-
 Cardiac Aftermath of Gestational Diabetes—From Intrauterine Impact to Lifelong Complications: A Systematic Review
Cardiac Aftermath of Gestational Diabetes—From Intrauterine Impact to Lifelong Complications: A Systematic Review -
 A Zebrafish Seizure Model of cblX Syndrome Reveals a Dose-Dependent Response to mTor Inhibition
A Zebrafish Seizure Model of cblX Syndrome Reveals a Dose-Dependent Response to mTor Inhibition -
 The Interplay of One-Carbon Metabolism, Mitochondrial Function, and Developmental Programming in Ruminant Livestock
The Interplay of One-Carbon Metabolism, Mitochondrial Function, and Developmental Programming in Ruminant Livestock
Journal Description
Journal of Developmental Biology
Journal of Developmental Biology
is an international, peer-reviewed, open access journal on the development of multicellular organisms at the molecule, cell, tissue, organ and whole organism levels published quarterly online by MDPI.
- Open Access— free for readers, with article processing charges (APC) paid by authors or their institutions.
- High Visibility: indexed within Scopus, ESCI (Web of Science), PubMed, PMC, PubAg, CAPlus / SciFinder, and other databases.
- Rapid Publication: manuscripts are peer-reviewed and a first decision is provided to authors approximately 27.1 days after submission; acceptance to publication is undertaken in 5.6 days (median values for papers published in this journal in the second half of 2025).
- Recognition of Reviewers: reviewers who provide timely, thorough peer-review reports receive vouchers entitling them to a discount on the APC of their next publication in any MDPI journal, in appreciation of the work done.
- Testimonials: See what our editors and authors say about Journal of Developmental Biology.
Impact Factor:
2.5 (2024);
5-Year Impact Factor:
2.8 (2024)
Latest Articles
Transformation of the Biological Paradigm in Bone Regeneration: An Integrative Review
J. Dev. Biol. 2026, 14(1), 14; https://doi.org/10.3390/jdb14010014 - 11 Mar 2026
Abstract
►
Show Figures
Bone tissue is among the most commonly transplanted tissues worldwide. The treatment of critical-sized bone defects remains a significant challenge, as there is currently no universally accepted experimental model or therapeutic standard. Recent advances in fundamental cell biology are driving a paradigm shift
[...] Read more.
Bone tissue is among the most commonly transplanted tissues worldwide. The treatment of critical-sized bone defects remains a significant challenge, as there is currently no universally accepted experimental model or therapeutic standard. Recent advances in fundamental cell biology are driving a paradigm shift in approaches to bone regeneration, highlighting the transformative potential of biofabrication technologies that integrate tissue engineering with personalized regenerative strategies. Three-dimensional (3D) bioprinting technology enables precise control over the architecture and spatial distribution of cellular and biologically active components, facilitating the creation of complex, personalized bone constructs. Central to this process are bioinks and biomaterials that mimic the extracellular matrix (ECM) and provide an optimal microenvironment for cellular function. Despite the substantial body of accumulated data, a comprehensive theoretical framework for functional bone biofabrication has not yet been fully established, emphasizing both the challenges and the innovative potential of the field. This integrative review synthesizes current knowledge on bone biology—from embryogenesis and cell–matrix interactions to molecular and neural regulation—and links it to the opportunities offered by biofabrication. Particular attention is given to bioinks as mediators between cell biology and engineering sciences, as well as to strategies for creating biomimetic ECM, optimizing scaffold design, and guiding future research toward clinically translatable bone regeneration.
Full article
Open AccessReview
SIX3 as a Regulator of Development and Disease
by
Ana Beatriz Matos, Laura Jesus Castro and Torcato Martins
J. Dev. Biol. 2026, 14(1), 13; https://doi.org/10.3390/jdb14010013 - 6 Mar 2026
Abstract
►▼
Show Figures
Transcriptional regulation is pivotal for developmental processes and cell fate specification in homeostasis. One particularly relevant group of transcription factors is the sine oculis homeobox (SIX) family, which is involved in a wide range of molecular processes from development to tissue maintenance. Within
[...] Read more.
Transcriptional regulation is pivotal for developmental processes and cell fate specification in homeostasis. One particularly relevant group of transcription factors is the sine oculis homeobox (SIX) family, which is involved in a wide range of molecular processes from development to tissue maintenance. Within this family, distinct subfamilies exhibit specific DNA-binding preferences and can function as transcriptional activators or repressors. In this review, we focus on the Optix/SIX3–SIX6 subfamily and discuss their roles as transcriptional regulators, as well as the consequences of their deregulation for neuronal and ocular development and for the maintenance of tissue homeostasis. We further examine how SIX3 can act either as a tumour suppressor or as a marker of poor prognosis in different cancer types. Moreover, we summarize recent findings on the role of SIX3 in pancreatic β cells and highlight emerging evidence that SIX2 also contributes to β-cell identity and regulatory stability. Downregulation of SIX2 and SIX3 alters gene regulatory programs associated with β-cell homeostasis and contributes to type 2 diabetes. As accumulating evidence links members of the SIX family to cancer and metabolic disease, it is crucial to characterize how these transcription factors regulate cell identity, with important implications for disease mechanisms and therapeutic strategies.
Full article

