed]
- Gold, J.A.W.; Derado, G.; Mody, R.K.; Benedict, K. Sporotrichosis-associated hospitalizations, United States, 2000–2013. Emerg. Infect. Dis. 2016, 22, 1817–1820. [Google Scholar] [CrossRef] [PubMed]
- Queiroz-Telles, F.; Fahal, A.H.; Falci, D.R.; Caceres, D.H.; Chiller, T.; Pasqualotto, A.C. Neglected endemic mycoses. Lancet Infect. Dis. 2017, 17, e367–e377. [Google Scholar] [CrossRef]
- Hernández-Castro, R.; Pinto-Almazán, R.; Arenas, R.; Sánchez-Cárdenas, C.D.; Espinosa-Hernández, V.M.; Sierra-Maeda, K.Y.; Conde-Cuevas, E.; Juárez-Durán, E.R.; Xicohtencatl-Cortes, J.; Carrillo-Casas, E.M.; et al. Epidemiology of clinical sporotrichosis in the Americas in the last ten years. J. Fungi 2022, 8, 588. [Google Scholar] [CrossRef]
- Rodrigues, A.M.; de Hoog, G.S.; Zhang, Y.; Camargo, Z.P. Emerging sporotrichosis is driven by clonal and recombinant Sporothrix species. Emerg. Microbes Infect. 2014, 3, e32. [Google Scholar] [CrossRef]
- Florez-Munoz, S.V.; Alzate, J.F.; Mesa-Arango, A.C. Molecular identification and antifungal susceptibility of clinical isolates of Sporothrix schenckii complex in Medellin, Colombia. Mycopathologia 2019, 184, 53–63. [Google Scholar] [CrossRef]
- Ramirez Soto, M.C. Sporotrichosis: The Story of an Endemic Region in Peru over 28 Years (1985 to 2012). PLoS ONE 2015, 10, e0127924. [Google Scholar] [CrossRef]
- Pappas, P.G.; Tellez, I.; Deep, A.E.; Nolasco, D.; Holgado, W.; Bustamante, B. Sporotrichosis in Peru: Description of an area of hyperendemicity. Clin. Infect Dis. 2000, 30, 65–70. [Google Scholar] [CrossRef] [PubMed]
- Camacho, E.; León-Navarro, I.; Rodríguez-Brito, S.; Mendoza, M.; Niño-Vega, G.A. Molecular epidemiology of human sporotrichosis in Venezuela reveals high frequency of Sporothrix globosa. BMC Infect Dis. 2015, 15, 94. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues, A.M.; de Hoog, G.S.; de Camargo, Z.P. Molecular diagnosis of pathogenic Sporothrix species. PLoS Negl. Trop. Dis. 2015, 9, e0004190. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues, A.M.; de Hoog, G.S.; de Cassia Pires, D.; Brilhante, R.S.N.; da Costa Sidrim, J.J.; Gadelha, M.F.; Colombo, A.L.; de Camargo, Z.P. Genetic diversity and antifungal susceptibility profiles in causative agents of sporotrichosis. BMC Infect. Dis. 2014, 14, 219. [Google Scholar] [CrossRef]
- Rodrigues, A.M.; de Hoog, S.; de Camargo, Z.P. Emergence of pathogenicity in the Sporothrix schenckii complex. Med. Mycol. 2013, 51, 405–412. [Google Scholar] [CrossRef]
- Rodrigues, A.M.; de Melo Teixeira, M.; de Hoog, G.S.; Schubach, T.M.P.; Pereira, S.A.; Fernandes, G.F.; Bezerra, L.M.L.; Felipe, M.S.; de Camargo, Z.P. Phylogenetic analysis reveals a high prevalence of Sporothrix brasiliensis in feline sporotrichosis outbreaks. PLoS Negl. Trop. Dis. 2013, 7, e2281. [Google Scholar] [CrossRef]
- Falcão, E.M.M.; de Lima Filho, J.B.; Campos, D.P.; Valle, A.C.F.d.; Bastos, F.I.; Gutierrez-Galhardo, M.C.; Freitas, D.F.S. [Hospitalizations and deaths related to sporotrichosis in Brazil (1992–2015)]. Cad. De Saude Publica 2019, 35, e00109218. [Google Scholar] [CrossRef]
- de Carvalho, J.A.; Beale, M.A.; Hagen, F.; Fisher, M.C.; Kano, R.; Bonifaz, A.; Toriello, C.; Negroni, R.; Rego, R.S.M.; Gremiao, I.D.F.; et al. Trends in the molecular epidemiology and population genetics of emerging Sporothrix species. Stud. Mycol. 2021, 100, 100129. [Google Scholar] [CrossRef]
- Araujo, A.K.L.; de Santana Leal, C.A. Feline sporotrichosis in the municipality of Bezerros, Agreste Pernambucano: Case report. Pubvet 2016, 10, 795–872. [Google Scholar] [CrossRef]
- Nunes, G.D.L.; dos Santos Carneiro, R.; Filgueira, K.D.; Filgueira, F.G.F.; Fernandes, T.H.T. Feline sporotrichosis in Itaporanga municipality, Paraíba state, Brazil: Case report. Arq. De Ciências Veterinárias E Zool. Da UNIPAR 2011, 14. [Google Scholar]
- Filgueira, K.D. Sporotrichosis in the canine species: A case report on city of Mossoro, RN. Cienc. Anim. Bras. 2009, 10, 673–677. [Google Scholar]
- de Oliveira Bento, A.; de Sena Costa, A.S.; Lima, S.L.; do Monte Alves, M.; de Azevedo Melo, A.S.; Rodrigues, A.M.; da Silva-Rocha, W.P.; Milan, E.P.; Chaves, G.M. The spread of cat-transmitted sporotrichosis due to Sporothrix brasiliensis in Brazil towards the Northeast region. PLoS Negl. Trop. Dis. 2021, 15, e0009693. [Google Scholar] [CrossRef] [PubMed]
- Schubach, T.M.; Schubach, A.; Okamoto, T.; Barros, M.B.; Figueiredo, F.B.; Cuzzi, T.; Fialho-Monteiro, P.C.; Reis, R.S.; Perez, M.A.; Wanke, B. Evaluation of an epidemic of sporotrichosis in cats: 347 cases (1998–2001). J. Am. Vet. Med. Assoc. 2004, 224, 1623–1629. [Google Scholar] [CrossRef]
- Schubach, A.O.; Schubach, T.M.; Barros, M.B. Epidemic cat-transmitted sporotrichosis. N. Engl. J. Med. 2005, 353, 1185–1186. [Google Scholar] [CrossRef] [PubMed]
- Falcao, E.M.M.; Pires, M.C.S.; Andrade, H.B.; Goncalves, M.L.C.; Almeida-Paes, R.; do Valle, A.C.F.; Bastos, F.I.; Gutierrez-Galhardo, M.C.; Freitas, D.F.S. Zoonotic sporotrichosis with greater severity in Rio de Janeiro, Brazil: 118 hospitalizations and 11 deaths in the last 2 decades in a reference institution. Med. Mycol. 2019, 58, 141–143. [Google Scholar] [CrossRef]
- Córdoba, S.; Isla, G.; Szusz, W.; Vivot, W.; Hevia, A.; Davel, G.; Canteros, C.E. Molecular identification and susceptibility profile of Sporothrix schenckii sensu lato isolated in Argentina. Mycoses 2018, 61, 441–448. [Google Scholar] [CrossRef] [PubMed]
- Etchecopaz, A.N.; Lanza, N.; Toscanini, M.A.; Devoto, T.B.; Pola, S.J.; Daneri, G.L.; Iovannitti, C.A.; Cuestas, M.L. Sporotrichosis caused by Sporothrix brasiliensis in Argentina: Case report, molecular identification and in vitro susceptibility pattern to antifungal drugs. J. De Mycol. Med. 2019, 30, 100908. [Google Scholar] [CrossRef]
- García Duarte, J.M.; Wattiez Acosta, V.R.; Fornerón Viera, P.M.L.; Aldama Caballero, A.; Gorostiaga Matiauda, G.A.; Rivelli de Oddone, V.B.; Pereira Brunelli, J.G. Sporotrichosis transmitted by domestic cat. A family case report. Rev. Del Nac. 2017, 9, 67–76. [Google Scholar] [CrossRef]
- Rios, M.E.; Suarez, J.M.D.; Moreno, J.; Vallee, J.; Moreno, J.P. Zoonotic sporotrichosis related to cat contact: First case report from Panama in Central America. Cureus 2018, 10, e2906. [Google Scholar] [CrossRef]
- PAHO. Sporothrix brasiliensis, an Emerging Fungal Pathogen, Notable for its Zoonotic Transmission and Epidemic Potential for Human and Animal Health in the Americas. Available online: https://www.someve.com.ar/images/noticias/2019/S-brasiliensis_lasAmericas_30082019_ES.pdf (accessed on 30 August 2019).
