The Role of Transforming Growth Factor-Beta in Retinal Ganglion Cells with Hyperglycemia and Oxidative Stress
Abstract
:1. Introduction
2. Results
2.1. Effects of Hyperglycemia on RGCs
2.2. Immunoblot Analysis of RGCs with Hyperglycemia
2.3. Immunofluorescence Analysis of RGCs with Hyperglycemia
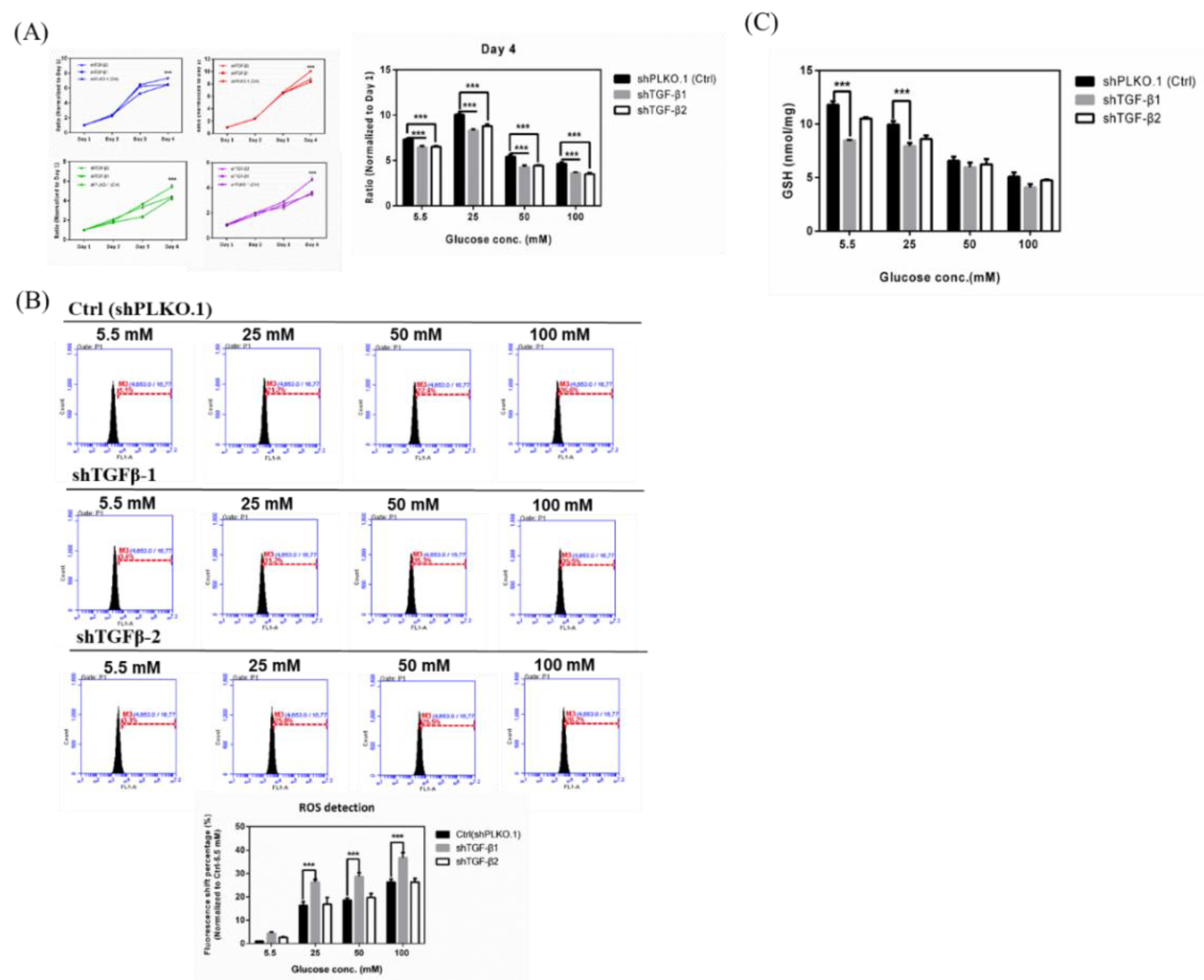
2.4. Effects in TGF-β1/2 Knockdown RGCs with Hyperglycemia
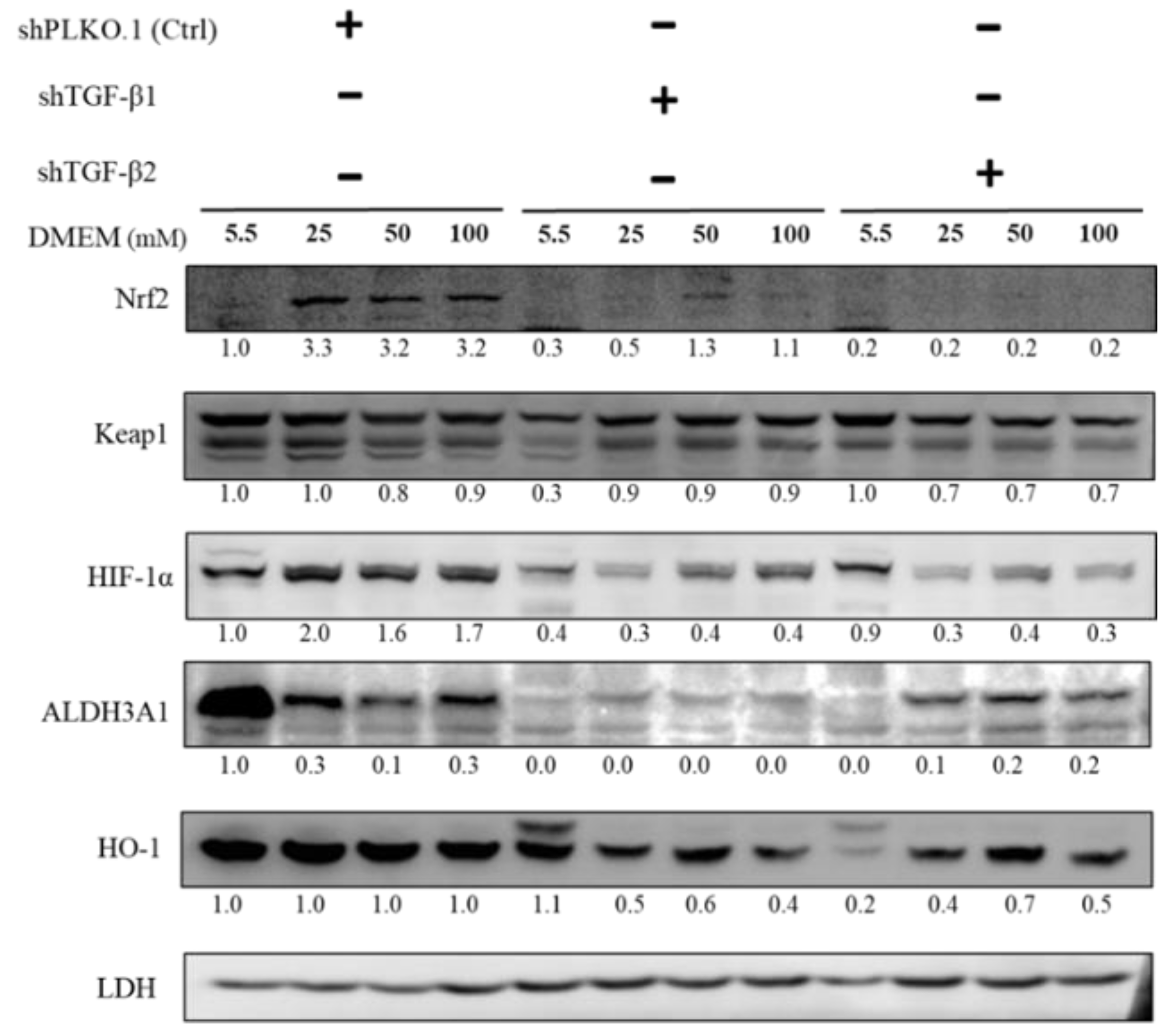
2.5. Immunoblot Analysis of Activation of Downstream Substrates in TGF-β1/2 Knockdown RGC-5 Cells Treated with Hyperglycemia
2.6. Effects in RGCs with Hyperglycemia with or w/o rhTGF-β1 Protein (5 ng/mL)
2.7. Immunoblot Analysis in RGCs with Hyperglycemia with or w/o Recombinant TGF-β1 Protein (5 ng/mL)
2.8. Effects in RGCs with Hyperglycemia with or without Hydrogen Peroxide for 1 h
2.9. Immunoblot Analysis in RGCs with Hyperglycemia with or without Hydrogen Peroxide (1 mM) for 1 h
2.10. A Hypothetical Model Detailing the Role of TGF-β in Hyperglycemia
3. Discussion
4. Materials and Methods
4.1. Cell Line and Cell Culture
4.2. Proliferation Assay
4.3. Cell Viability Assay
4.4. Detection of ROS
4.5. Immunoblotting Analysis via Western Blot
4.6. Immunofluorescence
4.7. Treatment of Cells with Recombinant Human TGF-β Protein1 (rhTGF-β1)
4.8. Measurement of Intracellular GSH Content
4.9. High-Resolution Respirometry
4.10. Statistical Analysis
Author Contributions
Funding
Conflicts of Interest
References
- Bansal, V.; Kalita, J.; Misra, K.U. Diabetic neuropathy. Postgrad. Med. J. 2006, 82, 95–100. [Google Scholar] [CrossRef] [PubMed]
- Lechner, J.; O’Leary, O.E.; Stitt, A.W. The pathology Associated with Diabetic Retinopathy. Vis. Res. 2017, 139, 7–14. [Google Scholar] [CrossRef] [PubMed]
- Wong, T.Y.; Cheung, C.M.; Larsen, M.; Sharma, S.; Simo, R. Diabetic retinopathy. Nat. Rev. Dis. Primers 2016, 2, 16012. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Lo, A.C.Y. Diabetic Retinopathy: Pathophysiology and Treatments. Int. J. Mol. Sci. 2018, 19, 1816. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kusuhara, S.; Fukushima, Y.; Ogura, S.; Inoue, N.; Uemura, A. Pathophysiology of Diabetic Retinopathy: The Old and the New. Diabetes Metab. J. 2018, 42, 364–376. [Google Scholar] [CrossRef] [PubMed]