Figure 1
Open AccessReview
Genomic Impacts of Biological Exposures
by
Amalia S. Parra and Christopher A. Johnston
J. Dev. Biol. 2026, 14(1), 12; https://doi.org/10.3390/jdb14010012 - 5 Mar 2026
Abstract
►▼
Show Figures
Development and maintenance of complex tissues depends on a number of coordinated steps from early development through adulthood. These processes are fundamentally controlled by highly regulated gene expression patterns. Although critical contributors during development, intrinsic changes in gene expression alone cannot fully explain
[...] Read more.
Development and maintenance of complex tissues depends on a number of coordinated steps from early development through adulthood. These processes are fundamentally controlled by highly regulated gene expression patterns. Although critical contributors during development, intrinsic changes in gene expression alone cannot fully explain the complicated pathways that control tissue homeostasis. Rather, tissues are continuously exposed to extrinsic factors that also influence essential cellular processes. These external environmental factors are collectively known as the exposome. Notably, how different exposures impact gene expression and protein function, as well as how certain exposures lead to disease states, is not well understood. To understand how internal and external factors influence organismal development and homeostasis, it is necessary to consider how genetic and nongenetic components interact to direct critical biochemical pathways. Doing so presents new avenues for precision medicine, understanding disease progression, identifying biological threats, and improving biological security concerns. In this review, we present recent advances in exposure biology, focusing on how these innovations can help identify novel biomarkers to better understand changing exposome components. We also discuss the need to integrate technologies and exposure research to better identify and predict threats.
Full article

Figure 1
Open AccessReview
Origins of Avian Hyperactive Mitochondria, Genome Compaction, and Air-Sac Physiology in Early Theropods During the Carnian Pluvial Episode
by
Takumi Satoh
J. Dev. Biol. 2026, 14(1), 11; https://doi.org/10.3390/jdb14010011 - 2 Mar 2026
Abstract
►▼
Show Figures
Extant birds and the earliest dinosaurs may share fundamental metabolic features essential for aerobic exercise, suggesting that the extraordinary physical performance typical of avian species originated when dinosaurs first appeared during the Carnian Pluvial Episode (CPE). This physiological adaptation is complemented by hyperactive
[...] Read more.
Extant birds and the earliest dinosaurs may share fundamental metabolic features essential for aerobic exercise, suggesting that the extraordinary physical performance typical of avian species originated when dinosaurs first appeared during the Carnian Pluvial Episode (CPE). This physiological adaptation is complemented by hyperactive mitochondria that exhibit high oxygen consumption and low reactive oxygen species production. Molecular genomics of fossils, the so-called “Jurassic Genome,” indicates that these early dinosaurs possessed compact genomes, 50–60% the size of the human genome, and small cells, implying a highly stringent metabolic regime. We suggest that hyperactive mitochondria, closely associated with compact genomes and small cells, drive theropod adaptation to the hot, dry, and hypoxic environments of the Late Triassic period, ultimately enabling their ecological dominance. Early dinosaurs such as Herrerasaurus are hypothesized to have possessed advanced physiological traits shared with modern birds, including hyperactive mitochondria, compact genomes, small cells, and a developing air-sac system. Collectively, these features most likely may have contributed to exceptional metabolic capacity, locomotor performance, and adaptation to the harsh environment of the CPE.
Full article

Figure 1
Open AccessSystematic Review
Evolutionary Restructuring and Systematic Review of the NBPF Gene Family: Comparative Genomics, Functional Divergence, and Disease-Linked Pathways
by
Manuel Escalona and Rosa Roy
J. Dev. Biol. 2026, 14(1), 10; https://doi.org/10.3390/jdb14010010 - 24 Feb 2026
Abstract
►▼
Show Figures
The Neuroblastoma Breakpoint Family (NBPF) consists of 23 genes, 9 of which are pseudogenes, and is characterized by extensive duplication events and species-specific diversification in Homo sapiens, as well as by the presence of a unique protein domain known as Olduvai (also
[...] Read more.
The Neuroblastoma Breakpoint Family (NBPF) consists of 23 genes, 9 of which are pseudogenes, and is characterized by extensive duplication events and species-specific diversification in Homo sapiens, as well as by the presence of a unique protein domain known as Olduvai (also referred to as DUF1220 or the NBPF domain). Previous studies have attempted to define subfamilies based on the presence of HLS triplet domains; however, this classification has become increasingly unclear with the identification of additional NBPF members. The family remains poorly understood, and the functions of many genes are still unknown, although several have been hypothesized to play key roles in cell proliferation and developmental processes, particularly in neural and skeletal tissues. In this study, we systematically analyzed all available data on the NBPF gene family using the PRISMA-S methodology to infer the biological functions in which these genes may be involved. We also generated multiple phylogenetic trees to support the creation of coherent subfamilies and to correlate the origin of each subfamily with homologous genes in our last common ancestor with the Pan genus, providing what we believe to be one of the most comprehensive phylogenetic reconstructions including all currently annotated NBPF members. Through comparative genomic and phylogenetic analyses, we propose that the NBPF may have originated from a duplication of the PDE4DIP gene, with NBPF26 representing the ancestral member from which the remaining NBPF genes diverged via lineage-specific segmental duplications. In this systematic review and comparative genomic study, we present the first integrative synthesis of our knowledge of the NBPF, encompassing its evolutionary origins, structural dynamics, expression across tissues, and clinical associations.
Full article

