Arsenic Secondary Methylation Capacity Is Inversely Associated with Arsenic Exposure-Related Muscle Mass Reduction
Abstract
:1. Introduction
2. Materials and Methods
2.1. Ethical Permission
2.2. Study Areas
2.3. Study Subjects
2.4. Questionnaire and Interview
2.5. Collection of Water and Measurement of Arsenic
2.6. Collection of Urine and Measurement of Urinary Arsenic Species
2.7. Blood Collection and Measurement of Serum Creatinine
2.8. Estimation of Lean Body Mass (LBM)
2.9. Estimation of HOMA-IR
2.10. Statistical Analyses
3. Results
3.1. Characteristics and Distribution of the Subjects Based on the Drinking Water Arsenic Levels and Urinary Arsenic Profiles
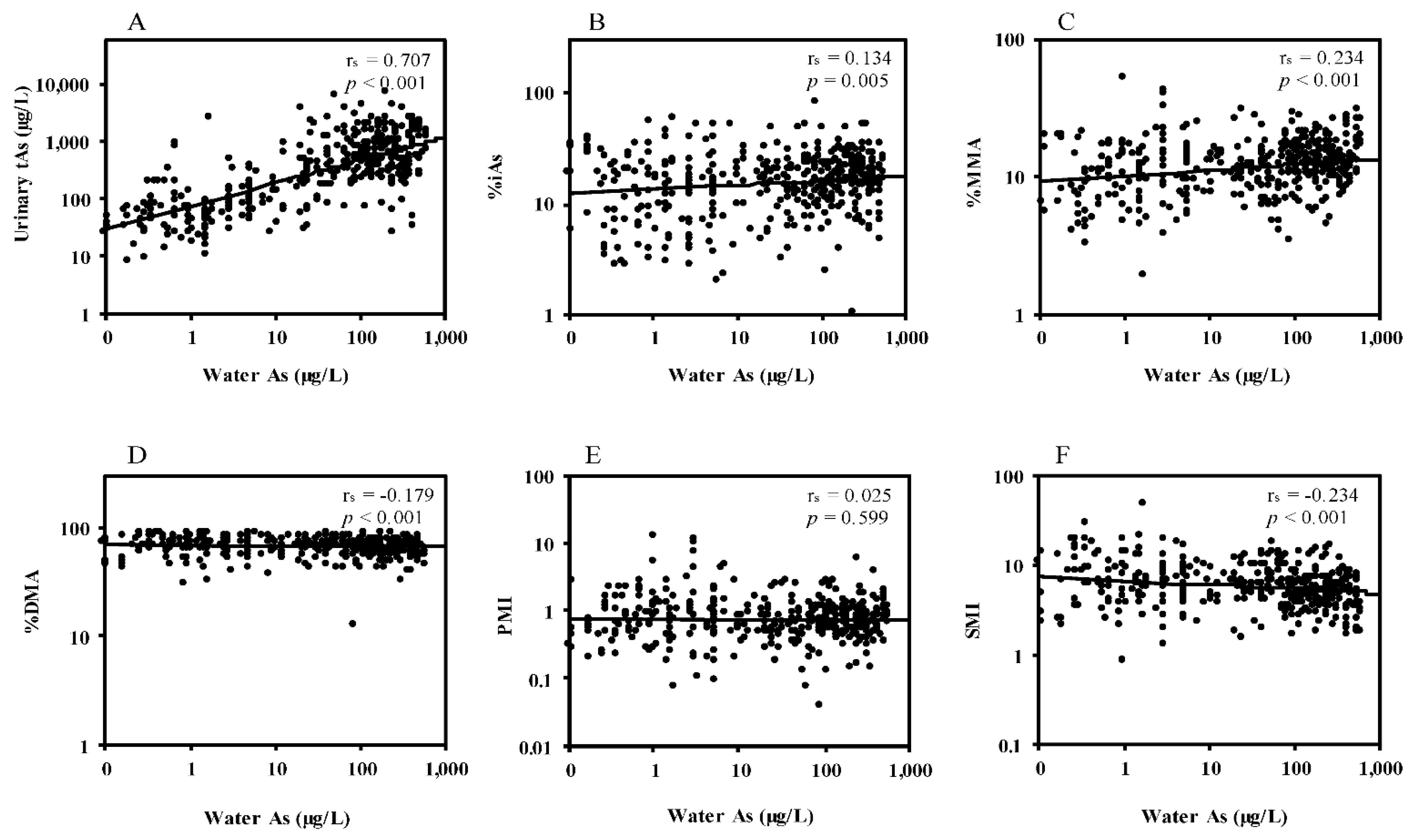
3.2. Correlations between Water Arsenic and Urinary Arsenic Species
3.3. Effects of Arsenic Exposure on Muscle Mass Measures and HOMA-IR
3.4. Associations between Urinary Arsenic Species and Muscle Mass Measures
3.5. Associations between Urinary Arsenic Species and HOMA-IR
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability statement
Conflicts of Interest
References
- Rahman, M.; Sohel, N.; Yunus, F.M.; Alam, N.; Nahar, Q.; Streatfield, P.K.; Yunus, M. Arsenic exposure and young adult’s mortality risk: A 13-year follow-up study in Matlab, Bangladesh. Environ. Int. 2019, 123, 358–367. [Google Scholar] [CrossRef] [PubMed]
- Rahman, M.; Sohel, N.; Yunus, M.; Chowdhury, M.E.; Hore, S.K.; Zaman, K.; Bhuiya, A.; Streatfield, P.K. Increased childhood mortality and arsenic in drinking water in Matlab, Bangladesh: A population-based cohort study. PLoS ONE 2013, 8, e55014. [Google Scholar]
- Chen, Y.; Graziano, J.H.; Parvez, F.; Liu, M.; Slavkovich, V.; Kalra, T.; Argos, M.; Islam, T.; Ahmed, A.; Rakibuz-Zaman, M.; et al. Arsenic exposure from drinking water and mortality from cardiovascular disease in Bangladesh: Prospective cohort study. BMJ 2011, 342, d2431. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Argos, M.; Kalra, T.; Rathouz, P.J.; Chen, Y.; Pierce, B.; Parvez, F.; Islam, T.; Ahmed, A.; Rakibuz-Zaman, M.; Hasan, R.; et al. Arsenic exposure from drinking water, and all-cause and chronic-disease mortalities in Bangladesh (HEALS): A prospective cohort study. Lancet 2010, 376, 252–258. [Google Scholar] [CrossRef] [Green Version]
- Sohel, N.; Persson, L.A.; Rahman, M.; Streatfield, P.K.; Yunus, M.; Ekström, E.C.; Vahter, M. Arsenic in drinking water and adult mortality: A population-based cohort study in rural Bangladesh. Epidemiology 2009, 20, 824–830. [Google Scholar] [CrossRef]
- Paul, S.K.; Islam, M.S.; Hasibuzzaman, M.M.; Hossain, F.; Anjum, A.; Saud, Z.A.; Haque, M.M.; Sultana, P.; Haque, A.; Andric, K.B.; et al. Higher risk of hyperglycemia with greater susceptibility in females in chronic arsenic-exposed individuals in Bangladesh. Sci. Total Environ. 2019, 668, 1004–1012. [Google Scholar] [CrossRef] [PubMed]
- Mondal, V.; Hosen, Z.; Hossen, F.; Siddique, A.E.; Tony, S.R.; Islam, Z.; Islam, M.S.; Hossain, S.; Islam, K.; Sarker, M.K.; et al. Arsenic exposure-related hyperglycemia is linked to insulin resistance with concomitant reduction of skeletal muscle mass. Environ. Int. 2020, 143, 105890. [Google Scholar] [CrossRef]
- Navas-Acien, A.; Silbergeld, E.K.; Streeter, R.A.; Clark, J.M.; Burke, T.A.; Guallar, E. Arsenic exposure and type 2 diabetes: A systematic review of the experimental and epidemiologic evidence. Environ. Health Perspect. 2006, 114, 641–648. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pan, W.C.; Seow, W.J.; Kile, M.L.; Hoffman, E.B.; Quamruzzaman, Q.; Rahman, M.; Mahiuddin, G.; Mostofa, G.; Lu, Q.; Christiani, D.C. Association of low to moderate levels of arsenic exposure with risk of type 2 diabetes in Bangladesh. Am. J. Epidemiol. 2013, 178, 1563–1570. [Google Scholar] [CrossRef] [Green Version]