- Montenegro, H.; Rodrigues, A.M.; Galvão Dias, M.A.; da Silva, E.A.; Bernardi, F.; Camargo, Z.P. Feline sporotrichosis due to Sporothrix brasiliensis: An emerging animal infection in São Paulo, Brazil. BMC Vet Res. 2014, 10, 269. [Google Scholar] [CrossRef]
- Zhou, X.; Rodrigues, A.M.; Feng, P.; Hoog, G.S. Global ITS diversity in the Sporothrix schenckii complex. Fungal Divers. 2014, 66, 153–165. [Google Scholar] [CrossRef]
- Rediguieri, B.C.; da Cruz Bahiense, I.; de Carvalho, J.A.; Leite, G.R.; Falqueto, A.; Rodrigues, A.M.; Gonçalves, S.S. Clinical, epidemiological, and epizootic features of Sporothrix brasiliensis in Espírito Santo, Brazil. Ecohealth 2022, 19, 124–134. [Google Scholar] [CrossRef] [PubMed]
- Maschio-Lima, T.; Marques, M.D.R.; Lemes, T.H.; Brizzotti-Mazuchi, N.S.; Caetano, M.H.; de Almeida, B.G.; Bianco, L.M.; Monteiro, R.C.; Rodrigues, A.M.; de Camargo, Z.P.; et al. Clinical and epidemiological aspects of feline sporotrichosis caused by Sporothrix brasiliensis and in vitro antifungal susceptibility. Vet. Res. Commun. 2021, 45, 171–179. [Google Scholar] [CrossRef]
- da Cruz Bahiense Rocha, I.; Terra, P.P.D.; Cardoso de Oliveira, R.; Lubianca Zanotti, R.; Falqueto, A.; de Camargo, Z.P.; Rodrigues, A.M.; Goncalves, S.S. Molecular-based assessment of diversity and population structure of Sporothrix spp. clinical isolates from Espirito Santo-Brazil. Mycoses 2021, 64, 420–427. [Google Scholar] [CrossRef] [PubMed]
- Macêdo-Sales, P.A.; Souza, L.O.P.; Della-Terra, P.P.; Lozoya-Pérez, N.E.; Machado, R.L.D.; Rocha, E.M.d.S.d.; Lopes-Bezerra, L.M.; Guimarães, A.J.; Rodrigues, A.M.; Mora-Montes, H.M.; et al. Coinfection of domestic felines by distinct Sporothrix brasiliensis in the Brazilian sporotrichosis hyperendemic area. Fungal Genet. Biol. 2020, 140, 103397. [Google Scholar] [CrossRef] [PubMed]
- Macêdo-Sales, P.A.; Souto, S.R.L.S.; Destefani, C.A.; Lucena, R.P.; Machado, R.L.D.; Pinto, M.R.; Rodrigues, A.M.; Lopes-Bezerra, L.M.; Rocha, E.M.S.; Baptista, A.R.S. Domestic feline contribution in the transmission of Sporothrix in Rio de Janeiro State, Brazil: A comparison between infected and non-infected populations. BMC Vet. Res. 2018, 14, 19. [Google Scholar] [CrossRef]
- Teixeira, M.d.M.; Rodrigues, A.M.; Tsui, C.K.M.; de Almeida, L.G.P.; Van Diepeningen, A.D.; Gerrits van den Ende, B.; Fernandes, G.F.; Kano, R.; Hamelin, R.C.; Lopes-Bezerra, L.M.; et al. Asexual propagation of a virulent clone complex in human and feline outbreak of sporotrichosis. Eukaryot Cell 2015, 14, 158–169. [Google Scholar] [CrossRef]
- Rodrigues, A.M.; Fernandes, G.F.; Araujo, L.M.; Della Terra, P.P.; Dos Santos, P.O.; Pereira, S.A.; Schubach, T.M.; Burger, E.; Lopes-Bezerra, L.M.; de Camargo, Z.P. Proteomics-based characterization of the humoral immune response in sporotrichosis: Toward discovery of potential diagnostic and vaccine antigens. PLoS Negl. Trop. Dis. 2015, 9, e0004016. [Google Scholar] [CrossRef]
- Freitas, D.C.; Migliano, M.F.; Zani Neto, L. Sporotrichosis. Observation of spontaneous case in domestic cat (Felis catus). Rev. Fac. Med. Vet. Univ. Sao Paulo 1956, 5, 601–604. [Google Scholar] [CrossRef]
- Freitas, D.C.; Moreno, G.; Saliba, A.M.; Botino, J.Á.; Mós, E.M. Sporotrichosis in dogs and cats. Rev. Fac. Med. Vet Univ. Sao Paulo 1965, 7, 381–387. [Google Scholar] [CrossRef]
- Almeida, F.; Sampaio, S.A.P.; Lacaz, C.S.; Fernandes, J.C. Statistical data on sporotrichosis. Analysis of 344 cases. An. Bras. De Dermatol. 1955, 30, 9–12. [Google Scholar]
- Lacaz, C.S.; Porto, E.; Martins, J.E.C.; Heins-Vaccari, E.M.; de Melo, N.T. (Eds.) Tratado de Micologia Médica, 9th ed.; Sarvier: São Paulo, Brazil, 2002; p. 1104. [Google Scholar]
- Marques, S.A.; Franco, S.R.; de Camargo, R.M.; Dias, L.D.; Haddad Junior, V.; Fabris, V.E. Sporotrichosis of the domestic cat (Felis catus): Human transmission. Rev. Do Inst. De Med. Trop. De Sao Paulo 1993, 35, 327–330. [Google Scholar]
- Baroni, F.; Campos, S.; Direito, G. Sporotrichosis in a cat (a case description). Rev. Bras. Med. Vet. 1998, 20, 25–27. [Google Scholar]
- Barros, M.B.d.L.; Schubach, T.P.; Coll, J.O.; Gremião, I.D.; Wanke, B.; Schubach, A. Sporotrichosis: Development and challenges of an epidemic. Rev. Panam. Salud Publica 2010, 27, 455–460. [Google Scholar] [CrossRef] [PubMed]
- Gremião, I.D.F.; Oliveira, M.M.E.; Monteiro de Miranda, L.H.; Saraiva Freitas, D.F.; Pereira, S.A. Geographic expansion of sporotrichosis, Brazil. Emerg. Infect. Dis. 2020, 26, 621–624. [Google Scholar] [CrossRef]
- Larsson, C.E.; Goncalves Mde, A.; Araujo, V.C.; Dagli, M.L.; Correa, B.; Fava Neto, C. Feline sporotrichosis: Clinical and zoonotic aspects. Rev. Do Inst. De Med. Trop. De Sao Paulo 1989, 31, 351–358. [Google Scholar]
- Borges, T.S.; Rossi, C.N.; Fedullo, J.D.; Taborda, C.P.; Larsson, C.E. Isolation of Sporothrix schenckii from the claws of domestic cats (indoor and outdoor) and in captivity in Sao Paulo (Brazil). Mycopathologia 2013, 176, 129–137. [Google Scholar] [CrossRef]
- Silva, C.E.; Valeriano, C.A.T.; Ferraz, C.E.; Neves, R.P.; Oliveira, M.M.E.; Silva, J.C.A.L.; Magalhães, V.; Lima-Neto, R.G. Epidemiological features and geographical expansion of sporotrichosis in the state of Pernambuco, northeastern Brazil. Future Microbiol. 2021, 16, 1371–1379. [Google Scholar] [CrossRef]
- Larsson, C.E. Sporotrichosis. Braz. J. Vet. Res. Anim. Sci. 2011, 48, 250–259. [Google Scholar] [CrossRef]
- Almeida, F.P. Micologia Médica: Estudo das Micoses Humanas e de Seus Cogumelos; Companhia Melhoramentos de São Paulo: São Paulo, Brazil, 1939; p. 710. [Google Scholar]
- Fernandes, C.G.N.; Moura, S.T.d.; Dantas, A.F.M.; Blatt, M.C.S. Feline sporotrichosis: Clinical epidemiological aspects: Case reports (Cuiabá, Mato Grosso, Brasil). MEDVEP. Rev. Cient. Med. Vet. 2004, 2, 39–43. [Google Scholar]
- SES-MS. Sporotrichosis Technical Note—Nº 1/2021-GTZ/CEVE/DGVS/SES. Available online: https://www.vs.saude.ms.gov.br/wp-content/uploads/2021/08/Nota-tecnica-esporotricose.pdf (accessed on 17 June 2022).
- CRMV-SE. Technical Note No. 01/2021. Available online: https://www.crmvse.org.br/wp-content/uploads/2021/06/Nota-Tecnica-01-2021.pdf (accessed on 23 June 2022).
- SES-AM. Joint Technical Note—Nº 006/2021—GEVEP/DEVAE/DAP/DRA/SUBGS. Available online: https://semsa.manaus.am.gov.br/wp-content/uploads/2021/05/Nota-Tecnica_Fluxo_APS_e_Vigilancia_e_Ficha_de_Notificacao_da_Esporotricose_assinadas.pdf (accessed on 17 June 2022).
- COVISA. Technical Note 09: DVE/DVZ/COVISA/2020—Surveillance and Clinical Management of Human Sporotrichosis in the Municipality of São Paulo. Available online: https://docs.bvsalud.org/biblioref/2020/08/1102196/nota-tecnica-09-dve-zoo-2020_esporotricose_v6-alterada-a-pedid_CBJA7E3.pdf (accessed on 17 June 2022).
