Fabrication of Hydroxyapatite with Bioglass Nanocomposite for Human Wharton’s-Jelly-Derived Mesenchymal Stem Cell Growing Substrate
Abstract
:1. Introduction
2. Results and Discussion
2.1. TEM Results for HAp/BG Nanocomposites
2.2. Cell Culture Evaluation In Vitro
2.2.1. Scanning Electron Microscopy Analysis of Cell Adherence and Morphology
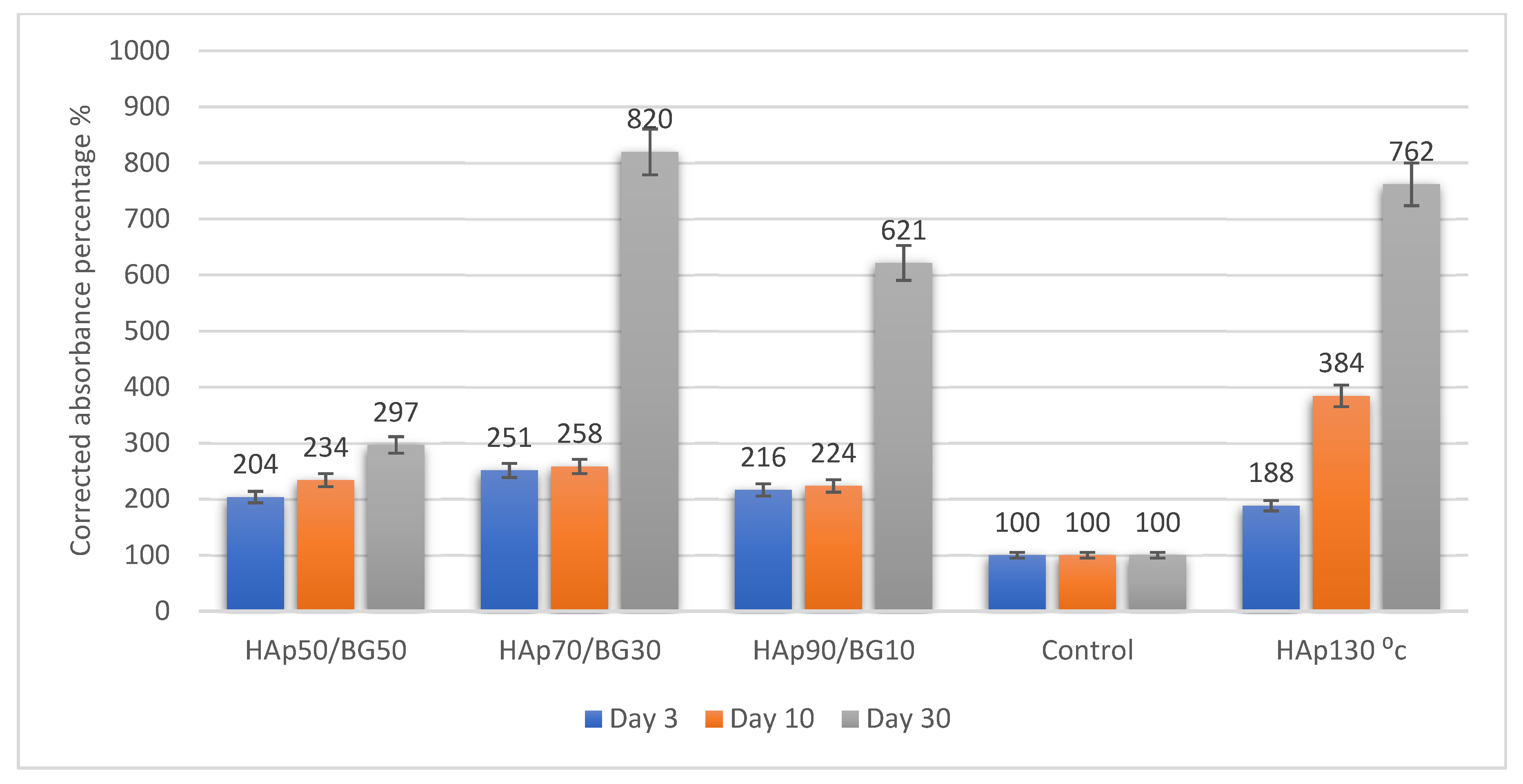
2.2.2. Cell Viability Assay Analysis
2.2.3. Cell Metabolic Activity in Scaffolds
2.3. SEM-Energy Dispersed X-ray (SEM-EDX) Analysis of Osteoblast Differentiation
3. Discussion
3.1. Effect of Crystal Size and Crystallinity on Cellular Response
3.2. Cell Culture on HAp and HAp/BG Nanocomposite Scaffolds
4. Materials and Methods
4.1. Preparation of HAp Nanopowder
4.2. Preparation of BG Nanopowder
4.3. Preparation of HAp/BG Nanocomposite Powder
4.4. Characterization
4.5. In Vitro Bioactivity Preparation
4.5.1. Harvest and Preparation of hWJMSCs
4.5.2. Cell Seeding Preparation on the Scaffold
4.6. Cell Culture Studies In Vitro
4.6.1. Cell Morphology Observation
4.6.2. Cell Viability Analysis
4.6.3. Cell Metabolic Activities on Scaffolds
4.6.4. Elemental Composition via EDX Analysis for Osteoblast Differentiation
4.6.5. Alkaline Phosphatase (ALP) Activity Measurement for Osteoblast Differentiation
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Sandeep, G.; Varma, H.; Kumary, T.; Babu, S.S.; John, A. Characterization of novel bioactive glass coated hydroxyapatite granules in correlation with in vitro and in vivo studies. Trends Biomater. Artif. Organs 2006, 19, 99–107. [Google Scholar]
- Oktar, F. Hydroxyapatite–TiO2 composites. Mater. Lett. 2006, 60, 2207–2210. [Google Scholar] [CrossRef]
- Pushpakanth, S.; Srinivasan, B.; Sreedhar, B.; Sastry, T. An in situ approach to prepare nanorods of titania–hydroxyapatite (TiO2–HAp) nanocomposite by microwave hydrothermal technique. Mater. Chem. Phys. 2008, 107, 492–498. [Google Scholar] [CrossRef]
- Xia, L.; Xie, Y.; Fang, B.; Wang, X.; Lin, K. In situ modulation of crystallinity and nano-structures to enhance the stability and osseointegration of hydroxyapatite coatings on Ti-6Al-4V implants. Chem. Eng. J. 2018, 347, 711–720. [Google Scholar] [CrossRef]
- Ravarian, R.; Moztarzadeh, F.; Hashjin, M.S.; Rabiee, S.; Khoshakhlagh, P.; Tahriri, M. Synthesis, characterization and bioactivity investigation of bioglass/hydroxyapatite composite. Ceram. Int. 2010, 36, 291–297. [Google Scholar] [CrossRef]
- Hench, L.L. The story of Bioglass®. J. Mater. Sci. Mater. Med. 2006, 17, 967–978. [Google Scholar] [CrossRef]
- Liu, X.; Morra, M.; Carpi, A.; Li, B. Bioactive calcium silicate ceramics and coatings. Biomed. Pharmacother. 2008, 62, 526–529. [Google Scholar] [CrossRef]
