The Quality and Fertilizing Potential of Red Deer (Cervus elaphus L.) Epididymal Spermatozoa Stored in a Liquid State
Abstract
1. Introduction
2. Results
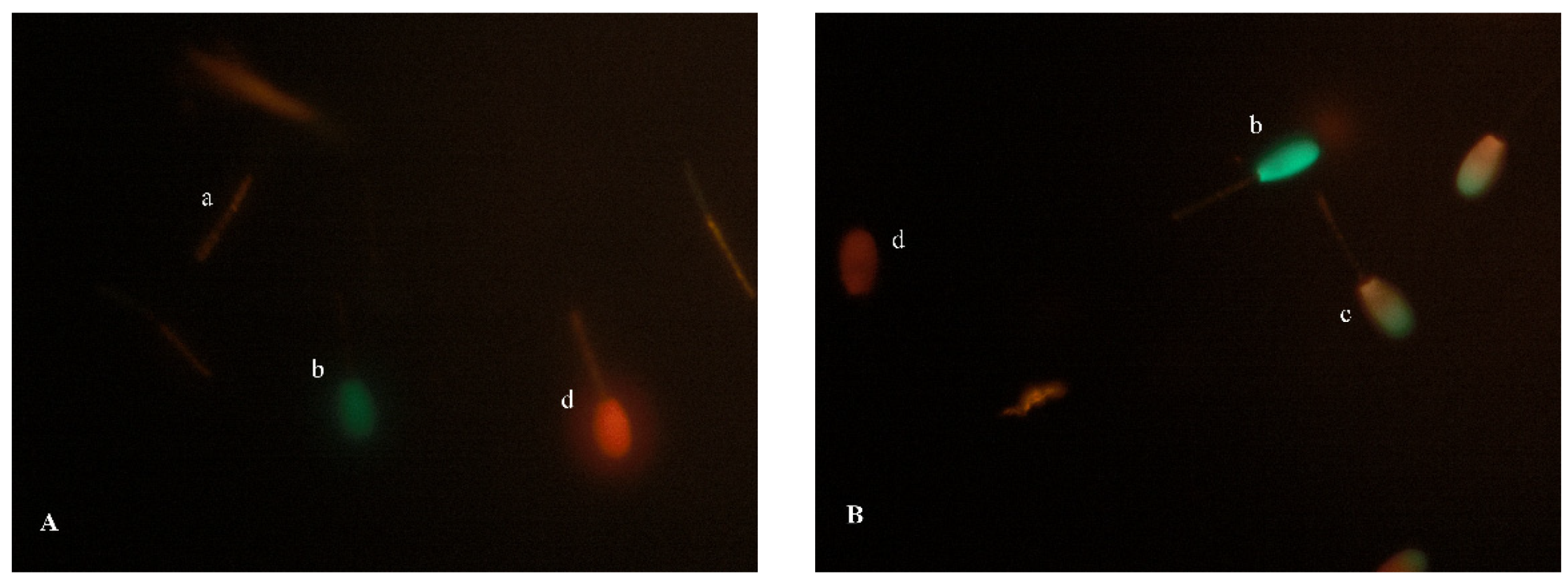
2.1. Experiment 1: The Effect of Storage Time (Days) on the Quality Parameters of Red Deer Epididymal Spermatozoa Stored in a Liquid State
2.2. Experiment 2: Fertilizing Potential of Red Deer Epididymal Spermatozoa Stored in a Liquid State
3. Discussion
4. Materials and Methods
4.1. Animals
4.2. Collection and Initial Evaluation of Epididymal Spermatozoa
4.3. Liquid Storage of Sperm
4.4. Experiment 1: Sperm Quality Analysis
4.4.1. Analysis of Sperm Motility Parameters
4.4.2. Sperm Morphology Assay
4.4.3. Fluorescence Assays
4.5. Experiment 2: Assessment of the Fertilizing Potential of Epididymal Spermatozoa
4.5.1. Induction of Estrus and Ovulation and Oocyte Collection
4.5.2. In Vitro Oocyte Maturation and In Vitro Fertilization
4.5.3. Artificial Insemination (AI) Procedure
4.5.4. Diagnosis of Pregnancy
4.6. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Garde, J.J.; Martínez-Pastor, F.; Gomendio, M.; Malo, A.F.; Soler, A.J.; Fernández-Santos, M.R.; Esteso, M.C.; García, A.J.; Anel, L.; Roldán, E.R.S. The application of reproductive technologies to natural populations of red deer. Reprod. Domest. Anim. 2006, 41, 93–102. [Google Scholar] [CrossRef]
- Morrow, C.J.; Penfold, L.M.; Wolfe, B.A. Artificial insemination in deer and non-domestic bovids. Theriogenology 2009, 71, 149–165. [Google Scholar] [CrossRef] [PubMed]
- Abella, D.F.; Da Costa, M.; Guérin, Y.; Dacheux, J.L. Fertility of undiluted ram epididymal spermatozoa stored for several days at 4 °C. Animal 2015, 9, 313–319. [Google Scholar] [CrossRef]
- Suzuki, C.; Yoshioka, K.; Itoh, S.; Kawarasaki, T.; Kikuchi, K. In vitro fertilization and subsequent development of porcine oocytes using cryopreserved and liquid-stored spermatozoa from various boars. Theriogenology 2005, 64, 1287–1296. [Google Scholar] [CrossRef] [PubMed]
- Cunha, A.T.M.; Carvalho, J.O.; Guimarães, A.L.S.; Leme, L.O.; Caixeta, F.M.; Viana, J.H.M.; Dode, M.A.N. Bovine epididymal spermatozoa treatment for in vitro fertilization: Heparin accelerates fertilization and enables a reduction in coincubation time. PLoS ONE 2019, 14, e0209692. [Google Scholar] [CrossRef] [PubMed]