- Bison, I. Feline Sporotrichosis: Literature Review. Bachelor’s Thesis, Federal University of Rondônia, Rolim de Moura, Rondônia, 2019. [Google Scholar]
- Boechat, J.S.; Oliveira, M.M.E.; Almeida-Paes, R.; Gremiao, I.D.F.; Machado, A.C.S.; Oliveira, R.V.C.; Figueiredo, A.B.F.; Rabello, V.B.S.; Silva, K.B.L.; Zancope-Oliveira, R.M.; et al. Feline sporotrichosis: Associations between clinical-epidemiological profiles and phenotypic-genotypic characteristics of the etiological agents in the Rio de Janeiro epizootic area. Mem. Do Inst. Oswaldo Cruz 2018, 113, 185–196. [Google Scholar] [CrossRef] [PubMed]
- de Souza, E.W.; Borba, C.M.; Pereira, S.A.; Gremiao, I.D.F.; Langohr, I.M.; Oliveira, M.M.E.; de Oliveira, R.V.C.; da Cunha, C.R.; Zancope-Oliveira, R.M.; de Miranda, L.H.M.; et al. Clinical features, fungal load, coinfections, histological skin changes, and itraconazole treatment response of cats with sporotrichosis caused by Sporothrix brasiliensis. Sci. Rep. 2018, 8, 9074. [Google Scholar] [CrossRef] [PubMed]
- de Araujo, M.L.; Rodrigues, A.M.; Fernandes, G.F.; de Camargo, Z.P.; de Hoog, G.S. Human sporotrichosis beyond the epidemic front reveals classical transmission types in Espírito Santo, Brazil. Mycoses 2015, 58, 485–490. [Google Scholar] [CrossRef] [PubMed]
- Colodel, M.M.; Jark, P.C.; Ramos, C.J.R.; Martins, V.M.V.; Schneider, A.F.; Pilati, C. Feline cutaneous sporotrichosis in the state of Santa Catarina: Case reports. Vet. Foco 2009, 7, 18–27. [Google Scholar]
- Cordeiro, F.N.; Bruno, C.B.; Paula, C.D.; Motta Jde, O. Familial occurrence of zoonotic sporotrichosis. An. Bras. De Dermatol. 2011, 86, S121–S124. [Google Scholar] [CrossRef] [PubMed]
- Silva, G.M.; Howes, J.C.F.; Leal, C.A.S.; Mesquita, E.P.; Pedrosa, C.M.; Oliveira, A.A.F.; Silva, L.B.G.; Mota, R.A. Feline sporotrichosis outbreak in metropolitan Recife. Pesqui. Vet. Bras. 2018, 38, 1767–1771. [Google Scholar] [CrossRef]
- Marques-Melo, E.H.; Lessa, D.F.d.S.; Garrido, L.H.A.; Nunes, A.C.B.T.; Chaves, K.P.; Porto, W.J.N.; Notomi, M. Domestic feline as sporotricosis transmitting agent for human: Report of the first case in the state of Alagoas. Rev. Baiana De Saúde Pública 2014, 38, 490–498. [Google Scholar]
- Madrid, I.M.; Mattei, A.; Martins, A.; Nobre, M.; Meireles, M. Feline sporotrichosis in the southern region of Rio Grande do Sul, Brazil: Clinical, zoonotic and therapeutic aspects. Zoonoses Public Health 2010, 57, 151–154. [Google Scholar] [CrossRef]
- Sanchotene, K.O.; Madrid, I.M.; Klafke, G.B.; Bergamashi, M.; Terra, P.P.D.; Rodrigues, A.M.; de Camargo, Z.P.; Xavier, M.O. Sporothrix brasiliensis outbreaks and the rapid emergence of feline sporotrichosis. Mycoses 2015, 58, 652–658. [Google Scholar] [CrossRef]
- Madrid, I.M.; Mattei, A.S.; Fernandes, C.G.; Oliveira Nobre, M.; Meireles, M.C.A. Epidemiological findings and laboratory evaluation of sporotrichosis: A description of 103 cases in cats and dogs in Southern Brazil. Mycopathologia 2012, 173, 265–273. [Google Scholar] [CrossRef]
- Eudes Filho, J.; Santos, I.B.D.; Reis, C.M.S.; Patané, J.S.L.; Paredes, V.; Bernardes, J.; Poggiani, S.; Castro, T.C.B.; Gomez, O.M.; Pereira, S.A.; et al. A novel Sporothrix brasiliensis genomic variant in Midwestern Brazil: Evidence for an older and wider sporotrichosis epidemic. Emerg. Microbes Infect. 2020, 9, 2515–2525. [Google Scholar] [CrossRef] [PubMed]
- Bernardes-Engemann, A.R.; Almeida, M.A.; Bison, I.; Rabello, V.B.S.; Ramos, M.L.M.; Pereira, S.A.; Almeida-Paes, R.; de Lima Brasil, A.W.; Zancopé-Oliveira, R.M. Anti-Sporothrix Antibody Detection in Domestic Cats as an Indicator of a Possible New Occurrence Area for Sporotrichosis in North Brazil. Mycopathologia 2022, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, P.M.; Lopez, R.R.; Collier, B.A. Survival, fecundity, and movements of free-roaming cats. J. Wildl. Manag. 2007, 71, 915–919. [Google Scholar] [CrossRef]
- Jongman, E.C. Adaptation of domestic cats to confinement. J. Vet. Behav. 2007, 2, 193–196. [Google Scholar] [CrossRef]
- Horn, J.A.; Mateus-Pinilla, N.; Warner, R.E.; Heske, E.J. Home range, habitat use, and activity patterns of free-roaming domestic cats. J. Wildl. Manag. 2011, 75, 1177–1185. [Google Scholar] [CrossRef]
- Keesing, F.; Belden, L.K.; Daszak, P.; Dobson, A.; Harvell, C.D.; Holt, R.D.; Hudson, P.; Jolles, A.; Jones, K.E.; Mitchell, C.E.; et al. Impacts of biodiversity on the emergence and transmission of infectious diseases. Nature 2010, 468, 647–652. [Google Scholar] [CrossRef]
- Jones, K.E.; Patel, N.G.; Levy, M.A.; Storeygard, A.; Balk, D.; Gittleman, J.L.; Daszak, P. Global trends in emerging infectious diseases. Nature 2008, 451, 990–993. [Google Scholar] [CrossRef]
- Shaheen, M.N.F. The concept of one health applied to the problem of zoonotic diseases. Rev. Med. Virol. 2022, 32, e2326. [Google Scholar] [CrossRef] [PubMed]
- Winck, G.R.; Raimundo, R.L.G.; Fernandes-Ferreira, H.; Bueno, M.G.; D’Andrea, P.S.; Rocha, F.L.; Cruz, G.L.T.; Vilar, E.M.; Brandão, M.; Cordeiro, J.L.P.; et al. Socioecological vulnerability and the risk of zoonotic disease emergence in Brazil. Sci. Adv. 2022, 8, eabo5774. [Google Scholar] [CrossRef]
- Bloom, D.E.; Cadarette, D. Infectious disease threats in the twenty-first century: Strengthening the global response. Front. Immunol. 2019, 10, 549. [Google Scholar] [CrossRef]
- Alzuguir, C.L.C.; Pereira, S.A.; Magalhães, M.A.F.M.; Almeida-Paes, R.; Freitas, D.F.S.; Oliveira, L.F.A.; Pimentel, M.I.F. Geo-epidemiology and socioeconomic aspects of human sporotrichosis in the municipality of Duque de Caxias, Rio de Janeiro, Brazil, between 2007 and 2016. Trans. R. Soc. Trop. Med. Hyg. 2020, 114, 99–106. [Google Scholar] [CrossRef] [PubMed]
- Silva, M.B.; Costa, M.M.; Torres, C.C.; Galhardo, M.C.; Valle, A.C.; Magalhaes Mde, A.; Sabroza, P.C.; Oliveira, R.M. [Urban sporotrichosis: A neglected epidemic in Rio de Janeiro, Brazil]. Cad. De Saude Publica 2012, 28, 1867–1880. [Google Scholar] [CrossRef] [PubMed]
- Veasey, J.V.; Carvalho, G.d.S.M.; Ruiz, L.R.B.; Neves Neto, M.F.; Zaitz, C. Epidemiological and geographical distribution profile of urban sporotrichosis in the city of São Paulo. An. Bras. De Dermatol. 2022, 97, 228–230. [Google Scholar] [CrossRef] [PubMed]
- Ellwanger, J.H.; Kulmann-Leal, B.; Kaminski, V.L.; Valverde-Villegas, J.M.; Veiga, A.; Spilki, F.R.; Fearnside, P.M.; Caesar, L.; Giatti, L.L.; Wallau, G.L.; et al. Beyond diversity loss and climate change: Impacts of Amazon deforestation on infectious diseases and public health. An. Da Acad. Bras. De Cienc. 2020, 92, e20191375. [Google Scholar] [CrossRef]
- Pörtner, H.-O.; Roberts, D.C.; Adams, H.; Adelekan, I.; Adler, C.; Adrian, R.; Aldunce, P.; Ali, E.; Ara Begum, R.; Bednar-Friedl, B.; et al. IPCC, 2022: Climate Change 2022: Impacts, Adaptation, and Vulnerability: Contribution of Working Group II to the Sixth Assessment Report of the Intergovernmental Panel on Climate Change; Pörtner, H.-O., Roberts, D.C., Tignor, M., Poloczanska, E.S., Mintenbeck, K., Alegría, A., Craig, M., Langsdorf, S., Löschke, S., Möller, V., et al., Eds.; Cambridge University Press: Cambridge, UK, 2022. [Google Scholar]
- Jones Bryony, A.; Grace, D.; Kock, R.; Alonso, S.; Rushton, J.; Said Mohammed, Y.; McKeever, D.; Mutua, F.; Young, J.; McDermott, J.; et al. Zoonosis emergence linked to agricultural intensification and environmental change. Proc. Natl. Acad. Sci. USA 2013, 110, 8399–8404. [Google Scholar] [CrossRef]
- Rabello, V.B.S.; Almeida-Silva, F.; Scramignon-Costa, B.d.S.; Motta, B.d.S.; de Macedo, P.M.; Teixeira, M.d.M.; Almeida-Paes, R.; Irinyi, L.; Meyer, W.; Zancopé-Oliveira, R.M. Environmental isolation of Sporothrix brasiliensis in an area with recurrent feline sporotrichosis cases. Front. Cell. Infect. Microbiol. 2022, 12, 894297. [Google Scholar] [CrossRef]
- Almeida-Silva, F.; Rabello, V.B.; Scramignon-Costa, B.D.; Zancopé-Oliveira, R.M.; de Macedo, P.M.; Almeida-Paes, R. Beyond domestic cats: Environmental detection of Sporothrix brasiliensis DNA in a hyperendemic area of sporotrichosis in Rio de Janeiro state, Brazil. J. Fungi 2022, 8, 604. [Google Scholar] [CrossRef]