Graphical abstract
Open AccessArticle
The Influence of Fluidic Flow Stress on the Development of the Secondary Palate
by
Masayo Nagata, Satoru Hayano, Ziyi Wang, Takahiro Kosami and Hiroshi Kamioka
J. Dev. Biol. 2026, 14(1), 9; https://doi.org/10.3390/jdb14010009 - 12 Feb 2026
Abstract
►▼
Show Figures
Craniofacial development is orchestrated by a finely regulated interplay of numerous genes and signaling pathways. Palatogenesis proceeds through a complex, stepwise process, in which endogenous mechanical stresses within tissues have been implicated. However, the impact of exogenous fluidic flow mechanical stress derived from
[...] Read more.
Craniofacial development is orchestrated by a finely regulated interplay of numerous genes and signaling pathways. Palatogenesis proceeds through a complex, stepwise process, in which endogenous mechanical stresses within tissues have been implicated. However, the impact of exogenous fluidic flow mechanical stress derived from maternal movement on palatal development remains unclear. In this study, we investigated the effect of exogenous fluidic flow mechanical stress on palatal morphogenesis, focusing on the horizontal outgrowth of palatal shelves after elevation. Palatal tissues dissected from mouse embryos were subjected to organ culture with or without mechanical loading (loaded and unloaded groups, respectively). Stress magnitude was quantified by calculating wave energy, and morphometric and molecular analyses were performed. Compared with the unloaded group, palatal shelves in the loaded group showed significant increases in thickness and volume, accompanied by enhanced cell proliferation, nuclear translocation of YAP and β-catenin, and upregulation of the osteogenic markers Osterix and Osteocalcin. No significant difference in apoptosis was observed. These findings indicate that exogenous mechanical stress promotes cell proliferation and osteogenic differentiation through the Hippo and WNT/β-catenin pathways in palate explants. Our results suggest that moderate maternal movement-induced mechanical stress contributes to normal palatogenesis, providing new insights into the mechanisms underlying cleft palate.
Full article

Figure 1
Open AccessArticle
Vestigial-like 4 Regulates Neurogenesis and Neural Crest Formation During Xenopus Development
by
Pierre Thiébaud, Emilie Simon, François Moisan, Sandrine Fedou, Hamid-Reza Rezvani and Nadine Thézé
J. Dev. Biol. 2026, 14(1), 8; https://doi.org/10.3390/jdb14010008 - 11 Feb 2026
Abstract
►▼
Show Figures
VESTIGIAL-LIKE proteins constitute a family of evolutionarily conserved proteins that act as cofactors in regulating gene expression through their binding to TEAD transcription factors. Among the four members of this family in vertebrates, VESTIGIAL-LIKE 4 has emerged as a tumor suppressor that competes
[...] Read more.
VESTIGIAL-LIKE proteins constitute a family of evolutionarily conserved proteins that act as cofactors in regulating gene expression through their binding to TEAD transcription factors. Among the four members of this family in vertebrates, VESTIGIAL-LIKE 4 has emerged as a tumor suppressor that competes with YAP in binding TEADs, thus inhibiting the HIPPO pathway downstream of YAP. Nevertheless, very few studies have addressed its function during early vertebrate development. Here, we used gain- and loss-of-function strategies to investigate the role of vestigial-like 4 during Xenopus laevis development. Our data show that vestigial-like 4 is a key regulator of neurogenesis and neural crest formation. In embryos depleted of vestigial-like 4, neurogenesis is severely impaired, and neither neurog1 nor neurod1 is able to stimulate neurogenesis. Vestigial-like 4 is also required for neural crest formation through pax3 and sox9 regulation, and this property does not necessarily require its interaction with tead. Collectively, our findings demonstrate that vestigial-like 4 is an important regulator of neurogenesis and neural crest formation. Although vestigial-like 4 can bind to tead proteins in the embryo, its function does not depend solely on this interaction, suggesting a complex level of regulation with which vestigial-like 4 regulates early steps in development and differentiation.
Full article

Figure 1
Open AccessArticle
Functional State of Lampbrush Chromosomes in Early Vitellogenic Oocytes of Hibernating Frogs Rana temporaria
by
Nadya V. Ilicheva and Olga I. Podgornaya
J. Dev. Biol. 2026, 14(1), 7; https://doi.org/10.3390/jdb14010007 - 2 Feb 2026
Abstract
►▼
Show Figures
Lampbrush chromosomes (LBCs) are a feature of amphibian oocytes and are typically associated with high levels of transcription during active oocyte growth. However, their state during winter hibernation has not been studied. Here, we investigated LBCs in early vitellogenic oocytes (early stage 4)
[...] Read more.
Lampbrush chromosomes (LBCs) are a feature of amphibian oocytes and are typically associated with high levels of transcription during active oocyte growth. However, their state during winter hibernation has not been studied. Here, we investigated LBCs in early vitellogenic oocytes (early stage 4) of the grass frog Rana temporaria during winter hibernation. We found that the chromosomes retained their lampbrush morphology, and the phosphorylated form of RNA polymerase II resided on the lateral loops. Transcription on the lateral loops was reduced but detectable at cold conditions and significantly increased when the oocytes were transferred at room temperature. Satellite S1a transcripts were detected at the lateral loops of the chromosomes by RNA FISH. The possible significance of maintaining chromosomes in the lampbrush form during hibernation is discussed.
Full article