- Duh, E.J.; Sun, J.K.; Stitt, A.W. Diabetic retinopathy: Current understanding, mechanisms, and treatment strategies. JCI Insight 2017, 2, 14. [Google Scholar] [CrossRef] [PubMed]
- Safi, S.Z.; Qvist, R.; Kumar, S.; Batumalaie, K.; Bin, I.S. Molecular mechanisms of diabetic retinopathy, general preventive strategies, and novel therapeutic targets. Biomed. Res. Int. 2014, 2014, 801269. [Google Scholar] [CrossRef] [Green Version]
- Maugeri, G.; D’Amico, A.G.; Rasà, D.M.; La Cognata, V.; Saccone, S.; Concetta, F.; Cavallaro, S.; D’Agata, V. Nicotine promotes blood retinal barrier damage in a model of human diabetic macular edema. Toxicol. In Vitro 2017, 44, 182–189. [Google Scholar] [CrossRef]
- Wang, L.; Wu, C.-G.; Fang, C.-Q.; Gao, J.; Liu, Y.-Z.; Chen, Y.; Chen, Y.-N.; Xu, Z.-G. The protective effect of alpha-Lipoic acid on mitochondria in the kidney of diabetic rats. Int. J. Clin. Exp. Med. 2013, 6, 90. [Google Scholar]
- Almasry, M.S.; Habib, K.E.; Elmansy, A.R.; Hassan, Z.A. Hyperglycemia Alters the Protein Levels of Prominin-1 and VEGFA in the Retina of Albino Rats. J. Histochem. Cytochem. 2018, 66, 33–45. [Google Scholar] [CrossRef]
- Brownlee, M. Biochemistry and molecular cell biology of diabetic complications. Nature 2001, 414, 813–820. [Google Scholar] [CrossRef] [PubMed]
- Nishikawa, T.; Edelstein, D.; Du, L.X.; Yamagishi, S.; Matsumura, T.; Kaneda, Y.; Yorek, M.A.; Beebe, D.; Oates, P.J.; Hammes, H.-P.; et al. Normalizing mitochondrial superoxide production blocks three pathways of hyperglycaemic damage. Nature 2000, 404, 787–790. [Google Scholar] [CrossRef] [PubMed]
- Tien, T.; Zhang, J.; Muto, T.; Kim, D.; Sarthy, V.P.; Roy, S. High Glucose Induces Mitochondrial Dysfunction in Retinal Muller Cells: Implications for Diabetic Retinopathy. Invest. Ophthalmol. Vis. Sci. 2017, 58, 2915–2921. [Google Scholar] [CrossRef] [PubMed]
- Boss, J.D.; Singh, P.K.; Pandya, H.K.; Tosi, J.; Kim, C.; Tewari, A.; Juzych, M.S.; Abrams, G.W.; Kumar, A. Assessment of Neurotrophins and Inflammatory Mediators in Vitreous of Patients With Diabetic Retinopathy. Invest. Ophthalmol. Vis. Sci. 2017, 58, 5594–5603. [Google Scholar] [CrossRef] [PubMed]