- Morris, L.; Tiplady, C.; Allen, W. The in Vivo Fertility of Cauda Epididymal Spermatozoa in the Horse. Theriogenology 2002, 58, 643–646. [Google Scholar] [CrossRef]
- Zomborszky, Z.; Zubor, T.; Tóth, J.; Horn, P. Sperm collection from shot red deer stags (Cervus elaphus) and the utilisation of sperm frozen and subsequently thawed. Acta Vet. Hung. 1999, 47, 263–270. [Google Scholar] [CrossRef]
- Comizzoli, P.; Mauget, R.; Mermillod, P. Assessment of in vitro fertility of deer spermatozoa by heterologous IVF with zona-free bovine oocytes. Theriogenology 2001, 56, 261–274. [Google Scholar] [CrossRef]
- Soler, A.J.; Poulin, N.; Fernández-Santos, M.R.; Cognie, Y.; Esteso, M.C.; Garde, J.J.; Mermillod, P. Heterologous in vitro fertility evaluation of cryopreserved Iberian red deer epididymal spermatozoa with zona-intact sheep oocytes and its relationship with the characteristics of thawed spermatozoa. Reprod. Domest. Anim. 2008, 43, 293–298. [Google Scholar] [CrossRef]
- Watson, P.F. The causes of reduced fertility with cryopreserved semen. Anim. Reprod. Sci. 2000, 60–61, 481–492. [Google Scholar] [CrossRef]
- Asher, G.W.; Berg, D.K.; Evans, G. Storage of semen and artificial insemination in deer. Anim. Reprod. Sci. 2000, 62, 195–211. [Google Scholar] [CrossRef]
- Soler, A.J.; Pérez-Guzmán, M.D.; Garde, J.J. Storage of red deer epididymides for four days at 5 degrees C: Effects on sperm motility, viability, and morphological integrity. J. Exp. Zool. A Comp. Exp. Biol. 2003, 295, 188–199. [Google Scholar] [CrossRef] [PubMed]
- Fernández-Santos, M.R.; Martínez-Pastor, F.; Matias, D.; Domínguez-Rebolledo, A.E.; Esteso, M.C.; Montoro, V.; Garde., J.J. Effects of long-term chilled storage of red deer epididymides on DNA integrity and motility of thawed spermatozoa. Anim. Reprod. Sci. 2009, 111, 93–104. [Google Scholar] [CrossRef] [PubMed]
- Dziekońska, A.; Niedźwiecka, E.; Niklewska, M.E.; Koziorowska-Gilun, M.; Kordan, W. Viability longevity and quality of epididymal sperm stored in the liquid state of European red deer (Cervus elaphus elaphus). Anim. Reprod. Sci. 2020, 213, 106269. [Google Scholar] [CrossRef] [PubMed]