Figure 1
Open AccessArticle
Female Aging Affects Coilin Pattern in Mouse Cumulus Cells
by
Alexey S. Anisimov, Dmitry S. Bogolyubov and Irina O. Bogolyubova
J. Dev. Biol. 2026, 14(1), 6; https://doi.org/10.3390/jdb14010006 - 15 Jan 2026
Abstract
►▼
Show Figures
Cumulus cells (CCs) are a distinct population of granulosa cells (GCs) that surround the developing and ovulated mammalian oocyte. The features of their structural organization and the expression pattern of key genes significantly affect oocyte viability. Changes in the functional activity of the
[...] Read more.
Cumulus cells (CCs) are a distinct population of granulosa cells (GCs) that surround the developing and ovulated mammalian oocyte. The features of their structural organization and the expression pattern of key genes significantly affect oocyte viability. Changes in the functional activity of the nucleus are often expressed in changes in the structure of nuclear bodies (NBs), including Cajal bodies (CBs). The diagnostic protein of CBs is coilin, which maintains their structural integrity. Using fluorescent and electron microscopy, we examined maternal aging-associated changes in coilin pattern in mouse CCs. We found that older mice had a decrease in the number of coilin-positive bodies, while external transcriptome data analysis revealed no significant changes in Coil and Smn1 gene expression. We hypothesized that the age-related dynamics of coilin-containing bodies are determined not by changes in the expression level of key components of these bodies, but by age-related changes in CC metabolism. Considering that CCs are a by-product of IVF protocols, making them available for analysis in sufficient quantities, age-related changes in the number and size of coilin-positive NBs in CCs may serve as a promising biomarker for assessing ovarian functional aging.
Full article

Graphical abstract
Open AccessArticle
Influence of Obstructive Uropathy on Cyst Formation and Nephrogenesis: Insights from a Fetal Lamb Model
by
Kohei Kawaguchi, Takuya Kawaguchi, Juma Obayashi, Yasuji Seki, Kunihide Tanaka, Kei Ohyama, Junki Koike, Shigeyuki Furuta, Kevin C. Pringle and Hiroaki Kitagawa
J. Dev. Biol. 2026, 14(1), 5; https://doi.org/10.3390/jdb14010005 - 9 Jan 2026
Abstract
Obstructive uropathy (OU) during fetal development induces a fetal cystic dysplastic kidney. The mechanisms of cyst formation and the onset of renal dysfunction remain unclear. Determining whether nephrogenic potential persists during fetal life may suggest whether early intervention could preserve renal development. We
[...] Read more.
Obstructive uropathy (OU) during fetal development induces a fetal cystic dysplastic kidney. The mechanisms of cyst formation and the onset of renal dysfunction remain unclear. Determining whether nephrogenic potential persists during fetal life may suggest whether early intervention could preserve renal development. We aimed to evaluate residual nephrogenic activity in fetal cystic dysplastic kidneys using β-catenin and CD10 immunostaining, and to assess whether the site of obstruction influences cystogenesis. After appropriate approval, 20 timed-gestation fetal lambs had OU created at 60 days. Males underwent urethral and urachal ligation (n = 8, 3 lost), and females underwent unilateral ureteric ligation (n = 8, 1 lost). Fetuses were sacrificed at 80 days (n = 6) and 140 days (term, n = 10), comparing kidneys with normal controls of the same gestational age using immunohistochemical staining for β-catenin and CD10. Developing fetal cystic dysplastic kidneys were identified at 80 days. β-catenin staining showed the absence of granular cytoplasmic expression in cystic regions, indicating arrested nephrogenesis. In male models, cysts originated exclusively from proximal tubules. Female models exhibited mixed proximal and distal tubular involvement. CD10 staining confirmed the loss of proximal tubular markers. Renal development remained arrested at term. Cyst formation disrupts renal development early in gestation, which persists until term. Differences in cystogenesis between the models suggest that the site of obstruction influences pathogenic mechanisms.
Full article
(This article belongs to the Special Issue Developmental Biology of the Kidney: From Molecular Mechanisms to Congenital Disorders)
►▼
Show Figures

Graphical abstract
Open AccessArticle
Discovery of New Markers for Haemogenic Endothelium and Haematopoietic Progenitors in the Mouse Yolk Sac
by
Guillermo Diez-Pinel, Alessandro Muratore, Christiana Ruhrberg and Giovanni Canu
J. Dev. Biol. 2026, 14(1), 4; https://doi.org/10.3390/jdb14010004 - 6 Jan 2026
Abstract
►▼
Show Figures
Erythro-myeloid progenitors (EMPs) originate from the haemogenic endothelium in the yolk sac via an endothelial-to-haematopoietic transition (EHT) to generate blood and immune cells that support embryo development. Yet, the transitory nature of EHT and the limited availability of molecular markers have constrained our
[...] Read more.
Erythro-myeloid progenitors (EMPs) originate from the haemogenic endothelium in the yolk sac via an endothelial-to-haematopoietic transition (EHT) to generate blood and immune cells that support embryo development. Yet, the transitory nature of EHT and the limited availability of molecular markers have constrained our understanding of the origin, identity, and differentiation dynamics of EMPs. Here, we have refined the annotation of yolk sac haemato-vascular populations in publicly available single-cell RNA sequencing (scRNAseq) datasets from mouse embryos to identify novel molecular markers of haemogenic endothelium and EMPs. By sub-clustering key cell populations followed by pseudotime analysis, we refined cluster annotations and then reconstructed differentiation trajectories. Subsequent differential gene expression analysis between clusters identified novel cell surface markers for haemogenic endothelial cells (Fxyd5 and Scarf1) and EMPs (Fcer1g, Tyrobp, and Mctp1). Further, we have identified candidate signalling and metabolic pathways that may regulate yolk sac haematopoietic emergence and differentiation. The specificity of FXYD5, SCARF1, and FCER1G for haemogenic endothelium and EMPs was validated by immunostaining of the mouse yolk sac. These insights into the transcriptional dynamics in the yolk sac should support future investigation of EHT and haematopoietic differentiation during early mammalian development.
Full article