- Sung, T.C.; Huang, J.W.; Guo, H.R. Association between arsenic exposure and diabetes: A meta-analysis. Biomed. Res. Int. 2015, 2015, 368087. [Google Scholar] [CrossRef]
- Kile, M.L.; Christiani, D.C. Environmental arsenic exposure and diabetes. JAMA 2008, 300, 845–846. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Khazem, S.; Itani, L.; Kreidieh, D.; El Masri, D.; Tannir, H.; Citarella, R.; El Ghoch, M. Reduced lean body mass and cardiometabolic diseases in adult males with overweight and obesity: A pilot study. Int. J. Environ. Res. Public Health 2018, 15, 2754. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Doherty, T.J. The influence of aging and sex on skeletal muscle mass and strength. Curr. Opin. Clin. Nutr. Metab. Care 2001, 4, 503–508. [Google Scholar] [CrossRef] [PubMed]
- Perkisas, S.; Lamers, S.; Degerickx, R.; Van Mieghem, E.; Vandewoude, M.; Verhoeven, V.; De Cock, A.M. The relation between mortality, intramuscular adipose tissue and sarcopenia in hospitalized geriatric patients. Eur. Geriatr. Med. 2018, 9, 801–807. [Google Scholar] [CrossRef]
- Miljkovic, I.; Zmuda, J.M. Epidemiology of myosteatosis. Curr. Opin. Clin. Nutr. Metab. Care 2010, 13, 260. [Google Scholar] [CrossRef]
- Correa-de-Araujo, R.; Addison, O.; Miljkovic, I.; Goodpaster, B.H.; Bergman, B.C.; Clark, R.V.; Elena, J.W.; Esser, K.A.; Ferrucci, L.; Harris-Love, M.O.; et al. Myosteatosis in the context of skeletal muscle function deficit: An interdisciplinary workshop at the national institute on aging. Front. Physiol. 2020, 11, 963. [Google Scholar] [CrossRef]
- Ryan, A.S.; Dobrovolny, C.L.; Smith, G.V.; Silver, K.H.; Macko, R.F. Hemiparetic muscle atrophy and increased intramuscular fat in stroke patients. Arch. Phys. Med. Rehabil. 2002, 83, 1703–1707. [Google Scholar] [CrossRef]
- Machann, J.; Häring, H.; Schick, F.; Stumvoll, M. Intramyocellular lipids and insulin resistance. Diabetes Obes. Metab. 2004, 6, 239–248. [Google Scholar] [CrossRef]
- Ambrosio, F.; Brown, E.; Stolz, D.; Ferrari, R.; Goodpaster, B.; Deasy, B.; Distefano, G.; Roperti, A.; Cheikhi, A.; Garciafigueroa, Y.; et al. Arsenic induces sustained impairment of skeletal muscle and muscle progenitor cell ultrastructure and bioenergetics. Free Radic. Biol. Med. 2014, 74, 64–73. [Google Scholar] [CrossRef] [Green Version]
- Anguiano, T.; Sahu, A.; Qian, B.; Tang, W.Y.; Ambrosio, F.; Barchowsky, A. Arsenic directs stem cell fate by imparting notch signaling into the extracellular matrix niche. Toxicol. Sci. 2020, 177, 494–505. [Google Scholar] [CrossRef]
- Zhang, C.; Ferrari, R.; Beezhold, K.; Stearns-Reider, K.; D’Amore, A.; Haschak, M.; Stolz, D.; Robbins, P.D.; Barchowsky, A.; Ambrosio, F. Arsenic promotes NF-Κb-mediated fibroblast dysfunction and matrix remodeling to impair muscle stem cell function. Stem Cells 2016, 34, 732–742. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, S.L.; Chang, F.H.; Liou, S.H.; Wang, H.J.; Li, W.F.; Hsieh, D.P. Inorganic arsenic exposure and its relation to metabolic syndrome in an industrial area of Taiwan. Environ. Int. 2007, 33, 805–811. [Google Scholar] [CrossRef] [PubMed]
- Tseng, C.H.; Huang, Y.K.; Huang, Y.L.; Chung, C.J.; Yang, M.H.; Chen, C.J.; Hsueh, Y.M. Arsenic exposure, urinary arsenic speciation, and peripheral vascular disease in blackfoot disease-hyperendemic villages in Taiwan. Toxicol. Appl. Pharmacol. 2005, 206, 299–308. [Google Scholar] [CrossRef]
- Chen, Y.; Wu, F.; Liu, M.; Parvez, F.; Slavkovich, V.; Eunus, M.; Ahmed, A.; Argos, M.; Islam, T.; Rakibuz-Zaman, M.; et al. A prospective study of arsenic exposure, arsenic methylation capacity, and risk of cardiovascular disease in Bangladesh. Environ. Health Perspect. 2013, 121, 832–838. [Google Scholar] [CrossRef] [Green Version]