- Navarrete, A.A.; Tsai, S.M.; Mendes, L.W.; Faust, K.; de Hollander, M.; Cassman, N.A.; Raes, J.; van Veen, J.A.; Kuramae, E.E. Soil microbiome responses to the short-term effects of Amazonian deforestation. Mol. Ecol. 2015, 24, 2433–2448. [Google Scholar] [CrossRef]
- Khan, M.A.W.; Bohannan, B.J.M.; Nüsslein, K.; Tiedje, J.M.; Tringe, S.G.; Parlade, E.; Barberán, A.; Rodrigues, J.L.M. Deforestation impacts network co-occurrence patterns of microbial communities in Amazon soils. FEMS Microbiol. Ecol. 2019, 95, fiy230. [Google Scholar] [CrossRef]
- Rosenberg, K.; Bertaux, J.; Krome, K.; Hartmann, A.; Scheu, S.; Bonkowski, M. Soil amoebae rapidly change bacterial community composition in the rhizosphere of Arabidopsis thaliana. ISME J. 2009, 3, 675–684. [Google Scholar] [CrossRef]
- Steenbergen, J.N.; Nosanchuk, J.D.; Malliaris, S.D.; Casadevall, A. Interaction of Blastomyces dermatitidis, Sporothrix schenckii, and Histoplasma capsulatum with Acanthamoeba castellanii. Infect. Immun. 2004, 72, 3478–3488. [Google Scholar] [CrossRef] [PubMed]
- Lemos Tavares, P.; Carvalho Ribeiro, A.; Kercher Berte, F.; da Silva Hellwig, A.H.; Machado Pagani, D.; Tavares de Souza, C.C.; Brittes Rott, M.; Scroferneker, M.L. The interaction between Sporothrix schenckii sensu stricto and Sporothrix brasiliensis with Acanthamoeba castellanii. Mycoses 2020, 63, 302–307. [Google Scholar] [CrossRef] [PubMed]
- Aqeel, Y.; Siddiqui, R.; Iftikhar, H.; Khan, N.A. The effect of different environmental conditions on the encystation of Acanthamoeba castellanii belonging to the T4 genotype. Exp. Parasitol. 2013, 135, 30–35. [Google Scholar] [CrossRef] [PubMed]
- Rossow, J.A.; Queiroz-Telles, F.; Caceres, D.H.; Beer, K.D.; Jackson, B.R.; Pereira, J.G.; Ferreira Gremião, I.D.; Pereira, S.A. A one health approach to combatting Sporothrix brasiliensis: Narrative review of an emerging zoonotic fungal pathogen in South America. J. Fungi 2020, 6, 247. [Google Scholar] [CrossRef] [PubMed]
- Mahajan, V.K. Sporotrichosis: An overview and therapeutic options. Dermatol. Res. Pract. 2014, 2014, 272376. [Google Scholar] [CrossRef] [PubMed]
- Lopes-Bezerra, L.M.; Mora-Montes, H.M.; Zhang, Y.; Nino-Vega, G.; Rodrigues, A.M.; de Camargo, Z.P.; de Hoog, S. Sporotrichosis between 1898 and 2017: The evolution of knowledge on a changeable disease and on emerging etiological agents. Med. Mycol. 2018, 56, 126–143. [Google Scholar] [CrossRef]
- Kwon-Chung, J.K.; Bennett, J.E. Medical Mycology; Lea & Febiger: Philadelphia, PA, USA, 1992. [Google Scholar]
- Rippon, J.W. Medical Mycology—The Pathogenic Fungi and the Pathogenic Actinomycetes, 3rd ed.; W. B. Saunders Company: Philadelphia, PA, USA, 1988. [Google Scholar]
- Rodrigues, A.M.; Orofino-Costa, R.; de Camargo, Z.P. Sporothrix spp. In Pocket Guide to Mycological Diagnosis, 1st ed.; Cordeiro Rde, A., Ed.; CRC Press: Boca Raton, FL, USA, 2019; pp. 99–113. [Google Scholar] [CrossRef]
- Rodrigues, A.M.; de Hoog, G.S.; de Camargo, Z.P. Feline Sporotrichosis. In Emerging and Epizootic Fungal Infections in Animals; Seyedmousavi, S., de Hoog, G.S., Guillot, J., Verweij, P.E., Eds.; Springer International Publishing: Cham, Switzerland, 2018; Volume 1, pp. 199–231. [Google Scholar]
- Miranda, L.H.; Quintella, L.P.; Menezes, R.C.; dos Santos, I.B.; Oliveira, R.V.; Figueiredo, F.B.; Lopes-Bezerra, L.M.; Schubach, T.M. Evaluation of immunohistochemistry for the diagnosis of sporotrichosis in dogs. Vet. J. 2011, 190, 408–411. [Google Scholar] [CrossRef]
- Bernardes-Engemann, A.R.; de Lima Barros, M.; Zeitune, T.; Russi, D.C.; Orofino-Costa, R.; Lopes-Bezerra, L.M. Validation of a serodiagnostic test for sporotrichosis: A follow-up study of patients related to the Rio de Janeiro zoonotic outbreak. Med. Mycol. 2015, 53, 28–33. [Google Scholar] [CrossRef]
- Gezuele, E.; Da Rosa, D. Importance of the sporotrichosis asteroid body for the rapid diagnosis of sporotrichosis. Rev. Iberoam. De Micol. 2005, 22, 147–150. [Google Scholar] [CrossRef]
- Schwarz, J. The diagnosis of deep mycoses by morphologic methods. Hum. Pathol. 1982, 13, 519–533. [Google Scholar] [CrossRef]
- Morris-Jones, R. Sporotrichosis. Clin. Exp. Dermatol. 2002, 27, 427–431. [Google Scholar] [CrossRef] [PubMed]
- de Lima Barros, M.B.; Schubach, A.O.; de Vasconcellos Carvalhaes de Oliveira, R.; Martins, E.B.; Teixeira, J.L.; Wanke, B. Treatment of cutaneous sporotrichosis with Itraconazole—Study of 645 patients. Clin. Infect. Dis. 2011, 52, e200–e206. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues, A.M.; Hagen, F.; de Camargo, Z.P. A spotlight on Sporothrix and sporotrichosis. Mycopathologia 2022, 1–5, in press. [Google Scholar] [CrossRef]
- Gonçalves, S.S.; Cano, J.F.; Stchigel, A.M.; Melo, A.S.; Godoy-Martinez, P.C.; Correa, B.; Guarro, J. Molecular phylogeny and phenotypic variability of clinical and environmental strains of Aspergillus flavus. Fungal Biol. 2012, 116, 1146–1155. [Google Scholar] [CrossRef] [PubMed]
- Silva, J.N.; Miranda, L.H.M.; Menezes, R.C.; Gremiao, I.D.F.; Oliveira, R.V.C.; Vieira, S.M.M.; Conceicao-Silva, F.; Ferreiro, L.; Pereira, S.A. Comparison of the sensitivity of three methods for the early diagnosis of sporotrichosis in cats. J. Comp. Pathol. 2018, 160, 72–78. [Google Scholar] [CrossRef]
- Rodrigues, A.M.; Fernandes, G.F.; de Camargo, Z.P. Sporotrichosis. In Emerging and Re-Emerging Infectious Diseases of Livestock; Bayry, J., Ed.; Springer: Berlin/Heidelberg, Germany, 2017; pp. 391–421. [Google Scholar] [CrossRef]
- Marimon, R.; Gené, J.; Cano, J.; Guarro, J. Sporothrix luriei: A rare fungus from clinical origin. Med. Mycol. 2008, 46, 621–625. [Google Scholar] [CrossRef]
- Oliveira, M.M.; Almeida-Paes, R.; Muniz, M.M.; Gutierrez-Galhardo, M.C.; Zancope-Oliveira, R.M. Phenotypic and molecular identification of Sporothrix isolates from an epidemic area of sporotrichosis in Brazil. Mycopathologia 2011, 172, 257–267. [Google Scholar] [CrossRef]
- Zhao, M.-d.; Zhou, X.; Liu, T.-t.; Yang, Z.-b. Morphological and physiological comparison of taxa comprising the Sporothrix schenckii complex. J. Zhejiang Univ. Sci. B 2015, 16, 940–947. [Google Scholar] [CrossRef]
- Vásquez-del-Mercado, E.; Arenas, R.; Padilla-Desgarenes, C. Sporotrichosis. Clin. Derm. 2012, 30, 437–443. [Google Scholar] [CrossRef]
- Richardson, M.D.; Warnock, D.W. Fungal Infection: Diagnosis and Management, 4th ed.; Wiley-Blackwell: Oxford, UK, 2012. [Google Scholar]
- Oyarce, J.A.; García, C.; Alave, J.; Bustamante, B. Epidemiological clinical and laboratory characterization of sporotrichosis in patients of a tertiary care hospital in Lima, Peru, from 1991 to 2014. Rev. Chil. Infectol. 2016, 33, 315–321. [Google Scholar] [CrossRef]
- Arenas, R.; Sanchez-Cardenas, C.D.; Ramirez-Hobak, L.; Ruiz Arriaga, L.F.; Vega Memije, M.E. Sporotrichosis: From KOH to molecular biology. J. Fungi 2018, 4, 62. [Google Scholar] [CrossRef]
- Quintella, L.P.; Passos, S.R.; do Vale, A.C.; Galhardo, M.C.; Barros, M.B.; Cuzzi, T.; Reis Rdos, S.; de Carvalho, M.H.; Zappa, M.B.; Schubach Ade, O. Histopathology of cutaneous sporotrichosis in Rio de Janeiro: A series of 119 consecutive cases. J. Cutan. Pathol. 2011, 38, 25–32. [Google Scholar] [CrossRef]
- Quintella, L.P.; Passos, S.R.; de Miranda, L.H.; Cuzzi, T.; Barros, M.B.; Francesconi-do-Vale, A.C.; Galhardo, M.C.; Madeira Mde, F.; Figueiredo de Carvalho, M.H.; Schubach Ade, O. Proposal of a histopathological predictive rule for the differential diagnosis between American tegumentary leishmaniasis and sporotrichosis skin lesions. Br. J. Dermatol. 2012, 167, 837–846. [Google Scholar] [CrossRef]
- da Rosa, W.D.; Gezuele, E.; Calegari, L.; Goni, F. Asteroid body in sporotrichosis. Yeast viability and biological significance within the host immune response. Med. Mycol. 2008, 46, 443–448. [Google Scholar] [CrossRef] [PubMed]
- Civila, E.S.; Bonasse, J.; Conti-Diaz, I.A.; Vignale, R.A. Importance of the direct fresh examination in the diagnosis of cutaneous sporotrichosis. Int. J. Derm. 2004, 43, 808–810. [Google Scholar] [CrossRef] [PubMed]