- Bainbridge, D.R.J.; Catt, S.L.; Evans, G.; Jabbour, H.N. Successful in vitro fertilization of in vivo matured oocytes aspirated laparoscopically from red deer hinds (Cervus elaphus). Theriogenology 1999, 51, 891–898. [Google Scholar] [CrossRef]
- Ros-Santaella, J.L.; Domínguez-Rebolledo, A.E.; Garde, J.J. Sperm flagellum volume determines freezability in red deer spermatozoa. PLoS ONE 2014, 9, e112382. [Google Scholar] [CrossRef]
- Malo, A.F.; Garde, J.J.; Soler, A.J.; Garcia, A.J.; Gomendio, M. Male fertility in natural populations of red deer is determined by sperm velocity and the proportion of normal spermatozoa. Biol. Reprod. 2005, 72, 822–829. [Google Scholar] [CrossRef]
- Ramón, M.; Martínez-Pastor, F.; García-Álvarez, O.; Maroto-Morales, A.; Soler, A.J.; Jiménez-Rabadán, P.; Fernández-Santos, M.R.; Bernabéu, R.; Garde, J.J. Taking advantage of the use of supervised learning methods for characterization of sperm population structure related with freezability in the Iberian red deer. Theriogenology 2012, 77, 1661–1672. [Google Scholar] [CrossRef]
- Fernández-Santos, M.R.; Esteso, M.C.; Soler, A.J.; Montoro, V.; Garde, J.J. Effects of egg yolk and cooling rate on the survival of refrigerated red deer (Cervus elaphus hispanicus) epididymal spermatozoa. Reprod. Domest. Anim. 2006, 41, 114–118. [Google Scholar] [CrossRef]
- Martinez-Pastor, F.; Guerra, C.; Kaabi, M.; Diaz, A.R.; Anel, E.; Herraez, P.; de Paz, P.; Anel, L. Decay of sperm obtained from epididymes of wild ruminants depending on postmortem time. Theriogenology 2005, 63, 24–40. [Google Scholar] [CrossRef]
- Cooper, T.G.; Yeung, C.H.; Fetic, S.; Sobhani, A.; Nieschlag, E. Cytoplasmic droplets are normal structures of human spermatozoa but are not well preserved by routine procedures for assessing sperm morphology. Hum. Reprod. 2004, 19, 2283–2288. [Google Scholar] [CrossRef] [PubMed]
- du Plessis, S.S.; Agarwal, A.; Mohanty, G.; van der Linde, M. Oxidative phosphorylation versus glycolysis: What fuel do spermatozoa use? Asian J. Androl. 2015, 17, 230–235. [Google Scholar] [CrossRef] [PubMed]