- Hsieh, F.I.; Hwang, T.S.; Hsieh, Y.C.; Lo, H.C.; Su, C.T.; Hsu, H.S.; Chiou, H.Y.; Chen, C.J. Risk of erectile dysfunction induced by arsenic exposure through well water consumption in Taiwan. Environ. Health Perspect. 2008, 116, 532–536. [Google Scholar] [CrossRef] [Green Version]
- Vahter, M.; Åkesson, A.; Liden, C.; Ceccatelli, S.; Berglund, M. Gender differences in the disposition and toxicity of metals. Environ. Res. 2007, 104, 85–95. [Google Scholar] [CrossRef]
- Zhang, Q.; Hou, Y.; Wang, D.; Xu, Y.; Wang, H.; Liu, J.; Xia, L.; Li, Y.; Tang, N.; Zheng, Q.; et al. Interactions of arsenic metabolism with arsenic exposure and individual factors on diabetes occurrence: Baseline findings from Arsenic and Non-Communicable disease cohort (AsNCD) in China. Environ. Pollut. 2020, 265, 114968. [Google Scholar] [CrossRef] [PubMed]
- Grau-Perez, M.; Kuo, C.C.; Gribble, M.O.; Balakrishnan, P.; Jones Spratlen, M.; Vaidya, D.; Francesconi, K.A.; Goessler, W.; Guallar, E.; Silbergeld, E.K.; et al. Association of low-moderate arsenic exposure and arsenic metabolism with incident diabetes and insulin resistance in the Strong Heart Family Study. Environ. Health Perspect. 2017, 125, 127004. [Google Scholar] [CrossRef]
- Spratlen, M.J.; Grau-Perez, M.; Best, L.G.; Yracheta, J.; Lazo, M.; Vaidya, D.; Balakrishnan, P.; Gamble, M.V.; Francesconi, K.A.; Goessler, W.; et al. The association of arsenic exposure and arsenic metabolism with the metabolic syndrome and its individual components: Prospective evidence from the Strong Heart Family Study. Am. J. Epidemiol. 2018, 187, 1598–1612. [Google Scholar] [CrossRef]
- Kile, M.L.; Hoffman, E.; Rodrigues, E.G.; Breton, C.V.; Quamruzzaman, Q.; RA, M.; Mahiuddin, G.; Hsueh, Y.M.; Christiani, D.C. A pathway-based analysis of urinary arsenic metabolites and skin lesions. Am. J. Epidemiol. 2011, 173, 778–786. [Google Scholar] [CrossRef] [Green Version]
- Zhang, Q.; Wang, D.; Zheng, Q.; Zheng, Y.; Wang, H.; Xu, Y.; Li, X.; Sun, G. Joint effects of urinary arsenic methylation capacity with potential modifiers on arsenicosis: A cross-sectional study from an endemic arsenism area in Huhhot Basin, northern China. Environ. Res. 2014, 132, 281–289. [Google Scholar] [CrossRef]
- Thomas, D.J.; Li, J.; Waters, S.B.; Xing, W.; Adair, B.M.; Drobna, Z.; Devesa, V.; Styblo, M. Arsenic (+3 oxidation state) methyltransferase and the methylation of arsenicals. Exp. Biol. Med. 2007, 232, 3–13. [Google Scholar]
- Stýblo, M.; Venkatratnam, A.; Fry, R.C.; Thomas, D.J. Origins, fate, and actions of methylated trivalent metabolites of inorganic arsenic: Progress and prospects. Arch. Toxicol. 2021, 95, 1547–1572. [Google Scholar] [CrossRef]
- Sakurai, T.; Kojima, C.; Kobayashi, Y.; Hirano, S.; Sakurai, M.H.; Waalkes, M.P.; Himeno, S. Toxicity of a trivalent organic arsenic compound, dimethylarsinous glutathione in a rat liver cell line (TRL 1215). Br. J. Pharmacol. 2006, 149, 888–897. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Watanabe, T.; Hirano, S. Metabolism of arsenic and its toxicological relevance. Arch. Toxicol. 2013, 87, 969–979. [Google Scholar] [CrossRef] [PubMed]
- Kuo, C.C.; Moon, K.A.; Wang, S.L.; Silbergeld, E.; Navas-Acien, A. The association of arsenic metabolism with cancer, cardiovascular disease, and diabetes: A systematic review of the epidemiological evidence. Environ. Health Perspect. 2017, 125, 087001. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Stump, C.S.; Henriksen, E.J.; Wei, Y.; Sowers, J.R. The metabolic syndrome: Role of skeletal muscle metabolism. Ann. Med. 2006, 38, 389–402. [Google Scholar] [CrossRef]
- British Geological Survey, Department of Public Health Engineering (Bangladesh). Arsenic Contamination of Groundwater in Bangladesh; Kinniburgh, D.G., Smedley, P.L., Eds.; Final Report British Geological Survey Report WC/00/19 British Geological Survey; Keyworth; 2001; Volume 2. Available online: https://www2.bgs.ac.uk/arsenic/bangladesh/ (accessed on 24 May 2021).