- Miranda, L.H.; Conceicao-Silva, F.; Quintella, L.P.; Kuraiem, B.P.; Pereira, S.A.; Schubach, T.M. Feline sporotrichosis: Histopathological profile of cutaneous lesions and their correlation with clinical presentation. Comp. Immunol. Microbiol. Infect. Dis. 2013, 36, 425–432. [Google Scholar] [CrossRef] [PubMed]
- Welsh, R.D. Sporotrichosis. J. Am. Vet. Med. Assoc. 2003, 223, 1123–1126. [Google Scholar] [CrossRef]
- Pereira, S.A.; Menezes, R.C.; Gremião, I.D.F.; Silva, J.N.; de O. Honse, C.; Figueiredo, C.; da Silva, D.T.; Kitada, A.A.B.; dos Reis, É.G.; Schubach, T.M.P. Sensitivity of cytopathological examination in the diagnosis of feline sporotrichosis. J. Feline Med. Surg. 2011, 13, 220–223. [Google Scholar] [CrossRef]
- Seyedmousavi, S.; Bosco, S.d.M.G.; de Hoog, S.; Ebel, F.; Elad, D.; Gomes, R.R.; Jacobsen, I.D.; Jensen, H.E.; Martel, A.; Mignon, B.; et al. Fungal infections in animals: A patchwork of different situations. Med. Mycol. 2018, 56, 165–187. [Google Scholar] [CrossRef]
- Gonsales, F.F.; Fernandes, N.; Mansho, W.; Montenegro, H.; Guerra, J.M.; de Araujo, L.J.T.; da Silva, S.M.P.; Benites, N.R. Feline Sporothrix spp. detection using cell blocks from brushings and fine-needle aspirates: Performance and comparisons with culture and histopathology. Vet. Clin. Pathol. 2019, 48, 143–147. [Google Scholar] [CrossRef]
- Zhang, Y.Q.; Xu, X.G.; Zhang, M.; Jiang, P.; Zhou, X.Y.; Li, Z.Z.; Zhang, M.F. Sporotrichosis: Clinical and histopathological manifestations. Am. J. Dermatopathol. 2011, 33, 296–302. [Google Scholar] [CrossRef] [PubMed]
- Hussein, M.R. Mucocutaneous Splendore-Hoeppli phenomenon. J. Cutan. Pathol. 2008, 35, 979–988. [Google Scholar] [CrossRef] [PubMed]
- Widal, F.; Aerami, P.; Joltrain, E.; Brissaud, E.T.; Weill, A. Serodiagnostic mycosique, applications au diagnostic de la sporotrichose et de l’actinomycose. Les coagulations et cofixations mycosiques. Ann. Inst. Pasteur. 1910, 24, 1–33. [Google Scholar]
- Ochoa, A.G.; Figueroa, E. Polisacaridos del Sporotrichum schenckii. Datos immunologicos: Intradermoreaccion en el diagnostico de la esporotrichosis. Rev. Inst. Salubr. Enferm. Trop. 1947, 8, 143–153. [Google Scholar]
- Blumer, S.O.; Kaufman, L.; Kaplan, W.; McLaughlin, D.W.; Kraft, D.E. Comparative evaluation of five serological methods for the diagnosis of sporotrichosis. Appl. Microbiol. 1973, 26, 4–8. [Google Scholar] [CrossRef]
- Velasco, O.; Ochoa, A.G. Sporotrichosis in patients with previous positive sporotrichin reaction. Rev. De Investig. En Salud Publica 1971, 31, 53–55. [Google Scholar]
- Ghosh, A.; Chakrabarti, A.; Sharma, V.K.; Singh, K.; Singh, A. Sporotrichosis in Himachal Pradesh (north India). Trans. R. Soc. Trop. Med. Hyg. 1999, 93, 41–45. [Google Scholar] [CrossRef]
- Bonifaz, A.; Vázquez-González, D. Sporotrichosis: An update. G Ital Derm. Venereol 2010, 145, 659–673. [Google Scholar]
- Kusuhara, M. Sporotrichosis and dematiaceous fungal skin infections. Nihon Ishinkin Gakkai Zasshi 2009, 50, 213–217. [Google Scholar] [CrossRef]
- Itoh, M.; Okamoto, S.; Kariya, H. Survey of 200 cases of sporotrichosis. Dermatologica 1986, 172, 209–213. [Google Scholar] [CrossRef]
- Kusuhara, M.; Hachisuka, H.; Sasai, Y. Statistical survey of 150 cases with sporotrichosis. Mycopathologia 1988, 102, 129–133. [Google Scholar] [CrossRef] [PubMed]
- Posada, H.R. Prueba cutanea con esporotricina. Mycopathologia 1968, 36, 42–54. [Google Scholar] [CrossRef]
- Bonifaz, A.; Toriello, C.; Araiza, J.; Ramírez-Soto, M.C.; Tirado-Sánchez, A. Sporotrichin skin test for the diagnosis of sporotrichosis. J. Fungi 2018, 4, 55. [Google Scholar] [CrossRef]
- Bernardes-Engemann, A.R.; Orofino Costa, R.C.; Miguens, B.P.; Penha, C.V.L.; Neves, E.; Pereira, B.A.S.; Dias, C.M.P.; Mattos, M.; Gutierrez, M.C.; Schubach, A.; et al. Development of an enzyme-linked immunosorbent assay for the serodiagnosis of several clinical forms of sporotrichosis. Med. Mycol. 2005, 43, 487–493. [Google Scholar] [CrossRef] [PubMed]
- López-Ribot, J.L.; Casanova, M.; Murgui, A.; Martínez, J.P. Antibody response to Candida albicans cell wall antigens. FEMS Immunol. Med. Microbiol. 2004, 41, 187–196. [Google Scholar] [CrossRef] [PubMed]
- Alba-Fierro, C.A.; Pérez-Torres, A.; López-Romero, E.; Cuéllar-Cruz, M.; Ruiz-Baca, E. Cell wall proteins of Sporothrix schenckii as immunoprotective agents. Rev. Iberoam. De Micol. 2014, 31, 86–89. [Google Scholar] [CrossRef]
- Fernandes, G.F.; Lopes-Bezerra, L.M.; Bernardes-Engemann, A.R.; Schubach, T.M.; Dias, M.A.; Pereira, S.A.; de Camargo, Z.P. Serodiagnosis of sporotrichosis infection in cats by enzyme-linked immunosorbent assay using a specific antigen, SsCBF, and crude exoantigens. Vet. Microbiol. 2011, 147, 445–449. [Google Scholar] [CrossRef]
- Penha, C.V.; Bezerra, L.M. Concanavalin A-binding cell wall antigens of Sporothrix schenckii: A serological study. Med. Mycol. 2000, 38, 1–7. [Google Scholar] [CrossRef][Green Version]
- Alvarado, P.; Ostos, A.; Franquiz, N.; Roschman-González, A.; Zambrano, E.A.; Mendoza, M. Serological diagnosis of sporotrichosis using an antigen of Sporothrix schenckii sensu stricto mycelium. Investig. Clin. 2015, 56, 111–122. [Google Scholar]
- Fernandes, G.F.; Amaral, C.C.D.; Sasaki, A.; Godoy, P.M.; De Camargo, Z.P. Heterogeneity of proteins expressed by Brazilian Sporothrix schenckii isolates. Med. Mycol. 2009, 47, 855–861. [Google Scholar] [CrossRef]
- Bernardes-Engemann, A.R.; Loureiro y Penha, C.V.; Benvenuto, F.; Braga, J.U.; Barros, M.L.; Orofino-Costa, R.; Lopes-Bezerra, L.M. A comparative serological study of the SsCBF antigenic fraction isolated from three Sporothrix schenckii strains. Med. Mycol. 2009, 47, 874–878. [Google Scholar] [CrossRef]
- Rodrigues, A.M.; Kubitschek-Barreira, P.H.; Fernandes, G.F.; de Almeida, S.R.; Lopes-Bezerra, L.M.; de Camargo, Z.P. Immunoproteomic analysis reveals a convergent humoral response signature in the Sporothrix schenckii complex. J. Proteom. 2015, 115, 8–22. [Google Scholar] [CrossRef] [PubMed]
- Martínez-Álvarez, J.A.; García-Carnero, L.C.; Kubitschek-Barreira, P.H.; Lozoya-Pérez, N.E.; Belmonte-Vázquez, J.L.; de Almeida, J.R.; Gómez-Infante, A.D.J.; Curty, N.; Villagómez-Castro, J.C.; Peña-Cabrera, E.; et al. Analysis of some immunogenic properties of the recombinant Sporothrix schenckii Gp70 expressed in Escherichia coli. Future Microbiol. 2019, 14, 397–410. [Google Scholar] [CrossRef]
- Baptista, V.S.; Mothé, G.B.; Santos, G.M.P.; Melivilu, C.S.I.; Santos, T.O.; Virginio, E.D.; de Macêdo-Sales, P.A.; Pinto, M.R.; Machado, R.L.D.; Rocha, E.M.S.; et al. Promising application of the SsCBF ELISA test to monitor the therapeutic response of feline sporotrichosis caused by Sporothrix brasiliensis from Brazilian epidemics. Braz. J. Microbiol. 2021, 52, 145–153. [Google Scholar] [CrossRef] [PubMed]
- Elias Costa, M.R.; Da Silva Lacaz, C.; Kawasaki, M.; De Camargo, Z.P. Conventional versus molecular diagnostic tests. Med. Mycol. 2000, 38 (Suppl. 1), 139–145. [Google Scholar] [CrossRef][Green Version]
- Ghosh, A.; Maity, P.K.; Hemashettar, B.M.; Sharma, V.K.; Chakrabarti, A. Physiological characters of Sporothrix schenckii isolates. Mycoses 2002, 45, 449–554. [Google Scholar]
- Mesa-Arango, A.C.; del Rocío Reyes-Montes, M.; Pérez-Mejía, A.; Navarro-Barranco, H.; Souza, V.; Zúñiga, G.; Toriello, C. Phenotyping and genotyping of Sporothrix schenckii isolates according to geographic origin and clinical form of sporotrichosis. J. Clin. Microbiol. 2002, 40, 3004–3011. [Google Scholar] [CrossRef]
- Lücking, R.; Aime, M.C.; Robbertse, B.; Miller, A.N.; Aoki, T.; Ariyawansa, H.A.; Cardinali, G.; Crous, P.W.; Druzhinina, I.S.; Geiser, D.M.; et al. Fungal taxonomy and sequence-based nomenclature. Nat. Microbiol. 2021, 6, 540–548. [Google Scholar] [CrossRef]
- Crous, P.W.; Lombard, L.; Sandoval-Denis, M.; Seifert, K.A.; Schroers, H.J.; Chaverri, P.; Gené, J.; Guarro, J.; Hirooka, Y.; Bensch, K.; et al. Fusarium: More than a node or a foot-shaped basal cell. Stud. Mycol. 2021, 98, 1–184. [Google Scholar] [CrossRef] [PubMed]