- Martínez-Pastor, F.; Fernández-Santos, M.R.; del Olmo, E.; Domínguez-Rebolledo, A.E.; Esteso, M.C.; Montoro, V.; Garde, J.J. Mitochondrial activity and forward scatter vary in necrotic, apoptotic and membrane-intact spermatozoan subpopulations. Reprod. Fertil. Dev. 2008, 20, 547–556. [Google Scholar] [CrossRef] [PubMed]
- Ardón, F.; Helms, D.; Sahin, E.; Bollwein, H.; Töpfer-Petersen, E.; Waberski, D. Chromatin-unstable boar spermatozoa have little chance of reaching oocytes in vivo. Reproduction 2008, 135, 461–470. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Larson-Cook, K.L.; Brannian, J.D.; Hansen, K.A.; Kasperson, K.M.; Aamold, E.D.; Evenson, D.P. Relationship between the outcomes of assisted reproductive techniques and sperm DNA fragmentation as measured by the sperm chromatin structure assay. Fertil. Steril. 2003, 80, 895–902. [Google Scholar] [CrossRef]
- Evenson, D.P.; Larson, K.L.; Jost, L.K. Sperm chromatin structure assay: Its clinical use for detecting sperm DNA fragmentation in male infertility and comparisons with other techniques. J. Androl. 2002, 23, 25–43. [Google Scholar] [CrossRef]
- Waberski, D.; Schapmann, E.; Henning, H.; Riesenbeck, A.; Brandt, H. Sperm chromatin structural integrity in normospermic boars is not related to semen storage and fertility after routine AI. Theriogenology 2011, 75, 337–345. [Google Scholar] [CrossRef]
- Golshan Iranpour, F.; Rezazadeh Valojerdi, M. The epididymal sperm viability, motility and DNA integrity in dead mice maintained at 4–6 °C. Iran. J. Reprod. Med. 2013, 11, 195–200. [Google Scholar]
- Demyda-Peyrás, S.; Dorado, J.; Hidalgo, M.; Anter, J.; De Luca, L.; Genero, E.; Moreno-Millán, M. Effects of oocyte quality, incubation time and maturation environment on the number of chromosomal abnormalities in IVF-derived early bovine embryos. Reprod. Fertil. Dev. 2013, 25, 1077. [Google Scholar] [CrossRef]
- Berg, D.; Thompson, J.; Asher, G. Development of in vitro embryo production systems for red deer (Cervus elaphus). Anim. Reprod. Sci. 2002, 70, 65–76. [Google Scholar] [CrossRef]
- Korzekwa, A.J.; Kotlarczyk, A.M.; Szczepańska, A.A.; Grzyb, M.; Siergiej, A.; Wocławek-Potocka, I. Antioxidative Potential of Red Deer Embryos Depends on Reproductive Stage of Hind as A Oocyte Donor. Animals 2020, 10, 1190. [Google Scholar] [CrossRef] [PubMed]