Figure 1
Open AccessReview
The Interplay of One-Carbon Metabolism, Mitochondrial Function, and Developmental Programming in Ruminant Livestock
by
Kazi Sarjana Safain, Kendall C. Swanson and Joel S. Caton
J. Dev. Biol. 2026, 14(1), 3; https://doi.org/10.3390/jdb14010003 - 3 Jan 2026
Abstract
Maternal nutrition during gestation profoundly influences fetal growth, organogenesis, and long-term offspring performance through developmental programming. Among the molecular mechanisms responsive to maternal nutrient availability, one-carbon metabolism plays a central role by integrating folate, methionine, choline, and vitamin B12 pathways that regulate
[...] Read more.
Maternal nutrition during gestation profoundly influences fetal growth, organogenesis, and long-term offspring performance through developmental programming. Among the molecular mechanisms responsive to maternal nutrient availability, one-carbon metabolism plays a central role by integrating folate, methionine, choline, and vitamin B12 pathways that regulate methylation, nucleotide synthesis, and antioxidant defense. These processes link maternal nutritional status to epigenetic remodeling, cellular proliferation, and redox balance during fetal development. Mitochondria act as nutrient sensors that translate maternal metabolic cues into bioenergetic and oxidative signals, shaping tissue differentiation and metabolic flexibility. Variations in maternal diet have been associated with shifts in fetal amino acid, lipid, and energy metabolism, suggesting adaptive responses to constrained intrauterine environments. This review focuses on the molecular interplay between one-carbon metabolism, mitochondrial function, and metabolomic adaptation in developmental programming of ruminant livestock. Understanding these mechanisms offers opportunities to design precision nutritional strategies that enhance fetal growth, offspring productivity, and long-term resilience in livestock production systems.
Full article
(This article belongs to the Special Issue Mitochondrial Function and Dysfunction in Developmental Biology Metabolism)
►▼
Show Figures
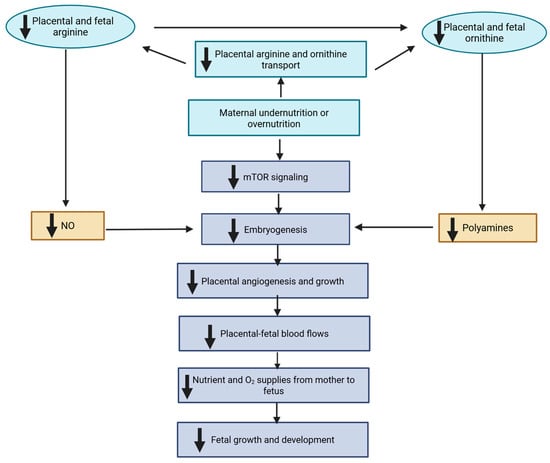
Figure 1
Open AccessArticle
A Zebrafish Seizure Model of cblX Syndrome Reveals a Dose-Dependent Response to mTor Inhibition
by
Claudia B. Gil, David Paz, Briana E. Pinales, Victoria L. Castro, Claire E. Perucho, Annalise Gonzales, Giulio Francia, Sepiso K. Masenga, Antentor Hinton, Jr. and Anita M. Quintana
J. Dev. Biol. 2026, 14(1), 2; https://doi.org/10.3390/jdb14010002 - 25 Dec 2025
Abstract
►▼
Show Figures
Mutations in the transcriptional co-factor HCFC1 cause methylmalonic aciduria and homocystinemia, cblX type (cblX) (MIM#309541), non-syndromic X-linked intellectual disability (XLID), and focal epilepsy. Zebrafish studies have revealed increased activation of the Akt/mTor signaling pathway after mutation of hcfc1a, one ortholog
[...] Read more.
Mutations in the transcriptional co-factor HCFC1 cause methylmalonic aciduria and homocystinemia, cblX type (cblX) (MIM#309541), non-syndromic X-linked intellectual disability (XLID), and focal epilepsy. Zebrafish studies have revealed increased activation of the Akt/mTor signaling pathway after mutation of hcfc1a, one ortholog of HCFC1. mTOR hyperactivation is linked to seizures, and its inhibition alleviates epilepsy in other preclinical models. We hypothesized that mTor overactivity in hcfc1a mutant zebrafish increases seizure susceptibility and/or severity. We employed a two-concentration model of the seizure-inducing agent, pentylenetetrazol (PTZ), with or without pretreatment of the mTor inhibitor, torin1. Mutation of hcfc1a did not alter the response to PTZ at sub-optimal concentrations, and the pharmaceutical inhibition of mTor using the compound Torin1 reduced response to 1 µM PTZ, but only in a dose-dependent manner. Higher doses of mTor inhibition did not reduce the seizure response in mutant larvae but were effective in wildtype siblings. These data suggest that inhibition of mTor in an hcfc1a-deficient background leads to a reaction that differs from the traditional response observed in wildtype siblings. Collectively, we present a model that can be used to test dose–response and the development of combinatorial treatment approaches in a high-throughput manner.
Full article