- Siddique, A.E.; Rahman, M.; Hossain, M.I.; Karim, Y.; Hasibuzzaman, M.M.; Biswas, S.; Islam, M.S.; Rahman, A.; Hossen, F.; Mondal, V.; et al. Association between chronic arsenic exposure and the characteristic features of asthma. Chemosphere 2020, 246, 125790. [Google Scholar] [CrossRef]
- Huda, N.; Hossain, S.; Rahman, M.; Karim, M.R.; Islam, K.; Mamun, A.A.; Hossain, M.I.; Mohanto, N.C.; Alam, S.; Aktar, S.; et al. Elevated levels of plasma uric acid and its relation to hypertension in arsenic-endemic human individuals in Bangladesh. Toxicol. Appl. Pharmacol. 2014, 281, 11–18. [Google Scholar] [CrossRef]
- Hasibuzzaman, M.M.; Hossain, S.; Islam, M.S.; Rahman, A.; Anjum, A.; Hossain, F.; Mohanto, N.C.; Karim, M.R.; Hoque, M.M.; Saud, Z.A.; et al. Association between arsenic exposure and soluble thrombomodulin: A cross sectional study in Bangladesh. PLoS ONE 2017, 12, e0175154. [Google Scholar] [CrossRef]
- Suzuki, Y.; Shimoda, Y.; Endo, Y.; Hata, A.; Yamanaka, K.; Endo, G. Rapid and effective speciation analysis of arsenic compounds in human urine using anion-exchange columns in HPLC-ICP-MS. J. Occup. Health 2009, 51, 380–385. [Google Scholar] [CrossRef]
- Islam, M.S.; Mohanto, N.C.; Karim, M.R.; Aktar, S.; Hoque, M.M.; Rahman, A.; Jahan, M.; Khatun, R.; Aziz, A.; Salam, K.A.; et al. Elevated concentrations of serum matrix metalloproteinase-2 and -9 and their associations with circulating markers of cardiovascular diseases in chronic arsenic-exposed individuals. Environ. Health 2015, 14, 92. [Google Scholar] [CrossRef] [Green Version]
- Boer, P. Estimated lean body mass as an index for normalization of body fluid volumes in humans. Am. J. Physiol. 1984, 247, F632–F636. [Google Scholar] [CrossRef]
- Caruso, D.; De Santis, D.; Rivosecchi, F.; Zerunian, M.; Panvini, N.; Montesano, M.; Biondi, T.; Bellini, D.; Rengo, M.; Laghi, A. Lean body weight-tailored iodinated contrast injection in obese patient: Boer versus James formula. Biomed. Res. Int. 2018, 2018, 8521893. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Patel, S.S.; Molnar, M.Z.; Tayek, J.A.; Ix, J.H.; Noori, N.; Benner, D.; Heymsfield, S.; Kopple, J.D.; Kovesdy, C.P.; Kalantar-Zadeh, K. Serum creatinine as a marker of muscle mass in chronic kidney disease: Results of a cross-sectional study and review of literature. J. Cachexia Sarcopenia Muscle 2013, 4, 19–29. [Google Scholar] [CrossRef] [PubMed]
- Janssen, I.; Heymsfield, S.B.; Wang, Z.M.; Ross, R. Skeletal muscle mass and distribution in 468 men and women aged 18–88 yr. J. Appl. Physiol. 2000, 89, 81–88. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Abe, T.; Kearns, C.F.; Fukunaga, T. Sex differences in whole body skeletal muscle mass measured by magnetic resonance imaging and its distribution in young Japanese adults. Br. J. Sports Med. 2003, 37, 436–440. [Google Scholar] [CrossRef] [Green Version]
- Shen, H.; Niu, Q.; Xu, M.; Rui, D.; Xu, S.; Feng, G.; Ding, Y.; Li, S.; Jing, M. Factors affecting arsenic methylation in arsenic-exposed humans: A systematic review and meta-analysis. Int. J. Environ. Res. Public Health 2016, 13, 205. [Google Scholar] [CrossRef] [Green Version]
- Correa-de-Araujo, R.; Harris-Love, M.O.; Miljkovic, I.; Fragala, M.S.; Anthony, B.W.; Manini, T.M. The need for standardized assessment of muscle quality inkeletal muscle function deficit and other aging-related muscle dysfunctions: Asymposium report. Front. Physiol. 2017, 8, 87. [Google Scholar] [CrossRef]
- Hu, H.; Nakagawa, T.; Honda, T.; Yamamoto, S.; Okazaki, H.; Yamamoto, M.; Miyamoto, T.; Eguchi, M.; Kochi, T.; Shimizu, M.; et al. Low serum creatinine and risk of diabetes: The Japan Epidemiology Collaboration on Occupational Health Study. J. Diabetes Investig. 2019, 10, 1209–1214. [Google Scholar] [CrossRef]
- Kashima, S.; Inoue, K.; Matsumoto, M.; Akimoto, K. Low serum creatinine is a type 2 diabetes risk factor in men and women: The Yuport Health Checkup Center cohort study. Diabetes Metab. 2017, 43, 460–464. [Google Scholar] [CrossRef] [PubMed]
- Harita, N.; Hayashi, T.; Sato, K.K.; Nakamura, Y.; Yoneda, T.; Endo, G.; Kambe, H. Lower serum creatinine is a new risk factor of type 2 diabetes: The Kansai healthcare study. Diabetes Care 2009, 32, 424–426. [Google Scholar] [CrossRef] [Green Version]
- Takeuchi, M.; Imano, H.; Muraki, I.; Shimizu, Y.; Hayama-Terada, M.; Kitamura, A.; Okada, T.; Kiyama, M.; Iso, H. Serum creatinine levels and risk of incident type 2 diabetes mellitus or dysglycemia in middle-aged Japanese men: A retrospective cohort study. Vet. Rec. 2018, 6, e000492. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Moon, J.S.; Lee, J.E.; Yoon, J.S. Variation in serum creatinine level is correlated to risk of type 2 diabetes. Endocrinol. Metab. 2013, 28, 207–213. [Google Scholar] [CrossRef] [Green Version]