- Anzar, M.; He, L.; Buhr, M.M.; Kroetsch, T.G.; Pauls, K.P. Sperm apoptosis in fresh and cryopreserved bull semen detected by flow cytometry and its relationship with fertility. Biol. Reprod. 2002, 66, 354–360. [Google Scholar] [CrossRef] [PubMed]
- Evenson, D.P. Loss of livestock breeding efficiency due to uncompensable sperm nuclear defects. Reprod. Fertil. Dev. 1999, 11, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Yin, Y.; Tang, L.; Zhang, P.; Kong, D.; Wang, Z.; Guan, J.; Song, G.; Tang, B.; Li, Z. Optimizing the Conditions for In Vitro Maturation and Artificial Activation of Sika Deer (Cervus nippon hortulorum) Oocytes. Reprod. Domest. Anim. 2013, 48, 27–32. [Google Scholar] [CrossRef] [PubMed]
- Uccheddu, S.; Pintus, E.; Garde, J.J.; Fleba, L.; Muzzeddu, M.; Pudda, F.; Bogliolo, L.; Strina, A.; Nieddu, S.; Ledda, S. Post-mortem recovery, in vitro maturation and fertilization of fallow deer (Dama dama, Linnaeus 1758) oocytes collected during reproductive and no reproductive season. Reprod. Domest. Anim. 2020, 55, 1294–1302. [Google Scholar] [CrossRef]
- Catalá, M.G.; Izquierdo, D.; Rodríguez-Prado, M.; Hammami, S.; Paramio, M.T. Effect of oocyte quality on blastocyst development after in vitro fertilization (IVF) and intracytoplasmic sperm injection (ICSI) in a sheep model. Fertil. Steril. 2012, 97, 1004–1008. [Google Scholar] [CrossRef]
- Vajta, G.; Rindom, N.; Peura, T.T.; Holm, P.; Greve, T.; Callesen, H. The effect of media, serum and temperature on in vitro survival of bovine blastocysts after Open Pulled Straw (OPS) vitrification. Theriogenology 1999, 52, 939–948. [Google Scholar] [CrossRef]
- Bilodeau-Goeseels, S.; Kastelic, J.P. Factors affecting embryo survival and strategies to reduce embryonic mortality in cattle. Can. J. Anim. Sci. 2003, 83, 659–671. [Google Scholar] [CrossRef]
- Givens, M.D.; Marley, M.S. Infectious causes of embryonic and fetal mortality. Theriogenology 2008, 70, 270–285. [Google Scholar] [CrossRef]
- Adams Waldorf, K.M.; McAdams, R.M. Influence of infection during pregnancy on fetal development. Reproduction 2013, 146, R151–R162. [Google Scholar] [CrossRef]