- Della Terra, P.P.; Gonsales, F.F.; de Carvalho, J.A.; Hagen, F.; Kano, R.; Bonifaz, A.; Camargo, Z.P.; Rodrigues, A.M. Development and evaluation of a multiplex qPCR assay for rapid diagnostics of emerging sporotrichosis. Transbound. Emerg. Dis. 2021, in press. [Google Scholar] [CrossRef] [PubMed]
- Pinheiro, B.G.; Hahn, R.C.; Camargo, Z.P.; Rodrigues, A.M. Molecular tools for detection and identification of Paracoccidioides species: Current status and future perspectives. J. Fungi 2020, 6, 293. [Google Scholar] [CrossRef] [PubMed]
- de Carvalho, J.A.; Monteiro, R.C.; Hagen, F.; Camargo, Z.P.; Rodrigues, A.M. Trends in molecular diagnostics and genotyping tools applied for emerging Sporothrix species. J. Fungi 2022. [Google Scholar]
- Hu, S.; Chung, W.-H.; Hung, S.-I.; Ho, H.-C.; Wang, Z.-W.; Chen, C.-H.; Lu, S.-C.; Kuo, T.-T.; Hong, H.-S. Detection of Sporothrix schenckii in clinical samples by a nested PCR assay. J. Clin. Microbiol. 2003, 41, 1414–1418. [Google Scholar] [CrossRef] [PubMed]
- Kano, R.; Nakamura, Y.; Watanabe, S.; Tsujimoto, H.; Hasegawa, A. Identification of Sporothrix schenckii based on sequences of the chitin synthase 1 gene. Mycoses 2001, 44, 261–265. [Google Scholar] [CrossRef] [PubMed]
- Kano, R.; Matsuoka, A.; Kashima, M.; Nakamura, Y.; Watanabe, S.; Mizoguchi, M.; Hasegawa, A. Detection of Sporothrix schenckii chitin synthase 1 (CHS1) gene in biopsy specimens from human patients with sporotrichosis. J. Derm. Sci. 2003, 33, 73–74. [Google Scholar] [CrossRef]
- Mendoza, M.; Brito, A.; Schaper, D.A.; Spooner, V.A.; Alvarado, P.; Castro, A.; Fernandez, A. Technical evaluation of nested PCR for the diagnosis of experimental sporotrichosis. Rev. Iberoam. De Micol. 2012, 29, 120–125. [Google Scholar] [CrossRef]
- Zhang, M.; Li, F.; Gong, J.; Yang, X.; Zhang, J.; Zhao, F. Development and evaluation of a real-time polymerase chain reaction for fast diagnosis of sporotrichosis caused by Sporothrix globosa. Med. Mycol. 2019, 58, 61–65. [Google Scholar] [CrossRef]
- Rodrigues, A.M.; de Hoog, G.S.; Camargo, Z.P. Genotyping species of the Sporothrix schenckii complex by PCR-RFLP of calmodulin. Diagn Microbiol. Infect. Dis. 2014, 78, 383–387. [Google Scholar] [CrossRef]
- Rodrigues, A.M.; Najafzadeh, M.J.; de Hoog, G.S.; de Camargo, Z.P. Rapid identification of emerging human-pathogenic Sporothrix species with rolling circle amplification. Front. Microbiol. 2015, 6, 1385. [Google Scholar] [CrossRef]
- Ramírez-Soto, M.C.; Aguilar-Ancori, E.G.; Quispe-Ricalde, M.A.; Muñiz-Duran, J.G.; Quispe-Florez, M.M.; Chinen, A. Molecular identification of Sporothrix species in a hyperendemic area in Peru. J. Infect. Public. Health. 2021, 14, 670–673. [Google Scholar] [CrossRef]
- Gonsales, F.F.; Fernandes, N.C.C.A.; Mansho, W.; Montenegro, H.; Benites, N.R. Direct PCR of lesions suggestive of sporotrichosis in felines. Arq. Bras. Med. Vet. Zootec. 2020, 72, 2002–2006. [Google Scholar] [CrossRef]
- Zhang, M.; Li, F.; Li, R.; Gong, J.; Zhao, F. Fast diagnosis of sporotrichosis caused by Sporothrix globosa, Sporothrix schenckii, and Sporothrix brasiliensis based on multiplex real-time PCR. PLoS Negl. Trop. Dis. 2019, 13, e0007219. [Google Scholar] [CrossRef]
- Madrid, H.; Gené, J.; Cano, J.; Silvera, C.; Guarro, J. Sporothrix brunneoviolacea and Sporothrix dimorphospora, two new members of the Ophiostoma stenoceras-Sporothrix schenckii complex. Mycologia 2010, 102, 1193–1203. [Google Scholar] [CrossRef] [PubMed]
- Rangel-Gamboa, L.; Martinez-Hernandez, F.; Maravilla, P.; Arenas-Guzman, R.; Flisser, A. Update of phylogenetic and genetic diversity of Sporothrix schenckii sensu lato. Med. Mycol. 2016, 54, 248–255. [Google Scholar] [CrossRef] [PubMed]
- de Carvalho, J.A.; Hagen, F.; Fisher, M.C.; de Camargo, Z.P.; Rodrigues, A.M. Genome-wide mapping using new AFLP markers to explore intraspecific variation among pathogenic Sporothrix species. PLoS Negl. Trop. Dis. 2020, 14, e0008330. [Google Scholar] [CrossRef]
- Restrepo, C.M.; Llanes, A.; Lleonart, R. Use of AFLP for the study of eukaryotic pathogens affecting humans. Infect. Genet. Evol. 2018, 63, 360–369. [Google Scholar] [CrossRef] [PubMed]
- Roberto, T.N.; De Carvalho, J.A.; Beale, M.A.; Hagen, F.; Fisher, M.C.; Hahn, R.C.; de Camargo, Z.P.; Rodrigues, A.M. Exploring genetic diversity, population structure, and phylogeography in Paracoccidioides species using AFLP markers. Stud. Mycol. 2021, 100, 100131. [Google Scholar] [CrossRef]
- Rodrigues, A.M.; Beale, M.A.; Hagen, F.; Fisher, M.C.; Terra, P.P.D.; de Hoog, S.; Brilhante, R.S.N.; de Aguiar Cordeiro, R.; de Souza Collares Maia Castelo-Branco, D.; Rocha, M.F.G.; et al. The global epidemiology of emerging Histoplasma species in recent years. Stud. Mycol. 2020, 97, 100095. [Google Scholar] [CrossRef]
- Singhal, N.; Kumar, M.; Kanaujia, P.K.; Virdi, J.S. MALDI-TOF mass spectrometry: An emerging technology for microbial identification and diagnosis. Front. Microbiol. 2015, 6, 791. [Google Scholar] [CrossRef]
- Murray, P.R. What is new in clinical microbiology-microbial identification by MALDI-TOF mass spectrometry: A paper from the 2011 William Beaumont Hospital Symposium on molecular pathology. J. Mol. Diagn. JMD 2012, 14, 419–423. [Google Scholar] [CrossRef]
- Oliveira, M.M.; Santos, C.; Sampaio, P.; Romeo, O.; Almeida-Paes, R.; Pais, C.; Lima, N.; Zancope-Oliveira, R.M. Development and optimization of a new MALDI-TOF protocol for identification of the Sporothrix species complex. Res. Microbiol. 2015, 166, 102–110. [Google Scholar] [CrossRef] [PubMed]
- Etchecopaz, A.; Toscanini, M.A.; Gisbert, A.; Mas, J.; Scarpa, M.; Iovannitti, C.A.; Bendezú, K.; Nusblat, A.D.; Iachini, R.; Cuestas, M.L. Sporothrix brasiliensis: A review of an emerging South American fungal pathogen, its related disease, presentation and spread in Argentina. J. Fungi 2021, 7, 170. [Google Scholar] [CrossRef] [PubMed]
- Matos, A.M.F.; Moreira, L.M.; Barczewski, B.F.; de Matos, L.X.; de Oliveira, J.B.V.; Pimentel, M.I.F.; Almeida-Paes, R.; Oliveira, M.G.; Pinto, T.C.A.; Lima, N.; et al. Identification by MALDI-TOF MS of Sporothrix brasiliensis isolated from a subconjunctival infiltrative lesion in an immunocompetent patient. Microorganisms 2019, 8, 22. [Google Scholar] [CrossRef] [PubMed]
- Espinel-Ingroff, A.; Abreu, D.P.B.; Almeida-Paes, R.; Brilhante, R.S.N.; Chakrabarti, A.; Chowdhary, A.; Hagen, F.; Cordoba, S.; Gonzalez, G.M.; Govender, N.P.; et al. Multicenter and international study of MIC/MEC distributions for definition of epidemiological cutoff values (ECVs) for species of Sporothrix identified by molecular methods. Antimicrob. Agents Chemother. 2017, 61, e01057-17. [Google Scholar] [CrossRef] [PubMed]
- Costa, R.O.; Macedo, P.M.; Carvalhal, A.; Bernardes-Engemann, A.R. Use of potassium iodide in dermatology: Updates on an old drug. An. Bras. De Dermatol. 2013, 88, 396–402. [Google Scholar] [CrossRef]
- Brilhante, R.S.N.; Silva, M.; Pereira, V.S.; de Oliveira, J.S.; Maciel, J.M.; Silva, I.; Garcia, L.G.S.; Guedes, G.M.M.; Cordeiro, R.A.; Pereira-Neto, W.A.; et al. Potassium iodide and miltefosine inhibit biofilms of Sporothrix schenckii species complex in yeast and filamentous forms. Med. Mycol. 2018, 57, 764–772. [Google Scholar] [CrossRef]
- Sharma, B.; Sharma, A.K.; Sharma, U. Sporotrichosis: A comprehensive review on recent drug-based therapeutics and management. Curr. Derm. Rep. 2022, 11, 110–119. [Google Scholar] [CrossRef]
- Gremião, I.D.F.; Miranda, L.H.M.d.; Pereira-Oliveira, G.R.; Menezes, R.C.; Machado, A.C.d.S.; Rodrigues, A.M.; Pereira, S.A. Advances and challenges in the management of feline sporotrichosis. Rev. Iberoam. Micol. 2022, S1130-1406(22)00021-3. [Google Scholar] [CrossRef]
- Borba-Santos, L.P.; Rodrigues, A.M.; Gagini, T.B.; Fernandes, G.F.; Castro, R.; de Camargo, Z.P.; Nucci, M.; Lopes-Bezerra, L.M.; Ishida, K.; Rozental, S. Susceptibility of Sporothrix brasiliensis isolates to amphotericin B, azoles, and terbinafine. Med. Mycol. 2015, 53, 178–188. [Google Scholar] [CrossRef]
- CDDI. Cortellis Drug Discovery Intelligence Database. Available online: https://www.cortellis.com/drugdiscovery/ (accessed on 4 June 2022).