- Ormazabal, V.; Nair, S.; Elfeky, O.; Aguayo, C.; Salomon, C.; Zuñiga, F.A. Association between insulin resistance and the development of cardiovascular disease. Cardiovasc. Diabetol. 2018, 17, 122. [Google Scholar] [CrossRef]
- Rask-Madsen, C.; King, G.L. Mechanisms of disease: Endothelial dysfunction in insulin resistance and diabetes. Nat. Clin. Pract. Endocrinol. Metab. 2007, 3, 46–56. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.K.; Tseng, C.H.; Huang, Y.L.; Yang, M.H.; Chen, C.J.; Hsueh, Y.M. Arsenic methylation capability and hypertension risk in subjects living in arseniasis-hyperendemic areas in southwestern Taiwan. Toxicol. Appl. Pharmacol. 2007, 218, 135–142. [Google Scholar] [CrossRef]
- Mendez, M.A.; González-Horta, C.; Sánchez-Ramírez, B.; Ballinas-Casarrubias, L.; Cerón, R.H.; Morales, D.V.; Terrazas, F.A.; Ishida, M.C.; Gutiérrez-Torres, D.S.; Saunders, R.J.; et al. Chronic exposure to arsenic and markers of cardiometabolic risk: A cross-sectional study in Chihuahua, Mexico. Environ. Health Perspect. 2016, 124, 104–111. [Google Scholar] [CrossRef] [Green Version]
- Lemaire, M.; Lemarié, C.A.; Molina, M.F. Exposure to moderate arsenic concentrations increases atherosclerosis in ApoE-/- mouse model. Toxicol. Sci. 2011, 122, 211–221. [Google Scholar] [CrossRef] [Green Version]
- Gribble, M.O.; Crainiceanu, C.M.; Howard, B.V.; Umans, J.G.; Francesconi, K.A.; Goessler, W.; Zhang, Y.; Silbergeld, E.K.; Guallar, E.; Navas-Acien, A. Body composition and arsenic metabolism: A cross-sectional analysis in the Strong Heart Study. Environ. Health 2013, 12, 107. [Google Scholar] [CrossRef] [Green Version]
- Abuawad, A.; Spratlen, M.J.; Parvez, F.; Slavkovich, V.; Ilievski, V.; Lomax-Luu, A.M.; Saxena, R.; Shahriar, H.; Nasir Uddin, M.; Islam, T.; et al. Association between body mass index and arsenic methylation in three studies of Bangladeshi adults and adolescents. Environ. Int. 2021, 149, 106401. [Google Scholar] [CrossRef]
- Del Razo, L.M.; García-Vargas, G.G.; Valenzuela, O.L.; Castellanos, E.H.; Sánchez-Peña, L.C.; Currier, J.M.; Drobná, Z.; Loomis, D.; Stýblo, M. Exposure to arsenic in drinking water is associated with increased prevalence of diabetes: A cross-sectional study in the Zimapán and Lagunera regions in Mexico. Environ. Health 2011, 10, 73. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kim, N.H.; Mason, C.C.; Nelson, R.G.; Afton, S.E.; Essader, A.S.; Medlin, J.E.; Levine, K.E.; Hoppin, J.A.; Lin, C.; Knowler, W.C.; et al. Arsenic exposure and incidence of type 2 diabetes in Southwestern American Indians. Am. J. Epidemiol. 2013, 177, 962–969. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kuo, C.C.; Howard, B.V.; Umans, J.G.; Gribble, M.O.; Best, L.G.; Francesconi, K.A.; Goessler, W.; Lee, E.; Guallar, E.; Navas-Acien, A. Arsenic exposure, arsenic metabolism, and incident diabetes in the Strong Heart study. Diabetes Care 2015, 38, 620–627. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lindberg, A.L.; Kumar, R.; Goessler, W.; Thirumaran, R.; Gurzau, E.; Koppova, K.; Rudnai, P.; Leonardi, G.; Fletcher, T.; Vahter, M. Metabolism of low-dose inorganic arsenic in a central European population: Influence of sex and genetic polymorphisms. Environ. Health Perspect. 2007, 115, 1081–1086. [Google Scholar] [CrossRef] [Green Version]
- Gomez-Rubio, P.; Roberge, J.; Arendell, L.; Harris, R.B.; O’Rourke, M.K.; Chen, Z.; Cantu-Soto, E.; Meza-Montenegro, M.M.; Billheimer, D.; Lu, Z.; et al. Association between body mass index and arsenic methylation efficiency in adult women from southwest U.S. and northwest Mexico. Toxicol. Appl. Pharmacol. 2011, 252, 176–182. [Google Scholar] [CrossRef] [Green Version]
- Ahsan, H.; Chen, Y.; Kibriya, M.G.; Slavkovich, V.; Parvez, F.; Jasmine, F.; Gamble, M.V.; Graziano, J.H. Arsenic metabolism, genetic susceptibility, and risk of premalignant skin lesions in Bangladesh. Cancer Epidemiol. Biomarkers Prev. 2007, 16, 1270–1278. [Google Scholar] [CrossRef] [Green Version]
- Xu, Y.Y.; Wang, Y.; Li, X.; He, M.; Xue, P.; Fu, J.Q.; Wang, H.H.; Sun, G.F. Variations in arsenic methylation capacity and oxidative DNA lesions over a 2-year period in a high arsenic-exposed population. Int. Arch. Occup. Environ. Health 2009, 82, 251–258. [Google Scholar] [CrossRef]
- Chen, Y.C.; Guo, Y.L.L.; Su, H.J.J.; Hsueh, Y.M.; Smith, T.J.; Ryan, L.M.; Lee, M.S.; Chao, S.C.; Lee, J.Y.Y.; Christiani, D.C. Arsenic methylation and skin cancer risk in southwestern Taiwan. J. Occup. Environ. Med. 2003, 45, 241–248. [Google Scholar] [CrossRef]
- Yen, Y.P.; Tsai, K.S.; Chen, Y.W.; Huang, C.F.; Yang, R.S.; Liu, S.H. Arsenic inhibits myogenic differentiation and muscle regeneration. Environ. Health Perspect. 2010, 118, 949–956. [Google Scholar] [CrossRef]
- Chen, C.M.; Chung, M.N.; Chiu, C.Y.; Liu, S.H.; Lan, K.C. Inorganic Arsenic Exposure Decreases Muscle Mass and Enhances Denervation-Induced Muscle Atrophy in Mice. Molecules 2020, 25, 3057. [Google Scholar] [CrossRef]