- Xia, W.; Chang, J. Preparation and characterization of nano-bioactive-glasses (NBG) by a quick alkali-mediated sol–gel method. Mater. Lett. 2007, 61, 3251–3253. [Google Scholar] [CrossRef]
- Šupová, M. Substituted hydroxyapatites for biomedical applications: A review. Ceram. Int. 2015, 41, 9203–9231. [Google Scholar] [CrossRef]
- Shamsi, M.; Salimi, A.; Ghallasi, M.; Halabian, R. Effect of synthetic biologically activated 45S5 glass nanoparticles on osteogenesis differentiation of mesenchymal human bone marrow. Sci. J. Iran Blood Transfus Organ 2018, 15, 272–286. [Google Scholar]
- Kokubo, T.; Takadama, H. How useful is SBF in predicting in vivo bone bioactivity? Biomaterials 2006, 27, 2907–2915. [Google Scholar] [CrossRef] [PubMed]
- Nabian, N.; Jahanshahi, M.; Rabiee, S.M. Synthesis of nano-bioactive glass–ceramic powders and its in vitro bioactivity study in bovine serum albumin protein. J. Mol. Struct. 2011, 998, 37–41. [Google Scholar] [CrossRef]
- Ghomi, H.; Fathi, M.; Edris, H. Effect of the composition of hydroxyapatite/bioactive glass nanocomposite foams on their bioactivity and mechanical properties. Mater. Res. Bull. 2012, 47, 3523–3532. [Google Scholar] [CrossRef]
- Shirdar, M.R.; Farajpour, N.; Shahbazian-Yassar, R.; Shokuhfar, T. Nanocomposite materials in orthopedic applications. Front. Chem. Sci. Eng. 2019, 13, 1–13. [Google Scholar] [CrossRef]
- Yadav, S.; Singh, P.; Pyare, R. Synthesis, characterization, mechanical and biological properties of biocomposite based on zirconia containing 1393 bioactive glass with hydroxyapatite. Ceram. Int. 2020, 46, 10442–10451. [Google Scholar] [CrossRef]
- Aminian, A.; Solati-Hashjin, M.; Samadikuchaksaraei, A.; Bakhshi, F.; Gorjipour, F.; Farzadi, A.; Schmücker, M. Synthesis of silicon-substituted hydroxyapatite by a hydrothermal method with two different phosphorous sources. Ceram. Int. 2011, 37, 1219–1229. [Google Scholar] [CrossRef]
- Taheriana, M.; Tamizifara, M.; Fathib, M.; Maleki Ghalehc, H. Modified sol-gel method for synthesis of hydroxyapatite-bioglass nanocomposite. In Proceedings of the 4th international conference on nanostructures (ICNS4), Institute for Nanoscience & Nanotechnology (INST), Sharif University of Technology, Tehran, Iran, 12–14 March 2012. [Google Scholar]
- Xuan Bui, V.; Quang Vo, M.; Anh Nguyen, T.; Thi Bui, H. Investigation of Bioactive Glass-Ceramic 60SiO2-30CaO-10P2O5 Prepared by Hydrothermal Method. Adv. Mater. Sci. Eng. 2019, 2019, 1528326. [Google Scholar] [CrossRef] [Green Version]