- Sasaki, M.; Ozawa, Y.; Kurihara, T.; Kubota, S.; Yuki, K.; Soda, S.; Kobayashi, S.; Ishida, S.; Tsubota, K. Neurodegenerative influence of oxidative stress in the retina of a murine model of diabetes. Diabetologia 2010, 53, 971–979. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pal, P.B.; Sinha, K.; Sil, P.C. Mangiferin attenuates diabetic nephropathy by inhibiting oxidative stress mediated signaling cascade, TNFalpha related and mitochondrial dependent apoptotic pathways in streptozotocin-induced diabetic rats. PLoS ONE 2014, 9, e107220. [Google Scholar] [CrossRef] [Green Version]
- Chen, Y.; Zhang, Y.; Jic, H.; Ji, Y.; Yang, J.; Huang, J.; Sun, D. Involvement of hypoxia-inducible factor-1alpha in the oxidative stress induced by advanced glycation end products in murine Leydig cells. Toxicol. In Vitro 2016, 32, 146–153. [Google Scholar] [CrossRef] [PubMed]
- Ishii, T.; Itoh, K.; Takahashi, S.; Sato, H.; Yanagawa, T.; Katoh, Y.; Bannai, S.; Yamamoto, M. Transcription factor Nrf2 coordinately regulates a group of oxidative stress-inducible genes in macrophages. J. Biol. Chem. 2000, 275, 16023–16029. [Google Scholar] [CrossRef] [Green Version]
- Sun, Q.; Shen, Z.-Y.; Meng, Q.-T.; Liu, H.-Z.; Duan, W.-N.; Xiaun, Z.-Y. The role of DJ-1/Nrf2 pathway in the pathogenesis of diabetic nephropathy in rats. Ren. Fail. 2016, 38, 294–304. [Google Scholar] [CrossRef] [Green Version]
- Uruno, A.; Furusawa, Y.; Yagishita, Y.; Fukutomi, T.; Muramatsu, H.; Negishi, T.; Sugawara, A.; Kensler, T.W.; Yamamoto, M. The Keap1-Nrf2 system prevents onset of diabetes mellitus. Mol. Cell Biol. 2013, 33, 2996–3010. [Google Scholar] [CrossRef] [Green Version]
- Abdo, S.; Zhang, S.L.; Chan, J.S. Reactive Oxygen Species and Nuclear Factor Erythroid 2-Related Factor 2 Activation in Diabetic Nephropathy: A Hidden Target. J. Diabetes Metab. 2015, 6. [Google Scholar] [CrossRef]
- Motohashi, H.; Yamamoto, M. Nrf2-Keap1 defines a physiologically important stress response mechanism. Trends Mol. Med. 2004, 10, 549–557. [Google Scholar] [CrossRef] [PubMed]
- Lawrence, D.A. Transforming growth factor-beta: A general review. Eur. Cytokine Netw. 1996, 7, 363–374. [Google Scholar] [PubMed]