- McCoy, C.R.; Stadelman, B.S.; Brumaghim, J.L.; Liu, J.T.; Bain, L.J. Arsenic and Its Methylated Metabolites Inhibit the Differentiation of Neural Plate Border Specifier Cells. Chem. Res. Toxicol. 2015, 28, 1409–1421. [Google Scholar] [CrossRef] [PubMed]
- Dávila-Esqueda, M.E.; Morales, J.M.; Jiménez-Capdeville, M.E.; De la Cruz, E.; Falcón-Escobedo, R.; Chi-Ahumada, E.; Martin-Pérez, S. Low-level subchronic arsenic exposure from prenatal developmental stages to adult life results in an impaired glucose homeostasis. Exp. Clin. Endocrinol. Diabetes 2011, 119, 613–617. [Google Scholar] [CrossRef]
- Ditzel, E.J.; Nguyen, T.; Parker, P.; Camenisch, T.D. Effects of arsenite exposure during fetal development on energy metabolism and susceptibility to diet-induced fatty liver disease in male mice. Environ. Health Perspect. 2016, 124, 201–209. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bonaventura, M.M.; Bourguignon, N.S.; Bizzozzero, M.; Rodriguez, D.; Ventura, C.; Cocca, C.; Libertun, C.; Lux-Lantos, V.A. Arsenite in drinking water produces glucose intolerance in pregnant rats and their female offspring. Food Chem. Toxicol. 2017, 100, 207–216. [Google Scholar] [CrossRef]
- Huang, M.C.; Douillet, C.; Dover, E.N.; Stýblo, M. Prenatal arsenic exposure and dietary folate and methylcobalamin supplementation alter the metabolic phenotype of C57BL/6J mice in a sex-specific manner. Arch. Toxicol. 2018, 92, 1925–1937. [Google Scholar] [CrossRef]
- Navas-Acien, A.; Spratlen, M.J.; Abuawad, A.; LoIacono, N.J.; Bozack, A.K.; Gamble, M.V. Early-life arsenic exposure, nutritional status, and adult diabetes risk. Curr. Diab. Rep. 2019, 19, 147. [Google Scholar] [CrossRef] [PubMed]
- Chung, C.J.; Huang, C.J.; Pu, Y.S.; Su, C.T.; Huang, Y.K.; Chen, Y.T.; Hsueh, Y.M. Urinary 8-hydroxydeoxyguanosine and urothelial carcinoma risk in low arsenic exposure area. Toxicol. Appl. Pharmacol. 2008, 226, 14–21. [Google Scholar] [CrossRef] [PubMed]
- Gamble, M.V.; Liu, X.; Ahsan, H.; Pilsner, J.R.; Ilievski, V.; Slavkovich, V.; Parvez, F.; Levy, D.; Factor-Litvak, P.; Graziano, J.H. Folate, homocysteine, and arsenic metabolism in arsenic-exposed individuals in Bangladesh. Environ. Health Perspect. 2005, 113, 1683–1688. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pu, Y.S.; Yang, S.M.; Huang, Y.K.; Chung, C.J.; Huang, S.K.; Chiu, A.W.H.; Yang, M.H.; Chen, C.J.; Hsueh, Y.M. Urinary arsenic profile affects the risk of urothelial carcinoma even at low arsenic exposure. Toxicol. Appl. Pharmacol. 2007, 218, 99–106. [Google Scholar] [CrossRef] [PubMed]
- Steinmaus, C.; Carrigan, K.; Kalman, D.; Atallah, R.; Yuan, Y.; Smith, A.H. Dietary intake and arsenic methylation in a US population. Environ. Health Perspect. 2005, 113, 1153–1159. [Google Scholar] [CrossRef] [Green Version]
- Steinmaus, C.; Moore, L.E.; Shipp, M.; Kalman, D.; Rey, O.A.; Biggs, M.L.; Hopenhayn, C.; Bates, M.N.; Zheng, S.; Wiencke, J.K.; et al. Genetic polymorphisms in MTHFR 677 and 1298, GSTM1 and T1, and metabolism of arsenic. J. Toxicol. Environ. Health 2007, 70, 159–170. [Google Scholar] [CrossRef]
- Chiou, H.Y.; Hsueh, Y.M.; Hsieh, L.L.; Hsu, L.I.; Hsu, Y.H.; Hsieh, F.I.; Wei, M.L.; Chen, H.C.; Yang, H.T.; Leu, L.C.; et al. Arsenic methylation capacity, body retention, and null genotypes of glutathione S-transferase M1 and T1 among current arsenic-exposed residents in Taiwan. Mutat. Res. 1997, 386, 197–207. [Google Scholar] [CrossRef]
- Kurttio, P.; Komulainen, H.; Hakala, E.; Kahelin, H.; Pekkanen, J. Urinary excretion of arsenic species after exposure to arsenic present in drinking water. Arch. Environ. Contam. Toxicol. 1998, 34, 297–305. [Google Scholar] [CrossRef]
- Thongprayoon, C.; Cheungpasitporn, W.; Kashani, K. Serum creatinine level, a surrogate of muscle mass, predicts mortality in critically ill patients. J. Thorac. Dis. 2016, 8, E305. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Heymsfield, S.B.; Arteaga, C.; McManus, C.; Smith, J.; Moffitt, S. Measurement of muscle mass in humans: Validity of the 24-h urinary creatinine method. Am. J. Clin. Nutr. 1983, 37, 478–494. [Google Scholar] [CrossRef] [PubMed]
- Park, J.; Mehrotra, R.; Rhee, C.M.; Molnar, M.Z.; Lukowsky, L.R.; Patel, S.S.; Nissenson, A.R.; Kopple, J.D.; Kovesdy, C.P.; Kalantar-Zadeh, K. Serum creatinine level, a surrogate of muscle mass, predicts mortality in peritoneal dialysis patients. Nephrol. Dial. Transplant. 2013, 28, 2146–2155. [Google Scholar] [CrossRef] [Green Version]
- Bozack, A.K.; Howe, C.G.; Hall, M.N.; Liu, X.; Slavkovich, V.; Ilievski, V.; Lomax-Luu, A.M.; Parvez, F.; Siddique, A.B.; Shahriar, H.; et al. Betaine and choline status modify the effects of folic acid and creatine supplementation on arsenic methylation in a randomized controlled trial of Bangladeshi adults. Eur. J. Nutr. 2021, 60, 1921–1934. [Google Scholar] [CrossRef]