- Mondal, S.; Hoang, G.; Manivasagan, P.; Moorthy, M.S.; Nguyen, T.P.; Phan, T.T.V.; Oh, J. Nano-hydroxyapatite bioactive glass composite scaffold with enhanced mechanical and biological performance for tissue engineering application. Ceram. Int. 2018, 44, 15735–15746. [Google Scholar] [CrossRef]
- Tham, A.Y.; Gandhimathi, C.; Praveena, J.; Venugopal, J.R.; Ramakrishna, S.; Kumar, S.D. Minocycline loaded hybrid composites nanoparticles for mesenchymal stem cells differentiation into osteogenesis. Int. J. Mol. Sci. 2016, 17, 1222. [Google Scholar] [CrossRef] [Green Version]
- Collart-Dutilleul, P.Y.; Panayotov, I.; Secret, E.; Cunin, F.; Gergely, C.; Cuisinier, F.; Martin, M. Initial stem cell adhesion on porous silicon surface: Molecular architecture of actin cytoskeleton and filopodial growth. Nanoscale Res. Lett. 2014, 9, 1–10. [Google Scholar] [CrossRef] [Green Version]
- Maruyama, K.; Henmi, A.; Okata, H.; Sasano, Y. Analysis of calcium, phosphorus, and carbon concentrations during developmental calcification of dentin and enamel in rat incisors using scanning electron microscopy with energy dispersive X-ray spectroscopy (SEM-EDX). J. Oral Biosci. 2016, 58, 173–179. [Google Scholar] [CrossRef]
- Mansourizadeh, F.; Miri, V.; Sagha, M.; Asadi, A.; Golmohammadi, M.G. A comparison of the Growth and Differentiation of the Human Umbilical Cord Mesenchymal Stem Cells on the Poly-L-lactic acid/Hydroxyapatite Composite Scaffold with Pure Poly-L-lactic Acid Scaffold. J. Maz. Univ. Med. Sci. 2015, 24, 133–147. [Google Scholar]
- Doostmohammadi, A.; Fathi, M.H.; Bigham Sadegh, A.; Tavakolizadeh, A.H.; Akbarian, H. In Vivo Evaluation of Bioactive Nano Bioceramics for Incorporation into and Production of Bone (an Animal Study). Iran. J. Orthop. Surg. 2012, 10, 67–76. [Google Scholar]
- Hu, Q.; Tan, Z.; Liu, Y.; Tao, J.; Cai, Y.; Zhang, M.; Tang, R. Effect of crystallinity of calcium phosphate nanoparticles on adhesion, proliferation, and differentiation of bone marrow mesenchymal stem cells. J. Mater. Chem. 2007, 17, 4690–4698. [Google Scholar] [CrossRef]
- Karataş, Ö. Investigation of Biocompatibility of Calcium Phosphate Based Materials and Cements. Master’s Thesis, Izmir Institute of Technology, Izmir, Turkey, 2014. [Google Scholar]
- Taghiabadi, E.; Nasri, S.; Shafieyan, S.; Firoozinezhad, S.J.; Aghdami, N. Fabrication and characterization of spongy denuded amniotic membrane based scaffold for tissue engineering. Cell J. 2015, 16, 476. [Google Scholar] [PubMed]
- Scimeca, M.; Bischetti, S.; Lamsira, H.K.; Bonfiglio, R.; Bonanno, E. Energy Dispersive X-ray (EDX) microanalysis: A powerful tool in biomedical research and diagnosis. Eur. J. Histochem. 2018, 62, 2841. [Google Scholar] [CrossRef] [PubMed]