- Funkenstein, B.; Olekh, E.; Jakowlew, S.B. Identification of a novel transforming growth factor-beta (TGF-beta6) gene in fish: Regulation in skeletal muscle by nutritional state. BMC Mol. Biol. 2010, 11, 37. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Roberts, A.B. Molecular and cell biology of TGF-beta. Miner. Electrolyte Metab. 1998, 24, 111–119. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Cui, M.; Wei, Y.; Kong, X.; Tang, L.; Xu, D. Inhibition of the expression of TGF-beta1 and CTGF in human mesangial cells by exendin-4, a glucagon-like peptide-1 receptor agonist. Cell Physiol. Biochem. 2012, 30, 749–757. [Google Scholar] [CrossRef]
- Yang, H.; Huang, Y.; Chen, X.; Liu, J.; Lu, Y.; Bu, L.; Xia, L.; Xiao, W.; Chen, M.; Nie, Q.; et al. The role of CTGF in the diabetic rat retina and its relationship with VEGF and TGF-beta(2), elucidated by treatment with CTGFsiRNA. Acta Ophthalmol. 2010, 88, 652–659. [Google Scholar] [CrossRef]
- Grigsby, J.; Betts, B.; Vidro-Kotchan, E.; Culbert, R.; Tsin, A. A possible role of acrolein in diabetic retinopathy: Involvement of a VEGF/TGFbeta signaling pathway of the retinal pigment epithelium in hyperglycemia. Curr. Eye Res. 2012, 37, 1045–1053. [Google Scholar] [CrossRef] [Green Version]
- Kita, T.; Hata, Y.; Kano, K.; Miura, M.; Nakao, S.; Noda, Y.; Shimokawa, H.; Ishibashi, T. Transforming growth factor-beta2 and connective tissue growth factor in proliferative vitreoretinal diseases: Possible involvement of hyalocytes and therapeutic potential of Rho kinase inhibitor. Diabetes 2007, 56, 231–238. [Google Scholar] [CrossRef] [Green Version]
- More, S.V.; Choi, D.K. Promising cannabinoid-based therapies for Parkinson’s disease: Motor symptoms to neuroprotection. Mol. Neurodegener. 2015, 10, 17. [Google Scholar] [CrossRef] [Green Version]
- Dobolyi, A.; Vincze, C.; Pal, G.; Lovas, G. The neuroprotective functions of transforming growth factor beta proteins. Int. J. Mol. Sci. 2012, 13, 8219–8258. [Google Scholar] [CrossRef] [PubMed]
- Prendes, M.A.; Harris, A.; Wirostko, B.M.; Gerber, A.L.; Siesky, B. The role of transforming growth factor beta in glaucoma and the therapeutic implications. Br. J. Ophthalmol. 2013, 97, 680–686. [Google Scholar] [CrossRef] [PubMed]