- Francesconi, G.; Francesconi do Valle, A.C.; Passos, S.L.; de Lima Barros, M.B.; de Almeida Paes, R.; Curi, A.L.; Liporage, J.; Porto, C.F.; Galhardo, M.C. Comparative study of 250 mg/day terbinafine and 100 mg/day itraconazole for the treatment of cutaneous sporotrichosis. Mycopathologia 2011, 171, 349–354. [Google Scholar] [CrossRef]
- Carolus, H.; Pierson, S.; Lagrou, K.; Van Dijck, P. Amphotericin B and other polyenes-discovery, clinical use, mode of action and drug resistance. J. Fungi 2020, 6, 321. [Google Scholar] [CrossRef]
- Institute, C.a.L.S. Reference Method for Broth Dilution Antifungal Susceptibility Testing of Yeast, M27, 4th ed.; Clinical and Laboratory Standards Institute: Wayne, PA, USA, 2017; p. 46. [Google Scholar]
- Institute, C.a.L.S. Reference Method for Broth Dilution Antifungal Susceptibility Testing of Filamentous Fungi, Approved Standard, M38, 3rd ed.; Clinical and Laboratory Standards Institute: Wayne, PA, USA, 2017; Volume 37, p. 46. [Google Scholar]
- Fernandez-Silva, F.; Capilla, J.; Mayayo, E.; Guarro, J. Modest efficacy of voriconazole against murine infections by Sporothrix schenckii and lack of efficacy against Sporothrix brasiliensis. Mycoses 2014, 57, 121–124. [Google Scholar] [CrossRef] [PubMed]
- Mario, D.N.; Guarro, J.; Santurio, J.M.; Alves, S.H.; Capilla, J. In vitro and in vivo efficacy of amphotericin B combined with posaconazole against experimental disseminated sporotrichosis. Antimicrob. Agents Chemother. 2015, 59, 5018–5021. [Google Scholar] [CrossRef] [PubMed]
- Fichman, V.; Valle, A.C.F.d.; de Macedo, P.M.; Freitas, D.F.S.; Oliveira, M.M.E.d.; Almeida-Paes, R.; Gutierrez-Galhardo, M.C. Cryosurgery for the treatment of cutaneous sporotrichosis in four pregnant women. PLoS Negl. Trop. Dis. 2018, 12, e0006434. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, C.P.; do Valle, A.C.; Freitas, D.F.; Reis, R.; Galhardo, M.C. Pregnancy during a sporotrichosis epidemic in Rio de Janeiro, Brazil. Int. J. Gynaecol. Obs. 2012, 117, 294–295. [Google Scholar] [CrossRef] [PubMed]
- Legabão, B.C.; Fernandes, J.A.; de Oliveira Barbosa, G.F.; Bonfim-Mendonça, P.S.; Svidzinski, T.I.E. The zoonosis sporotrichosis can be successfully treated by photodynamic therapy: A scoping review. Acta Trop. 2022, 228, 106341. [Google Scholar] [CrossRef]
- Borba-Santos, L.P.; Gagini, T.; Ishida, K.; de Souza, W.; Rozental, S. Miltefosine is active against Sporothrix brasiliensis isolates with in vitro low susceptibility to amphotericin B or itraconazole. J. Med. Microbiol. 2015, 64, 415–422. [Google Scholar] [CrossRef]
- Fischman Gompertz, O.; Rodrigues, A.M.; Fernandes, G.F.; Bentubo, H.D.; de Camargo, Z.P.; Petri, V. Atypical clinical presentation of sporotrichosis caused by Sporothrix globosa resistant to itraconazole. Am. J. Trop. Med. Hyg. 2016, 94, 1218–1222. [Google Scholar] [CrossRef]
- Nakasu, C.C.T.; Waller, S.B.; Ripoll, M.K.; Ferreira, M.R.A.; Conceição, F.R.; Gomes, A.D.R.; Osório, L.D.G.; de Faria, R.O.; Cleff, M.B. Feline sporotrichosis: A case series of itraconazole-resistant Sporothrix brasiliensis infection. Braz. J. Microbiol. 2021, 52, 163–171. [Google Scholar] [CrossRef]
- Waller, S.B.; Dalla Lana, D.F.; Quatrin, P.M.; Ferreira, M.R.A.; Fuentefria, A.M.; Mezzari, A. Antifungal resistance on Sporothrix species: An overview. Braz. J. Microbiol. 2021, 52, 73–80. [Google Scholar] [CrossRef]
- Lyra, M.R.; Sokoloski, V.; de Macedo, P.M.; Azevedo, A.C.P. Sporotrichosis refractory to conventional treatment: Therapeutic success with potassium iodide. An. Bras. De Dermatol. 2021, 96, 231–233. [Google Scholar] [CrossRef] [PubMed]
- Poester, V.R.; Basso, R.P.; Stevens, D.A.; Munhoz, L.S.; de Souza Rabello, V.B.; Almeida-Paes, R.; Zancopé-Oliveira, R.M.; Zanchi, M.; Benelli, J.L.; Xavier, M.O. Treatment of human sporotrichosis caused by Sporothrix brasiliensis. J. Fungi 2022, 8, 70. [Google Scholar] [CrossRef]
- Bernardes-Engemann, A.R.; Tomki, G.F.; Rabello, V.B.d.S.; Almeida-Silva, F.; Freitas, D.F.S.; Gutierrez-Galhardo, M.C.; Almeida-Paes, R.; Zancopé-Oliveira, R.M. Sporotrichosis caused by non-wild type Sporothrix brasiliensis strains. Front. Cell. Infect. Microbiol. 2022, 12, 893501. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.; Puumala, E.; Robbins, N.; Cowen, L.E. Antifungal drug resistance: Molecular mechanisms in Candida albicans and beyond. Chem. Rev. 2021, 121, 3390–3411. [Google Scholar] [CrossRef]
- Koehler, A.; Pagani, D.M.; da Silva Hellwig, A.H.; Scroferneker, M.L. In-vitro antifungal susceptibility of the genus Sporothrix and correlation with treatment options for sporotrichosis: A systematic review. Rev. Med. Microbiol. 2021, 32, 219–227. [Google Scholar] [CrossRef]
- Gagini, T.; Borba-Santos, L.P.; Messias Rodrigues, A.; Pires de Camargo, Z.; Rozental, S. Clotrimazole is highly effective in vitro against feline Sporothrix brasiliensis isolates. J. Med. Microbiol. 2017, 66, 1573–1580. [Google Scholar] [CrossRef] [PubMed]
- Fichman, V.; Marques de Macedo, P.; Francis Saraiva Freitas, D.; Carlos Francesconi do Valle, A.; Almeida-Silva, F.; Reis Bernardes-Engemann, A.; Zancopé-Oliveira, R.M.; Almeida-Paes, R.; Clara Gutierrez-Galhardo, M. Zoonotic sporotrichosis in renal transplant recipients from Rio de Janeiro, Brazil. Transpl. Infect. Dis. 2021, 23, e13485. [Google Scholar] [CrossRef] [PubMed]
- Georgopoulos, A.; Petranyi, G.; Mieth, H.; Drews, J. In vitro activity of naftifine, a new antifungal agent. Antimicrob. Agents Chemother. 1981, 19, 386–389. [Google Scholar] [CrossRef] [PubMed]
- Artunduaga Bonilla, J.J.; Honorato, L.; Haranahalli, K.; Gremião, I.D.F.; Pereira, S.A.; Guimarães, A.; Baptista, A.R.S.; Patricia, d.M.T.; Rodrigues, M.L.; Miranda, K.; et al. Antifungal activity of Acylhydrazone derivatives against Sporothrix spp. Antimicrob. Agents Chemother. 2021, 65, e02593-20. [Google Scholar] [CrossRef] [PubMed]
- Borba-Santos, L.P.; Visbal, G.; Braga, T.G.; Rodrigues, A.M.; De Camargo, Z.P.; Bezerra, L.L.; Ishida, K.; De Souza, W.; Rozental, S. ∆24-sterol methyltransferase plays an important role in the growth and development of Sporothrix schenckii and Sporothrix brasiliensis. Front. Microbiol. 2016, 7, 311. [Google Scholar] [CrossRef]
- Borba-Santos, L.P.; Nucci, M.; Ferreira-Pereira, A.; Rozental, S. Anti-Sporothrix activity of ibuprofen combined with antifungal. Braz. J. Microbiol. 2021, 52, 101–106. [Google Scholar] [CrossRef] [PubMed]
- Borba-Santos, L.P.; Ishida, K.; Calogeropoulou, T.; Souza, W.; Rozental, S. Adamantylidene-substituted alkylphosphocholine TCAN26 is more active against Sporothrix schenckii than miltefosine. Mem. Do Inst. Oswaldo Cruz 2016, 111, 523–527. [Google Scholar] [CrossRef]
- Asquith, C.R.M.; Machado, A.C.S.; de Miranda, L.H.M.; Konstantinova, L.S.; Almeida-Paes, R.; Rakitin, O.A.; Pereira, S.A. Synthesis and identification of pentathiepin-based inhibitors of Sporothrix brasiliensis. Antibiotics 2019, 8, 249. [Google Scholar] [CrossRef]