- Panzavolta, S.; Torricelli, P.; Sturba, L.; Bracci, B.; Giardino, R.; Bigi, A. Setting properties and in vitro bioactivity of strontium-enriched gelatin–calcium phosphate bone cements. J. Biomed. Mater. Res. Part A 2008, 84, 965–972. [Google Scholar] [CrossRef]
- Kotian, R.; Rao, P.P.; Madhyastha, P. X-ray diffraction analysis of hydroxyapatite-coated in different plasma gas atmosphere on Ti and Ti-6Al-4V. Eur. J. Dent. 2017, 11, 438–446. [Google Scholar] [CrossRef] [Green Version]
- Fang, B.; Wan, Y.Z.; Tang, T.T.; Gao, C.; Dai, K.R. Proliferation and osteoblastic differentiation of human bone marrow stromal cells on hydroxyapatite/bacterial cellulose nanocomposite scaffolds. Tissue Eng. Part A 2009, 15, 1091–1098. [Google Scholar] [CrossRef] [PubMed]







| Sample | Component 1 A: BG | Component 2 B: HAp | HAp/BG Crystal Size | HAp/BG Degree of Crystallinity (%) |
|---|---|---|---|---|
| 1 | 0 | 100 | 38.5 | 80 |
| 2 | 10 | 90 | 37.5 | 77.15 |
| 3 | 30 | 70 | 33.12 | 75.84 |
| 4 | 50 | 50 | 31.58 | 67.98 |
| HAp90/BG10 | HAp70/BG30 | HAp50/BG50 | HAp | |
|---|---|---|---|---|
| Size | 31.4 nm | 30.12 nm | 25.1 nm | 37.5 nm |
| Sample | Component 1 A: BG | Component 2 B: HAp |
|---|---|---|
| 1 | 0 | 100 |
| 2 | 10 | 90 |
| 3 | 30 | 70 |
| 4 | 50 | 50 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ebrahimi, S.; Hanim, Y.U.; Sipaut, C.S.; Jan, N.b.A.; Arshad, S.E.; How, S.E. Fabrication of Hydroxyapatite with Bioglass Nanocomposite for Human Wharton’s-Jelly-Derived Mesenchymal Stem Cell Growing Substrate. Int. J. Mol. Sci. 2021, 22, 9637. https://doi.org/10.3390/ijms22179637
Ebrahimi S, Hanim YU, Sipaut CS, Jan NbA, Arshad SE, How SE. Fabrication of Hydroxyapatite with Bioglass Nanocomposite for Human Wharton’s-Jelly-Derived Mesenchymal Stem Cell Growing Substrate. International Journal of Molecular Sciences. 2021; 22(17):9637. https://doi.org/10.3390/ijms22179637
Chicago/Turabian StyleEbrahimi, Shamsi, Yusoff Umul Hanim, Coswald Stephen Sipaut, Norsazlina binti Ahmad Jan, Sazmal E. Arshad, and Siew Eng How. 2021. "Fabrication of Hydroxyapatite with Bioglass Nanocomposite for Human Wharton’s-Jelly-Derived Mesenchymal Stem Cell Growing Substrate" International Journal of Molecular Sciences 22, no. 17: 9637. https://doi.org/10.3390/ijms22179637
APA StyleEbrahimi, S., Hanim, Y. U., Sipaut, C. S., Jan, N. b. A., Arshad, S. E., & How, S. E. (2021). Fabrication of Hydroxyapatite with Bioglass Nanocomposite for Human Wharton’s-Jelly-Derived Mesenchymal Stem Cell Growing Substrate. International Journal of Molecular Sciences, 22(17), 9637. https://doi.org/10.3390/ijms22179637