- Bicalho, M.L.; Lima, F.S.; Machado, V.S.; Meira, E.B., Jr.; Ganda, E.K.; Foditsch, C.; Bicalho, R.C.; Gilbert, R.O. Associations among Trueperella pyogenes, endometritis diagnosis, and pregnancy outcomes in dairy cows. Theriogenology 2016, 85, 267–274. [Google Scholar] [CrossRef] [PubMed]
- Watson, P.F. Use of a Giemsa stain to detect changes in acrosomes of frozen ram spermatozoa. Vet. Rec. 1975, 97, 12–15. [Google Scholar] [CrossRef] [PubMed]
- Garner, D.L.; Johnson, L.A. Viability assessment of mammalian sperm using Sybr-14 and propidium iodide. Biol. Reprod. 1995, 53, 276–284. [Google Scholar] [CrossRef]
- Trzcińska, M.; Bryła, M. Apoptotic-like changes of boar spermatozoa in freezing media supplemented with different antioxidants. Pol. J. Vet. Sci. 2015, 18, 473–480. [Google Scholar] [CrossRef] [PubMed]
- Partyka, A.; Rodak, O.; Bajzert, J.; Kochan, J.; Niżański, W. The Effect of L-Carnitine, Hypotaurine, and Taurine Supplementation on the Quality of Cryopreserved Chicken Semen. Biomed. Res. Int. 2017, 2017, 7279341. [Google Scholar] [CrossRef]
- Kotlarczyk, A.M.; Grzyb, M.; Korzekwa, A.J. Regulation of uterine function during estrous cycle, anestrus phase and pregnancy by steroids in red deer (Cervus elaphus L.). Sci. Rep. 2021, 11, 20109. [Google Scholar] [CrossRef]
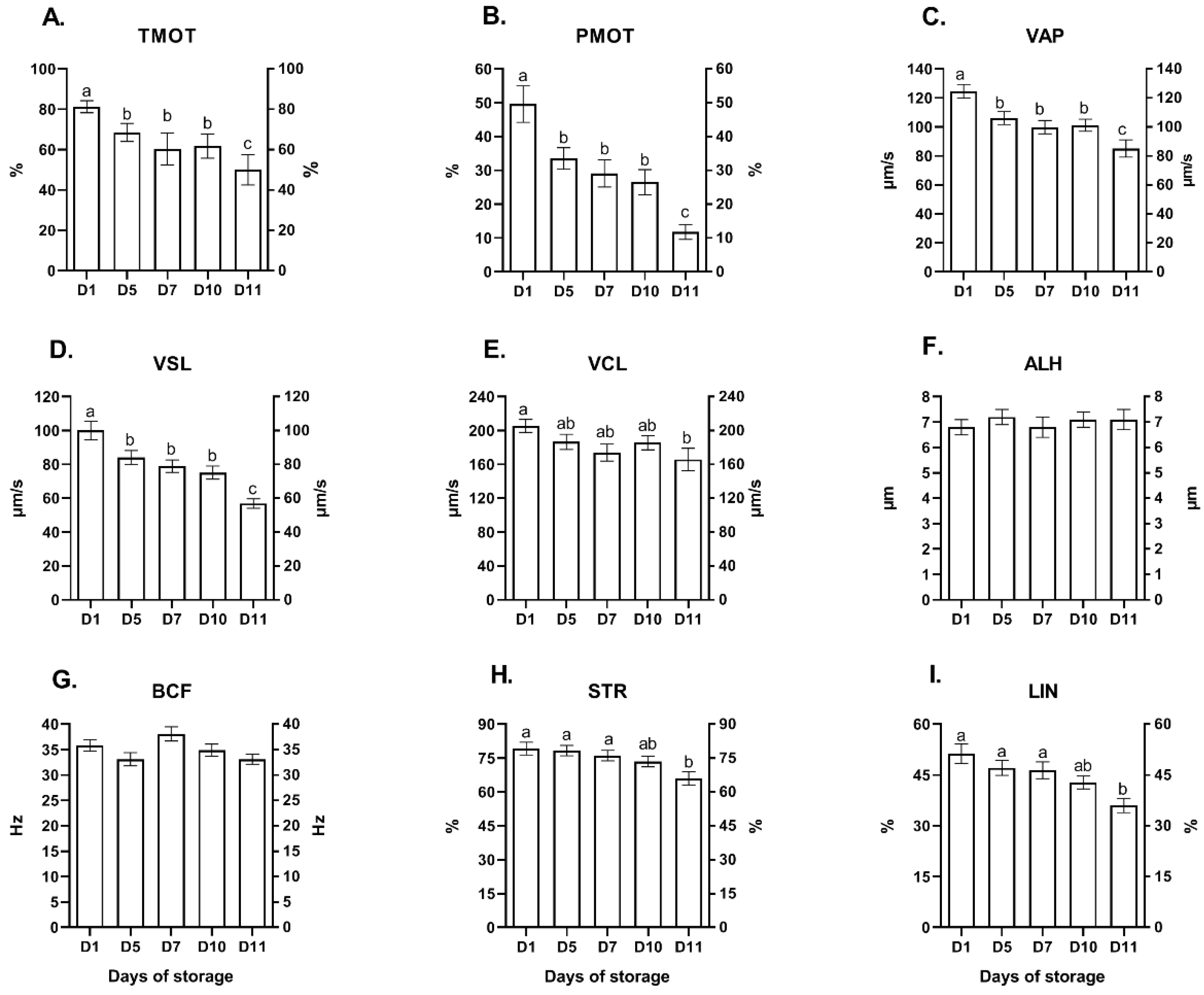


| Sperm Quality Parameters | Storage Time (Days) | ||||
|---|---|---|---|---|---|
| Day 1 | Day 5 | Day 7 | Day 10 | Day 11 | |
| MOR (%) | 75.6 ± 2.6 a | 67.6 ± 2.8 b | 66.8 ± 2.6 b | 63.5 ± 3.1 bc | 56.3 ± 3.8 c |
| NAR (%) | 89.0 ± 1.4 a | 85.8 ± 1.4 ab | 84.8 ± 1.4 ab | 81.2 ± 1.6 b | 79.8 ± 2.3 b |
| HD (%) | 5.5 ± 1.6 a | 6.2 ± 1.7 a | 7.5 ± 0.9 a | 7.1 ± 2.1 a | 20.9 ± 2.8 b |
| MD (%) | 1.7 ± 0.8 a | 1.3 ± 0.4 a | 1.2 ± 2.9 a | 1.1 ± 0.3 a | 9.2 ± 2.7 b |
| TD (%) | 7.0 ± 1.0 a | 14.8 ± 2.9 b | 17.8 ± 0.1 b | 17.1 ± 2.1 b | 21.2 ± 7.2 b |
| PNA−/PI− (%) | 79.8 ± 3.8 a | 79.1 ± 1.8 a | 77.0 ± 4.1 a | 70.9 ± 3.4 b | 60.7 ± 5.4 b |
| SYBR+/PI− (%) | 87.9 ± 1.8 a | 85.8 ± 1.4 a | 86.9 ± 2.2 a | 81.2 ± 1.6 b | 79.8 ± 2.3 b |
| MMP (%) | 88.6 ± 1.7 a | 83.1 ± 1.4 b | 86.0 ± 2.6 ab | 78.4 ± 2.3 b | 77.9 ± 5.5 b |
| AO (%) | 95.3 ± 0.9 | 91.8 ± 2.4 | 93.8 ± 1.3 | 92.0 ± 1.8 | 90.2 ± 1.3 |
| YOPRO−/PI− (%) | 86.6 ± 2.1 a | 70.5 ± 3.7 b | 71.4 ± 4.3 b | 66.6 ± 3.5 bc | 58.6 ± 4.0 c |