Graphical abstract
Open AccessArticle
The Epithelial Egg Tooth of the Chicken Shares Protein Markers with the Embryonic Subperiderm and Feathers
by
Attila Placido Sachslehner, Julia Steinbinder, Claudia Hess, Veronika Mlitz and Leopold Eckhart
J. Dev. Biol. 2026, 14(1), 1; https://doi.org/10.3390/jdb14010001 - 22 Dec 2025
Abstract
The epithelial egg tooth is used by birds to open the eggshell for hatching. This ectodermal structure consists of a multilayered periderm and a hard cornified portion, the caruncle or actual egg tooth. Here, we determined the protein composition of the egg tooth
[...] Read more.
The epithelial egg tooth is used by birds to open the eggshell for hatching. This ectodermal structure consists of a multilayered periderm and a hard cornified portion, the caruncle or actual egg tooth. Here, we determined the protein composition of the egg tooth of the chicken and compared the proteins to markers of other epithelia identified in previous studies. The egg tooth and the upper beak of chicken embryos of Hamburger and Hamilton (HH) stage 44 were subjected to mass spectrometry-based proteomics. We found that scaffoldin, a marker of the embryonic periderm and the feather sheath, was enriched in the egg tooth relative to the beak. Likewise, Epidermal Differentiation protein containing DPCC Motifs (EDDM) and Epidermal Differentiation protein starting with a MTF motif and rich in Histidine (EDMTFH), which had previously been characterized as markers of the subperiderm on embryonic scutate scales and the barbs of feathers, were also enriched in the egg tooth. The expression of EDDM and EDMTFH was confirmed RT-PCR analysis. Our data suggest that the epithelial egg tooth is related to the subperiderm and feathers, a hypothesis with potentially important implications for the evolution of the avian integument.
Full article
(This article belongs to the Special Issue Feature Papers from Journal of Developmental Biology Reviewers, 2nd Edition)
►▼
Show Figures

Figure 1
Open AccessReview
Pathophysiology and Management of Placenta Accreta Spectrum
by
Lana Shteynman, Genevieve Monanian, Gilberto Torres, Giancarlo Sabetta, Deborah M. Li, Zhaosheng Jin, Tiffany Angelo, Bahaa E. Daoud and Morgane Factor
J. Dev. Biol. 2025, 13(4), 45; https://doi.org/10.3390/jdb13040045 - 10 Dec 2025
Cited by 1
Abstract
►▼
Show Figures
Placenta Accreta Spectrum (PAS) disorders, including placenta accreta, increta, and percreta, are serious obstetric conditions characterized by abnormal placental adherence to the uterine wall. With increasing incidence, PAS poses significant risks, primarily through massive hemorrhage during or after delivery, often necessitating hysterectomy. Key
[...] Read more.
Placenta Accreta Spectrum (PAS) disorders, including placenta accreta, increta, and percreta, are serious obstetric conditions characterized by abnormal placental adherence to the uterine wall. With increasing incidence, PAS poses significant risks, primarily through massive hemorrhage during or after delivery, often necessitating hysterectomy. Key risk factors include prior cesarean sections, uterine surgery, and placenta previa diagnosis. In this review, we will examine the pathophysiology of PAS, with a focus on the mechanisms underlying abnormal trophoblast invasion and defective decidualization. We will highlight the role of uterine scarring, extracellular matrix remodeling, dysregulated signaling pathways, and immune and vascular alterations in disrupting the maternal-fetal interface, ultimately predisposing to morbid placentation and delivery complications. We will also discuss the life-threatening complications of PAS, such as shock and multi-organ failure, which require urgent multidisciplinary intensive care, as well as the optimization of management through preoperative planning and intraoperative blood loss control to reduce maternal morbidity and mortality.
Full article