- Hall, M.N.; Gamble, M.V. Nutritional manipulation of one-carbon metabolism: Effects on arsenic methylation and toxicity. J. Toxicol. 2012, 2012, 595307. [Google Scholar] [CrossRef] [Green Version]
- Gribble, M.O.; Howard, B.V.; Umans, J.G.; Shara, N.M.; Francesconi, K.A.; Goessler, W.; Crainiceanu, C.M.; Silbergeld, E.K.; Guallar, E.; Navas-Acien, A. Arsenic exposure, diabetes prevalence, and diabetes control in the Strong Heart Study. Am. J. Epidemiol. 2012, 176, 865–874. [Google Scholar] [CrossRef] [Green Version]
| Variables | [n, %] | Water As (µg/L) | Urinary tAs (µg/L) | %iAs | %MMA | %DMA | PMI | SMI | |
|---|---|---|---|---|---|---|---|---|---|
| Mean (SD) | Mean (SD) | Mean (SD) | Mean (SD) | Mean (SD) | Mean (SD) | Mean (SD) | |||
| All | 437 | 128.44 (165.87) | 635.89 (950.03) | 18.35 (10.74) | 12.87 (5.81) | 68.78 (12.32) | 1.02 (1.19) | 6.78 (4.46) | |
| Sex b | Female | 207 (47.4) | 127.08 (161.96) | 727.32 (1122.23) | 19.36 (11.87) | 12.96 (6.93) | 67.69 (13.77) | 1.08 (1.55) | 7.13 (5.4) |
| Male | 230 (52.6) | 129.67 (169.65) | 553.6 (755.73) | 17.45 (9.55) | 12.79 (4.57) | 69.76 (10.79) | 0.98 (0.72) | 6.46 (3.37) | |
| p-value | 0.871 | 0.061 | 0.067 | 0.769 | 0.082 | 0.384 | 0.127 | ||
| Age, years a | 18−31 | 151 (34.5) | 131.64(187.43) | 586.3 (643.4) | 19.57 (9.45) | 12.84 (5.42) | 67.59 (12.38) | 0.84 (0.65) | 6.85 (4.65) |
| 32−42 | 142 (32.5) | 126.99 (147.59) | 596.99 (766.9) | 19.35 (11.73) | 12.62 (5.6) | 68.03 (12.38) | 1 (1.26) | 6.75 (4.9) | |
| 43−60 | 144 (33) | 126.51 (159.81) | 726.25 (1313.54) | 16.09 (10.72) | 13.15 (6.4) | 70.77 (12.05) | 1.25 (1.48) | 6.73 (3.76) | |
| p-value | 0.958 | 0.378 | 0.008 | 0.747 | 0.058 | 0.01 | 0.97 | ||
| BMI a | 13.67−19.10 | 146 (33.4) | 129.24 (146.15) | 525.79 (635.11) | 19.9 (9.63) | 13.11 (4.89) | 66.99 (10.96) | 0.84 (0.65) | 6.07 (3.11) |
| 19.11−22.20 | 145 (33.2) | 124.54 (147.96) | 658.25 (913.85) | 16.61 (9.27) | 12.88 (5.22) | 70.51 (10.96) | 1.13 (1.19) | 6.73 (3.93) | |
| 22.22−36.70 | 146 (33.4) | 131.52 (199) | 723.78 (1208.58) | 18.53 (12.78) | 12.62 (7.09) | 68.85 (14.52) | 1.11 (1.53) | 7.53 (5.8) | |
| p-value | 0.936 | 0.193 | 0.032 | 0.767 | 0.051 | 0.07 | 0.019 | ||
| Income, USD a | 9.33−22.12 | 142 (32.5) | 137.3 (163.78) | 590.4 (980.4) | 18.5 (9.67) | 12.93 (5.43) | 68.58 (11.56) | 0.96 (1.08) | 6.58 (3.98) |
| 22.13−26.64 | 149 (34.1) | 111.58 (142.7) | 595.21 (943.88) | 17.74 (11.45) | 12.56 (6.22) | 69.7 (12.62) | 1.1 (1.35) | 7.32 (5.52) | |
| 56.67−83.30 | 146 (33.4) | 135.48 (185.59) | 725.42 (966.37) | 18.88 (11.04) | 13.19 (5.95) | 67.93 (12.77) | 1.02 (1.2) | 6.45 (3.84) | |
| p-value | 0.379 | 0.4 | 0.675 | 0.675 | 0.492 | 0.626 | 0.227 | ||
| Education a | No formal education | 242 (55.4) | 130.01 (166.65) | 614.21 (1035.91) | 17.43 (10.7) | 12.34 (5.94) | 70.23 (11.94) | 1.09 (1.36) | 7.17 (4.22) |
| Primary | 127 (29.1) | 121.94 (151.52) | 656.96 (882.17) | 19.72 (10.68) | 13.12 (5.05) | 67.16 (12.49) | 0.87 (0.61) | 6.51 (5.22) | |
| Above | 68 (15.6) | 135.02 (189.3) | 673.67 (738.79) | 19.07 (10.84) | 14.29 (6.43) | 66.64 (12.83) | 1.08 (1.34) | 5.9 (3.49) | |
| p-value | 0.851 | 0.863 | 0.128 | 0.042 | 0.022 | 0.217 | 0.082 | ||
| Occupation a | Housewife | 195 (44.6) | 120.95 (153.5) | 720.2 (1132.63) | 19.43 (12.02) | 12. 91 (6.99) | 67.66 (13.79) | 1.08 (1.57) | 7.18 (5.48) |
| Farmer | 180 (41.2) | 117.95 (140.23) | 492.76 (582.76) | 17.56 (9.71) | 12.67 (4.41) | 69.77 (10.69) | 0.94 (0.6) | 6.45 (3.26) | |
| others c | 62 (14.2) | 182.45 (246.04) | 786.23 (1126.25) | 17.25 (9.03) | 13.34 (5.30) | 69.42 (11.74) | 1.11 (1.08) | 6.48 (3.79) | |
| p-value | 0.021 | 0.027 | 0.166 | 0.731 | 0.232 | 0.457 | 0.247 | ||
| Smoking b | No | 340 (77.8) | 130.21 (169.83) | 618.69 (939.08) | 19.2 (11.29) | 13.05 (6.17) | 67.75 (12.75) | 1.01 (1.27) | 6.73 (4.68) |
| Yes | 97 (22.2) | 122.23 (151.83) | 696.16 (990.09) | 15.36 (7.89) | 12.25 (4.26) | 72.39 (9.92) | 1.07 (0.85) | 6.96 (3.58) | |
| p-value | 0.676 | 0.479 | <0.001 | 0.147 | <0.001 | 0.701 | 0.645 | ||
| Skin symptoms b | No | 216 (49.4) | 74.35 (129.75) | 376.15 (692.86) | 18.26 (11.13) | 12.6 (6.61) | 69.14 (13.09) | 1.14 (1.61) | 7.40 (5.44) |
| Yes | 221 (50.6) | 181.23 (182.7) | 890.85 (1110.21) | 18.62 (10.35) | 13.23 (5.05) | 68.15 (11.58) | 0.92 (0.62) | 6.14 (3.19) | |
| p-value | <0.001 | <0.001 | 0.733 | 0.276 | 0.414 | 0.07 | 0.005 | ||
| Variables | Area | Male | Female | ||
|---|---|---|---|---|---|
| n | Mean ± SD | n | Mean ± SD | ||
| Water As (µg/L) | Low | 41 | 2.48 ± 3.4 | 55 | 2.84 ± 4.76 |
| High | 189 | 157.25 ± 175.4 # | 152 | 172.04 ± 167.69 # | |
| Serum creatinine (mg/dL) | Low | 41 | 0.96 ± 0.16 | 55 | 0.86 ± 0.18 |
| High | 189 | 0.80 ± 0.18 # | 151 | 0.77 ± 0.16 # | |
| LBM (kg) | Low | 41 | 43.98 ± 6.96 | 55 | 36.58 ± 5.7 |