| YOPRO+/PI− (%) | 3.2 ± 1.0 a | 12.4 ± 5.4 b | 12.1 ± 5.6 b | 10.5 ± 5.1 b | 16.3 ± 5.4 b |
| Fertilization Potential | |||
|---|---|---|---|
| Sperm Stored One Day (Group 1) | Sperm Stored Seven Days (Group 2) | Sperm Stored Ten Days (Group 3) | |
| TMOT (%) | 82.6 ± 1.7 a | 71.0 ± 4.1 b | 64.8 ± 3.7 b |
| PMOT (%) | 42.8 ± 3.3 a | 34.8 ± 4.2 b | 21.6 ± 4.5 b |
| Total oocytes | 115 | 83 | 44 |
| Immature oocytes | 7 | 8 | 4 |
| Mature oocytes | 108/115 (93.9%) | 75/83 (90.4%) | 40/44 (90.9%) |
| Fertilized oocytes | 69/108 (63.9%) | 59/83 (71.1%) | 38/44 (86.4%) |
| Cleaved embryos | 39/69 (56.5%) a | 16/59 (27.1%) b | 5/38 (13.2%) b |
| Blastocysts | 12/39 (17.4%) | 5/16 (8.5%) | 4/38 (10.5%) |
| Pregnancy rate | 3/6 (50%) | 2/6 (33%) | 6/6 (100%) |
| Birth rate | 1/6 (17%) | 2/6 (33%) | 2/6 (33%) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dziekońska, A.; Koziorowska-Gilun, M.; Kordan, W.; Neuman, N.M.; Kotlarczyk, A.M.; Korzekwa, A.J. The Quality and Fertilizing Potential of Red Deer (Cervus elaphus L.) Epididymal Spermatozoa Stored in a Liquid State. Int. J. Mol. Sci. 2022, 23, 14591. https://doi.org/10.3390/ijms232314591
Dziekońska A, Koziorowska-Gilun M, Kordan W, Neuman NM, Kotlarczyk AM, Korzekwa AJ. The Quality and Fertilizing Potential of Red Deer (Cervus elaphus L.) Epididymal Spermatozoa Stored in a Liquid State. International Journal of Molecular Sciences. 2022; 23(23):14591. https://doi.org/10.3390/ijms232314591
Chicago/Turabian StyleDziekońska, Anna, Magdalena Koziorowska-Gilun, Władysław Kordan, Nicoletta M. Neuman, Angelika M. Kotlarczyk, and Anna J. Korzekwa. 2022. "The Quality and Fertilizing Potential of Red Deer (Cervus elaphus L.) Epididymal Spermatozoa Stored in a Liquid State" International Journal of Molecular Sciences 23, no. 23: 14591. https://doi.org/10.3390/ijms232314591
APA StyleDziekońska, A., Koziorowska-Gilun, M., Kordan, W., Neuman, N. M., Kotlarczyk, A. M., & Korzekwa, A. J. (2022). The Quality and Fertilizing Potential of Red Deer (Cervus elaphus L.) Epididymal Spermatozoa Stored in a Liquid State. International Journal of Molecular Sciences, 23(23), 14591. https://doi.org/10.3390/ijms232314591