- Agarwal, N. RGC-5 cells. Invest Ophthalmol. Vis. Sci. 2013, 54, 1–11. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tsukamoto, T.; Kajiwara, K.; Nada, S.; Okada, M. Src mediates TGF-beta-induced intraocular pressure elevation in glaucoma. J. Cell Physiol. 2019, 234, 1730–1744. [Google Scholar] [CrossRef]
- Giacco, F.; Brownlee, M. Oxidative stress and diabetic complications. Circ Res. 2010, 107, 1058–1070. [Google Scholar] [CrossRef] [Green Version]
- Reddy, M.A.; Zhang, E.; Natarajan, R. Epigenetic mechanisms in diabetic complications and metabolic memory. Diabetologia 2015, 58, 443–455. [Google Scholar] [CrossRef] [Green Version]
- Candiloros, H.; Muller, S.; Zeghari, N.; Donner, M.; Drouin, P.; Ziegler, O. Decreased erythrocyte membrane fluidity in poorly controlled IDDM. Influence of ketone bodies. Diabetes Care 1995, 18, 549–551. [Google Scholar] [CrossRef]
- Saydah, S.H.; Miret, M.; Sung, J.; Varas, C.; Gause, D.; Brancati, F.L. Postchallenge hyperglycemia and mortality in a national sample of U.S. adults. Diabetes Care 2001, 24, 1397–1402. [Google Scholar] [CrossRef] [Green Version]
- Jouven, X.; Lemaitre, R.N.; Rea, T.D.; Sotoodehnia, N.; Empana, J.P.; Siscovick, D.S. Diabetes, glucose level, and risk of sudden cardiac death. Eur. Heart J. 2005, 26, 2142–2147. [Google Scholar] [CrossRef] [Green Version]
- Cai, L.; Li, W.; Wang, G.; Guo, L.; Jiang, Y.; Kang, Y.J. Hyperglycemia-induced apoptosis in mouse myocardium: Mitochondrial cytochrome C-mediated caspase-3 activation pathway. Diabetes 2002, 51, 1938–1948. [Google Scholar] [CrossRef] [Green Version]
- Tamai, K.; Mizushima, T.; Wu, X.; Inoue, A.; Ota, M.; Yokoyama, Y.; Miyoshi, N.; Haraguchi, N.; Takahashi, H.; Nishimura, J.; et al. Photodynamic Therapy Using Indocyanine Green Loaded on Super Carbonate Apatite as Minimally Invasive Cancer Treatment. Mol. Cancer Ther. 2018, 17, 1613–1622. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, J.M.; Tseng, C.W.; Lin, C.C.; Law, C.H.; Chien, Y.A.; Kuo, W.H.; Chou, H.C.; Wang, W.C.; Chan, H.L. Upregulation of LGALS1 is associated with oral cancer metastasis. Ther. Adv. Med. Oncol. 2018, 10, 1758835918794622. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Guha, P.; Dey, A.; Sen, R.; Chatterjee, M.; Chattopadhyay, S.; Bandyopadhyay, S.K. Intracellular GSH depletion triggered mitochondrial Bax translocation to accomplish resveratrol-induced apoptosis in the U937 cell line. J. Pharmacol. Exp. Ther. 2011, 336, 206–214. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, Y.C.; Cheng, T.H.; Lin, W.L.; Chen, C.L.; Yang, W.Y.; Blackstone, C.; Chang, C.R. Srv2 Is a Pro-fission Factor that Modulates Yeast Mitochondrial Morphology and Respiration by Regulating Actin Assembly. Iscience 2019, 11, 305–317. [Google Scholar] [CrossRef] [PubMed] [Green Version]










| Antibody | Molecular Weights (kDa) | Company | Monoclonal/Polyclonal | Working Concentration |
|---|---|---|---|---|
| Nrf2 | 75/130 | Abclonal A1244 | rab pAb | 1:2000 |
| Keap1 | 70 | Abclonal-A17062 | rab pAb | 1:2000 |
| HIF-1α | 93 | Abclonal A11945 | rab pAb | 1:2000 |
| ALDH3A1 | 50 | Abclonal A13275 | rab pAb | 1:2000 |
| HO-1 | 33 | GeneTex-GTX101147 | rab pAb | 1:2000 |
| TGFβ-1 | 46 | Abclonal-A2124 | rab pAb | 1:2000 |
| TGFβ-2 | 48/55 | Abclonal A3640 | rab pAb | 1:2000 |
| LDH (Lactate Dehydrogenase) | 36 | GeneTex-GTX101416 | rab pAb | 1:2000 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, H.-Y.; Ho, Y.-J.; Chou, H.-C.; Liao, E.-C.; Tsai, Y.-T.; Wei, Y.-S.; Lin, L.-H.; Lin, M.-W.; Wang, Y.-S.; Ko, M.-L.; et al. The Role of Transforming Growth Factor-Beta in Retinal Ganglion Cells with Hyperglycemia and Oxidative Stress. Int. J. Mol. Sci. 2020, 21, 6482. https://doi.org/10.3390/ijms21186482
Chen H-Y, Ho Y-J, Chou H-C, Liao E-C, Tsai Y-T, Wei Y-S, Lin L-H, Lin M-W, Wang Y-S, Ko M-L, et al. The Role of Transforming Growth Factor-Beta in Retinal Ganglion Cells with Hyperglycemia and Oxidative Stress. International Journal of Molecular Sciences. 2020; 21(18):6482. https://doi.org/10.3390/ijms21186482
Chicago/Turabian StyleChen, Hsin-Yi, Yi-Jung Ho, Hsiu-Chuan Chou, En-Chi Liao, Yi-Ting Tsai, Yu-Shan Wei, Li-Hsun Lin, Meng-Wei Lin, Yi-Shiuan Wang, Mei-Lan Ko, and et al. 2020. "The Role of Transforming Growth Factor-Beta in Retinal Ganglion Cells with Hyperglycemia and Oxidative Stress" International Journal of Molecular Sciences 21, no. 18: 6482. https://doi.org/10.3390/ijms21186482





