- Article
Computational Screening of AI-Generated Antihypertensive Virtual Leads for Polypharmacological Anticancer Potential
- Uche A. K. Chude-Okonkwo and
- Mokete Motente
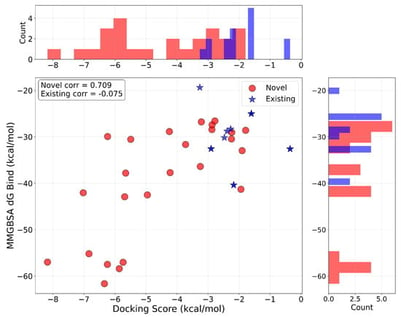
Background: The growing recognition of shared molecular pathways and molecular signatures between cardiovascular diseases and cancer has motivated interest in exploring antihypertensive-associated chemical space for oncological applications. Concurrently, artificial intelligence (AI)-driven molecular generation has enabled the rapid creation of virtual lead candidates for specific therapeutic indications, although their broader biological interaction profiles often remain unexplored. Methods: In this paper, we explore the computational screening of a library of AI-generated antihypertensive virtual lead compounds to evaluate their polypharmacological anticancer potential. The compounds were originally designed and prioritized for modulating -adrenergic receptors but are here re-evaluated in a cancer-focused context using a multi-stage in silico approach. We chose five (5) known cancer target proteins and performed compound profiling for drug-likeness, pharmacokinetic suitability, and safety. Docking simulations, binding free energy estimates, molecular interaction mapping, and pharmacophore modeling were used to evaluate the molecules’ interactions with the cancer-linked protein targets. We employed the binding free energy estimates of the ligand–protein complexes to determine compounds with polypharmacological anticancer potential. In addition, molecular dynamics simulations of some of the compounds with polypharmacological anticancer potential were employed to evaluate binding stability and dynamic behavior of selected ligand–target complexes. Results: Several compounds showed good docking scores, physicochemical characteristics, and pharmacokinetic profiles. Also, the results reveal that several AI-generated antihypertensive virtual leads exhibit favorable multi-target binding profiles, with consistent docking affinities and stable interaction networks across multiple cancer-related targets. Conclusions: Our findings suggest that several of the hypothetically evaluated compounds exhibit favorable physicochemical properties, acceptable predicted pharmacokinetic and safety profiles, and consistent predicted binding affinities across multiple cancer-relevant targets.
19 February 2026