Figure 1
Open AccessSystematic Review
Cardiac Aftermath of Gestational Diabetes—From Intrauterine Impact to Lifelong Complications: A Systematic Review
by
Sophia Tsokkou, Ioannis Konstantinidis, Antonios Keramas, Vasileios Anastasiou, Alkis Matsas, Maria Florou, Alexandra Arvanitaki, Emmanouela Peteinidou, Theodoros Karamitsos, George Giannakoulas, Themistoklis Dagklis, Theodora Papamitsou, Antonios Ziakas and Vasileios Kamperidis
J. Dev. Biol. 2025, 13(4), 44; https://doi.org/10.3390/jdb13040044 - 8 Dec 2025
Abstract
►▼
Show Figures
Background. Gestational diabetes mellitus (GDM) induces maternal hyperglycemia, which may alter fetal cardiac structure and function, increasing short- and long-term cardiovascular risks. Purpose. To systematically review the evidence on the fetal cardiac structural and functional effects of GDM, to explore the
[...] Read more.
Background. Gestational diabetes mellitus (GDM) induces maternal hyperglycemia, which may alter fetal cardiac structure and function, increasing short- and long-term cardiovascular risks. Purpose. To systematically review the evidence on the fetal cardiac structural and functional effects of GDM, to explore the diagnostic role of novel imaging and biochemical biomarkers, and to summarize the long-term cardiovascular complications associated with GDM. Materials and Methods. A systematic search of PubMed, Scopus, and Cochrane Library was conducted according to the PRISMA guidelines. All studies comparing cardiac outcomes in GDM and non-GDM pregnancies were included. Data on myocardial hypertrophy, diastolic and systolic function, imaging modalities, and biomarkers were extracted and qualitatively synthesized. Results. A total of twelve eligible studies were identified. Fetal cardiac hypertrophy and diastolic and early systolic dysfunction are common among GDM pregnancies and can be detected by dual-gate Doppler and speckle-tracking echocardiography. Abnormalities are observed in indices such as the myocardial performance index, E/A, E/e′ ratios, and global longitudinal and circumferential strain in fetuses and may persist in the neonatal period. Alterations may be more pronounced for the right ventricle compared to the left. Septal hypertrophy is associated with elevated umbilical cord pro-brain natriuretic peptide. The risk of early-onset cardiovascular disease in the progeny of diabetic mothers is 29% higher, as evidenced by population-based cohort data. Conclusions. GDM is linked to fetal cardiac remodeling and an increased long-term cardiovascular risk. Early detection and customized interventions to reduce adverse outcomes may be achieved by integrating advanced echocardiographic techniques and biomarkers into prenatal surveillance.
Full article
Figure 1
Open AccessReview
How Cytoskeletal Disorders Contribute to Errors in the Chromosomal Segregation of Oocytes and Cleavage Stage Embryos
by
Stefka Delimitreva and Irina Chakarova
J. Dev. Biol. 2025, 13(4), 43; https://doi.org/10.3390/jdb13040043 - 2 Dec 2025
Abstract
►▼
Show Figures
Observations of the processes of oogenesis, fertilization, and the earliest embryonic development have given us the opportunity to estimate the importance of chromosomal distribution errors for the success of mammalian reproduction. It is now known that in the large volume of oocytes, zygotes
[...] Read more.
Observations of the processes of oogenesis, fertilization, and the earliest embryonic development have given us the opportunity to estimate the importance of chromosomal distribution errors for the success of mammalian reproduction. It is now known that in the large volume of oocytes, zygotes and the first embryonic cells, the rearrangement of chromatin is associated with a complex rearrangement of cytoskeletal structures, which creates specific problems. This review discusses two main issues critical to the success of early embryos: Why oocyte meiosis is too frequently wrong in chromosomal segregation? Why the first zygotic mitoses are too frequently wrong in chromosomal segregation? We concluded the following: (1) The main cytoskeletal defects that disturb oocyte meiosis are a problematic connection between cytoskeleton and nucleoskeleton, unsuccessful movement of the spindle to the oocyte periphery, unstable anchoring of the spindle to oolemma, and deviations in meiotic spindle morphology; (2) The main cytoskeletal defects that disturb pronuclear unification are nonfunctional male centriole, unsuccessful forming of microtubule aster around the sperm centrosome, problematic movement of the two pronuclei towards each other and inappropriate contacts between centrosomes, microtubules and nuclear pore complexes; (3) Cytoskeletal defects that disturb zygote mitosis are unsuccessful forming of bipolar mitotic spindle, non-synchronized congression of maternal and paternal chromosomes, and unsuccessful attachment of kinetochores to microtubules.
Full article

Figure 1
Open AccessArticle
Dynamic Alterations in Testicular Autophagy in Prepubertal Mice
by
Dong Zhang, Xiaoyun Pang, Zhenxing Yan, Weitao Dong, Zihao Fang, Jincheng Yang, Yanyan Wang, Li Xue, Jiahao Zhang, Chen Xue, Hongwei Duan, Xianghong Du and Yuxuan He
J. Dev. Biol. 2025, 13(4), 42; https://doi.org/10.3390/jdb13040042 - 18 Nov 2025
Abstract
►▼
Show Figures
Autophagy has a potential regulatory effect on spermatogenesis and testicular development. Dynamic alterations in the testicular autophagy of prepubertal mice were analyzed, and the relationship between autophagy levels and testicular development was clarified using C57BL/6 mice aged 1, 2, 4, 6, and 8
[...] Read more.
Autophagy has a potential regulatory effect on spermatogenesis and testicular development. Dynamic alterations in the testicular autophagy of prepubertal mice were analyzed, and the relationship between autophagy levels and testicular development was clarified using C57BL/6 mice aged 1, 2, 4, 6, and 8 weeks. Transmission electron microscopy was used to identify autophagic vacuoles. The expression of autophagy-related proteins and PI3K/AKT/mTOR signaling pathway-related proteins was determined using Western blotting. Localization of microtubule-associated protein light chain 3 (LC3) and sequestosome 1 (p62) in testicular tissues was determined using immunofluorescence and immunohistochemistry. Autophagic vacuoles in spermatogenic cells increased gradually from weeks 1 to 4, peaked at 2 weeks, decreased sharply at 6 weeks, and were undetectable at 8 weeks. The expression of Beclin 1 autophagy-related protein, LC3-II, and p62 was highest at 2 weeks among the five age groups, whereas LC3-II and p62 were mainly localized in spermatogonia and spermatocytes. Moreover, low mTOR expression and its increased expression were detected at 1–2 weeks and 2–8 weeks, respectively. These results show that testicular autophagic levels exhibit a dynamic pattern of “increase (1–2 weeks) followed by a decrease (2–8 weeks),” providing a reference in determining the relationship between autophagy levels and testicular development.
Full article