| High | 189 | 43.57 ± 6.15 | 152 | 33.13 ± 8.08 # | |
| The subjects with the HOMA-IR measurement (n = 271) $ | |||||
| Water As (µg/L) | Low | 29 | 2.35 ± 3.35 | 39 | 2.88 ± 4.84 |
| High | 105 | 162.9 ± 156.77 # | 98 | 186.74 ± 156.77 # | |
| Serum creatinine (mg/dL) | Low | 29 | 0.99 ± 0.14 | 39 | 0.9 ± 0.15 |
| High | 105 | 0.78 ± 0.16 # | 98 | 0.77 ± 0.13 # | |
| LBM (kg) | Low | 29 | 43.42 ± 6.89 | 39 | 37.24 ± 5.93 |
| High | 105 | 42.84± 7.47 | 98 | 31.29 ± 8.09 # | |
| HOMA-IR | Low | 29 | 0.81 ± 0.16 | 39 | 0.91 ± 0.37 |
| High | 105 | 1.57 ± 0.8 # | 98 | 2.35 ± 1.88 # | |
| Variables | All a | Male b | Female c | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| LBM (kg) | p-Value | Serum Creatinine d (mg/dL) | p-Value | LBM (kg) | p-Value | Serum Creatinine e (mg/dL) | p-Value | LBM (kg) | p-Value | Serum Creatinine f (mg/dL) | p-Value | |
| β (95% CI) | β (95% CI) | β (95% CI) | β (95% CI) | β (95% CI) | β (95% CI) | |||||||
| Urinary tAs (µg/L) | −0.03 (−0.044, −0.016) | <0.001 | −0.042 (−0.057, −0.026) | <0.001 | −0.012 (−0.028, 0.004) | 0.149 | −0.056 (−0.079, −0.032) | <0.001 | −0.046 (−0.069, −0.024) | <0.001 | −0.029 (−0.05, −0.009) | 0.005 |
| Sex | −0.107 (−0.129, −0.085) | <0.001 | ||||||||||
| %iAs | −0.049 (−0.08, −0.019) | 0.002 | −0.065 (−0.099, −0.031) | <0.001 | −0.041 (−0.077, −0.006) | 0.024 | −0.088 (−0.142, −0.033) | 0.002 | −0.055 (−0.104, −0.005) | 0.03 | −0.050 (−0.094, −0.007) | 0.024 |
| Sex | −0.108 (−0.13, −0.086) | <0.001 | ||||||||||
| %MMA | −0.16 (−0.202, −0.118) | <0.001 | −0.141 (−0.189, −0.092) | <0.001 | −0.141 (−0.193, −0.089) | <0.001 | −0.114 (−0.197, −0.031) | 0.007 | −0.173 (−0.239, −0.107) | <0.001 | −0.155 (−0.213, −0.097) | <0.001 |
| Sex | −0.113 (−0.134, −0.091) | <0.001 | ||||||||||
| %DMA | 0.231 (0.135, 0.327) | <0.001 | 0.228 (0.12, 0.337) | <0.001 | 0.211 (0.088, 0.333) | 0.001 | 0.327 (0.141, 0.513) | 0.001 | 0.243 (0.096, 0.389) | 0.001 | 0.177 (0.046, 0.308) | 0.008 |
| Sex | −0.106 (−0.128, −0.084) | <0.001 | ||||||||||
| PMI | −0.022 (−0.051, 0.006) | 0.119 | −0.001 (−0.033,- 0.03) | 0.927 | −0.017 (−0.05, 0.017) | 0.327 | 0.033 (−0.018, 0.084) | 0.205 | −0.027 (−0.072, 0.018) | 0.236 | −0.023 (−0.062, 0.017) | 0.26 |
| Sex | −0.109 (−0.132, −0.087) | <0.001 | ||||||||||
| SMI | 0.133 (0.099, 0.166) | <0.001 | 0.121 (0.082, 0.159) | <0.001 | 0.119 (0.077, 0.16) | <0.001 | 0.116 (0.05, 0.182) | 0.001 | 0.142 (0.09, 0.194) | <0.001 | 0.122 (0.076, 0.168) | <0.001 |
| Sex | −0.11 (−0.132, −0.089) | <0.001 | ||||||||||
| Variables | HOMA-IR | |||||
|---|---|---|---|---|---|---|
| All a | p-Value | Male b | p-Value | Female c | p-Value | |
| Unstandardized β (95% CI) | Unstandardized β (95% CI) | Unstandardized β (95% CI) | ||||
| Urinary tAs (µg/L) | 0.212 (0.17, 0.254) | <0.001 | 0.2 (0.14, 0.261) | <0.001 | 0.219 (0.159, 0.279) | <0.001 |
| Sex | 0.091 (0.021, 0.162) | 0.011 | ||||
| %iAs | 0.143 (0.035, 0.251) | 0.009 | 0.132 (−0.012, 0.276) | 0.073 | 0.145 (−0.016, 0.306) | 0.078 |
| Sex | 0.107 (0.025, 0.19) | 0.011 | ||||
| %MMA | 0.376 (0.223, 0.53) | <0.001 | 0.18 (−0.057, 0.416) | 0.135 | 0.471 (0.263, 0.678) | <0.001 |
| Sex | 0.135 (0.057, 0.214) | 0.001 | ||||
| %DMA | −0.447 (−0.778, −0.117) | 0.008 | −0.534 (−1.079, 0.011) | 0.055 | −0.393 (−0.832, 0.046) | 0.079 |
| Sex | 0.107 (0.024, 0.189) | 0.011 | ||||
| PMI | 0.026 (−0.073, 0.126) | 0.605 | −0.061 (−0.203, 0.081) | 0.395 | 0.082 (−0.059, 0.223) | 0.254 |
| Sex | 0.132 (0.048, 0.215) | 0.002 | ||||
| SMI | −0.305 (−0.428, −0.183) | <0.001 | −0.174 (−0.359, 0.012) | 0.067 | −0.369 (−0.539, −0.201) | <0.001 |
| Sex | 0.12 (0.041, 0.198) | 0.003 | ||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sarker, M.K.; Tony, S.R.; Siddique, A.E.; Karim, M.R.; Haque, N.; Islam, Z.; Islam, M.S.; Khatun, M.; Islam, J.; Hossain, S.; et al. Arsenic Secondary Methylation Capacity Is Inversely Associated with Arsenic Exposure-Related Muscle Mass Reduction. Int. J. Environ. Res. Public Health 2021, 18, 9730. https://doi.org/10.3390/ijerph18189730
Sarker MK, Tony SR, Siddique AE, Karim MR, Haque N, Islam Z, Islam MS, Khatun M, Islam J, Hossain S, et al. Arsenic Secondary Methylation Capacity Is Inversely Associated with Arsenic Exposure-Related Muscle Mass Reduction. International Journal of Environmental Research and Public Health. 2021; 18(18):9730. https://doi.org/10.3390/ijerph18189730
Chicago/Turabian StyleSarker, Md. Khalequzzaman, Selim Reza Tony, Abu Eabrahim Siddique, Md. Rezaul Karim, Nazmul Haque, Zohurul Islam, Md. Shofikul Islam, Moriom Khatun, Jahidul Islam, Shakhawoat Hossain, and et al. 2021. "Arsenic Secondary Methylation Capacity Is Inversely Associated with Arsenic Exposure-Related Muscle Mass Reduction" International Journal of Environmental Research and Public Health 18, no. 18: 9730. https://doi.org/10.3390/ijerph18189730