- Gopinath, P.; Yadav, R.K.; Shukla, P.K.; Srivastava, K.; Puri, S.K.; Muraleedharan, K.M. Broad spectrum anti-infective properties of benzisothiazolones and the parallels in their anti-bacterial and anti-fungal effects. Bioorg. Med. Chem. Lett. 2017, 27, 1291–1295. [Google Scholar] [CrossRef] [PubMed]
- Azevedo-França, J.A.; Granado, R.; de Macedo Silva, S.T.; Santos-Silva, G.D.; Scapin, S.; Borba-Santos, L.P.; Rozental, S.; de Souza, W.; Martins-Duarte É, S.; Barrias, E.; et al. Synthesis and biological activity of novel zinc-itraconazole complexes in protozoan parasites and Sporothrix spp. Antimicrob. Agents Chemother. 2020, 64, e01980-19. [Google Scholar] [CrossRef] [PubMed]
- de Azevedo-França, J.A.; Borba-Santos, L.P.; de Almeida Pimentel, G.; Franco, C.H.J.; Souza, C.; de Almeida Celestino, J.; de Menezes, E.F.; Dos Santos, N.P.; Vieira, E.G.; Ferreira, A.; et al. Antifungal promising agents of zinc(II) and copper(II) derivatives based on azole drug. J. Inorg. Biochem. 2021, 219, 111401. [Google Scholar] [CrossRef]
- Gagini, T.; Colina-Vegas, L.; Villarreal, W.; Borba-Santos, L.P.; de Souza Pereira, C.; Batista, A.A.; Kneip Fleury, M.; de Souza, W.; Rozental, S.; Costa, L.A.S.; et al. Metal–azole fungistatic drug complexes as anti-Sporothrix spp. agents. New J. Chem. 2018, 42, 13641–13650. [Google Scholar] [CrossRef]
- Tandon, V.K.; Kumar, S.; Mishra, N.N.; Shukla, P.K. Micelles catalyzed chemo- and regio-selective one pot and one step synthesis of 2,3,5,6-tetrakis(alkyl and arylsulfanyl)-1,4-benzoquinones and 2,5-diaminosubstituted-1,4-benzoquinones “In-Water” and their biological evaluation as antibacterial and antifungal agents. Eur. J. Med. Chem. 2012, 56, 375–386. [Google Scholar] [CrossRef]
- Brilhante, R.S.; Silva, N.F.; Marques, F.J.; Castelo-Branco Dde, S.; de Lima, R.A.; Malaquias, A.D.; Caetano, E.P.; Barbosa, G.R.; de Camargo, Z.P.; Rodrigues, A.M.; et al. In vitro inhibitory activity of terpenic derivatives against clinical and environmental strains of the Sporothrix schenkii complex. Med. Mycol. 2015, 53, 93–98. [Google Scholar] [CrossRef]
- Brilhante, R.S.; Malaquias, A.D.; Caetano, E.P.; Castelo-Branco Dde, S.; Lima, R.A.; Marques, F.J.; Silva, N.F.; Alencar, L.P.; Monteiro, A.J.; Camargo, Z.P.; et al. In vitro inhibitory effect of miltefosine against strains of Histoplasma capsulatum var. capsulatum and Sporothrix spp. Med. Mycol. 2014, 52, 320–325. [Google Scholar] [CrossRef][Green Version]
- Borba-Santos, L.P.; Vila, T.; Rozental, S. Identification of two potential inhibitors of Sporothrix brasiliensis and Sporothrix schenckii in the Pathogen Box collection. PLoS ONE 2020, 15, e0240658. [Google Scholar] [CrossRef]
- Borba-Santos, L.P.; Barreto, T.L.; Vila, T.; Chi, K.D.; Dos Santos Monti, F.; de Farias, M.R.; Alviano, D.S.; Alviano, C.S.; Futuro, D.O.; Ferreira, V.; et al. In vitro and in vivo antifungal activity of buparvaquone against Sporothrix brasiliensis. Antimicrob. Agents Chemother. 2021, 65, e0069921. [Google Scholar] [CrossRef] [PubMed]
- Brilhante, R.S.; Pereira, V.S.; Oliveira, J.S.; Lopes, R.G.; Rodrigues, A.M.; Camargo, Z.P.; Pereira-Neto, W.A.; Castelo-Branco, D.S.; Cordeiro, R.A.; Sidrim, J.J.; et al. Pentamidine inhibits the growth of Sporothrix schenckii complex and exhibits synergism with antifungal agents. Future Microbiol. 2018, 13, 1129–1140. [Google Scholar] [CrossRef] [PubMed]






| In Vitro Antifungal Activity a | ||||
|---|---|---|---|---|
| High (MIC ≤ 1 µg/mL) | Moderate (1 < MIC ≤ 4 µg/mL) | Low (MIC > 4 µg/mL) | Reference | |
| Polyenes | ||||
| Amphotericin B | [262] | |||
| Azoles | ||||
| Albaconazole | [286] | |||
| Clotrimazole | [287] | |||
| Eberconazole | [286] | |||
| Fluconazole | [286] | |||
| Itraconazole | [262] | |||
| Isavuconazole | [288] | |||
| Ketoconazole | [262] | |||
| Miconazole | [286] | |||
| Posaconazole | [262] | |||
| Ravuconazole | [286] | |||
| Voriconazole | [262] | |||
| Allylamines | ||||
| Terbinafine | [262] | |||
| Naftifine | [289] | |||
| Echinocandins | ||||
| Anidulafungin | [286] | |||
| Caspofungin | [286] | |||
| Micafungin | [286] | |||
| Pirimidine | ||||
| Flucytosine | [286] | |||
| Group | Compound | Minimum Inhibitory Concentration a | Antifungal Effect or Mechanism of Action | Reference |
|---|---|---|---|---|
| Hydrazone derivatives | 22-hydrazone-imidazolin-2-yl-chol-5-ene-3β-ol | 0.01–0.5 μg/mL | Inhibition of ergosterol biosynthesis | [291] |
| 4-bromo-N′-(3,5-dibromo-2-hydroxybenzylidene)-benzohydrazide | 0.12–1 μg/mL | Inhibition of vesicular transport and cell cycle progression | [290] | |
| Oxadiazole | N-[5-(4-chlorophenyl)-1,3,4-oxadiazol-2-yl]-3-(trifluoromethyl)benzamide | 0.25–0.5 μM | Cell membrane disruption and neutral lipid accumulation | [292] |
| Alkylphospholipid analogc | TCAN26 | 0.25–2 μg/mL | Cell membrane disruption | [293] |
| Pentathiepin | 23 | 0.5–1 μg/mL | Unknown | [294] |
| Benzisothiazolone | 1.9 | 0.5 μg/mL | Apoptosis induction | [295] |
| Metal complex | Zn(itraconazole)2Cl2 | 0.08 μM | Unknown | [296] |
| Zn(ketoconazole)2(Ac)2⋅H2O | 0.125 μM | Unknown | [297] | |
| [Cu(PPh3)2(ketoconazole)2]NO3 | 0.006 μM | Unknown | [298] | |
| Naphthoquinone derivative | 2,5-dichloro-3,6-bis(4-methylpiperazin-1-yl)cyclohexa-2,5-diene-1,4-dione | 1.56 µg/mL | Unknown | [299] |
| Natural products | farnesol | 0.003–0.222 μg/mL | Unknown | [300] |
| Repositionable drugs | Miltefosine | 1–2 µg/mL | Cell membrane disruption | [278] |
| 0.25–2 μg/mL | [301] | |||
| Iodoquinol | 0.5–1 μM | Cell membrane disruption | [302] | |
| Buparvaquone b | 0.005–0.16 μg/mL | Mitochondrial dysfunction | [303] | |
| Ibuprofen | 0.03–0.5 μg/mL c | Cell membrane disruption and ROS accumulation | [292] | |
| Pentamidine | 0.06–0.25 μg/mL | DNA intercalation | [304] | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rodrigues, A.M.; Gonçalves, S.S.; de Carvalho, J.A.; Borba-Santos, L.P.; Rozental, S.; Camargo, Z.P.d. Current Progress on Epidemiology, Diagnosis, and Treatment of Sporotrichosis and Their Future Trends. J. Fungi 2022, 8, 776. https://doi.org/10.3390/jof8080776
Rodrigues AM, Gonçalves SS, de Carvalho JA, Borba-Santos LP, Rozental S, Camargo ZPd. Current Progress on Epidemiology, Diagnosis, and Treatment of Sporotrichosis and Their Future Trends. Journal of Fungi. 2022; 8(8):776. https://doi.org/10.3390/jof8080776
Chicago/Turabian StyleRodrigues, Anderson Messias, Sarah Santos Gonçalves, Jamile Ambrósio de Carvalho, Luana P. Borba-Santos, Sonia Rozental, and Zoilo Pires de Camargo. 2022. "Current Progress on Epidemiology, Diagnosis, and Treatment of Sporotrichosis and Their Future Trends" Journal of Fungi 8, no. 8: 776. https://doi.org/10.3390/jof8080776
APA StyleRodrigues, A. M., Gonçalves, S. S., de Carvalho, J. A., Borba-Santos, L. P., Rozental, S., & Camargo, Z. P. d. (2022). Current Progress on Epidemiology, Diagnosis, and Treatment of Sporotrichosis and Their Future Trends. Journal of Fungi, 8(8), 776. https://doi.org/10.3390/jof8080776