Figure 1
Open AccessArticle
Cloned Pig Fetuses Have a High Placental Lysophosphatidylcholine Level That Inhibits Trophoblast Cell Activity
by
Junkun Lai, Xiaoyu Gao, Guke Zhang, Xiao Wu, Yiqian Zhang, Shunbo Wang, Zhenfang Wu, Zicong Li and Zheng Xu
J. Dev. Biol. 2025, 13(4), 41; https://doi.org/10.3390/jdb13040041 - 12 Nov 2025
Abstract
►▼
Show Figures
Somatic cell nuclear transfer (SCNT) or cloning technology is widely used in agriculture and biomedicine. However, the application of this technology is limited by the low developmental competence of cloned embryos or fetuses, which frequently exhibit abnormal development of trophoblast cells or placentas.
[...] Read more.
Somatic cell nuclear transfer (SCNT) or cloning technology is widely used in agriculture and biomedicine. However, the application of this technology is limited by the low developmental competence of cloned embryos or fetuses, which frequently exhibit abnormal development of trophoblast cells or placentas. The purpose of this study was to investigate the possible causes of the erroneous placental development of SCNT-derived pig fetuses. The placental transcriptomic and lipidomic profiles were compared between 30-day-old SCNT- and artificial insemination (AI)-produced pig fetuses. Differentially expressed lipid metabolites between two groups of placentas were selected to test their effects on porcine trophoblast cell activity. The results showed that SCNT placentas exhibit impaired lipid metabolism and function. The level of a metabolite, lysophosphatidylcholine (LPC), in the glycerophospholipid metabolism pathway was substantially increased in SCNT placentas, compared with AI placentas. The elevation in LPC content may lead to impaired placental development in cloned pig fetuses, as LPC inhibited the proliferation and migration of porcine trophoblast cells. This study discovers a main cause of erroneous development of cloned pig fetuses, which will be beneficial for understanding the regulation of SCNT embryo development, as well as developing new methods to improve the efficiency of pig cloning.
Full article

Figure 1
Open AccessReview
Defective Neural Stem and Progenitor Cell Proliferation in Neurodevelopmental Disorders
by
Aki Shigenaka, Eri Nitta, Tadashi Nakagawa, Makiko Nakagawa and Toru Hosoi
J. Dev. Biol. 2025, 13(4), 40; https://doi.org/10.3390/jdb13040040 - 7 Nov 2025
Cited by 2
Abstract
►▼
Show Figures
Neurodevelopmental disorders (NDDs), including autism spectrum disorder, intellectual disability, and attention deficit hyperactivity disorder, are increasingly recognized as disorders of early brain construction arising from defects in neural stem and progenitor cell (NSPC) proliferation. NSPCs are responsible for generating the diverse neuronal and
[...] Read more.
Neurodevelopmental disorders (NDDs), including autism spectrum disorder, intellectual disability, and attention deficit hyperactivity disorder, are increasingly recognized as disorders of early brain construction arising from defects in neural stem and progenitor cell (NSPC) proliferation. NSPCs are responsible for generating the diverse neuronal and glial lineages that establish cortical architecture and neural circuitry; thus, their expansion must be tightly coordinated by intrinsic cell cycle regulators and extrinsic niche-derived cues. Disruption of these mechanisms—through genetic mutations, epigenetic dysregulation, or environmental insults—can perturb the balance between NSPC self-renewal and differentiation, resulting in aberrant brain size and connectivity. Recent advances using animal models and human pluripotent stem cell-derived brain organoids have identified key signaling pathways, including Notch, Wnt, SHH, and PI3K–mTOR, as central hubs integrating proliferative cues, while transcriptional and chromatin regulators such as PAX6, CHD8, SETD5, and ANKRD11 govern gene expression essential for proper NSPC cycling. Furthermore, prenatal exposure to teratogens such as Zika virus infection, valproic acid, or metabolic stress in phenylketonuria can recapitulate proliferation defects and microcephaly, underscoring the vulnerability of NSPCs to environmental perturbation. This review summarizes emerging insights into the molecular and cellular mechanisms by which defective NSPC proliferation contributes to NDD pathogenesis, highlighting convergence among genetic and environmental factors on cell cycle control. A deeper understanding of these pathways may uncover shared therapeutic targets to restore neurodevelopmental trajectories and mitigate disease burden.
Full article

Figure 1
Highly Accessed Articles
Latest Books
E-Mail Alert
News
Topics

Special Issues
Special Issue in
JDB
Feature Papers from Journal of Developmental Biology Reviewers, 2nd Edition
Guest Editors: Christopher A. Johnston, Junichi IwataDeadline: 25 March 2026
Special Issue in
JDB
Genetic and Epigenetic Mechanisms in Gametogenesis and Early Development: Insights from Models, Stem Cells, and Human Disorders
Guest Editors: Lon J. van Winkle, Philip Iannaccone, Rebecca Jean RyznarDeadline: 15 August 2026
Special Issue in
JDB
Mechanisms of Morphogenesis, Degeneration, and Regeneration
Guest Editors: Tsutomu Nohno, Hideyo Ohuchi, Naoyuki WadaDeadline: 16 September 2026
Special Issue in
JDB
Mitochondrial Function and Dysfunction in Developmental Biology Metabolism
Guest Editor: Simon J. ConwayDeadline: 15 October 2026
Topical Collections
Topical Collection in
JDB
Hedgehog Signaling in Embryogenesis
Collection Editors: Henk Roelink, Kay Grobe









